Isobutyric Anhydride: A Deep Dive into the Science and Its Impact
Historical Development
Back in the early twentieth century, the field of organic chemistry saw a surge in the demand for novel anhydrides. Isobutyric anhydride entered the stage as researchers chased after efficient reagents to drive acylation reactions. Industrial synthesis scaled up around the mid-century, responding to advancements in plastics, pharmaceuticals, and agrochemicals. The chemical world craved reliable intermediates that held up under manufacturing stresses, and isobutyric anhydride fit this bill. Its early use shaped a pattern of exploration and risk-taking that remains in chemistry today. Those days relied on curiosity and trial, leading to many modern industrial chemicals, not through accidental discovery, but through persistent inquiry.
Product Overview
Isobutyric anhydride pops up in labs and factories as a pungent, almost oily-looking liquid. The sharp odor betrays its reactive nature. Producers sell it chiefly through chemical vendors catering to research, synthesis, and specialty applications. Bottles and bulk drums, tightly sealed, line shelves from university storage lockers to vast chemical warehouses. Many see it as a workhorse acylating agent—handy for adding bulk and branching to molecules where direct acid or chloride methods might stumble. Beyond lab benches, it feeds into the messy business of crafting intermediates for pharmaceuticals, polymers, and fragrance additives.
Physical & Chemical Properties
With a clear, colorless appearance, isobutyric anhydride delivers a characteristic, biting aroma. Its molecular formula (C8H14O3) reveals a structure packed with two linked isobutyric acid residues. A boiling point around 177°C means it stands up to moderate heat without breaking down, but its volatility demands care in open-air work. Its density and solubility differ from similar anhydrides, which sometimes affects reaction efficiency. This compound doesn’t just add an acyl group; it actually transforms the physical landscape of molecules, thanks to its steric bulk and high reactivity. In my work, using this anhydride meant always checking venting and cooling, as the vapor could overwhelm even an experienced chemist’s nose.
Technical Specifications & Labeling
Chemical supply labels slap on warnings: flammable liquid, corrosive, and avoid skin or eye contact. Most bottles list assay percentage—almost always above 98% for research—and provide key identifiers like CAS number 97-72-3. The packaging survives solvent attack and restricts vapors, using thick-walled amber glass or steel drums. A safety data sheet offers guidance, but anyone working with isobutyric anhydride learns quickly that even a small spill can clear a room. Companies specify storage under inert gas or dry nitrogen, a nod to the compound’s reactive thirst for water in the air.
Preparation Method
Manufacturers prefer two routes: treating isobutyric acid with phosphorus pentachloride to yield the acid chloride, then reacting this with the acid’s sodium salt to make isobutyric anhydride; or using acetic anhydride directly with isobutyric acid in a dehydration reaction. Each process juggles temperature, solvent choice, and workup complexity. I’ve watched teams opt for acetic anhydride out of convenience and safety, despite the need to handle excess reagents and solvent waste later. The drive isn’t elegance alone; it’s also an economic march toward scalable, consistent batches across global production sites.
Chemical Reactions & Modifications
Isobutyric anhydride rarely sits dormant. Its main work goes toward acylation—hooking up with alcohols, amines, or phenols to make isobutyrylated products. These reactions feed pharmaceutical syntheses, giving rise to esters and amides with bulk and protection against unwanted side reactions. The compound’s branched structure adds hydrophobicity to target molecules, steering their solubility profiles. I’ve seen its use expand into the shape-shifting world of custom molecules, where small tweaks create new properties or improved performance in final products. Reactions tap into the clever interplay between kinetics and thermodynamics, a reminder of how skill shapes outcomes in synthetic chemistry.
Synonyms & Product Names
Walk through a catalog—or even a patent filing—and you’ll find isobutyric anhydride listed as 2-Methylpropanoic anhydride, Propanoic acid, 2-methyl-, anhydride, or simply Isobutyryl anhydride. Some vendors toss in historical or localized names, reflecting shifts in nomenclature over time. This muddle can sow confusion in sourcing and compliance, so professionals keep CAS 97-72-3 close at hand. Each synonym anchors chemists and procurement managers in a web of documentation, ensuring traceability and regulatory alignment.
Safety & Operational Standards
Every chemist knows the sting of isobutyric anhydride’s vapors—eyes water, nose burns, and gloves must stay snug. Handling calls for chemical splash goggles, respirators in tight or poorly ventilated settings, and thick nitrile gloves. Labs install fume hoods and ventilated cabinets, aiming to keep exposures well below recommended occupational limits. Emergency response teams train for quick neutralization, often using dilute alkali to mop up spills. Industry trends show increasing automation in closed systems, reducing skin and inhalation risks. Safety outcomes don’t rest only on good habits; they weave together equipment, policies, and a shared sense that one careless act could spark a serious incident.
Application Area
Pharmaceutical companies lean on isobutyric anhydride to manufacture intermediates where bulky ester or amide protection blocks are crucial. Beyond drugs, certain polymers get their needed side chains from this molecule, tweaking resilience or handling traits. Agrochemical designers grab it for building blocks in selective herbicides and fungicides. Flavors and fragrances industries use it—sparingly, given the odor and regulatory flags—to introduce complexity in synthetic blends. Universities and research institutes incorporate it into exploration of new catalysts, molecular machines, or even responsive surfaces. My own projects used it to create hard-to-find reference materials—a testament to its role in filling synthesis gaps and enabling custom chemistry challenges.
Research & Development
Academic labs keep asking tough questions, often using isobutyric anhydride to probe structure-activity relationships in drug discovery and polymer design. Journals fill with studies where delicate manipulations of the acyl group unlock properties or probe unknown reaction mechanisms. Some teams point to greener synthetic pathways, replacing hazardous reagents or seeking catalytic methods that trim waste. Industrial R&D outfits investigate automated, continuous-flow production that minimizes exposure and ups efficiency. The conversation around analytical standards and traceability gets louder every year—especially as complex molecules demand ever-tighter control of intermediates.
Toxicity Research
Safety studies point out that isobutyric anhydride doesn’t just irritate; it crosses the line into real risk with prolonged exposures, especially through inhalation or skin absorption. Standard test models show acute effects ranging from coughing and eye redness to, in high doses, pulmonary or dermal toxicity. The compound hydrolyzes swiftly in water, breaking down into isobutyric acid, but this process releases heat and acid vapors, further complicating remediation. Researchers look for data gaps, feeding toxicology reviews that shape safe handling policies. Real-world incidents—accidental splashes or leaks—support a consensus for robust training and rapid response readiness.
Future Prospects
Industry keeps evolving, and pressure mounts to cut hazardous waste and improve yield. Green chemistry principles now guide choices in acetylation chemistry, with isobutyric anhydride facing scrutiny for its emissions profile and ecological impact. I see research avenues exploring enzymatic or bio-based alternatives that mimic isobutyric anhydride’s role, reducing regulatory hurdles while keeping costs manageable. As new application areas like advanced materials and custom pharmaceuticals stretch chemical boundaries, demand remains strong for unique, tailored acylation agents. Yet, the future may reward those who rethink traditional protocols, streamlining reactions and slashing waste. With steady investment in safer, more sustainable chemical processes, this workhorse reagent will adapt, finding a place in both entrenched and emerging production strategies.
What is Isobutyric Anhydride used for?
Where Does Isobutyric Anhydride Fit In?
Isobutyric anhydride doesn’t get much credit outside of labs or factory floors, but its presence quietly shapes a slice of what ends up in manufactured goods. Shops selling chemicals label it as a specialty reagent, and most people never see the name pop up on a product label. Still, trace it back, and you’ll notice this compound impacting pharmaceuticals, pesticides, and flavors used by food companies.
Key Uses Across Industries
Pharmaceutical developers often rely on isobutyric anhydride when making complex compounds. Take certain anti-inflammatory medications or antibiotics as examples. The molecule brings its special reactivity—basically, it helps connect other chemical building blocks more efficiently. It’s more than just a helper; without it, some drugs become cost-prohibitive or simply too difficult to produce in large batches. This matters for patients on tight budgets and supply chains that need reliable sources.
Agricultural labs have found similar value in isobutyric anhydride. When designing crop protectants or new types of pesticides, the goal is to ensure potency and stability. Isobutyric anhydride steps in during synthesis, helping lock in the chemical features that prevent pests from damaging crops. Farmers depend on these advances for healthy, market-ready yields, and ultimately, it means more reliable harvests in the face of changing climate conditions.
I remember walking into a friend’s flavorings lab once and catching that strange, sharp whiff as they mixed up a batch to replicate “butter” for popcorn. Turns out, isobutyric anhydride helped create some of the foundational artificial flavors. In tiny amounts and under tightly controlled conditions, this chemical brings a tart note to flavor profiles that mimic tangy cheese or creamy butter. Food technologists have warned that purity matters since even small contaminants can shift food safety and taste. That care reflects a real priority for anyone eating processed or flavored products and points to the value of tight oversight.
Safety and Environmental Impacts
Not every tool comes without caution. While manufacturers gain value from isobutyric anhydride, improper handling can cause real harm. Its vapors irritate skin and lungs if workers don’t stay protected. Regulations around storage, waste disposal, and air quality monitoring keep this risk in check. I’ve seen protocols in place at chemical plants—mandatory air filtration, protective clothing, regular safety briefings. Smart companies take these controls seriously, not just to avoid fines, but to keep skilled people healthy and able to work day-in, day-out.
Environmentalists raise concerns about any chemical that could leach into soil or water. For isobutyric anhydride, responsible disposal matters more than almost any other point in its life cycle. Waste management teams capture residues before they reach the outside world, and tighter emission standards address potential air release. Effective oversight usually depends on local regulations and inspections—the stronger the rules, the safer the outcome.
Pushing for Smarter Solutions
Green chemistry research keeps pushing toward replacements and cleaner processes. Some labs now experiment with bio-based alternatives or new synthesis strategies to cut hazardous byproducts, hoping to bring costs and risks down. Companies willing to innovate or invest in next-generation technology can often streamline production, cut waste, and keep both workers and neighbors safer. For those of us living near factories or relying on finished goods, the effort shapes how safe the entire supply chain feels.
Isobutyric anhydride may never become a household name, but people who care about medicine access, food safety, and sustainable farming have a stake in how it’s made and managed. Watching where science and industry meet, small changes in a single chemical’s journey can ripple out to touch many lives.
What are the safety precautions when handling Isobutyric Anhydride?
Getting Real About Chemical Safety
Plenty of folks working in labs or manufacturing run into unfamiliar chemicals every week. Some get attention for being flammable, some smell terrible, and others just sneak up on you if you don’t read the label. Isobutyric anhydride falls into that “don’t cut corners” category. It’s known for its strong, sharp odor and its ability to irritate skin, eyes, and lungs in a hurry. You get one whiff and immediately know it means business.
Protect Yourself First
Nobody enjoys spending extra time suiting up, but personal protective equipment saves discomfort and potential doctor visits. Splash-proof goggles never won any fashion contests, but one drop in the eye can change your outlook fast. Nitrile gloves and a lab coat protect skin from spills. Using a chemical fume hood isn’t fancy — it's all about not breathing in vapors that can burn the inside of your nose and throat. The data back this up: inhaling isobutyric anhydride vapors can cause coughing, burning sensations, and at higher exposures, more severe damage.
Ventilation Is Your Friend
Cracking a window doesn’t cut it. Vapors build up quickly, especially when transferring the chemical from bulk containers or weighing out larger batches. Room ventilation systems marked as suitable for corrosives or organics pull the air away from your face so you’re not the canary in the coal mine. A functioning fume hood goes a long way, and investing in proper air filtration helps keep everyone safe.
Purchase, Storage, and Spill Management
I’ve seen rushed days lead people to store reactive chemicals next to ordinary cleaning supplies or in glass containers with questionable integrity. Isobutyric anhydride reacts with water, releasing heat and acids. That rules out open containers, leaky stoppers, and metal shelves near sinks. Dry, tightly sealed bottles kept in designated, well-ventilated cabinets cut down on accidents. These cabinets should be labeled so anyone walking by knows what’s inside.
I’ve had to deal with a minor anhydride spill before, and it’s not something you want to improvise. Absorbent pads and neutralizing agents like sodium bicarbonate help limit the impact. Nobody reaches for standard mop buckets — those just create more vapors and mix water in, which makes it worse.
Training Makes a Difference
Some say you can’t train common sense, but you can teach respect for hazardous materials. I watched a seasoned chemist demonstrate just how fast vapors travel on a cool day, using a plume of smoke near the hood’s edge. That sticks with you. It pays off to walk through the steps before using a new chemical, with coworkers doing practice runs with water. A team that’s clear on escape routes and spill procedures reacts faster and prevents mistakes from getting out of hand.
Know What To Do In Emergencies
It only takes one exposure to give someone a lifelong memory. Eye wash stations and safety showers look like background props until you reach for one in a hurry. Everyone in the room should know where these are, and routes should stay clear — clutter can cost seconds that matter. Emergency procedures taped in plain sight beat a thick binder locked in an office drawer. It’s practical. If you see symptoms after exposure — coughing, burning, confusion — get help quickly.
The Stakes Are Personal
Working around chemicals like isobutyric anhydride has sharpened my focus on routine details. Nobody remembers the days where everything goes right, but the small steps — gloves, airflow, eyes on the labels — add up. People often downplay the risks of chemicals they've seen before, but routine builds safety. Protecting yourself and your colleagues means respecting every substance, no matter how familiar it seems.
What is the chemical formula of Isobutyric Anhydride?
Why Chemical Formulas Matter
Getting the chemical formula right in the lab matters—a lot. Over the years, I’ve lost track of how many stories I’ve heard from colleagues mixing up a substance because the formula seemed confusing or only described in technical jargon. Knowing exactly what you’re working with protects you and anyone else handling these chemicals. In a world where safety and transparency guide good science, getting nitty-gritty details right becomes a habit you hold onto.
Diving Into Isobutyric Anhydride
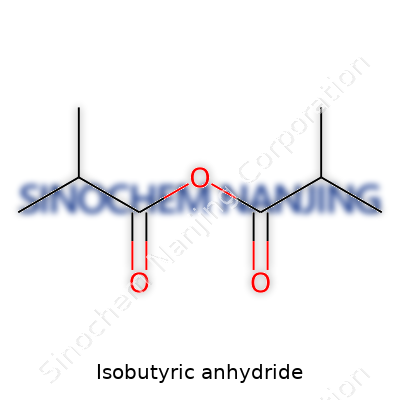
Isobutyric anhydride comes with the chemical formula (CH3)2CHCO)₂O, which you can also write as C8H14O3. This formula tells you it’s an anhydride formed from two molecules of isobutyric acid, minus one water molecule. A quick glance at the formula, and you’ll notice those eight carbons and three oxygens right away—a clear signal that you’re dealing with something well past a simple acid.
I’ve handled anhydrides like this in synthesis work, especially in pharmaceuticals, where slight changes in molecular structure shape the outcome. The unique reactivity of isobutyric anhydride comes from the anhydride linkage, which makes it a prime choice in creating esters or as an acylating agent.
The Importance of Handling and Understanding the Chemical
If you’ve ever opened a bottle of isobutyric anhydride, you know it’s nothing to take lightly. Its pungent odor and toxic vapor remind you why chemical literacy matters. A mistake not only slows your work but can land you in real danger. Over the last decade, emergency room visits tied to mishandling organic anhydrides haven’t dropped much, according to public health data. Almost every one of those accidents tied back to either improper training or a misunderstanding of what these chemicals actually do inside the body. Isobutyric anhydride isn’t just a tool in chemical manufacturing—it’s a reminder to respect every step and every number you read on a label.
This is where principles like Google’s E-E-A-T come into play: chemicals demand experience, expertise, trustworthiness, and integrity. Sharing the formula without knowledge shortchanges anyone reading. Real-world problems need clear answers.
Application and Safer Practices
In my own work, keeping up with safety data sheets and using personal protective equipment wasn’t an afterthought. Using isobutyric anhydride means ensuring ventilation, double-checking formulas, and preparing neutralizing agents just in case. Many labs have moved toward closed-system transfers for anhydrides precisely because one splash or wayward vapor can ruin a day, or a lab, or a life.
Modern chemistry labs increasingly use digital tracking for reagents. One missed decimal or swapped chemical name, and both the chemist and the experiment land in jeopardy. Knowing the correct chemical formula ensures compliance, reproducibility, and traceability.
Supporting Better Chemistry Education
The best lesson I learned early on came from a mentor: explain every step, double-check each label, and show curiosity about each formula. Sharing the structure and formula of isobutyric anhydride, I pass along the same confidence I got in those first years. Anyone picking up a bottle with C8H14O3 on the label deserves clear and honest information. That makes all the difference between a risky transaction and responsible chemistry.
How should Isobutyric Anhydride be stored?
Understanding the Risks
Anyone who’s handled chemicals like Isobutyric Anhydride knows just how quickly things can go wrong without the right precautions. Unlike household cleaners, Isobutyric Anhydride gives off strong, eye-watering fumes and reacts violently to water. Its low flash point and high reactivity practically invite trouble if stored carelessly. One bad whiff lingers in your lungs, and a wet spot can spark a chemical reaction faster than most folks realize.
Practical Storage Steps
First lesson I picked up in labs: never trust luck with chemicals like this. Tight, chemical-resistant metal containers with secure seals give peace of mind. Polyethylene and certain plastics, common standbys, start to break down here. So, stainless steel wins out. Any dribble on the outer surface signals sloppy handling—wiping down after use saves headaches later.
Keep Isobutyric Anhydride somewhere cool and dark, well away from sunlight or any heat sources. Heat speeds up dangerous decomposition, and I’ve seen entire storerooms have to evacuate after a bottle sat too close to a sunny window. Ambient temperatures below 25°C remain ideal. Dryness matters most, with humidity tempting disaster. Moisture in the air, or heaven forbid from a leaky pipe, starts breaking the chemical down and leads to corrosive fumes before long.
Fire safety cuts right to the center of the storage issue. I always keep Isobutyric Anhydride away from oxidizers, acids, and strong bases. A fire-resistant cabinet designed for flammable liquids acts as an insurance policy. Some folks let their bottles mingle with less volatile goods, but experience says segregating by compatibility lowers the odds of runaway reactions. Even static discharge or a dropped tool can trigger an accident if incompatible materials mingle by mistake.
Ventilation and Detection
A good chemical storage room breathes. If the air inside feels stuffy, trouble hides in stale corners. All facilities I’ve worked in run continuous ventilation, so one unnoticed spill never grows into a cloud. Chemical fume hoods, even small benchtop ones, cut down on vapor exposure. Don’t trust your nose—installing gas detectors provides the early warning I’d trust my safety on.
Training: The Human Element
Safe storage isn’t just engineering—it’s habit. I’ve seen newcomers grab the wrong bottle or forget to check caps by the end of a long shift. Regular refresher courses, honest safety discussions, and posted reminder sheets reinforce good habits. Posting proper hazard data sheets nearby also keeps everybody sharp.
Selecting the right personal protective equipment means more than just ticking boxes. Full goggles, gloves rated for acyl chlorides, and chemical-resistant aprons have saved skin and prevented emergency room trips more than once. No storage protocol covers lazy habits, so group accountability counts.
Preparing for Trouble
Accidents strike when least expected, so spill kits belong near storage sites. Neutralizing agents—sodium bicarbonate works well for small-scale cleanup—and extra absorbent pads make a difference. Familiarity with emergency wash stations and exits can mean the difference between minor accidents and serious harm.
For businesses or schools, keeping only as much stock as immediately needed always makes sense. Lower inventory cuts risks, paperwork, and headaches for everyone involved. Over-ordering might feel like saving effort, but nothing burns trust faster than unnecessary hazards.
Final Thoughts
Years in chemical workplaces have taught one lesson above all: respect the danger. Isobutyric Anhydride punishes complacency. With the right containers, climate, and people, you put safety on your side and focus on the good work chemicals can do, not their potential for harm.
What are the hazards associated with Isobutyric Anhydride?
Why Isobutyric Anhydride Matters in the Workplace
It’s easy to overlook chemicals like Isobutyric Anhydride if you’re not spending time in a lab or factory. For those who work around it, though, the hazards deserve serious respect. I’ve seen workers get too comfortable after years on the job, and that’s when trouble usually starts. Isobutyric Anhydride serves plenty of uses—from chemical synthesis to pharmaceuticals. But that usefulness brings some very real risk.
The Real Hazards: More Than Skin Deep
Many folks notice the sharp, acrid odor as soon as a container opens. That smell is your body’s first warning. Direct contact with this chemical can burn your skin or eyes, causing blisters or severe irritation. Even brief exposure sometimes triggers intense coughing, wheezing, or shortness of breath. It forms a cloud when spilled, and if inhaled, it quietly goes to work damaging the lining of your airways. Long-term exposure ramps up the risk of asthma or chronic respiratory disease.
I’ve heard about a case where a minor splash landed on someone’s hand, and they figured rinsing for a few seconds would do the trick. That error led to raw, painful patches that didn’t heal for days. Eyes are even more vulnerable—permanent vision loss isn’t out of the question in a bad spill.
Chemical Reactivity: The Fire Danger
Mixing Isobutyric Anhydride with water or humid air kicks off a violent reaction, releasing flammable, choking gases. If it leaks out, just the moisture in a room can start a reaction. Most workplaces keep it away from open flames, but not everyone thinks about static sparks or hot surfaces. Fires start fast with this chemical, generating thick smoke that contains highly toxic byproducts.
The EPA and OSHA have both listed Isobutyric Anhydride as a hazardous substance, requiring strict labeling and storage. No one wants to think about evacuation plans, but anyone near this stuff needs to know exactly what to do if the air suddenly fills with fumes.
Keeping People Safe: Real Solutions
I’ve learned that constant training saves more injuries than even the best safety gear. Workers need gloves, goggles, and protective clothing designed for organic acids—not just flimsy latex or cotton. Fume hoods and exhaust systems must run at full capacity every time the chemical comes out. Emergency showers and eyewash stations should stay clean, easy to reach, and tested every week. These aren’t just boxes to check—they save lives.
Spill response kits packed with neutralizing agents need regular inventory checks. People sometimes neglect maintenance, not wanting to waste supplies. But I’ve witnessed what happens when companies treat emergency equipment like a formality. It takes only one accident to prove how wrong that mindset is.
The Role of Good Reporting and Accountability
Open communication makes a difference. When someone sees a leak, cracked storage drum, or failing vent, speaking up must feel safe—no fear of blame. Supervisors should walk the floor, inspect storage, and bring up safety at every meeting. Regulators also play a crucial part, carrying out unannounced inspections and demanding solid records. No shortcuts here—solid habits grow out of real accountability, not paperwork for compliance alone.
Isobutyric Anhydride poses more than a technical challenge. It puts health and safety culture to the test. Respect for the risks keeps people out of harm’s way—and sometimes, it’s the only thing standing between business as usual and a full-blown emergency.
| Names | |
| Preferred IUPAC name | 2-Methylpropanoic anhydride |
| Other names |
2,2-Dimethylpropanoic anhydride Isobutyric acid anhydride Isobutanoic anhydride |
| Pronunciation | /ˌaɪsəˈbjuːtɪrɪk ænˈhaɪdraɪd/ |
| Identifiers | |
| CAS Number | ['97-28-3'] |
| Beilstein Reference | Beilstein Reference 741118 |
| ChEBI | CHEBI:39176 |
| ChEMBL | CHEMBL1681680 |
| ChemSpider | 64670 |
| DrugBank | DB14004 |
| ECHA InfoCard | 100.001.152 |
| EC Number | 211-663-0 |
| Gmelin Reference | 133937 |
| KEGG | C18673 |
| MeSH | D011842 |
| PubChem CID | 8744 |
| RTECS number | NL1575000 |
| UNII | J9P35835V5 |
| UN number | UN2480 |
| CompTox Dashboard (EPA) | DTXSID8021538 |
| Properties | |
| Chemical formula | C8H14O3 |
| Molar mass | 158.18 g/mol |
| Appearance | Colorless liquid |
| Odor | Pungent |
| Density | 0.96 g/cm³ |
| Solubility in water | Reacts with water |
| log P | 0.83 |
| Vapor pressure | 0.7 mmHg (20°C) |
| Acidity (pKa) | pKa = 4.86 |
| Magnetic susceptibility (χ) | -6.2×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.388 |
| Viscosity | 1.015 mPa·s (25 °C) |
| Dipole moment | 2.60 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 342.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -632.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1795.6 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02,GHS05 |
| Signal word | Danger |
| Hazard statements | H302, H314, H317, H332 |
| Precautionary statements | P261, P264, P271, P280, P301+P330+P331, P303+P361+P353, P305+P351+P338, P304+P340, P312, P337+P313, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-3-2-W |
| Flash point | 75 °C (167 °F; 348 K) |
| Autoignition temperature | 434 °C |
| Explosive limits | Explosive limits: 1.6–7.0% |
| Lethal dose or concentration | Lethal dose or concentration of Isobutyric Anhydride: "LD50 oral rat 2,700 mg/kg |
| LD50 (median dose) | LD50 (median dose): 4.5 g/kg (rat, oral) |
| NIOSH | RN7650000 |
| PEL (Permissible) | PEL: 50 ppm |
| REL (Recommended) | 1 mg/m³ |
| IDLH (Immediate danger) | IDLH: 100 ppm |
| Related compounds | |
| Related compounds |
Acetic anhydride Propionic anhydride Butyric anhydride Isobutyric acid Isovaleric anhydride |

