Looking at 2,2'-Azobisisobutyronitrile: Realities Behind a Key Industrial Chemical
Historical Development
Stepping into the history of 2,2'-Azobisisobutyronitrile, better known as AIBN, brings up the steady progress of practical organic chemistry over the past century. Chemists in the 1940s, particularly in Japan, searched for effective radical initiators to improve polymer production. Before AIBN came onto the scene, peroxides often filled that role but carried a fair share of safety risks and handling difficulties. The discovery of AIBN’s unique abilities to decompose predictably and generate nitrogen gas without leaving behind troublesome byproducts signaled a big change. Over time, countless industries have relied on this compound, trusting its properties to drive crucial reactions—especially in polymerization and organic synthesis. My own awareness of AIBN’s importance hit home during work in a research lab, watching its impact on yields and process reliability, making it clear why its discovery shaped not only synthetic chemistry but also daily manufacturing practices around the globe.
Product Overview and Synonyms
Take a stroll through a modern laboratory or a busy industrial facility, and references to AIBN jump out under different names—Vazo 64, 2,2’-azobis(2-methylpropionitrile), or simply “azo initiator.” Sticking with clear terminology keeps processes streamlined, especially when trades cross borders or shift between academic and industrial settings. AIBN often comes as a white, crystalline powder, and after decades of fine-tuning, suppliers now assure strong quality control matching technical specifications that fit demanding tasks. Knowing the different labels for this molecule matters, since any mix-up with names can trip up ordering, waste precious resources, and risk mishandling—a headache I’ve seen disrupt even seasoned labs.
Physical & Chemical Properties
Ask someone in polymer chemistry what they look for in a radical initiator, and stability plus predictability land near the top of the list. AIBN checks those boxes. At room temperature, it holds steady as a colorless solid, favoring longevity on the shelf if kept dry and cool. The molecular formula, C8H12N4, supports a moderate molecular weight, and the nitrile groups help set its decomposition pattern. AIBN’s standout feature comes from its thermal breakdown near 65°C, releasing nitrogen gas and two stable isobutyronitrile radicals. This specific property allows for cleaner reactions compared to alternatives like benzoyl peroxide, which can release unwanted acids and contaminate final products. Its solubility in many organic solvents, from acetone to toluene, gives process engineers options for different manufacturing routes. The compound’s lack of strong odor and easy handling—so long as operators mind the temperature limits—lets technicians focus more on the chemistry at hand rather than the logistics.
Technical Specifications & Labeling
Anyone who’s ordered AIBN for a project knows suppliers tout purity levels that usually exceed 98%, a mark critical for high-precision synthesis or controlled radical polymerizations. Labels also warn of decomposition temperature and outline exact storage instructions: shield from sunlight, keep below 40°C, and always cap the container tightly. Shipping codes align with global transport regulations, since AIBN’s classification as a flammable solid requires respected hazard controls. Some countries’ labeling rules force a clear GHS hazard pictogram marking its irritant potential and environmental risk. From an operational perspective, these rules mean more than just bureaucracy; they remind everyone down the line to respect the power—and hazards—packed into each bottle. Slipping up here, whether through sloppy labeling or guesswork, brings real-world consequences, not just paperwork headaches.
Preparation Method
Crafting AIBN starts with the reaction of isobutyronitrile and hydrazine derivatives, a process tuned over decades for yield and scalability. Sodium hypochlorite or similar oxidizing agents set off the azo linkage formation. This pathway, though conceptually simple, requires precise temperature and pH controls; stray from those and you may end up with a messy mixture difficult to separate. On an industrial scale, optimizing these steps balances output and purity with a sharp eye on worker safety and environmental discharge. Plants producing AIBN invest heavily in process controls and emission capture, since any misstep can lead to dangerous gas releases or heat buildup. Watching large-scale AIBN synthesis up close, the coordination between chemical engineering and safety oversight stands out, showing that behind every bottle is a network of hands-on expertise.
Chemical Reactions & Modifications
AIBN’s value ties closely to its predictability in generating free radicals. Heat sparks its breakdown, cleaving the molecule and spitting out a pair of carbon-centered radicals beside a burst of nitrogen gas. Chemists depend on those radicals to kickstart polymerizations, cross-link organic frameworks, and drive modifications that would otherwise limp along or stall at room temperature. Some research teams tweak AIBN, swapping in functionalized side chains, to shape the rate or selectivity of radical generation, making it more tailored for specialty applications. Concerns about byproducts and waste keep popping up, especially as environmental standards tighten, pushing companies to invent cleaner post-reaction purification methods. Still, AIBN’s base chemistry stays a mainstay—its reliability outweighing the flashier but less-proven competitors flooding the market every few years.
Safety & Operational Standards
No discussion of AIBN gets far without flagging its safety side. At the right temperatures, it behaves nicely, but let it overheat, and decomposition accelerates into runaway reactions. This risk gets magnified in bulk storage or during summer months, a lesson driven home after several high-profile incidents in poorly ventilated or climate-ignored storage sites. Handling demands gloves, goggles, and ventilation, plain and simple, not as “best practice” but as non-negotiable basics for working with crystalline azos. AIBN’s fire risk runs higher in dust or mixed with flammable solvents—two conditions never far from most industrial settings. Regulatory bodies in North America and Europe push for regular refresher training on “hot work” with AIBN and call for emergency protocols that do more than check boxes. Even with all this, it’s not unusual to find complacency slip in, especially with old hands who have handled AIBN for years without mishap.
Application Area
It’s nearly impossible to name a sector untouched by the reach of AIBN-triggered radical chemistry. Plastics—PVC, ABS, polyacrylonitrile—owe much of their backbone to AIBN polymerizations. Beyond plastics, the pharmaceutical world taps AIBN-induced reactions to craft intermediates too complex for simple routes. Laboratory-scale work also leverages AIBN for fine-tuning small molecule transformations. I’ve seen startups lean on AIBN to pilot new materials with unique surface properties, counting on reproducibility and low contaminant formation. With environmental regulations getting tougher, AIBN-based processes attract renewed attention for their relatively “clean” byproducts and the ease of integrating nitrogen venting into existing plant air-handling systems. Sectors leaning on its radical chemistry will likely keep growing, even as alternatives get a closer look.
Research & Development
AIBN still draws keen interest in research circles. Newer work focuses on more efficient initiation methods, coaxing down temperature thresholds to reduce energy costs. Teams are exploring “masked” AIBN derivatives releasing radicals under specific conditions for targeted reactions, a clever route especially appealing in the pharma and materials science worlds. Environmental pushback against legacy chemicals has nudged many to re-examine AIBN’s lifecycle—scrutinizing every aspect, from synthesis waste to end-product decomposition. There’s ongoing debate about greener alternatives, but few candidates offer the same reliability and affordability. My time spent looking through scientific journals makes one fact obvious: while the textbook uses of AIBN stay constant, the creative tweaks and repurposed applications grow year after year.
Toxicity Research
Toxicology doesn’t look past AIBN. Lab studies show AIBN acts as an irritant; swallow or inhale enough, and symptoms like headache, dizziness, or even central nervous system effects show up. Long-term risks, though less well-understood, draw concern, especially after breakdown into reactive byproducts. Environmental studies highlight its moderate aquatic toxicity, which pushes operators to capture any waste streams before discharge. Some parts of the world hold stricter limits than others; I’ve seen facilities face shutdowns over unmonitored AIBN emissions. Toxicologists keep pressing the point: respect AIBN’s hazards and support ongoing research, lest overlooked risks come back to bite industry and communities alike.
Future Prospects
Looking ahead, AIBN stands at a crossroads. Cleaner chemistry attracts investment, and companies keep searching for initiators leaving an even lighter environmental mark. On the other hand, few can challenge AIBN’s price point paired with dependable results. Regulatory changes may squeeze margins or throttle production in regions lagging on air and water quality enforcement. Yet, research pipelines brim with smarter applications—think triggers for bio-based polymers or in controlled drug delivery systems. The ongoing shift toward “green” chemistry may one day supplant AIBN, but for now, its mix of safety, reliability, and performance keeps it central to countless industries. Those invested in its supply or use would do well to follow both regulatory trends and technological developments, since the next breakthrough in radical chemistry could rewrite more than a few process manuals.
What is 2,2'-Azobisisobutyronitrile used for?
The Story Behind the Chemical
Scrolling through the industrial catalogs, you bump into some chemicals with tongue-twisting names. 2,2'-Azobisisobutyronitrile, usually written as AIBN, popped up in my undergraduate days in the lab. The professor brought out a bright white powder and asked who could kick-start a polymer reaction. Hands shot up. Someone mentioned “free radicals.”
AIBN shows up a lot where plastics and polymers are born. People searching for stronger, lighter, cheaper materials often turn to science for help. Think of the millions of bottles, toys, car parts, and wiring sleeves made with synthetic polymers every year. Polymers transform the raw ingredients—monomers—into new stuff built to last or stretch or bend. Most of that work depends on getting the right reaction started, and AIBN delivers as a key initiator.
Sparking the Reaction
AIBN breaks down and tosses off nitrogen gas and two reactive fragments, which nudge the monomers to join a chain. Unlike some other starters, AIBN doesn’t spit out harsh acids or metals. Safety counts, both for workers and the environment. Manufacturers mix AIBN into vinyl chloride to make PVC, but it also shows up in acrylics, polyacrylonitrile, and ABS plastics. It offers a little more control over the structure of these polymers, so the finished products have the right toughness or flexibility or transparency needed for real-world uses.
It’s Not Just Plastics
Curiosity about chemistry doesn’t stop with plastics. Organic chemists use AIBN in the lab to start all sorts of chain reactions. I’ve seen it spark a reaction called the Wohl-Ziegler bromination, which sticks bromine onto hydrocarbons. This helps in building more complicated molecules, from drugs to dyes. You’ll find journal articles where researchers favor AIBN to avoid messy byproducts and keep their mixtures cleaner. Cleaner reactions mean less separation later and, sometimes, better yields. In pharmaceuticals, people need absolute purity. Every shortcut matters for both cost and safety.
Downsides and Precautions
Handling AIBN can’t be taken lightly. Like so many chemical powders, it’s classified as hazardous. Breathing in the dust or spilling it isn’t an option. Its biggest risk comes from the way it can break down to produce nitrogen gas—and pressure. Too much makes it prone to explosion if overheated or confined. Storage means strict temperature control. Trained staff, proper ventilation, gloves, and goggles all make up the daily grind when working near it. Factories and research labs keep close watch with sensors and safety gear.
What’s Next: Greener Approaches?
Scientists and engineers look for alternatives to AIBN and related initiators. The aim is to reduce environmental impact without giving up what works in today’s production lines. Using sunlight or small amounts of more natural substances as initiators gets attention. More research happens every year, with groups testing newer systems that need less hazardous waste cleanup. Safer choices matter for workers, communities, and landfills alike.
For now, AIBN holds a central place in everything from burger wrappers to car dashboards. Understanding and reimagining its role can help us make the products we depend on better, safer, and a little greener.
What safety precautions should be taken when handling 2,2'-Azobisisobutyronitrile?
Getting Real about Risks
2,2'-Azobisisobutyronitrile, usually called AIBN in the lab, doesn’t announce itself with a strong smell or flashy warning signs, but that doesn’t mean it sits quietly on the shelf. AIBN starts breaking down at fairly low temperatures and literally pops off nitrogen gas. In the right conditions, it can go off hard enough to send broken glass or plastic flying. Looking at hospital records, most chemical injuries involve cutting corners—usually someone leaves out the basics because they’re in a rush or feel like they know the stuff already.
Honest Precautions: Practical Steps
Nobody wants to dress up like an astronaut for every single task, but working with AIBN means dressing for the job. Cotton lab coats beat synthetics because melted polyester sticks. Gloves matter—regular nitrile gloves handle short contact, but once the powder spills, folks need to wash up and change gloves. Safety goggles save eyes, yet I’ve watched even experienced chemists grab safety glasses off the counter and not bother with side shields. Nitrogen gas escapes fast, so always open containers in a fume hood. The gas won’t smell, but it stays deadly in tight spots.
Powder can float off a spatula during weighing. Lining the balance and bench with clean paper cuts down on spilled dust, and as soon as weighing ends, sealing everything keeps the air and skin clean. One of my team members learned this the hard way—a little puff traveled up and left him with a light rash. Messes happen, but extra time on setup shortens cleanup and stops real harm.
Storage Details That Matter
AIBN doesn’t love warm afternoons or direct sun. Flammable chemical cabinets with a stable, cool climate stretch shelf life and slow down unwanted reactions. Forget the crowded benches; every time AIBN sits near acids or metals, tiny lumps and bumps accelerate its breakdown. Keep those bottles by themselves, far from curious hands and heat sources. Label clear, with a bold “explosive” note, and set a reminder to check the bottle each quarter. More than once I’ve seen the powder turn off-white— a warning to toss the rest and call in waste services.
Training Everyone, Not Just the “Experts”
Lab training usually focuses on big hazards, but AIBN tends to sneak through safety classes because it isn’t always dramatic like ether fires or HF acid burns. Anyone who stocks shelves or empties bins needs to hear about its hazards, not just the PhDs and the postdocs. Regular refreshers, with real stories of accidents, stick in people’s minds more than a factsheet hidden in a drawer.
Emergency Plans: Get Specific, Not Generic
Explosions seem remote—right up until they aren’t. I’ve watched a curriculum overhaul after a bottle popped in a neighboring lab and sent project notes flying. Now, every new hire learns the exact steps for dealing with airborne powder, minor spills, and unwanted heating. There’s a bucket of sand marked for chemical fires, never water, set within arm’s reach for anyone scaling up reactions. The university brings in medical pros for hand-on training in chemical exposure and injury response.
Building Long-Term Habits
Safely working with AIBN isn’t about memorizing numbers. It’s about building thoughtful habits through repeated drills, practical clothing, honest discussions, and checks beyond bare compliance. None of it takes much time but skipping a step can cost a lot more than a simple lab mess.
How should 2,2'-Azobisisobutyronitrile be stored?
Understanding the Risks
Anyone who has handled chemicals like 2,2'-Azobisisobutyronitrile knows it commands respect. This powder, familiar to anyone who’s spent time in a polymer or research lab, comes with a real risk factor. Its main danger isn’t something dramatic or invisible—heat is the problem. Once this material gets warm, it can break down fast, producing nitrogen gas and heat. This runaway reaction rarely ends well. Stories of ruined batches, near-misses, and close calls fill online forums and word-of-mouth safety briefings.
Checks for a Proper Storage Spot
Labs often face space shortages. I’ve seen makeshift cabinets in basements and sheds crammed with all sorts of substances—it never turns out well. For 2,2'-Azobisisobutyronitrile, the site has to be cool, dry, and out of direct sunlight. A refrigerator designed for chemicals—not for snacks and lunch boxes—works best. General-purpose fridges may sound convenient, but they pose fire and contamination risks. I’ve watched staff make excuses about using home fridges, only to deal with leaks and accidental contact with food.
The Right Container
Original packaging, sealed and labelled, always beats improvised jars and recycled bags. Manufacturers know the best way to contain chemicals, so it pays to leave powders in their supplied bottles. Double containment isn’t about paranoia—it’s about lowering the stakes of a spill or accidental exposure. I remember a coworker relying on flimsy zipper bags once. A few days later, the powder had seeped out, contaminating a drawer. It cost hours in cleanup and forced the purchase of costly cleanup supplies. Lesson learned.
Humidity and Moisture
Some assume powder means safety from water—but moisture wreaks havoc. Water vapor in the air triggers slow decomposition and changes how the chemical behaves in experiments. Desiccant packs tucked inside storage cabinets help a lot. Silica gel works reliably and costs little. Professionals check those packs every few weeks and swap them out before they lose effectiveness.
Access Control and Labeling
Access isn’t just about locking doors. Keep logs and restrict entry to trained staff. Accidental mix-ups happen when chemicals are left on communal benches. Bold, clear labels with hazard symbols keep even rushed workers from careless mistakes. I once saw a temp confuse this compound with a harmless buffer because someone scribbled the name on masking tape. Proper labelling would have prevented that scare.
Fire and Ventilation
No one expects a fire until something starts smoking. Fire-resistant cabinets are a must for chemicals like this. Some places cut corners and store high-risk powders in wooden cabinets; insurance claims and safety audits later, protocols get strict. Good airflow, such as a well-maintained fume hood, dissipates any fumes that escape. A strong exhaust system can turn a potentially deadly mistake into a harmless lesson.
How the Real World Fixes Storage Problems
Some issues trace back to training gaps and pressure to save time. Continuing education for lab staff makes a real difference. Posting up-to-date safety sheets near storage areas has saved me and my coworkers more than once from dangerous shortcuts. Budget restraints stop some labs from upgrading storage. In these cases, pooling resources in shared facilities improves both access and safety.
Facts Above Assumptions
Chemical manufacturing and research always come down to trust—trust in your equipment, your colleagues, and the habits drilled into your day. Choosing the right storage strategy for 2,2'-Azobisisobutyronitrile cuts risk and saves money in the long run. In my experience, strict policies and a respect for the material mean fewer emergencies, better results, and safer workplaces for everyone.
What is the molecular formula of 2,2'-Azobisisobutyronitrile?
Why Learning About Chemical Formulas Still Matters
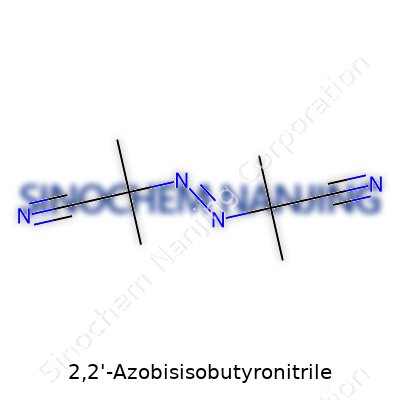
A lot of folks see long chemical names and glaze over. But the formula for 2,2'-Azobisisobutyronitrile, or AIBN, packs more punch in the real world than most would expect. It’s C8H12N4. That tidy arrangement isn’t just trivia for chemists; it unlocks the door to how this compound jump-starts plastic production and even affects safety in many labs.
The Role of AIBN in Industry and Daily Life
Most plastics and rubbers start from small molecules stitched together into giant chains. AIBN makes these chains possible by serving as a free-radical initiator. Pop the cork on a bottle of this stuff, and the molecules break into little fragments that set off the chain reaction to build polymers. Thanks to its chemical structure, AIBN leaves little nitrogen gas as a by-product, which gets vented away.
Polyvinyl chloride, or PVC, comes to mind right away—it’s used in pipes, window frames, even some medical gear. Without AIBN’s precise ratio of carbons, hydrogens, and nitrogens, the process would stall. Factories save money and cut down on the number of chemicals involved, making AIBN a steady favorite since the 20th century.
Understanding Risk: Safety and Environmental Impact
There’s another side to this molecule. AIBN has a reputation: it can be unstable, especially when heated or mishandled. That means explosions can happen if storage doesn’t follow strict rules. I’ve seen whole labs sent scrambling because someone ignored the warnings about temperature or containment.
Rules exist for a reason. European regulators treat AIBN as a hazardous material, and the National Institute for Occupational Safety and Health (NIOSH) flags it as a health hazard. The dust or powder can irritate skin and eyes. Anyone who has ever cleaned up after a spill can share a story about stubborn blue stains and stinging hands. Safety training places much more stress on not just knowing the formula, but respecting what it means to work with nitrogen-rich compounds.
How to Make Use of Chemical Knowledge
Real understanding of what “C8H12N4” represents means looking past the letters and numbers. Anyone working in labs or manufacturing should start with real-world habits: double-check your personal protective equipment, keep stock away from sunlight, and know where the nearest emergency shower sits. No textbook replaces keeping your nose and eyes clear.
People in the future fields of green chemistry are eyeing ways to swap AIBN for safer initiators. Some research groups in Japan and the US report success with alternatives that break down to water and CO2. Right now, most companies rely on AIBN because it’s reliable and cheap, but pressure keeps growing to cut the footprint left from nitrile compounds.
A Few Steps Forward
At its core, the molecular formula of AIBN signals a crossroads between utility and caution. If more labs respect the weight that comes with each subscript and symbol, fewer accidents and smoother production will follow. Experts who keep learning about each chemical’s story raise standards for the whole field. General chemistry class may seem far off, but one glance at a chemical’s formula can still tip you off to the story worth knowing.
Is 2,2'-Azobisisobutyronitrile hazardous to health or the environment?
Understanding What We’re Dealing With
In the plastics and chemical world, 2,2'-Azobisisobutyronitrile – usually shortened to AIBN – stands out. Folks making plastics, rubber, and even pharmaceuticals often turn to AIBN for its knack for kicking off chemical reactions. It’s a pale, crystalline powder, easy to blend in, and loved as an initiator in polymer production. This chemical looks harmless resting in a jar, but that first impression doesn’t tell the whole story.
A Real Risk to Human Health
Look at any serious safety sheet or scientific review and dangers pop up quickly. AIBN breaks down into toxic gases, especially when heated or exposed to shock. One of those gases? Hydrogen cyanide. Even a whiff of hydrogen cyanide can cause trouble, attacking the body’s ability to use oxygen. Common sense tells you that a dusty chemical which might puff out cyanide gas doesn’t belong anywhere near your nose or mouth. Lab workers always pull out goggles, gloves, and masks just to be safe.
Breathe in AIBN dust, and you invite headaches and dizziness. Swallowing it – not that anyone sane would do so – leads to stomach trouble, weakness, even confusion. Occupational studies show repeated exposure brings bigger risks: skin reactions, allergic responses, irritation in the lungs. Some countries force companies to keep levels in the air below strict limits, but accidents still happen, especially if someone skips the safety rules just once.
What About the Environment?
Left unchecked, AIBN causes a stir in local ecosystems too. Here’s where environmental scientists put their boots on: AIBN biodegrades slowly. Spilled or dumped AIBN leeches into soil and water, where sunlight and bacteria struggle to break it down. Traces then drift into streams or rivers, where aquatic life starts to feel the effects. Fish and water insects show stress signs, and toxins from AIBN’s breakdown can stay active for months. Small mistakes upstream linger in the water supply, and the story doesn’t end with fish — toxic breakdown byproducts work their way up the food chain.
Factories know these risks and face real pressure to keep emissions low. Wastewater rules get tighter every year, driven by the proven danger to wildlife and future water sources. AIBN isn’t the only hazardous chemical in play, but it doesn’t do the environment any favors either.
Shaping Sensible Solutions
AIBN keeps showing up in industrial processes for a reason: it works fast and reliably. That said, health and ecological risks give people every reason to look for new paths forward. One smart approach finds companies swapping AIBN for safer alternatives which don’t produce cyanide, or switching to closed systems that nearly eliminate leaks and worker exposure. Some research teams develop new initiators built from plant-derived chemicals, though old habits and cost concerns slow adoption.
Strict training for staff and doubling down on ventilation and protective equipment put a real dent in accidents. Proactive cleanup plans for spills help, too. It’s not about outlawing chemistry — it’s about showing respect for what these compounds can do, both good and bad. In the end, responsible use mixed with steady innovation lets us take advantage of AIBN’s power, while sidestepping the worst hazards it brings along.
| Names | |
| Preferred IUPAC name | 2,2'-Azanediyl bis(2-methylpropanenitrile) |
| Other names |
AIBN Azobis(isobutyronitrile) Vazo 64 Chemabsolute AIBN |
| Pronunciation | /ˌeɪzoʊˌɪsəˌboʊ.tɪr.oʊˈnaɪ.trɪl/ |
| Identifiers | |
| CAS Number | 78-67-1 |
| Beilstein Reference | 1208734 |
| ChEBI | CHEBI:38703 |
| ChEMBL | CHEMBL1779 |
| ChemSpider | 6736 |
| DrugBank | DB06813 |
| ECHA InfoCard | 03b193a5-4c95-4378-82b4-3eb3b771d6eb |
| EC Number | 208-912-9 |
| Gmelin Reference | 74584 |
| KEGG | C06421 |
| MeSH | D000375 |
| PubChem CID | 8265 |
| RTECS number | U03500900 |
| UNII | OJ7C5O6NLN |
| UN number | UN1324 |
| Properties | |
| Chemical formula | C8H12N4 |
| Molar mass | 164.21 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.1 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 0.67 |
| Vapor pressure | 0.08 mmHg (at 20 °C) |
| Acidity (pKa) | 18.1 |
| Basicity (pKb) | pKb 5.1 (25 °C) |
| Magnetic susceptibility (χ) | -637.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.3667 |
| Viscosity | 1.52 mPa·s (at 25 °C) |
| Dipole moment | 4.37 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 229.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -35.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2096 kJ/mol |
| Hazards | |
| GHS labelling | GHS02|GHS07|GHS09 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H225, H302, H317, H334, H335, H400, H410 |
| Precautionary statements | P210, P220, P221, P261, P273, P280, P305+P351+P338, P308+P313, P342+P311, P370+P378 |
| NFPA 704 (fire diamond) | 3-3-2-SPECIAL:OX |
| Flash point | 77 °C (171 °F) |
| Autoignition temperature | 525 °C |
| Explosive limits | Explosive limits: 1 - 7% |
| Lethal dose or concentration | LD50 oral rat 640 mg/kg |
| LD50 (median dose) | 640 mg/kg (rat, oral) |
| NIOSH | TE9275000 |
| PEL (Permissible) | PEL: 5 mg/m³ |
| REL (Recommended) | 0.3 mg/m³ |
| IDLH (Immediate danger) | 500 mg/m3 |
| Related compounds | |
| Related compounds |
Hydrazine Azobenzene Isobutyronitrile Azodicarbonamide Azobisformamide |

