Isophorone Diisocyanate: Under the Lab Lens and in the Real World
Historical Development
Plastics didn’t always hold together well in the early twentieth century. After years of tinkering with organic compounds, chemists in Germany stumbled on isophorone diisocyanate (IPDI) in their search for more rugged polymers. The drive came from wartime demand for surfaces that could shrug off chemicals and physical abuse. Companies picked up IPDI for its unique ring-shaped backbone, harnessing its power to deliver clear, long-lasting coatings. Over the decades, its use found a firm foothold in industries needing performance beyond just durability—people wanted resistance to sunlight, stains, and heat too. As cities grew and cars hit the streets in greater numbers, IPDI-based coatings shaped roads, bridges, and vehicles.
Product Overview
IPDI stands out among diisocyanates because of its cycloaliphatic structure. You can spot the difference by the products that rely on it—vehicle topcoats that don’t yellow after years under the sun, and floor finishes that push back against scratches and chemical spills. Manufacturers have leaned on its lower viscosity and stable reactivity. Rather than just filling a niche, it continues to bridge gaps in protective coatings and polyurethane elastomers.
Physical & Chemical Properties
Anyone who has worked with IPDI recognizes its faint, sharp odor—not pleasant but unmistakably industrial. It remains a pale liquid at room temperature with a boiling point floating near 158°C at 5 mmHg. The cycloaliphatic ring in its backbone gives it a chemical toughness, showing up as both higher lightfastness and resistance to chlorination compared to aromatic diisocyanates like TDI or MDI. It’s soluble in standard organic solvents—think toluene, acetone, and ethyl acetate—but steers clear of water, as moisture triggers polymerization with awkward vigor.
Technical Specifications & Labeling
Chemical suppliers typically bottle IPDI under clear hazard labeling. Each container sports the CAS Number 4098-71-9, with purity (usually upwards of 99%), and batch-specific details listed on the front. The material safety data sheets spell out the handling concerns, stating reactivity with alcohols, amines, and acids, and the strict need for personal protective equipment. The labeling calls out United Nations numbers for shipping, and flammability warnings that remind even seasoned operators to keep ventilation and fire suppression close at hand.
Preparation Method
Unlike other commodity diisocyanates, synthesizing IPDI calls for a careful balance. Most factory processes begin with hydrogenating isophorone, a byproduct from acetone chemistry, to create isophorone diamine. This diamine meets phosgene under controlled conditions, producing IPDI and releasing gaseous byproducts—chief among them, hydrochloric acid. The process looks straightforward on a flow diagram, but in practice, engineers must control temperatures, monitor phosgene levels, and ensure the product doesn’t crystallize in the pipes or tanks.
Chemical Reactions & Modifications
Chemists reach for IPDI mainly to build polyurethanes. It reacts vigorously with polyols, creating a dense network that companies spin into coatings and elastomers. Its reactivity doesn’t stop there. Formulators tailor-make adducts or prepolymers using IPDI to dictate properties like curing speed, film hardness, and flexibility without the yellowing many other isocyanates show after ultraviolet exposure. Specialty waterborne polyurethane dispersions count on IPDI to deliver chemical stability without sacrificing clarity. Even in adhesives, IPDI finds favor for bonding surfaces subject to regular flexing or outdoor exposure.
Synonyms & Product Names
Isophorone diisocyanate appears on chemical manifests under several monikers. You can find it listed as IPDI, 3-Isocyanatomethyl-3,5,5-trimethylcyclohexyl isocyanate, or even by trade names such as Desmodur® I, depending on the manufacturer. On safety sheets and billing forms, these synonyms serve to reduce confusion but often point to the same pale yellowish liquid prized for specialty applications.
Safety & Operational Standards
People dealing with IPDI in factories or research labs learn fast that it brings real health hazards. The compound acts as a strong respiratory sensitizer—scrubbing every surface and airing out rooms after a spill becomes standard routine. Regulatory agencies impose strict exposure limits; for example, the American Conference of Governmental Industrial Hygienists sets a threshold limit value of 0.005 ppm for IPDI, recognizing its potential to trigger occupational asthma. Workers suit up in gloves, goggles, and full-face respirators, running air recirculation fans to minimize fume buildup. Training sessions and medical monitoring help employers catch early signs of exposure and prevent chronic health issues. Fire procedures get special attention because IPDI reacts with water, even the humidity in the air, releasing heat and hazardous gases. Emergency response plans include neutralizing agents and isolated cleanup stations.
Application Area
IPDI supports a broad range of real-world applications. In automotive repair shops, topcoat clear finishes enriched with IPDI maintain their gloss years after regular sun exposure. Industrial floors see endless forklifts, chemical spills, and hard impacts; IPDI-based polyurethanes have proven their value keeping these surfaces from pitting or chalking. Medical device manufacturers use IPDI-derived coatings on equipment housings, looking for wipe-clean safety and minimal chemical absorption. Sporting goods, electronics, and even marine deck coverings benefit from the toughness and clarity IPDI brings to the table.
Research & Development
Research teams have delved into using IPDI for aqueous polyurethane dispersions, which cut down on volatile organic compounds and support stricter regulatory requirements. Scientists keep exploring catalysts and new polymer structures to strengthen UV resistance, flexibility, or surface hardness without driving up costs. In my own work with coatings companies, even small tweaks in the polymer backbone produced by IPDI change the weathering results on metal test coupons after only a season. Pharmaceutical and biomedical engineers dive into IPDI’s biocompatibility for wound dressings and diagnostic gear, always running new toxicity panels to prove safety.
Toxicity Research
Decades of animal and human data show that unprotected IPDI exposure can cause allergic responses, including contact dermatitis and respiratory sensitization. It doesn’t build up in body fat like some organic solvents, but repeated low-level contact does sensitize workers at a higher rate than other urethane chemicals. Chronic studies in rodents and in cell cultures have revealed that the eyes, throat, and lungs are especially vulnerable, prompting numerous updates to handling guidelines and occupational exposure limits worldwide. Safety teams teach workers the warning signs—red, itchy skin or tightness in the chest—so they can seek prompt treatment and avoid serious long-term consequences.
Future Prospects
With ongoing pressure to reduce emissions and sharpen performance, chemical companies now scout for production methods that cut down on waste and minimize hazards. IPDI, being a high-value niche product, will see more work in closed-loop systems and greener catalysts as the industry shifts to smaller carbon footprints. Research labs are already tinkering with plant-based polyols and alternative diisocyanates to meet end-users’ demand for sustainable and less hazardous choices. At the same time, regulators weigh stricter workplace controls and tighter emissions measurement to keep communities and ecosystems out of harm’s way. It’s easy to see IPDI continuing to play a role in demanding applications, but its long-term track popularity will depend on how well producers deliver safer, cleaner processes and whether emerging competitors can match its strengths under stress and sunlight.
What is Isophorone Diisocyanate used for?
Everyday Life Connects to Industrial Chemistry
People might not talk about isophorone diisocyanate, or IPDI, at work or over dinner. Most folks wouldn’t recognize it by name, though plenty handle the products that trace back to it every day. IPDI plays a central role in making tough coatings and resilient plastics. It shows up on floors, cars, sneakers, and rooftops. My uncle’s old garage, which stood up to years of spilled oil and dragging tires, owed its glossy, scratch-resistant surface to the science behind IPDI.
Why Manufacturers Reach for IPDI
If you work around construction crews or DIY circles, you'll hear praise for polyurethane finishes. The secret sauce often starts with isophorone diisocyanate. Paint factories value it for reliable performance under everything from Florida heat to Minnesota frost. It’s mixed into coatings designed to stay slick, not sticky, long after other options would yellow or crack.
Automotive shops rely on IPDI-based clear coats to shield car hoods and bumpers from pebbles, salt, and sunlight. I’ve helped a neighbor check over his fender after months of winter roads, and that deep shine always survived the worst. The outer layer owes its staying power to how these chemicals bind and harden, resisting the elements.
In the world of sports equipment and outdoor furniture, manufacturers want gear to last. Think about bicycle frames that get tossed in a trunk, or sneakers splattered with mud and left to dry. IPDI-based polyurethane keeps materials flexible but tough, never brittle from cold or soft from heat.
Why Attention to Safety Matters
Plenty of folks in factories have learned that just because a substance makes life easier, it doesn’t remove risk. Isophorone diisocyanate isn’t something you’d want in your lungs or on your skin, and direct exposure brings well-known hazards. People with asthma or other lung problems feel the sting more than most, and the chemical has secured its spot on workplace safety data sheets. Proper gloves, masks, and ventilation all matter — and the stories from shop floors back up every warning in the manual.
The U.S. Environmental Protection Agency focuses on isophorone diisocyanate in its risk evaluations, recognizing its value but also tracking its risks. Workers have the most contact, but rules about ventilation and protective clothing protect everyone down the line, including the people handling finished furniture or store-bought tools.
Paths Toward Safer and Smarter Use
Industries chase new ways to cut exposure risks. Manufacturers and chemists keep searching for safer substitutes or better ways to handle materials. When alternatives fall short, more training and careful engineering controls make a difference. I’ve met shop managers who budget for air filters and regular health screenings, knowing each dollar spent keeps longtime staff around and healthy. Customers ask more questions than ever before about what goes into their purchases. Increased transparency puts pressure on companies to address worker safety and environmental impact head-on.
A clear finish on a desk or the glossy side mirrors on a new car might not reveal the chemical work behind them. Still, talking through the ways isophorone diisocyanate shapes both products and workplaces gives everyone a better grasp of the science in the background. The effort to create safer, longer-lasting coatings means constant improvements — not just to the products, but to the lives of those making and using them, too.
What safety precautions are needed when handling Isophorone Diisocyanate?
Why This Matters
Isophorone diisocyanate helps build tough coatings, adhesives, and elastomers found in many industrial sites. Yet, handling this chemical goes far beyond donning a simple pair of gloves. Those without the right approach could face skin irritation, breathing problems, or dangerous chemical burns. None of these outcomes can be brushed off, especially when lives and long-term health hang in the balance. Strong chemical exposure can leave lasting damage, and too many stories start with someone cutting a corner or skipping a simple step.
Solid Basics Go a Long Way
People in factories or labs soon learn the old wisdom: layering up matters more than bravado. Gloves rated for chemical work, splash-resistant goggles, and protective aprons take the brunt of the hazard. Even familiar exposures can hit hard, and those worn bits of skin near a wrist gap will remind anyone who rushes through prep. To truly cut risk, good gear fits tight and never leaves skin exposed.
Poor Ventilation Can’t Stay Hidden
Take air quality seriously. Diisocyanates linger as vapor, and one whiff won’t warn someone fast enough. Local exhaust hoods or high-quality respirators become the last line of defense, blocking fumes from ever reaching the lungs. Respiratory irritation leads to chronic conditions in workers caught unprotected over time. It takes action from managers to keep air circulating and to replace filters before they fail. No one should wait for the first person to cough.
Personal Experience Counts
Years spent around paint plants and resins taught me the dangers of shortcutting safety. One shift, a coworker handled a small batch without sleeves down, thinking splashing “never happens on small drums.” Half an hour later, hives forced a hospital visit and weeks off the floor. It became a hard lesson for us: even the smallest lapse brings big risks. Fresh workers and seasoned hands both improve when companies enforce frequent reminders and hands-on demonstrations.
Skin and Eye Contact: A Real Threat
Isophorone diisocyanate eats through skin’s defenses quickly. All rings, watches, and exposed zippers bring weak spots. Face shields work alongside goggles because splashes move sideways—not just straight-on like lab tests suggest. A spill means flushing with water immediately and seeking medical care. Getting complacent just once can lead to pain or long-term scarring.
Emergency Response Still Matters
Training without real practice leaves gaps. Regular drills for spills or exposures prepare teams to react without hesitation. An eyewash station in the far corner won’t help if blocked by boxes or forgotten under grime. Workers checking their routes and practicing once a season keeps everyone ready. Every step should feel second-nature, not something to puzzle out in a panic.
Management’s Role
Managers carrying clipboards do more than scouting for rules—they set the tone for caution and respect. If a team leader shrugs off a leaking valve or keeps quiet about running low on gloves, everyone follows suit. People do the right thing when a top-down culture demands it. Clear labeling, visible material safety sheets, and checklists drive habits. Above all, an open line for staff to report hazards keeps problems from turning into news headlines.
What are the health risks associated with Isophorone Diisocyanate exposure?
What Is Isophorone Diisocyanate?
Isophorone Diisocyanate, or IPDI, shows up in laboratories and factories where people deal with polyurethane products. Think about coatings, paints, adhesives. Science classes often gloss over chemicals and their dangers, but real-world experience brings out the ugly side of not paying attention. IPDI can sneak into places workers or bystanders never expect, and the impacts on health stack up fast if vigilance slips.
Breathing Problems and Asthma
The trouble with IPDI starts as soon as it hits the air. Breathing vapors triggers coughing, shortness of breath, tightness in the chest. For someone with asthma or a history of breathing issues, the impact ramps up quickly. The muscle memory of every breath feels different after inhaling things like IPDI. Workers at painting or plastics plants know that exposure to these vapors leads to chronic coughs or worsens lung disease. Repeated exposure can cause occupational asthma that doesn’t quit, turning every hallway or stairwell into a challenge.
Skin and Eye Irritation
It doesn’t just stop at the lungs. Touching liquid or vapor irritates the skin, sometimes causing itchy rashes or chemical burns. Anyone who has worked long days with protective gloves and still gotten a patch of redness can share stories about underestimating a thin film of chemical on a surface. Splashes in the eyes bring on redness, pain, blurry vision. The body’s natural alarm bells start ringing, warning about danger before a real injury sets in. Over time, even low-level contact hardens skin or brings on allergic reactions. Anyone who has worked hands-on with strong chemicals develops a sixth sense for tingling on the skin; it tells you to stop and wash up before things get worse.
Long-term Risks and Sensitization
Short exposures cause trouble, but repeated contact brings on long-haul problems. Sensitization means that after being exposed for a while, even a trace of IPDI kicks the immune system into gear. One day, working with a small amount triggers headaches, fatigue, tightness in the chest, and skin eruptions. Ordinary cleanup tasks at the job become impossible. This risk sticks with people for years after they leave a factory or workshop, making life complicated even outside work.
What Science Tells Us
Nobody should need to rely only on experience to recognize danger. Research links isocyanates in general, including IPDI, to occupational asthma across multiple industries. The U.S. National Institute for Occupational Safety and Health describes the connection between long-term isocyanate exposure and chronic lung issues. Other studies from Europe and across Asia match those findings, stressing the need for policies to protect workers. The challenge isn’t just finding the right filter masks or gloves. It means pushing companies to install proper ventilation, running real air quality tests during every shift, and training staff to spot early warning signs.
Reducing the Risks
Getting around IPDI exposure takes real investment and personal responsibility. Wearing a mask, checking fit every morning, and switching it out on time makes a difference. Company owners can swap out more dangerous compounds for less toxic ones wherever possible, but the change rarely happens overnight. Giving workers regular health checks, posting emergency wash stations in every corner, and taking complaints seriously all save lives. Putting real pressure behind safety meetings and responding to every report matters.
Safe work is everyone’s job, whether you work with chemicals or supervise from afar. Knowing about IPDI shifts choices people make at work and at home. Telling these stories helps others stay alert to dangers hidden in everyday products.
How should Isophorone Diisocyanate be stored and transported?
The Risks Nobody Should Ignore
Isophorone diisocyanate, a mouthful to say, turns out to be a dangerous substance if left unattended. People working with this chemical quickly realize how it reacts with water to create toxic gases. I remember seeing a drum with a rusty cap at a plant, and all it took was a rainy day for that thing to start hissing. That moment reminded everyone there how just a drop in the wrong place could send you to the emergency room.
Key Conditions for Safe Storage
No one wants to come into work and face a toxic cloud. That’s why clear steps must be followed. Only steel or lined drums rated for hazardous chemicals should hold isophorone diisocyanate. Fancy coatings or cheap plastics break down after a few months. Keep it in a cool, dry, well-ventilated area. Moisture sneaks in from leaky roofs, sweaty pipes, or cracked seals. Watch for condensation during winter and high humidity in summer. It pays to double check seals and inspect storage areas, especially after a big storm or batch changeover.
I’ve seen old warehouses that haven’t had their fans replaced in a decade. One malfunction, and fumes build up before you know it. A local ventilation system that keeps any vapors away from workers can make the difference between a near-miss and a full-blown incident. Smoke detectors and carbon monoxide alarms offer zero protection against this chemical. Handheld air monitors should be part of normal rounds.
Transport Never Means Relaxation
Truck drivers don’t get enough credit. They move these chemicals in bulk, across thousands of miles, sometimes through the heart of a city. Tanks must meet strict standards; that much goes without saying. But after years on the road, it becomes clear drivers deserve detailed training. How do you spot a slow leak? What do you do if traffic pins you under a highway overpass in hot weather and the pressure gauge starts to climb? When crews skimp on security seals, or skip the checklists, it’s a ticking bomb on wheels.
Many shippers use RFID tags and old-fashioned paper logs, making sure everyone can trace the location and condition of the cargo. Emergency responders want to know exactly what they’re dealing with. Labels in clear view, shipping papers on hand—that’s how you avoid confusion and get the right help fast.
Lessons Learned and Smarter Strategies
This isn’t just about obeying regulations. People have families to go home to. I believe real safety culture comes from sharing what goes wrong, not hiding mistakes. If someone finds a leaking valve, the fix should go in the next company-wide safety talk. Chemical producers now set up buddy systems for double-checking storage and loading procedures. No single person can remember every checklist—teams pick up the slack for each other.
Investing in environmental controls and personal safety gear, like splash goggles and heavy gloves, costs less than an accident. Spills in the best-run facilities still happen—having hardened floors, quick access to absorbent socks, and neutralizing solutions on hand can spell the difference between clean-up and catastrophe. Look out for each other. That mindset keeps isophorone diisocyanate—dangerous as it is—out of headlines and off the accident reports.
Is Isophorone Diisocyanate hazardous to the environment?
Looking at What’s in the Barrel
People use isophorone diisocyanate (IPDI) to make certain plastics, paints, and coatings tougher and more durable. That comes in handy, especially in construction and automotive repair, but it doesn’t mean the chemical carries no risks. When I first walked into a warehouse stacked with barrels of IPDI, I remember the workers handling those shipments with thick gloves, face coverings, and a sense of respect — not out of superstition, but hard-learned experience. The stuff can bite if spilled, inhaled, or left to drift into the air or water.
A Breath of Concern
Folks working around IPDI often talk about its harsh smell, the quick irritation in the nose and throat. That’s not just discomfort. Breathing in vapors can cause asthma, coughing, and, after enough exposure, more serious lung problems. Animal tests back this up, showing that IPDI can trigger inflammation in lungs and throats. Plants and animals living close to factories using lots of IPDI get more than a whiff — accidental releases or poor storage can put entire water systems at risk, and local wildlife doesn’t get a warning sign.
Spills and Stormwater
I’ve seen runoff from industrial yards after heavy rain, and it’s never just mud and water. If barrels leak, or if cleaning processes use too much water, IPDI can start to move offsite. It likes to hang around in water and sticks to soil, which means it can end up near creeks, wetlands, or even further. Fish and other aquatic creatures pick up traces and can struggle with toxicity. IPDI breaks down, but not quickly — so it lingers a while. Drinking water sources nearby could carry a risk, especially in areas where industrial oversight runs thin.
People Pay the Cost
Local communities around big chemical plants have fought to get tighter monitoring of air and water. I grew up near a river downstream from several factories, and we always watched for dead fish floating near the shore after plant accidents. Even with the regulations in place, not every spill makes headlines, and not every cleanup hits the mark. People who hunt or fish nearby hold real fear about what ends up on their dinner table. Studies show that isocyanates like IPDI can show up in drinking water and food when spills aren’t caught fast enough.
Doing It Better: Stopping Trouble Before It Starts
Switching to less hazardous ingredients often tops industry wish lists, but the truth is, many jobs still rely on IPDI. Until true alternatives exist, strong handling rules matter. Storage tanks need triple-checks, spill kits should never gather dust. Emergency staff and local clinics need real training about the chemical, not just paperwork. Open communication helps: factories post real-time air and water quality data so neighbors don’t need to guess about what’s drifting into their air, soil, or water. Regular inspections, quick repairs, and honest reporting bring down the risk of small leaks turning into big messes.
Trust Built on Action
Public trust depends on visible action, not just technical language in safety sheets. Workers handling IPDI deserve regular health checks and gear that actually fits. Neighbors deserve to see how the risks are managed. Each time I’ve visited communities facing chemical hazards, the message rings clear: people notice who steps up with accountability, and who waits for a problem to unfold. If industry leaders show their work, and governments keep the pressure on with unannounced inspections and strong penalties, IPDI’s dangers don’t have to define neighborhoods. Safety grows from people caring enough to stop trouble before it hits, and listening when folks speak up with their concerns.
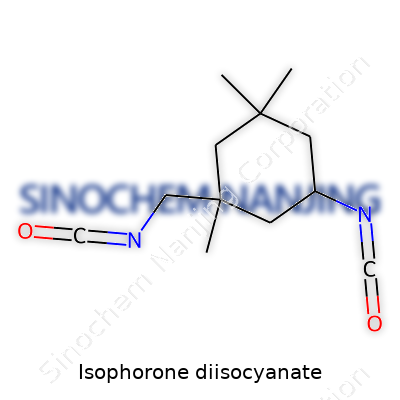
| Names | |
| Preferred IUPAC name | 3-Isocyanatomethyl-3,5,5-trimethylcyclohexyl isocyanate |
| Other names |
IPDI 3-Isocyanatomethyl-3,5,5-trimethylcyclohexyl isocyanate Isocyanic acid, 3-isocyanatomethyl-3,5,5-trimethylcyclohexyl ester Bis(isocyanatomethyl)trimethylcyclohexane |
| Pronunciation | /ˌaɪsəˈfɔːroʊn daɪˌaɪsoʊˈsaɪəneɪt/ |
| Identifiers | |
| CAS Number | 4098-71-9 |
| Beilstein Reference | 835873 |
| ChEBI | CHEBI:53304 |
| ChEMBL | CHEMBL1200892 |
| ChemSpider | 10209 |
| DrugBank | DB16735 |
| ECHA InfoCard | 07a8a4f4-3602-4b46-9e74-d8199d88066e |
| EC Number | 4098-71-9 |
| Gmelin Reference | 128181 |
| KEGG | C14268 |
| MeSH | D016439 |
| PubChem CID | 10771 |
| RTECS number | NR0175000 |
| UNII | 92V6SC438E |
| UN number | 2281 |
| Properties | |
| Chemical formula | C12H18N2O2 |
| Molar mass | 222.28 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | Sharp, musty odor |
| Density | 1.06 g/cm³ |
| Solubility in water | Reacts with water |
| log P | 3.18 |
| Vapor pressure | 0.02 mmHg (25°C) |
| Acidity (pKa) | 13.7 |
| Basicity (pKb) | 12.4 |
| Magnetic susceptibility (χ) | -5.91×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.427 |
| Viscosity | 12 mPa·s (25 °C) |
| Dipole moment | 4.62 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 489.70 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -546.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3895 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS02,GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H302, H312, H314, H317, H319, H330, H334, H335, H351, H410 |
| Precautionary statements | P210, P260, P264, P271, P280, P285, P302+P352, P304+P340, P305+P351+P338, P308+P313, P310, P312, P321, P341, P342+P311, P362, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-1-W |
| Flash point | 89 °C |
| Autoignition temperature | 230°C |
| Explosive limits | 1.0% - 10.0% |
| Lethal dose or concentration | LD50 (oral, rat): 3,900 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1230 mg/kg (oral, rat) |
| NIOSH | KWK795 |
| PEL (Permissible) | 0.005 ppm |
| REL (Recommended) | 0.005 ppm |
| IDLH (Immediate danger) | IDLH: 5 ppm |
| Related compounds | |
| Related compounds |
Isophorone cyanate Isophorone diamine Isophorone |

