Isooctene: Fueling Progress in Chemistry and Industry
Historical Development
Isooctene has found its way into industrial chemistry after years of research into hydrocarbon fuels and their performance. Petroleum refining throughout the early twentieth century drove chemists to investigate branched alkenes, aiming for molecules that packed a punch in fuel quality. Once tetraethyllead became a standard gasoline additive, demand grew for clean-burning alternatives like isooctene. This molecule began to turn heads in the 1930s and 40s, especially when engine knock problems highlighted the weaknesses of earlier fuels. Through the Second World War and into the motorbooming decades, isooctene became a benchmark in determining fuel octane numbers, marking a big leap in the petroleum industry’s drive for higher standards.
Product Overview
As a member of the C8H16 hydrocarbon family, isooctene plays a distinct role in the world of octenes. Industrial producers value its high octane rating, not just as a compound but as a reference point for blending and testing gasoline. Its stability in storage and handling also matters for both manufacturers and end users. Unlike some hydrocarbons that degrade or react unpleasantly, isooctene’s robust backbone holds up under a wide range of conditions, letting refineries trust it in everyday use.
Physical & Chemical Properties
Isooctene comes as a colorless liquid with a faint, sweet odor common to light alkenes. It sits right around a boiling point of 121–126°C and features a density of roughly 0.69 g/cm³ at room temperature. This means it floats on water and spreads quickly if spilled. Chemically, its stability owes to a branched structure, with a double bond that grants useful reactivity for further chemical transformations. Its flash point, volatility, and tendency to evaporate at ambient temperature create safety challenges that professionals know to control in a lab or factory setting.
Technical Specifications & Labeling
Manufacturers list isooctene by purity—often upwards of 98 percent for most technical grades—indicating levels of common contaminants like sulfur, water, and oxygenates. Products ship with detailed certificates that cite assay results for compliance with national and international standards, including Safety Data Sheets and hazard warnings. Labels display UN numbers, recommended handling procedures, and clear expiration dates, all spelled out so workers don’t have to guess on a busy floor or in a noisy plant.
Preparation Method
Large-scale production usually goes through alkylation, where isobutene and isobutane react over acid catalysts like sulfuric or hydrofluoric acid. Some methods dabble in dimerization of isobutene, followed by controlled hydrogenation steps to nudge the product toward the right structural isomer. Producers tackle leftovers and waste streams by supporting these processes with sophisticated distillation and gas scrubbing technologies, keeping downstream units primed for next-batch efficiency.
Chemical Reactions & Modifications
Isooctene likes to play with electrophilic additions, often picking up halogens or acids at its double bond. Hydrogenation smooths out its double bond, converting it into isooctane—a gold standard for “clean” hydrocarbon reference fuels. Chemists can coax it into a wide range of oxidation and polymerization reactions, expanding its role in specialty chemicals well beyond gasoline.
Synonyms & Product Names
Isooctene goes by a long list: 2,4,4-Trimethyl-1-pentene, 2,4,4-Trimethylpent-1-ene, and occasionally even as TMP1 in technical catalogs. Familiarity with these names can save a chemist from costly mix-ups at the ordering desk or during safety trainings. Sometimes it pops up as an ingredient in complex blends, where alternative labels keep old and new product lines connected through paperwork and regulatory filings.
Safety & Operational Standards
Handling isooctene means dealing with something flammable and volatile. National and local safety codes call for explosion-proof equipment in storage and transfer zones and rigorous safety training for every worker involved. Emergency procedures get rehearsed, even if incidents remain rare. Ventilation standards in blending facilities, regular PPE checks, and grounding of tanks and transfer lines set the baseline in responsible operations. Environmental managers monitor for leaks to air and groundwater, since spills travel fast and pull regulatory attention.
Application Area
Gasoline blending stands at the core of isooctene’s industrial use. Refineries control fuel performance by fine-tuning octane ratings with isooctene, helping high-compression engines run cleaner. Lubricant and additive companies use it as an intermediate, drawing on its double bond for further chemistry. Some specialty polymers and surfactant products include isooctene residues in their manufacturing histories, showing how far the molecule’s reach extends into daily life—often without anyone noticing.
Research & Development
Chemists keep searching for more sustainable production paths, moving away from corrosive acid catalysts and looking for solid-acid or enzyme-catalyzed methods. Some labs work on boosting selectivity and yield, aiming to minimize byproducts and curb waste. Advances in catalysis open doors to cleaner, more energy-efficient processes, something that lines up well with both corporate goals and new environmental regulations. Academic teams examine thermal stability, searching for additives or process tweaks that help isooctene perform better both in storage tanks and in combustion chambers.
Toxicity Research
Exposure studies show that isooctene, while less toxic than heavier hydrocarbons, still presents direct risks through inhalation or skin contact. Chronic exposure stories in refinery workers and tank operators have informed regulatory action, leading to stronger air monitoring and use of respirators. Animal studies look for long-term health impacts, focusing on organ systems and potential for bioaccumulation. Industry-driven and independent results support standard-setting agencies in shaping better rules, ultimately tightening workplace limits and shaping future licensing.
Future Prospects
Future demand for isooctene will ride on changes in transportation, especially as the shift to cleaner fuels speeds up. Some see potential in biomass-derived versions of isooctene, aiming to lower the carbon footprint of fuel blending. Technology innovations could see isooctene as a model substrate for new types of green catalytic transformations or as a feedstock in high-value specialty chemicals. The push for higher efficiency, greener synthesis, and safer use puts isooctene right at the intersection of classic hydrocarbon chemistry and modern sustainability efforts, pointing to a future with more refined control and broader applications.
What is Isooctene used for?
Why Isooctene Matters
Any talk about fuel advancements eventually circles back to Isooctene. This clear liquid doesn’t get the spotlight often, but it leaves a lasting mark on the way engines run and how fuels get made. Isooctene, or 2,4,4-trimethyl-1-pentene if you’re digging into a chemistry textbook, stands out because it shapes how gasoline gets its octane punch. My first job at a rural fueling station gave me a front-row seat to fuel quality issues. One lesson stuck with me: not all gasoline burns the same way, and many headaches trace back to poorly blended fuels. Turns out, Isooctene plays a part in smoothing those rough edges.
Boosting Octane Ratings
Most regular drivers glance over the “octane” number at the pump. Behind that number, there’s a mix of science and sharp business choices. Isooctene steps up by giving gasoline a higher octane rating, preventing engine knocking that can wear down parts fast and drain performance. According to studies from energy institutes, higher-octane fuels can even improve efficiency for direct injection engines. For refineries looking to make strong, cleaner-burning fuel blends, Isooctene is a key ingredient.
Role in Modern Fuel Formulation
Technicians often pick Isooctene as a reference fuel component for research. Its structure models real-world engine conditions well, letting scientists predict how engines will behave during new blends. While working with researchers on a project about alternative fuels, I came across cases where Isooctene was mixed to simulate extreme combustion conditions. Without reliable compounds like Isooctene, those test engines would sound like lawnmowers rather than luxury sedans.
Beyond Gasoline: Other Industry Uses
Isooctene’s use doesn’t stop at car engines. The chemical industry taps into its structure for making specialty chemicals, such as certain plasticizers and surfactants. Asphalt producers sometimes rely on its properties to tweak road materials for durability. Take one street in my hometown, for example: the city quietly used additives, some related to chemicals like Isooctene, that finally stopped potholes from forming after every spring thaw. Improvements like this rarely make headlines, but they keep infrastructure costs down in the long run.
Tackling Safety and Environmental Concerns
No honest discussion skips over health and safety. The American Chemical Society flags Isooctene as flammable, and short-term exposure to the vapors might cause headaches or dizziness. Refineries and labs invest in tight safety rules for storing and handling it. Environmental impacts draw concern, especially if large spills hit water systems. Tech advances in monitoring and containment help, but strict standards set by regulators keep incidents low. Giving workers up-to-date training and making sure spill response kits stay stocked builds a safer workplace.
Room for Greener Alternatives
Push for cleaner and safer fuel production remains strong. Some startups test bio-derived sources hoping to mirror Isooctene’s benefits without crude oil. Embracing green chemistry could lower emissions and make supply chains less vulnerable to oil price swings. These are early days, though—a switch to green alternatives calls for investment, research, and patience as energy companies balance cost and performance with new materials.
What are the safety precautions for handling Isooctene?
Looking Up Close at Isooctene Risks
Isooctene shows up in chemical plants and labs often, especially around fuel production and research. One thing I've learned over the years is this: flammable liquids don’t always look risky until things go sideways. And isooctene lands squarely in the high-alert category.
Working in a facility where we've had our fair share of flammable liquids, the first rule drilled into us was to keep ignition sources far away. Isooctene vapor can catch fire in a heartbeat, and normal office equipment like computers, coffee warmers, or even a static spark can be the culprit. That’s not just a checklist item from a safety manual. It’s the difference between a routine workday and a sudden emergency.
Ventilation and Storage: Not Just For Show
Open windows don’t cut it with isooctene. Local exhaust ventilation makes all the difference. Without it, vapor builds up, setting the scene for an explosive accident. In older labs where the extraction fans lagged behind, folks had close calls—headaches, nausea, or simply noticing that odd, sharp odor hanging in the air. Once you suspect a leak, getting everyone out and calling the emergency team straight away—there’s no room for hesitation.
Locking up isooctene behind proper fireproof doors, away from flammable chemicals and oxidizers, gives everyone peace of mind. Metal drums with tight seals keep the vapors in check. My team leaned on dedicated solvent cabinets—less temptation to stack things side by side or cut corners. Just because two bottles fit on the same shelf doesn’t mean they get along.
Personal Protective Equipment: No Substitute for Good Habits
Goggles and gloves serve more than just appearances. Isooctene will do a number on your skin or eyes, sometimes before you realize it. Labs provided splash-proof aprons and thick nitrile gloves for a reason—not the thin latex ones. Even with all the gear, folks learned to double-check cuffs, and never skipped face shields during transfers.
Anyone working with isooctene started each session with a team talk. We discussed what we were about to do, who’d handle which part, and where the nearest eyewash was. Even the most experienced chemists made mistakes when rushing or getting distracted.
Spill Response and Fire Readiness—Plan Beats Panic
One of my earliest safety drills involved an isooctene spill. Powdered absorbents, not rags or sawdust, worked best. Soaked-up isooctene needed its own metal disposal bin—no tossing it into the regular trash. Fire extinguishers in these rooms were always the correct class—dry chemical or CO2. I’ll never forget the tension when someone tried to use a water extinguisher on a chemical fire in training. Water just spreads the flames, making things worse.
Looking Ahead: Culture Matters as Much as Rules
Posting warning signs or handing out gloves won’t stop people from getting careless. What works is building a culture where folks look out for each other, step up if something seems off, and speak up about leaks or broken hoods. Regular workshops, open conversations about near-misses, and peer-to-peer reminders offer more protection than any instruction manual.
There’s always pressure to cut a corner if you’re running late or short-handed. Keeping safety personal—reminding each other that it means getting home in one piece—keeps everyone on their toes. Solid habits, teamwork, and honest communication pay off every single shift.
What is the chemical formula of Isooctene?
Unlocking What Isooctene Really Is
Isooctene shows up in the world of organic chemistry as a hydrocarbon, a member of the alkene family. Most people run into isooctane during conversations about fuel and gasoline, but isooctene often flies under the radar. The chemical formula for isooctene is C8H16. Just eight carbon atoms linked together, topped off with sixteen hydrogens, but its structure gives it special properties. The “iso” prefix signals a branched molecule, and the “ene” suffix tells chemists there’s at least one double bond somewhere among those carbon atoms.
Why Isooctene Matters—Even If You Don’t Think About It
Isooctene matters for more than just trivia night. It steps up as a building block for high-octane fuels, making engines run smoother. Without it, gasolines would struggle to handle modern engines that push performance and efficiency. The “octene” tells us there’s more than just fuel hidden inside. It’s a raw starting material for making a range of chemicals—everything from plasticizers to specialty lubricants. This isn’t some mysterious lab creation: refineries rely on isooctene on a regular basis.
Structure Drives Its Role
Isooctene doesn’t just look good on a molecular model. Its formula, C8H16, represents a molecule where double bonds matter. That double bond changes how the molecule reacts with catalysts, temperature, and pressure. Chemistry textbooks talk about Markovnikov’s rule and addition reactions, but the practical side comes out in refineries where tweaks to the carbon backbone create new chemicals for different uses. This isn’t just science-for-science's sake—altering the backbone gives us components for additives that prevent engine knocking, helping vehicles burn fuel more cleanly and efficiently.
An Industry Perspective: Opportunity and Hurdles
My first chemistry lab job focused on separating hydrocarbon mixtures. Isooctene stood out because it hit that sweet spot: not too volatile, not too inert. Colleagues in petrochemical research tried to tailor its structure for greater efficiency. The challenge comes in controlling those reactions. Too much heat or the wrong catalyst and you get unwanted byproducts, wasting feedstock and money. Real-world operations deal with wastewater, energy costs, and handling safety. These concerns require tight oversight, especially with rising regulatory pressure around air pollution and greenhouse gases.
Finding a Smarter Path Forward
To get more out of isooctene, the spotlight should turn to green chemistry. Better catalyst design, more efficient separation processes, and cleaner raw materials help lower environmental impacts. Some companies test out bio-based routes to isooctene, slicing carbon emissions compared to old-school oil refining. Reliable data show that more efficient catalytic converters pull out higher yields with lower waste, trimming excess costs for manufacturers and reducing byproducts that pollute the air and water.
Why This Chemistry Doesn’t Stay in the Lab
While something like C8H16 might sound like it belongs on a test, in practice isooctene shapes fuels, plastics, and everyday products. Chemists, engineers, and policymakers all share responsibility to keep exploring smarter ways to use hydrocarbons like isooctene. This approach builds better fuels and safer products while protecting the environment, all starting with a simple formula.
Is Isooctene flammable?
Not Just Another Chemical
Dealing with chemicals day in and day out, certain names come up that make people pause. Isooctene rings bells in labs and industrial sites. Folks working in fuel production or chemical manufacturing might know it for its use in blending or as an intermediate. To most people, though, it’s just another tongue-twister on a safety sheet. The reality: isooctene lights up with striking ease.
Flammability Is No Joke
Isooctene catches fire at a low temperature. The flash point, the number that tells us how quickly a liquid releases flammable vapors, sits around -12 degrees Celsius. Leave a bottle open near a spark and the risk runs high, even if the room feels chilly. This isn’t academic. Colleagues have seen spills go south with just static from a jumper or a stray tool.
Firemen train for stuff like this, but in any regular plant, not everyone stays alert. Many chemical fires start with someone underestimating flammable vapors or thinking a cold warehouse means safety. Isooctene evaporates easily. Vapors can travel away from where the spill happened and ignite somewhere else. Factories with poor ventilation or cluttered storage get hit the hardest.
What Makes Isooctene Stand Out
Among chemicals on a shelf, isooctene isn’t the only one prone to burns. What I’ve noticed, though, is that isooctene reminds even seasoned workers of gasoline—the same kind of fumes, the same headaches, the same flashing danger. Some folks stumble into trouble because they expect something that thick and oily to be safer. They get a real surprise.
To put numbers on it: isooctene’s lower explosive limit sits around 0.9% by volume in air. That means a small leak can charge up a room fast. Tests show that a spark, static shock, or hot surface brings trouble almost instantly. In a famous accident from a Midwest refinery, a forgotten unplugged lamp sparked off a flash fire that knocked out half a unit for weeks.
Lessons from the Shop Floor
What keeps people safe isn’t high-tech gear. In my experience, real prevention starts with habits. Workers should not trust sense of smell—the nose stops registering fumes after a while. Spill trays, spark-proof fans, and keeping containers tightly closed do more for safety than warning stickers.
Training goes a long way. Quick-fire drills, clear walkways, and regular checks on storage areas keep everyone sharp. Supervisors should welcome questions, not treat them as interruptions. I remember times when a new guy’s curiosity uncovered a leaky valve nobody noticed. That saved days of clean-up and a probable fire call.
Solutions: Backed by Facts and Lives Saved
Fires with isooctene grab headlines when they happen. They also leave marks in insurance claims, environmental fines, and injured pride. Standards from outfits like OSHA say properly grounded equipment and explosion-proof lights aren’t negotiable. In my years around tanks and transfer pumps, the safest yards always followed these rules day in, day out.
Online databases confirm the danger: the National Fire Protection Association labels isooctene as a serious fire risk, similar to gasoline. The solution involves more than locking up a drum—safe design, simple habits, and sharp training shape outcomes.
Bigger Picture
Isooctene stands as a reminder that flammability warnings aren’t hollow cautions. Whether someone stocks shelves, runs pumps, or just reads about the newest blend for fuels, real danger travels with the material. It’s up to everyone around it to treat that risk with respect and a willingness to act before something lights up.
How should Isooctene be stored?
Isooctene Calls for Real Vigilance
Isooctene doesn’t forgive carelessness. Used in fuel blending and chemical manufacturing, folks know this hydrocarbon always brings a flammable risk. I still remember a fire marshal at a plant stopping production for half a day just to swap out a leaky gasket on a storage tank. The sweat on his brow wasn’t just from the summer heat. Flammable liquids can end a production schedule and rewrite local news for all the wrong reasons.
Fire Risk Isn’t a Footnote
A vapor cloud near hot machinery or a stray spark, and the worst-case scenario becomes real. Isooctene storage should always happen away from ignition sources. Smoking, open flames, or even machines with hot surfaces can flip control into chaos. I once watched a team rewire an entire circuit to keep it 20 feet from a designated storage barrel—no shortcuts, no excuses.
Choosing the right container matters just as much. Isooctene generally finds a home in tightly sealed drum containers or aboveground tanks made from steel. Most people take the easy path—store it next to other chemicals—but that invites trouble. I’ve seen solvent storage rooms crammed shoulder-to-shoulder, and the smell alone warned of bad choices. Segregate it. Not just for process safety officers or compliance, but because one accident becomes everyone’s problem.
Temperature and Ventilation: Always High on the Checklist
Some chemicals can handle a drafty old shed in the yard. Isooctene wants climate control. Warm rooms push up vapor pressure inside the container. A little heat can send vapors seeping through pressure relief valves. Even without a spark, these invisible fumes can harm lungs and ruin air quality. Proper storage rooms always feature controlled temperatures and mechanical ventilation pulling fumes away from workers and equipment.
I once talked with an engineer who swore by storing isooctene below room temperature in summer—he checked the thermometer more than his phone. He said lower temperatures keep containers from swelling or venting, a lesson he learned after dealing with a pressurized drum sweating vapor under a sweltering roof.
What Safety Data Sheets Spell Out
Most companies have binders full of chemical safety paperwork, but reading those shouldn’t be an afterthought. Isooctene’s sheet warns about static electricity. Put a plastic funnel over the wrong metal can and sparks are possible. Grounding and bonding containers before transfer stops this from ever beginning. One plant manager updated his team’s training, showing a short video of a static discharge event. No Hollywood special effects needed—the crew paid more attention after that.
Personal protective equipment makes up another part of this story. Goggles and chemical-resistant gloves aren’t optional extras. Spills happen, hands slip, and isooctene can cause skin and eye irritation. Cleanup kits should never sit out of reach.
Practice Never Goes Out of Style
I’ve visited facilities where monthly safety drills weren’t just a line item for the record books. They cleared out rooms, reviewed response steps, and checked gear firsthand. Some might see it as overkill; those on the ground see families and coworkers behind every checklist.
Taking Responsibility Seriously
Proper isooctene storage isn’t a bureaucratic burden. It’s about people, jobs, and keeping the machinery humming. If you find yourself working near isooctene, pay attention—not just for your benefit, but for everyone around you. Respect chemistry’s real-world edges, and the lab coat becomes a shield, not a shroud.
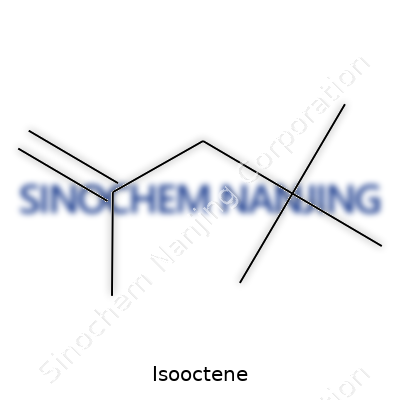
| Names | |
| Preferred IUPAC name | 4-Methylhex-1-ene |
| Other names |
2,4,4-Trimethyl-1-pentene Isooctylene |
| Pronunciation | /ˌaɪsəˈɒktiːn/ |
| Identifiers | |
| CAS Number | [ "3074-00-6" ] |
| Beilstein Reference | 1718734 |
| ChEBI | CHEBI:75364 |
| ChEMBL | CHEMBL159468 |
| ChemSpider | 58898 |
| DrugBank | DB13874 |
| ECHA InfoCard | 200-586-1 |
| EC Number | 270-994-1 |
| Gmelin Reference | 126069 |
| KEGG | C08320 |
| MeSH | D007517 |
| PubChem CID | 10467 |
| RTECS number | SQ1750000 |
| UNII | L0U920739D |
| UN number | UN2047 |
| Properties | |
| Chemical formula | C8H16 |
| Molar mass | 112.21 g/mol |
| Appearance | Colorless liquid |
| Odor | Odorless |
| Density | 0.690 g/cm3 |
| Solubility in water | Insoluble |
| log P | 2.9 |
| Vapor pressure | 2.9 kPa (at 20 °C) |
| Acidity (pKa) | 17.1 |
| Basicity (pKb) | Isooctene has no measurable basicity (pKb); it is considered non-basic. |
| Refractive index (nD) | 1.409 |
| Viscosity | 0.61 mPa·s (20°C) |
| Dipole moment | 0.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 184.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -27.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4714.7 kJ/mol |
| Pharmacology | |
| ATC code | Isooctene does not have an ATC code. |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02 |
| Signal word | Warning |
| Hazard statements | H225, H304, H315, H336, H411 |
| Precautionary statements | P210, P243, P261, P273, P280, P303+P361+P353, P304+P340, P305+P351+P338, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 1-4-2 |
| Flash point | -6 °C |
| Autoignition temperature | 415 °C |
| Explosive limits | 1.1-6.0% |
| Lethal dose or concentration | Lethal dose or concentration for Isooctene: "LD50 (oral, rat) > 2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 2,500 mg/kg |
| NIOSH | SNF62500 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Isooctene: 500 ppm |
| REL (Recommended) | 50 mg/m³ |
| IDLH (Immediate danger) | IDLH: 800 ppm |
| Related compounds | |
| Related compounds |
Isooctane 2-Methyl-2-heptene 2,4,4-Trimethyl-1-pentene Alkenes Branched hydrocarbons |

