Isohexene: Exploring Its Role and Impact
Historical Development of Isohexene
Isohexene first cropped up on the industrial radar during the rise of the petrochemical age. Chemists in the early twentieth century saw hydrocarbons like isohexene as building blocks for fuels and solvents, at a time when the global hunger for lighter, more reactive molecules was picking up speed. Researchers pushed forward by studying alkene structures, learning how branching and placement of double bonds shaped both reactivity and usefulness. Over decades, synthetic strategies moved from crude cracking processes to more directed catalytic techniques, offering higher purity and tailored isomer types. As demand for higher-octane fuels, plasticizers, and specialty monomers grew, isohexene’s presence locked itself into both research circles and industrial supply chains.
Product Overview
Isohexene covers a family of isomeric hydrocarbons, all revolving around a six-carbon chain with a single double bond and methyl branching. Among these, 2-methyl-1-pentene and 3-methyl-1-pentene turn up often in industrial production. These molecules show up in refinery streams, chemical feedstocks, and as lab reagents. Refineries extract isohexene to upgrade fuel quality, deliver chemical intermediates, and serve as a stepping-stone for making value-added compounds. Behind the scenes, isohexene stands as a proof point that even simple, colorless liquids can carry plenty of industrial weight. Since these isomers broadly resemble their parent alkenes, separating and identifying them demands tight process control and analytical care.
Physical & Chemical Properties
Isohexene falls into the family of alkenes with boiling points that hover between 62°C and 68°C depending on the particular isomer. Slightly lighter than water, its density ranges around 0.7 grams per cubic centimeter. The double bond shapes isohexene’s physical behavior, turning it into a reactive target for addition reactions, polymerization, and oxidation. In clean air, isohexene forms flammable vapors. As a non-polar molecule, it slips easily into most organic solvents and has little time for water. The liquid has a faint, gasoline-like odor, giving users a sensory cue to its volatility even at room temperature. This simple blend of characteristics has made isohexene a utility player in several transformation pathways.
Technical Specifications & Labeling
Commercial suppliers focus on purity and isomer ratio. Typical technical specs quote purity above 98%, with residual water or oxygenated impurities kept under fractions of a percent. Labels usually list CAS numbers for each recognized isomer—for example, 2-methyl-1-pentene as CAS 763-29-1. Alongside this, users spot hazard symbols for flammability and instructions for storage under nitrogen or argon. Drum labeling and safety data sheets always call out the health and fire risks, track batch numbers, and reference compliance with global transport standards. These granular details help storage managers and lab users keep safe practices tight and avoid cross-contamination in bigger process settings.
Preparation Method
Refiners typically draw isohexene from the catalytic cracking and isomerization of longer straight-chain alkanes, often working with crude n-hexane stocks as feed. Zeolite catalysts and controlled heat break carbon-carbon bonds, opening up routes for rearrangement and dehydrogenation. Skilled process engineers work to boost the yields of branched alkenes over linear ones, nudged along by tweaks in temperature, catalyst composition, and reaction time. In many labs, chemists also lean on dehydration of specific alcohols to yield pure isomers, swapping water out for a double bond. Each approach balances the scale—wide-open refinery streams or narrow small-scale synthesis—while keeping costs in line with downstream demand.
Chemical Reactions & Modifications
Isohexene reacts with halogens, acids, and hydrogen in ways that let chemists push its structure in new directions. Its double bond opens up in the face of hydrogenation, giving cyclohexane derivatives or more stable saturated hydrocarbon chains. Under the right conditions, isohexene polymerizes, helping build specialty plastics or modify resin backbones. In the presence of oxidants, the molecule transforms into epoxides, ketones, or even carboxylic acids, each with unique applications in coatings, adhesives, and flavor chemistry. Branching off from basic chemistry, functionalization steps add groups or linkers, offering a launchpad into drug intermediates or sensor reagents. Its reactivity gives industrial chemists plenty of room to invent new uses, fine-tuning reaction controls for yield, selectivity, and environmental impact.
Synonyms & Product Names
Industry catalogs refer to isohexene by names tied to its most common isomers, such as 2-methyl-1-pentene, 2-methyl-2-pentene, and 3-methyl-1-pentene. Other designations include branched hexenes, dimethylbutenes (in some global regions), or simple alkene shorthand used internally by blend technicians. Import-export paperwork sometimes lists “branched hexene isomers” if purity or separation details prove difficult to certify. Synonym confusion has flagged some safety recalls in the past; clear product naming and accurate labeling help cut down on mix-ups in large warehouses or multinational shipments.
Safety & Operational Standards
Working with isohexene always brings a need for paying attention to safety. These liquids burn fast, and their vapors catch fire from distant sparks or stray heat sources. Inhaling high concentrations causes dizziness, headaches, or even loss of consciousness, especially in closed spaces where people get careless about ventilation. Protective gear, closed storage drums, flame-proof cabinets, and gas monitoring in confined spaces all become part of the daily drill for refinery and lab users. Experience teaches that regular spill drills and up-to-date safety training keep staff aware and protect both human life and facility uptime. Compliance with workplace exposure limits—like 300 ppm in air, per many national standards—puts real teeth behind labeling and personnel monitoring.
Application Area
Isohexene pops up in several everyday industries. In refining, its branched structure helps uplift gasoline to higher octane ratings, reducing engine knock and supporting cleaner combustion. Chemical manufacturers use its reactivity as a springboard into plasticizer additives, surfactants, or building-block monomers for specialty rubbers. Laboratories often reach for isohexene as a model alkene in reaction studies, or to spike analytical standards when calibrating instruments. In the agrochemical space, isohexene derivatives play roles in chemical synthesis pathways for pesticides and herbicide adjuvants. Some adhesives and sealants pull on modified isohexene chains to give flexibility, water resistance, or improved sticking power. This variety stems from a blend of cost, availability, and easy transformation—qualities that make isohexene more than a background chemical.
Research & Development
Academic teams and corporate labs never stop looking for new uses and greener production routes for isohexene. One current thread involves biocatalytic routes, searching for enzymes that could coax renewable raw materials into branched hexenes without heavy energy spending or toxic byproducts. Another focus area zooms in on reaction selectivity, chasing after routes that limit waste and push reaction efficiency to new highs. Environmental scientists study how isohexene reacts with sunlight and air to form ground-level ozone or secondary pollutants—finding ways to limit atmospheric releases, especially near chemical parks and major shipping terminals. Partnerships between research groups, funded by both public grants and private contracts, now channel attention into cleaner, scalable technologies that cut carbon footprints and track environmental discharge.
Toxicity Research
Toxicology studies on isohexene and its close relatives reveal that acute exposures can cause irritation to eyes, nose, and respiratory tracts. Animal studies, mostly drawing on rodents, show mild central nervous system depression after inhalation at higher doses. Chronic exposure data read thinner, but regulatory agencies assign strict workplace exposure limits out of caution—recognizing both immediate and long-term health risks. Liquid isohexene sometimes defats the skin with repeated contact, leading to dermatitis or cracking, which underscores the value of gloves, goggles, and prompt washing after accidental splashes. So far, no strong evidence connects isohexene directly to mutagenicity, carcinogenicity, or reproductive risks, but knowledge gaps keep researchers and regulators watchful, especially as newer isomers or derivatives move out of the lab and into supply chains.
Future Prospects
The road ahead for isohexene looks like a mix of opportunity and challenge. With pressure mounting on refineries and chemical producers to lower emissions, the search intensifies for processes that make isohexene with less energy, less waste, and sharper control over isomer ratios. Synthetic biology could shift production away from petroleum, giving bio-based isohexene a shot via microbial fermentations. At the same time, stricter controls on volatile organic compounds and air quality push development toward closed-loop systems and improved leak detection. Engineers and business leaders watching consumer shifts—from more efficient vehicles to greener packaging—now keep isohexene on their lists for ongoing innovation. Growth depends on blending tradition with new technology, and on keeping a close eye on health, safety, and environmental respect.
What is Isohexene used for?
Chemical Foundation and Everyday Life
Isohexene crops up in plenty of places outside of the lab. As a hydrocarbon, this clear, volatile liquid sits among the many molecules that keep industrial chemistry busy. Researchers see isohexene as a building block. It’s not famous like some solvents, but its flexibility makes a difference in manufacturing.
Role in Industrial Chemistry
Isohexene shows up in chemical synthesis as an intermediate. I first spotted the name while working with a coatings manufacturer. The plant focused on producing resins. Their team used isohexene to tweak viscosity before final blending. Many coatings need fine-tuning to spread evenly and dry right. Isohexene offered the balance they needed—helping control spreadability without adding unwanted residues.
Factories lean on isohexene during the process of creating plasticizers—chemicals added to plastics for flexibility and durability. Vinyl flooring, food packaging, and wire insulation all look and feel different because of these tweaks. It’s tempting to overlook the steps before a final product lands in your house, but isohexene often plays its part long before you see a logo or label.
Applications in Fuel and Additives
Refineries look to isohexene as well. Engineers know that gasoline needs certain hydrocarbons for the engines we drive. Isohexene becomes part of fuel blends to raise the octane rating. That number matters. High octane keeps engines knocking less and running smoother, improving mileage and reducing engine wear. In the past, leaded gasoline addressed performance, but lead left a legacy of health problems. Today, refiners turn to cleaner additives, and isohexene is one piece in the ever-evolving puzzle.
Joining the Fragrance and Flavor Industry
The world of scents and flavors relies on countless chemicals. Isohexene works as a starting point for synthetic fragrances. Skilled chemists build longer or more complex molecules from it, introducing create citrusy notes or leafy green nuances for use in personal care or cleaning products. Each trend in perfumes or detergents can send buyers on a chase for a slightly different aroma, and isohexene often starts the story.
Supply Chain and Environmental Challenges
Responsible sourcing has become part of every conversation. Chemical producers now face pressure from buyers and regulators to track where and how raw materials move through the chain. Years ago at a regulatory meeting, I watched industry scientists debate air quality standards. Volatile organic compounds (VOCs) like isohexene evaporate and may contribute to smog. Cities already fighting air pollution set tighter rules, creating real tension between economic growth and public health.
Lowering emissions starts with monitoring leaks and changing system design. Secondary containment, vapor recovery systems, and better training help keep more isohexene where it belongs—in the factory or barrel, not the air. Consumers and smaller businesses need education too. Training staff not to spill, storing drums properly, and disposing of old stock right all reduce risks.
Looking Ahead
Chemistry rarely sits still. Research teams look for ways to recycle or substitute traditional ingredients like isohexene. Bio-based hydrocarbon production is slowly catching on, although costs often run higher. Green chemistry approaches—using less energy, making less waste—reshape the field brick by brick. Staying open to change helps protect both the industry’s future and the planet we all share.
What are the chemical properties of Isohexene?
Digging Into Isohexene’s Build
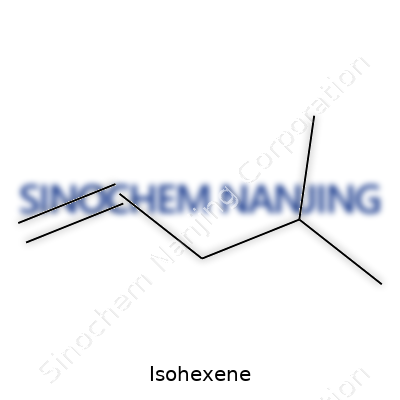
Isohexene belongs to a group of hydrocarbons called alkenes. It's not one specific molecule, but a set of isomers—all sharing the same formula: C6H12. The word “iso” just tells you that its carbon skeleton branches off in a way that’s a bit off ordinary straight chains. This branching comes into play for both chemical reactivity and how we use it in industry.
Why Structure Shapes Everything
You find a double bond in Isohexene, which means this molecule doesn’t stay still. The double bond grabs chemicals like acids and halogens out of the mix pretty fast. Isohexene’s most common isomer carries its double bond on the second carbon. This spot gets the molecule into reactions that pop up all over the place—like adding hydrogen to turn it into a simple alkane for fuel or sticking halogens onto it for specialty chemicals.
Biofuels, petrochemicals, even some flavors and fragrances—Isohexene keeps popping up. The double bond brings along higher reactivity compared to alkanes (the saturated guys). That means chemists like it for building other things, sometimes through polymerization or making intermediates for detergents. If you’ve ever wondered how some company whips up soft plastics or the base of synthetic lubricants, Isohexene’s chemistry sits somewhere in the middle steps.
Reading Its Behavior in the Lab
Isohexene behaves like other alkenes, showing a tendency toward addition reactions. If you drop some bromine water into a sample, the deep red color vanishes. That’s the double bond doing its job, sucking up the bromine. This type of reaction isn’t just for show—it’s how chemists check for double bonds in unknown samples, right in the lab.
It evaporates easily, spreading a smell you might recognize from some gasolines or solvents. In fact, volatility isn’t just an inconvenience in storage; it leads to exposure risks for workers, so anyone handling it should stay alert and use solid ventilation. The molecules don’t dissolve in water, but they spread happily in organic solvents, connecting with other oily chemicals in mixtures.
Fire, Fumes, and Responsible Handling
It’s easy to underestimate Isohexene because its clear liquid looks harmless. Burn it, and it goes up in a flash, often producing dense smoke. The same stuff that makes it precious for making fuels means there’s a big safety question wherever it’s stored or used. That double bond makes it easier to ignite than heavier or more saturated hydrocarbons, so spills and leaks need fast attention.
Breathing in the fumes isn’t exactly pleasant, either. I’ve spent afternoons working near open drums of branched alkenes, including Isohexene. The fumes sting a bit, leave a headache by dinnertime, and remind you that chemical knowledge has a real impact on well-being. Given current science, wearing organic vapor cartridges and keeping exposures limited are basic steps—not just guidelines but everyday survival on the shop floor.
Solutions and Smarter Practices
Better practices around Isohexene start with real information. Label those drums, seal the containers tight, and teach everyone around the area to treat even small leaks seriously. Upgrading ventilation saves money and health in the long run. On the research side, better catalysts and greener pathways for using Isohexene can cut down byproducts and reduce toxic waste. Industry groups publish safety data, but local leadership usually makes the difference.
Handling chemicals like Isohexene with respect keeps workers, products, and the environment out of unnecessary trouble. We’ve learned a lot from mishaps, and each new insight brings a safer, more effective use of these energetic molecules.
Is Isohexene safe to handle and store?
Why People Use Isohexene and What’s at Stake
Isohexene shows up in a lot of places — folks in labs work with it, manufacturers use it for things like solvents and chemical synthesis, and it winds up in many everyday applications. You can tell a lot about a chemical from the way people react to the name: procedures, warnings, gloves on hands, and fume hoods running full blast. That nervous approach isn’t just paranoia. Isohexene belongs to a family of hydrocarbons called alkenes, which puts it in the category of flammable substances that need respect.
Flashpoints and Fumes: Hazards Folks Shouldn’t Ignore
Let’s get the obvious fact on the table: isohexene catches fire easily. Its flash point — that’s the temperature where vapors can ignite — sits well below room temperature. That means the risk isn’t just about a spilled beaker near an open flame. A static spark, a hot light bulb, or even a poorly ventilated workspace can turn an accident into a real emergency. Vapors build up before anyone even smells them.
If you look at the safety data sheets, you’ll see repeated warnings about health effects. Breathing in vapors brings headaches, dizziness, and longer exposures may lead to problems with the nervous system. Some people shrug off short-term headaches, but chronic exposure has no upside. Deliberately skipping protection leaves people vulnerable. Splashing it on skin can cause irritation, and getting it in your eyes isn’t a mistake anyone makes twice.
Storage: No Room for Complacency
The real problem comes from treating isohexene like a harmless everyday product. Experience has taught me that people sometimes cut corners, stashing chemicals on a high shelf or letting half-sealed jugs pile up in storerooms. Isohexene doesn't take kindly to lazy handling. Storing it near oxidizers or heat sources can kick off dangerous reactions or even explosions.
Regulations recommend storing isohexene in tightly sealed containers, away from sunlight and sources of heat. Containers specifically rated for flammable substances help a lot — I’ve seen coworkers suffer minor burns just because thin-walled plastic bottles softened and leaked. Even temp swings in a garage or shed matter, so sticking to a controlled environment with fire suppression and good ventilation makes sense.
PPE Is Non-Negotiable
Not all workplaces take personal protective equipment seriously, but anyone who’s worked with volatile chemicals knows the value of goggles, gloves, and flame-resistant lab coats. I knew a guy who thought goggles were for “nervous types” — spent a night in the ER because a splash did permanent damage to his eye. You only mess up once with some chemicals.
Spills happen. The difference is cleanup becomes much safer if folks wear chemical-resistant gloves and avoid open drains, reducing the chance of inhaling fumes or sending stuff into the water supply. Fume hoods earn their keep; they’re not just accessories or extras for big labs, but essential for high-vapor work.
Better Habits, Fewer Close Calls
Incidents come from shortcuts. A training session costs less than an emergency room visit or a shutdown for a fire inspection. My own experience — seeing someone learn the hard way — taught me that clear labeling, well-maintained equipment, and regular safety drills work much better than playing catch-up during a crisis.
It’s worth paying attention to reports from regulatory bodies and occupational health groups. Hazard communication standards protect not just those in labs, but anyone downstream. Companies and universities adopting modern storage and handling protocols — like better ventilation and monitored storerooms — find their incident rates drop.
Isohexene isn’t some exotic chemical that only specialists touch. Anyone working around it earns real peace of mind by respecting its hazards. Good habits don’t just protect property; they protect lives.
What industries commonly use Isohexene?
Isohexene’s Footprint Beyond the Lab
Most people skip past technical ingredients on product labels, but ask a chemist, and they’ll tell you certain compounds keep popping up behind the scenes. Isohexene is one of those unsung compounds. It turns up in more places than most folks realize, especially across manufacturing and consumer goods.
Cosmetics and Personal Care: A Trusted Building Block
Isohexene enters the spotlight on the ingredient lists of cosmetics and skincare. Companies pick it for its light feel and quick evaporation. In my experience developing products in the beauty space, formulators want lightweight, fast-drying bases. Isohexene gives lotions and sprays that silky slip without clogging pores or leaving heavy residue. It also helps create that typical “instant dry” feel makeup primers and sprays often promise. Safety matters a lot—isohexene passes with flying colors in reviews by toxicologists and regulators, which eases concerns from consumers and product safety teams. Statistics from the Personal Care Products Council show steady yearly demand for volatile hydrocarbons like isohexene in new formulations.
Industrial Applications: Reliability Under Pressure
The paint and coatings business uses isohexene for its solvency. Formulators turn to it for blending additives and thinning agents that evaporate as a fresh layer dries. On job sites, a contractor can’t wait hours for a surface to cure. Isohexene makes quick recoats possible because it speeds up drying, meaning the crew moves on to the next project sooner. Paint technicians appreciate it, too, since it prevents pigment streaks and clumping. The same story holds for inks used in printing—print shops rely on isohexene’s volatility so pages don’t stick or smudge, keeping production lines flowing.
Pharmaceuticals: An Essential Step in Manufacturing
Even in medicine, isohexene gets its turn. It’s used as a solvent and process aid when producing active pharmaceutical ingredients, often helping draw out the purest compound possible during extraction or crystallization. Those in pharmaceutical operations trust it to pull off these delicate steps—missing the purity benchmark ruins batches and delays clinical trials. In production lines I’ve observed at contract manufacturing firms, keeping a steady supply of isohexene limits downtime and avoids regulatory headaches tied to impurity levels.
Adhesives and Sealants: Sticking Things Together
From home repairs to automotive assembly, adhesives often owe their performance to isohexene. It helps adhesives spread evenly and flash off at just the right moment. Without it, you end up fussing with sticky hands or waiting too long for a part to bond. Whether you’re patching up a shoe or assembling dashboards, you find isohexene lurking in the background, making sure the product behaves the way it should. As demand for faster-curing, lower-smell adhesives climbs, isohexene holds a solid place in these modern blends.
Looking at Safer Handling and Sustainability
Every time someone mentions industrial solvents, the conversation shifts to safety and air quality. Isohexene is flammable and needs careful storage, especially in large factories. The push for greener chemistry also encourages research into creating bio-based or less volatile alternatives. Some companies now invest in recycling processes for solvents, cutting waste and cost. As regulations evolve to limit emissions, tracking isohexene consumption becomes part of quality control. More transparency about sourcing and lifecycle impacts gives everyone—workers, end users, and managers—clear choices and peace of mind.
How is Isohexene different from other hexenes?
Getting to Know Isohexene
Most people hear the word “hexene” and think of any typical six-carbon hydrocarbon. The truth is, not all hexenes act or even look the same on a molecular level. Isohexene is a good example of how a slight change in structure changes how it behaves. Let’s break this down: isohexene doesn’t follow the straight-chain formula you’d see in 1-hexene or 2-hexene. Instead, it has a branching point—something that chemists refer to as an “iso” configuration. This small twist in the molecular structure leads to meaningful differences, both in the lab and in manufacturing settings.
Why Structure Shapes Everything
Isohexene packs a branch near the end of its carbon chain. That shift seems minor but speaks volumes about its reactivity and stability. Double bonds in molecules don’t just mean “more reactive”; their position creates different outcomes. Take 1-hexene: its open chain makes it easy to target specific chemical reactions, such as polymerization for plastic production. Isohexene’s branch, on the other hand, makes it less attractive for those exact applications but gives it an edge in places where more stability and unique reaction patterns matter.
I spent a few years working in a lab that synthesized various hexene isomers. One challenge always came up: predicting how different isomers responded to temperature shifts and catalysts. Isohexene stood out for resisting certain reactions better than its linear cousins. That resistance isn’t an inconvenience; it’s a hidden strength if you want to fine-tune a reaction or prevent unwanted side products in specialty chemicals.
Physical Properties and Real-World Impact
Anyone who’s handled isohexene in the lab will recall its distinctive boiling point and volatility profile. The branch in its structure squeezes the molecule into a more compact form, lowering the boiling point. Manufacturers tracking every last degree of energy use pay attention to that detail. Lower boiling points translate to changes in storage needs and transport strategies. For large-scale users, this adds up to real savings but also requires care with containment and fire protection.
Take gasoline blending. Linear hexenes mix in a predictable way, but isohexene’s branching alters combustion properties. Fuels with a touch of branched isomers like isohexene burn cleaner, which helps meet strict environmental standards. Oil refiners chase these branched molecules for that reason—getting just the right blend goes a long way toward making fuels that both perform and pollute less.
Challenges and Opportunities
Sourcing isohexene and separating it from other similar hydrocarbons doesn’t always go smoothly. It calls for sharper distillation cuts and sometimes more complex separation methods. These steps raise costs or slow production. Still, chemists see value in isohexene’s unique qualities—its branch lets it play a role that straight-chain hexenes rarely fill. It’s not just filler; it’s a molecule with options if you know how to work with it.
Developing new catalysts and refining separation processes stands out as a clear path forward. Research points to advanced membranes or green chemistry techniques that waste less and save energy. Bringing more efficient methods online could lower prices and expand isohexene’s role in everything from specialty plastics to high-octane fuels. Every small advance here benefits manufacturers and end-users who rely on performance and cost control in equal measure.
Why Knowing the Difference Matters
Small differences in molecular structure shape everything from chemical behavior to price and safety. For anyone working with hydrocarbons, spotting these details means the difference between just getting by and pushing the science further. Isohexene is a reminder that not every molecule plays by the same rules—and knowing those rules pays off every day in industry and research.
| Names | |
| Preferred IUPAC name | 3-Methylpent-1-ene |
| Other names |
Isohexylene 3-Methyl-2-pentene |
| Pronunciation | /ˌaɪ.səʊˈhɛk.sin/ |
| Identifiers | |
| CAS Number | 4098-32-0 |
| Beilstein Reference | 1720231 |
| ChEBI | CHEBI:136619 |
| ChEMBL | CHEMBL15427 |
| ChemSpider | 159489 |
| DrugBank | DB13836 |
| ECHA InfoCard | ECHA InfoCard: 100.117.435 |
| EC Number | 256-948-3 |
| Gmelin Reference | 163073 |
| KEGG | C14072 |
| MeSH | D007026 |
| PubChem CID | 11537 |
| RTECS number | NI4370000 |
| UNII | 9QY3TU327D |
| UN number | UN3295 |
| CompTox Dashboard (EPA) | DTXSID4020883 |
| Properties | |
| Chemical formula | C6H12 |
| Molar mass | 82.15 g/mol |
| Appearance | Colorless liquid |
| Odor | gasoline |
| Density | 0.673 g/cm³ |
| Solubility in water | Insoluble |
| log P | 2.8 |
| Vapor pressure | 3.33 kPa (at 20 °C) |
| Acidity (pKa) | 16.4 |
| Basicity (pKb) | 6.48 |
| Magnetic susceptibility (χ) | -77.9·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.3900 |
| Viscosity | 0.66 mPa·s (25 °C) |
| Dipole moment | 0.70 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 217.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -161.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3894.1 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H225, H315, H336, H411 |
| Precautionary statements | P210, P243, P261, P273, P280, P301+P310, P303+P361+P353, P304+P340, P308+P313, P331, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 1-3-0-F |
| Flash point | -6 °C |
| Autoignition temperature | 415 °C |
| Explosive limits | Explosive limits of Isohexene: "1.1-6.0% |
| Lethal dose or concentration | LD50 (oral, rat): > 5000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 5 g/kg (oral, rat) |
| NIOSH | NV1750000 |
| PEL (Permissible) | 500 ppm |
| REL (Recommended) | 5 ppm |
| IDLH (Immediate danger) | IDLH: 1,100 ppm |
| Related compounds | |
| Related compounds |
Isohexane Hexene Isooctane 2-Methylpent-1-ene 2-Methylpent-2-ene Cyclohexene |

