Isoheptene: A Down-to-Earth Commentary on Its Journey and Use
Historical Development
Isoheptene’s background isn’t talked about at kitchen tables, but for those in fields that touch chemistry, the compound has a story woven through advancements in hydrocarbons. Chemists first learned how splitting and rearranging crude oil’s building blocks could turn up molecules like isoheptene. Cracking processes during the 20th century opened up a wave of smaller, branched alkenes, and industries from gasoline to plastics were on the lookout for fuel-efficient and versatile compounds. Isoheptene—sometimes overlooked beside isooctane—became an example of how tweaking branching on carbon chains changes both performance as a fuel and its reaction profile. Over the decades, growing demand in specialty chemicals and additives prompted new ways to purify, store, and produce this molecule. I’ve seen technical literature get thicker over the years as chemists zeroed in on how isoheptene works under the hood, both literally and figuratively.
Product Overview
Isoheptene steps into the spotlight mainly as a colorless, volatile liquid, showing up in industrial pipelines as a raw material. Its role isn’t glamorous. It serves in fuel blending, intermediates for synthesis, and as a solvent for engineers who care less about a molecule’s fame than how it handles under pressure and inside closed systems. Cost, reactivity, and safety rule the day for procurement managers — every property balanced against line-item budgets and production yields.
Physical & Chemical Properties
You pick up a bottle of isoheptene, and, straight off, the smell tells you it’s an alkene — sharp and faintly sweet — reminding me of lab days when organic solvents lingered in the air. Isoheptene has a molecular formula of C7H14, with double bonds causing that “ene” ending, making it more reactive than its saturated cousins. Its boiling point hovers close to gasoline’s lighter fractions, making it manageable in refinery setups but also demanding airtight storage. It’s lighter than water, doesn’t dissolve in it, and moves easily from liquid to vapor. The compound lights up quickly — in the literal sense — so fire precautions run high in operations. The branched structure makes it stable for transport, but the double bond in the tail invites all sorts of modifications in chemical synthesis, from polymerization to halogenation.
Technical Specifications & Labeling
Producers use standard methods to report isoheptene’s purity — typically through gas chromatography — and most commercial samples claim purity above 95%. Take a look at a drum, and labeling checks off safety information, recommended storage temperatures, and shipping regulations under the UN/NA code for flammable liquids. That labeling covers international transport and regulatory compliance. Specifications often lay out water content, sulfur, and potential stabilizers added to minimize gumming or reaction during longer storage. This isn’t just a paperwork detail — trace impurities dog downstream reactions and can ruin equipment or final product quality.
Preparation Method
Refineries don’t stumble across isoheptene by chance. Oil crackers break open big hydrocarbon chains and use heat, pressure, or catalysts—often zeolites or clay—to coax out the right fragments. The process never gives just one product, so operators fine-tune conditions to maximize yields. Fractional distillation, a workhorse since the early days of oil, separates the isoheptene from similar molecules based on boiling point. Industrial plants have since adopted catalytic dehydrogenation, isomerization, or even targeted synthesis from smaller alkenes to make the process cleaner and more reliable. These upgrades don’t just affect output — they trim waste, save energy, and let operators tune the molecule for new niches.
Chemical Reactions & Modifications
Isoheptene’s double bond opens doors for modifications that keep organic chemists busy. Halogenation, hydroformylation, or alkylation can turn isoheptene into longer-chain molecules or precursors for flavors, fragrances, and advanced fuels. In one plant I toured, engineers used acid catalysts to tack on functional groups, making new plasticizers or surfactants. Polymer chemists use it to introduce branching into plastics, tuning flexibility or rigidity. Hazard comes if left unchecked — that double bond grabs radicals easily, triggering runaway reactions if stabilizers aren’t present. Watching line operators double-check temperature and pressure during exothermic runs gave me respect for their skill in keeping chemical processes safe.
Synonyms & Product Names
Isoheptene doesn’t go by just one name. Manufacturers and buyers swap terms, with “1-methylhexene,” “3-methylhexene,” or just “branched heptene” cropping up on invoices. Sometimes, trade names slip in if a blend contains antioxidants or stabilizers. This lack of consistency can confuse newcomers — I remember chasing down a mislabeled drum that turned out to be a related alkene. Laboratory and industrial buyers both keep up-to-date hazardous materials sheets and reference enterprise databases to avoid cross-orders. For regulatory filings and labels, the correct IUPAC name counts most, as improper reporting can land companies with fines or rejected shipments.
Safety & Operational Standards
Handling isoheptene isn’t for greenhorns. Flammable vapors spell hazard in large quantities, so plants enforce strict grounding, ventilation, and vapor detection practices. National and international standards dictate storage — always away from food, heat, oxidizers, or static electricity sources. Workers don flame-resistant gear, rely on sensors, and review Material Safety Data Sheets before the first drum gets opened. In my experience, operators who skip routine tank checks or ignore tiny leaks often trigger emergencies. Many facilities train repeatedly on fire and spill scenarios. Waste and washing waters never go down drains until treated, both for worker health and to keep solvents from leaching through landfills. Staying up-to-date with OSHA, EPA, and European REACH regulations keeps operations legal and builds public trust.
Application Area
Life outside the lab doesn’t see isoheptene directly, but its fingerprints show up everywhere. Fuel makers blend it into components to smooth out combustion in car engines, chasing higher octane numbers for power and efficiency. Industries making specialty surfactants and lubricants rely on isoheptene as a core building block — the same carbon skeleton that boosts performance in airplane oils or synthetic rubber for tires. I’ve also come across patents using isoheptene derivatives in pharmaceutical synthesis, chasing new routes for active ingredients. Coatings, adhesives, and paint thinners draw on its solvency and reactivity, while a smaller crowd of flavor chemists use certain downstream products for food-safe aromas. Demand shifts with trends in energy, automotives, and green chemistry, as designers search for alternatives to petroleum-heavy supply chains.
Research & Development
Academic and industry labs keep tinkering with production and new uses for isoheptene. Development often targets greener, more energy-efficient ways to make and modify the molecule, such as biocatalysis, renewable raw materials, or direct synthesis from CO2. Some papers in the last decade point to using genetic engineering in yeast or bacteria to turn plant sugars into alkenes similar to isoheptene. In polymer science, chemists look at how small tweaks in branching change performance, seeking material with exact stiffness, temperature resistance, or biodegradability. Researchers continue to study advanced purification, looking to save energy and keep impurities at bay, especially for pharmaceutical end uses. Investment in R&D often comes from public grants and private partnerships focused on circular economy goals and reducing greenhouse gas emissions.
Toxicity Research
Exposure to isoheptene at work can trigger headaches, dizziness, and irritation, especially in tight spaces where vapors linger. Toxicologists track short- and long-term effects using animal models and monitoring programs for chemical workers. Most data puts isoheptene in a moderate risk class — not acutely poisonous, but not benign either. Long-term exposure studies examine cancer risk and respiratory health, especially among workers exposed for decades. Safer concentrations and exposure durations shape workplace limits enforced by OSHA and similar agencies worldwide. Chemists designing new applications face ongoing pressure to provide safer byproducts, modify formulations for consumer goods, and design containment systems that sharply reduce leaks and spills in both factories and transit.
Future Prospects
Isoheptene won’t replace household names in fuel or plastic production, but its unique balance of reactivity, stability, and availability make it a staple in chemical toolkits. Cleaner synthesis, bio-based sources, and more selective catalysts stand to trim environmental impact and strengthen value in a world where sustainability and safety sell products just as much as performance. I’ve seen engineers pitch new ideas for storage and blending to boost efficiency, while policy makers push regulations nudging chemists toward less polluting materials. Growing demand in electronics, medical tech, and renewable sectors means isoheptene will stick around, though its finished forms may look different in coming generations. Young scientists entering the industry will bring fresh thinking, but lessons learned from the old hands — about care, caution, and creative problem solving — will always have a place alongside the lab bench.
What is Isoheptene used for?
What Isoheptene Brings to the Table
Isoheptene might not turn many heads at first mention, but its role in manufacturing and science runs deeper than many realize. In my early days working alongside a team in a small specialty chemistry lab, I remember handling isoheptene as just another name on a long list. Over time, I understood what makes it helpful and, in some industries, even essential.
Making Things Work in the World of Chemicals
Isoheptene belongs to the alkene family. Its molecular structure, with seven carbon atoms and a double bond, helps it take part in reactions that build more complex chemicals. Companies put isoheptene to work in the production of some specialty solvents and as an intermediate for other organic compounds. Experience in a manufacturing plant showed me how tough it can be to find the right substance for a reaction. Isoheptene, with its unique structure, lets chemists fine-tune chain length and offer products with just the right properties for adhesives, coatings, or fuels.
Automotive and petrochemical sectors press isoheptene into service in octane improvement. Gasoline standards keep getting stricter, and additives that boost engine performance without piling up harmful emissions matter more year after year. Isoheptene, blended carefully, forms part of tailored fuel solutions helping engines run smoother and longer.
Pharmaceuticals: The Less-Seen Side
People talk a lot about blockbuster drugs and fancy active ingredients, but background chemicals like isoheptene help set up some of those successes. Isoheptene serves as a building block during the synthesis of certain pharmaceutical agents. In small batches, researchers use it in labs to make molecular tweaks that could change how well a medicine works or how likely it is to break down safely inside the body.
Challenges and Safety
Isoheptene does not escape the usual risks that come with hydrocarbons. Flammability, proper storage, and exposure limits always top the list of concerns. In my hands-on experience, even small leaks can lead to strong fumes, so good ventilation, airtight containers, and regular safety checks become essential. Over the years, I have seen that many accidents trace back to rushed handling or not respecting those basic protocols.
Sustainable Paths Forward
Production of isoheptene relies mainly on petroleum processes. The chemistry world faces growing calls to cut environmental costs. For isoheptene, this could mean developing more energy-efficient synthesis methods or trying routes that start from plant-derived feedstocks. Green chemistry isn’t always simple, but making even small switches can lead to safer workplaces and a lighter carbon footprint.
Learning from Use, Not Just Theory
Anyone who has watched a process line grind to a halt because of the wrong chemical grade understands how every link in the supply chain counts. Isoheptene deserves its spot, not because of star power, but because it solves specific, real problems in real factories and labs. It shows that even low-profile compounds can carry big responsibilities in the world of chemical production, fuel, and medical research.
What are the side effects of Isoheptene?
Understanding Isoheptene in Everyday Terms
People who look for headache relief sometimes come across isoheptene, often found in combination migraine medicines. Doctors used to prescribe it more often, but the medical community now has more choices with fewer problems. Even if this drug seems like a quick fix for sharp, pounding headache pain, it brings a handful of side effects that deserve honest discussion.
How Isoheptene Affects the Body
Isoheptene contracts blood vessels in the brain. That action eases the throbbing sensation migraine sufferers know too well. Tightening those vessels can cause trouble, especially for people with blood pressure issues. Some folks notice their heart pounding or racing. Maybe sweat breaks out above their eyebrows. Sometimes nerves get jumpy, with shaky hands and a sense of restlessness. Even if these reactions pass quickly, they get your attention.
Heart and Blood Pressure Issues
Heart palpitations draw the most complaints. Your chest might thump and your pulse skip in a way that feels hard to ignore. Because isoheptene narrows blood vessels, it may push blood pressure higher, so people who struggle to control their numbers should be especially cautious. I’ve seen people take a migraine medicine in a hurry, only to regret it as they nervously watch their pulse shoot up.
Effects on Mood and Body
The nervousness and jitters can bleed into your mood. Some people become irritable or anxious after taking isoheptene. Sleep may go out the window for a few hours, thanks to the stimulant effect. Some folks notice dry mouth or lose their appetite. Rarely, blurred vision, ringing ears, or numbness in fingers and toes shows up—definitely signals to stop taking the medicine and talk to a doctor.
Rare but Serious Risks
Though less common, isoheptene may cause bigger headaches than it solves. People with a history of heart trouble, chest pain, or stroke face higher risks. Combining this drug with certain antidepressants or other stimulants can create dangerous spikes in blood pressure. Once, a neighbor with a family history of stroke felt his arm go numb after two doses; his doctor told him to stay away for good.
Safe Use and Alternatives
Some medical societies no longer recommend combination migraine medicines with isoheptene. Safer and more modern prescription options get favored now. If you pick up an over-the-counter migraine remedy, review the label and talk with your pharmacist. It pays to track how your body responds. Share any weird side effects with your healthcare provider—especially if you feel faint, dizzy, or can’t shake heart palpitations.
What Can Be Done?
Awareness matters most. People should talk frankly with their doctors about migraines or headaches before reaching for a fast fix. Medical professionals should keep up with guidelines and avoid old drugs with a high side effect burden. For those looking for safer alternatives, lifestyle shifts, stress-reduction tactics, and newer medication classes offer more hope than isoheptene in a rush. Head pain deserves real solutions, not just another round of unwelcome side effects.
How should Isoheptene be taken or administered?
A Look at Isoheptene and Its Uses
Isoheptene often gets prescribed for the treatment of certain kinds of headaches, like migraines or tension headaches. It belongs to a group of substances known for their effect on blood vessels. Doctors trust it to help constrict dilated vessels and dial down the relentless pain that some people face during tough headache episodes. The directions for taking this type of medicine come down to getting a clear plan from a healthcare provider who actually knows your health story.
Routes and Dosage: Why the Details Matter
Doctors usually recommend swallowing isoheptene by mouth with a glass of water. This is not something offered in an injection or patch. Pills or tablets make for easy, familiar dosing. Most adults will recognize the routine: open the bottle, count out a tablet or two, take it with water, and get on with the day.
Taking more than directed doesn’t speed relief and can lead to real trouble: pounding heart, anxiety, shaky hands, even dangerous spikes in blood pressure. Years ago, I learned from a relative with chronic migraines that doubling up on these types of drugs to “get ahead of the pain” offers nothing except extra side effects. Following prescription directions is more important than some may think.
Why Supervision Is Not Just a Recommendation
Isoheptene carries risks, especially in people with a history of heart disease, high blood pressure, or certain mood disorders. It turns out a lot of us walk around with risk factors we don’t even know about until we confront them. Because this medicine acts on blood vessels, a doctor needs to check for hidden problems first. Ignoring these details or borrowing someone else’s prescription can land a person in the ER.
Drug interactions make things even trickier. Isoheptene reacts with other medications, especially ones that raise blood pressure, or drugs for depression. I remember a neighbor who once started a new migraine drug and ended up with scary side effects because no one warned him about the clash with his daily blood pressure medicine. Conversations with your doctor and pharmacist really do matter. Keep an accurate list of everything you take, including herbal or over-the-counter remedies.
Tips to Reduce Risk
I tell anyone who listens: do not split pills, crush, or try to mix isoheptene with food or drink unless your doctor says it’s fine. Consistency wins over creativity. Set reminders. Track when and how much medicine you take, especially during stressful weeks when headaches feel nonstop. For people who have memory issues or a busy lifestyle, pill organizers or a friend who checks in can help prevent mistakes.
Most important, speak up if anything feels off. Chest pain, racing heart, confusion, or new symptoms deserve quick attention. There’s no sense in toughing it out or waiting for things to get worse.
Responsible Use and Community Knowledge
Modern medicine advances fast, but the basics—clear instructions, follow-up appointments, honesty about side effects—have never lost their power. Those facing migraines or tough headaches rely on isoheptene because it offers real relief. Still, safe use relies on respect for the process, a willingness to listen, and a little old-fashioned caution. Health comes from partnership, not guessing games.
Are there any contraindications for Isoheptene?
Understanding What Isoheptene Means for Health
I’ve spent years reading research and watching how drugs like isoheptene get used and talked about. Folks often find themselves stumbling across the name while dealing with migraine headaches, cluster headaches, or sinus pain. Isoheptene works as a vasoconstrictor, tightening blood vessels to ease pain caused by swelling and pressure. This usefulness draws attention, but it also raises medical questions. Who shouldn’t get near it, and why?
People Dealing with High Blood Pressure
If there’s one group who consistently pops up in warnings, it’s those with hypertension. That tightness isoheptene brings to blood vessels can help headaches, but it also can bump up blood pressure. Studies connect vasoconstrictors to higher risks of stroke, heart attack, and other cardiovascular events for people with preexisting high blood pressure. I’ve watched patients experience spikes even after a single dose, which can turn a solution into a serious problem. Doctors routinely flag this as a clear contraindication.
Heart Disease and Circulation Problems
Heart disease doesn’t mix with vasoconstrictors. I’ve seen doctors dig deep into patient histories, asking questions about angina, hardening of arteries, and previous heart attacks. Isoheptene, by tightening vessels, raises the strain on the heart. For folks with existing circulation issues, this risk can outweigh any benefit. Research covering several vasoconstrictors—including isoheptene—makes the same point: Heart and peripheral artery disease put people in the high-risk group, and the decision tilts heavily toward avoidance.
Overactive Thyroid (Hyperthyroidism)
People living with hyperthyroidism face a unique risk. Overactive thyroid means the system is already running in overdrive, often making pulse and blood pressure unpredictable. In my own circle, I’ve seen folks with hyperthyroidism react oddly to medicines that stimulate the body. Isoheptene can make these symptoms worse, jumpstarting the metabolism and heart further. Endocrinologists often say no to this drug for thyroid patients.
Other Significant Warnings
Glaucoma patients may run into problems with isoheptene, too. The drug tightens tiny blood vessels in the eye and can raise pressure, a dangerous thing with glaucoma. Also, people who’ve suffered recent strokes or who have certain kidney disorders fall under the watchful eyes of pharmacists wary of medicine-induced flare-ups.
Drug Interactions and Age Considerations
Interactions can sneak up quickly. Those taking other medications affecting blood pressure, antidepressants like MAO inhibitors, and some stimulants can play poorly with isoheptene. Older adults, specifically, can see an outsized effect—a little goes a long way, tipping blood pressure or bringing on confusion. Caregivers and family members I’ve talked to usually need close supervision to catch problems early.
What Works Instead?
Doctors do have choices beyond isoheptene when the risks pile up. For migraine pain, options like triptans, caffeine combinations, or even some nerve blocker devices give relief with lower risk for people in high-risk groups. Reading up on these and talking directly with a medical professional has helped many around me sidestep dangers while still managing pain.
Why It All Matters
No drug comes without trade-offs. Seeing firsthand how folks can slip through the cracks, I respect the power of a quick risk check, a deeper conversation with a doctor, and paying close attention to health history. With a drug like isoheptene, knowledge can be the truest safeguard.
Is a prescription required to obtain Isoheptene?
Understanding Isoheptene and Its Uses
Isoheptene gained attention over the years as a component in headache and migraine relief, often combined in products with other pain relievers or caffeine. Most people ran into it by way of old-school formulations, especially in certain combination headache medications. Many patients relied on these formulations for quick, reliable relief. Anyone who's suffered a sudden migraine understands how valuable that can be.
Prescription Rules and Why They Matter
In the United States, the regulatory environment for headache medications changed a lot since the 2000s. Mainstream pharmacies stopped stocking isoheptene-containing products on store shelves. By 2018, the FDA decided that drugs with isoheptene as an ingredient should come under stricter oversight. The rationale ties back to safety and risk of misuse. Isoheptene acts as a vasoconstrictor, narrowing blood vessels. That means potential heart risks, especially for people with blood pressure issues. Given these health concerns, government agencies want a doctor to review a patient’s history and factor in possible drug interactions.
Today, in the US and much of the world, you need a prescription for any medication containing isoheptene. It doesn’t matter whether it’s pure isoheptene or mixed with other drugs – you need medical clearance. If you try searching for products with isoheptene in pharmacies, a pharmacist will ask for a valid prescription. Online pharmacies also follow the law, blocking access unless you confirm you’ve seen a healthcare provider first.
Experience and Safety Concerns
Having worked with patients battling migraines and vascular headaches, I’ve seen the struggles first-hand. People often want fast solutions because pain disrupts their work, their families, their mood. In the past, patients would sometimes bypass consultation, self-dosing with over-the-counter pills without understanding the risks. Some would later come in with elevated blood pressure or strange side effects. Regulation wasn’t designed to create hurdles for people with pain, but to protect against avoidable harm.
Beyond individual risk, there’s a broader public health goal here. Medications with vasoconstrictive action can cause problems if mixed with other drugs, or if people have hidden health conditions. A healthcare provider doesn’t just write a prescription—she asks about your full medical picture. This protects you, and it stops inappropriate use that might jar other systems in the body.
Access Challenges and What Can Help
The prescription-only status causes real problems for those who relied on isoheptene-based drugs for years. Not every alternative works for everyone. Some patients tell me they feel “stuck” navigating insurance coverage or appointments just to get basic relief. Here lies a real opportunity for improvement. Easier telemedicine visits could cut down on wait times for necessary prescriptions. Insurance can do better at approving those visits, or ensuring migraine patients aren’t steered toward less-effective or more costly medications.
Doctors who listen, spend time explaining drug risks, and offer practical solutions help the most. Pharmacists also play a big role—many can recommend options or direct patients to clinics where they’ll actually get the right help the first time. With more open communication and a health system tuned to patient experience, we can meet safety goals without walling off relief from the people who need it most.
Finding the Right Help
Anyone needing relief from headaches or migraines should start with professional guidance. While self-medicating at home sometimes worked decades ago, risks are clearer now and drug interactions grow more complex as people age. Modern healthcare leans toward safety with prescription requirements for good reason. If you’re missing effective relief, connect with a provider who understands your journey. Health shouldn’t feel out of reach—or out of your hands.
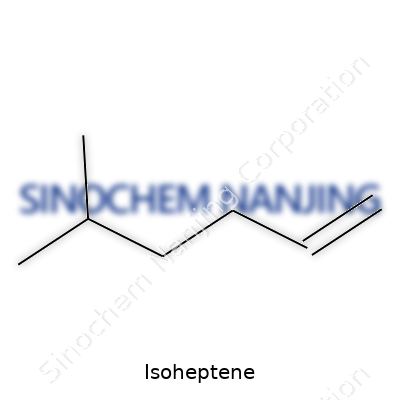
| Names | |
| Preferred IUPAC name | 3-Methylhex-1-ene |
| Other names |
1-Heptene Heptylene n-Heptene |
| Pronunciation | /ˌaɪ.soʊˈhɛpˌtiːn/ |
| Identifiers | |
| CAS Number | 7756-98-7 |
| 3D model (JSmol) | `Isoheptene` (also known as 4-methyl-2-hexene), JSmol 3D model string: ``` CCCC(C)C=C ``` This is the SMILES string representation commonly used to feed into JSmol for 3D rendering. |
| Beilstein Reference | 1740846 |
| ChEBI | CHEBI:44682 |
| ChEMBL | CHEMBL1531337 |
| ChemSpider | 54698 |
| DrugBank | DB08797 |
| ECHA InfoCard | 100.029.920 |
| EC Number | 203-624-3 |
| Gmelin Reference | 166073 |
| KEGG | C08313 |
| MeSH | D017355 |
| PubChem CID | 638051 |
| RTECS number | NJ6950000 |
| UNII | TLA8A2S6S3 |
| UN number | UN2049 |
| Properties | |
| Chemical formula | C7H14 |
| Molar mass | 96.17 g/mol |
| Appearance | Colorless liquid |
| Odor | mild odor |
| Density | 0.705 g/mL at 20 °C |
| Solubility in water | insoluble |
| log P | 2.96 |
| Vapor pressure | 286 mmHg (20 °C) |
| Basicity (pKb) | ISOHEPTENE does not have a defined pKb as it is a hydrocarbon and not a base. |
| Magnetic susceptibility (χ) | -7.44e-6 |
| Refractive index (nD) | 1.3960 |
| Viscosity | 0.545 mPa·s at 25°C |
| Dipole moment | 0.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 221.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –29.72 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4166.7 kJ/mol |
| Pharmacology | |
| ATC code | R02AD02 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H225, H304, H315, H336, H411 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P280, P303+P361+P353, P304+P340, P370+P378, P403+P235 |
| NFPA 704 (fire diamond) | 2-4-2 |
| Flash point | -6 °C |
| Autoignition temperature | 254 °C |
| Explosive limits | 1.1–7.1% |
| Lethal dose or concentration | LD₅₀ (oral, rat): 5000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 370 mg/kg (oral, rat) |
| NIOSH | NM9100000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.2 mg/m³ |
| IDLH (Immediate danger) | IDLH: 800 ppm |
| Related compounds | |
| Related compounds |
Heptene Methylhexene Isooctene |

