Isobutyraldehyde: A Down-to-Earth Commentary on a Chemical Staple
Historical Development
Folks working in chemical plants trace their manufacturing roots back decades, and isobutyraldehyde serves as a good example of persistent industrial interest. Since the early twentieth century, its story began in the shadows of World War I’s rapid industrial expansion. Technological shifts like the oxo process became regular sights for chemical engineers, who saw the opportunity in hydroformylation—adding carbon monoxide and hydrogen to propylene to create this compound. These methods supplied growing demand for plastics, paints, and flavors, giving isobutyraldehyde a permanent spot on chemical production lines. Older batch processes, while simple, gave way to continuous methods, improving yields and safety, and allowing for industrial-scale use. Each decade, refinements in catalysis shed light on greener, cleaner ways to synthesize and recover isobutyraldehyde, making it more available and less costly for factories around the world.
Product Overview
Isobutyraldehyde goes by several names—2-methylpropanal for those who read labels closely, or “IBA” for folks grabbing drums off a warehouse shelf. Its punchy aroma sometimes reminds people of the solvent aisle, yet chemists and product formulators look past the sharp scent. What draws attention is its role as a building block. Modern industry depends on these basic chemicals, whether you’re running a coatings facility, mixing up herbicides, or supplying big pharmaceutical companies. This colorless liquid isn’t flashy, but its influence stretches far into supply chains, connecting small labs to global brands.
Physical & Chemical Properties
Anyone who has spent time in a lab quickly learns that isobutyraldehyde looks like water—crystal clear, but with a volatile essence and an unmistakable irritation when inhaled. Boiling at 63 degrees Celsius, it evaporates fast if you leave the cap off, and its vapor can hang heavy without good ventilation. It carries a molecular weight of roughly 72 grams per mole, density near 0.8 g/cm³, and dissolves only sparingly in water. The molecule has a well-placed carbonyl group that draws reactions much like bees to honey, making it a go-to reagent in organic chemistry settings.
Technical Specifications & Labeling
Drums labeled for transport show more than just the name—there’s often a warning diamond, purity percentage (commonly above 98%), and details like UN1990, signaling hazardous status during shipping. Spec sheets from reliable suppliers spell out acid content, moisture limits, and recommended storage temperatures, all to guarantee safety and keep nasty side products at bay. Labels may also note ignition temperatures and flash points, important for those working in plants with busy solvent systems. Reputable suppliers back up shipments with certificates of analysis, confirming batch purity and helping buyers meet internal quality checks.
Preparation Method
Experienced chemical operators rely on hydroformylation for making isobutyraldehyde. Propylene meets a catalyst—often cobalt or rhodium-based—in reactors feeding carbon monoxide and hydrogen under pressure. Some manufacturers work toward higher atom efficiency and less waste by tuning deposits, reaction times, and pressures. After separation and purification, waste minimization and recycling sit on any plant supervisor’s checklist. Companies investing in this area get an edge, since improving catalyst life and increasing selectivity drives production costs down and shrinks a facility’s environmental footprint.
Chemical Reactions & Modifications
Isobutyraldehyde supports busy downstream operations because it reacts with plenty of compounds. Aldol condensations, for instance, stack carbon skeletons for new intermediates, while hydrogenation turns it into isobutanol, a solvent and fuel additive. The carbonyl group makes functional group interconversions straightforward, opening doors to producing pharmaceutical actives, agricultural chemicals, and plasticizers. Labs often explore routes for higher-value derivatives, leveraging the accessibility of the aldehyde group.
Synonyms & Product Names
Chemists and plant workers see isobutyraldehyde under several names—2-methylpropanal, isobutanal, or IBA. Some safety sheets prefer the IUPAC version. Walk down a plant’s chemical row and you’ll spot these names, alerting seasoned operators that they’re dealing with a volatile, reactive aldehyde requiring care and planning.
Safety & Operational Standards
Pure isobutyraldehyde demands respect and firm protocols. The liquid ignites at low temperatures and emits vapor that irritates the lungs and eyes. Routine workplace standards involve fume hoods, chemical goggles, gloves, and reliable air monitoring. Threshold limit values from groups like OSHA and ACGIH set exposure at ten parts per million over an eight-hour shift. Spill response plans, fire extinguishers, and well-ventilated handling rooms address both health and fire risks. Employees stay current with annual safety training to prevent injuries and chemical releases. The drum and equipment cleaning cycles, leak checks, and careful drum stacking keep mishaps and product losses to a minimum.
Application Area
Factories convert isobutyraldehyde into isobutanol, plasticizers, and coatings found everywhere—from car interiors to clear finish on wood floors. Pesticide manufacturers depend on it for herbicide intermediates, while fragrance and flavor firms use tiny doses for blending aromas and tastes in foods. Over-the-counter medicines and some vitamins stem from chemical transformations starting with this intermediate. As the push for bio-based chemicals grows, more companies investigate using renewable propylene and green hydrogen to feed their plants, moving the industry closer to sustainability goals.
Research & Development
Research teams at chemical companies and universities constantly look for better catalysts, greener solvents, and higher atom economy. Some labs pursue enzymatic or microbial production routes, hoping biology can rival chemistry’s throughput without high-pressure reactors or heavy metals. Low-energy processes and continuous flow reactors get closer scrutiny now due to energy costs and environmental regulations. Peer-reviewed studies drive innovation by showing how tweaks at the molecular or reactor level yield major gains in selectivity and safety. Cross-sector partnerships help apply lessons from the lab to factory-size production, turning pilot successes into industry standards.
Toxicity Research
Toxicologists have studied isobutyraldehyde enough to map both acute and chronic effects. Short-term exposure causes similar issues to other low molecular weight aldehydes—eye and respiratory tract irritation and possible headaches or nausea with higher concentrations. Rodent studies inform safe-level checks, supporting guidelines by regulatory agencies. Chronic exposure data remain patchy, spurring calls for broader studies on long-term workplace risks and neighborhood impacts near chemical plants. Wastewater and off-gas monitoring keep watch for risks to aquatic and terrestrial wildlife, reinforcing the need for high-efficiency scrubbers and careful wastewater management.
Future Prospects
Job training programs and public-private collaborations shape the workforce needed to experiment and build the greener plants of tomorrow. The global pivot to circular economies invites engineers to design biobased and closed-loop processes for isobutyraldehyde. Next-generation catalysts and smart plant controls may drive down energy consumption and open new chemistries that lower waste. Vigilance on health risks, transparent emission reporting, and partnerships with communities nearby keep public trust and safeguard both worker welfare and the environment. As legislation tightens and consumer demand for responsible sourcing grows, producers who invest in safer, cleaner, and more efficient systems likely find themselves ahead of the curve, shaping the legacy of isobutyraldehyde for decades to come.
What is Isobutyraldehyde used for?
Everyday Industry and Everyday Life
Isobutyraldehyde sounds like something you’d only see in the back room of a research lab. In reality, it plays a bigger role than most realize, touching everything from the fuel we pump at gas stations to the vitamins tucked in the back of our cabinets. Most folks probably never think about this chemical. I didn’t either, until I started reading product labels a little more closely and talking to people who’ve worked at chemical plants.
Behind the Scenes in Manufacturing
Factories count on isobutyraldehyde for one big thing: making raw materials that go on to become products people interact with every day. One of its most prominent uses is in the production of isobutanol. Isobutanol ends up as part of many solvents and coatings. Walk through any hardware store and the shelves lined with paints and varnishes owe some of their performance and drying characteristics to chemicals built around isobutyraldehyde.
This chemical also finds its way into the process for making neopentyl glycol. That’s a mouthful, but basically, it’s a key ingredient in making plastics and resins tougher. The same clear, hard finish you find on durable tabletops or floors often gets its backbone from those resins. Industry folks tell me that producing this stuff efficiently—without cutting corners—really matters for keeping the supply chain moving during high-demand seasons.
Helping out in the Fields and Pharmacies
Farmers might not realize it, but isobutyraldehyde plays a part in their world, too. Chemical companies use it when crafting certain pesticides and herbicides. It acts as an intermediate, meaning it’s a step in the assembly line, helping build molecules crafted to protect crops from destructive pests. Even if the final product doesn’t include isobutyraldehyde, getting to that end results involves it somewhere along the journey.
If you’ve ever taken a vitamin B3 (niacin) supplement, you could be holding a piece of this chemical’s story right in your hand. Isobutyraldehyde lands in the process of synthesizing niacin, which helps thousands of people with nutritional needs every year. The pharmaceutical field relies on these building block chemicals, and quality control matters—nobody wants to take a vitamin full of impurities. That means constant oversight and layers of certification at every step.
Looking at the Big Picture: Safety and Supply
Facilities working with isobutyraldehyde have a tough job to do. The chemical itself carries health hazards, so safety protocols run deep. Plant operators wear respirators, gloves, and work under strict ventilation guidelines so workers go home healthy after every shift. That kind of commitment has real consequences—messing up even once can ripple out, affecting local communities and company reputations for years.
Sometimes, disruptions in the supply chain for chemicals like isobutyraldehyde lead to shortages in downstream products. Some paint producers or plastic factories have cut back production or raised prices. Building in backup plans, like diversifying suppliers or investing in new production methods, often helps avoid the worst of these ripple effects. Next-generation manufacturing may rely on greener ways to make chemicals with less environmental baggage, which could mean less reliance on older processes someday.
Where We Go from Here
Isobutyraldehyde might not be a household name. Its real story lives in the products we see, use, and depend on every day. Vigilance about safe handling, investment in innovation, and transparency in manufacturing will keep its legacy more beneficial than burdensome—not just for industry, but for everyone driving, farming, or even just opening a bottle of vitamins.
What are the safety precautions for handling Isobutyraldehyde?
Understanding the Risks
Breathing in isobutyraldehyde feels sharp, harsh, and wrong. That scent comes with a punch for your eyes and nose, and can make your lungs burn. I’ve watched colleagues underestimate these fumes and land with splitting headaches, and it isn’t just a nuisance—prolonged exposure damages the liver and irritates respiratory passages. Even handling a small bottle gets risky when you don’t respect the flammability; one spark, and the gas catches with a surprising speed. The Occupational Safety and Health Administration (OSHA) gives clear limits for exposure—50 ppm—and that number serves as a warning not a recommendation.
Gear Matters—And So Does Training
Wearing the right gloves (nitrile beats latex every time with isobutyraldehyde) saves your skin. Putting on chemical splash goggles has to be a habit, not an afterthought, since fumes drift fast. Any time I work with volatile chemicals, a flame-resistant lab coat and closed-toe shoes get priority. Over time, I learned that even a quick five-minute task deserves full protective gear. Many injuries I’ve seen happened in those moments workers thought they’d “just be quick.”
Ventilation Saves Lives
Proper ventilation means more than just a cracked window. Fume hoods or dedicated extraction fans need to run every time the bottle opens. A fan that pulls even ten air changes an hour can mean all the difference. I spent years in labs where budget cuts threatened good ventilation; every time it failed, complaints about headaches and nausea went up. If you feel it, it’s too late—the air’s already saturated. Monitoring the environment with sensors supports early detection.
Storage and Handling
Storing isobutyraldehyde in tightly sealed containers, marked with clear hazard labels, protects more people than just the handler. The liquid wants to escape, so secondary containment trays catch drips. Keep it far from oxidizers or acids; mixing accidents cause fires and toxic gas. Locking up flammable chemicals in ventilated cabinets, away from direct sunlight and heat sources, has always made me feel more secure in the work space. Spills do happen—you’ll want to have sand or other absorbents close by. Above all, don’t trust your memory; keep written procedures posted right on the cabinet.
Disposal Reminders
After finishing up, leftover isobutyraldehyde can’t just go down the drain. It heads to approved hazardous waste containers, which specialized waste disposal crews handle. Mishandling disposal led to environmental fines and dangerous reactions at several plants I’ve visited. I joined in regular disposal training sessions and learned to double-check everything, cutting down on mistakes that cost the company and the environment.
Caring for Each Other
Working with hazardous materials always carries a risk, but a strong safety culture protects everyone. No shortcut stands up to repeated reminders, team training, spot checks, or peer support. Injuries drop when workers watch out for each other, remind each other, and refuse to let small mistakes slide. Building that trust turned even the riskiest lab jobs into places where we all made it home safe. The story of isobutyraldehyde isn’t just about a fiery danger in a bottle, but about the choice to do daily work with care for yourself and everyone sharing the lab or plant floor.
What is the chemical formula and structure of Isobutyraldehyde?
Formula and Structure
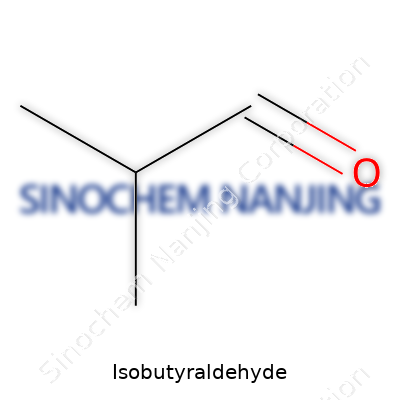
Isobutyraldehyde holds the chemical formula C4H8O. Sketched out on paper, its structure tends to stand out from its straight-chained cousin, butyraldehyde. At the core, isobutyraldehyde has a branching pattern: three carbons linked in a row, with a fourth carbon branching from the middle. Attach an aldehyde group (-CHO) at the end, and the molecule shows its personality. This branching carries real weight in how it behaves and what folks use it for. Chemists represent it as (CH3)2CHCHO. That might look cryptic, but anyone who’s ever assembled a model kit of molecules knows the value of branching versus a simple line. The structure leads to unique properties in boiling point, reactivity, and, most importantly, the roles it plays in industrial and laboratory settings.
Why the Structure Matters
Looking at organic chemistry from a practical lens, not all four-carbon aldehydes are made equal. That extra branch on isobutyraldehyde shifts the boiling point down and makes it a little more volatile compared to n-butyraldehyde. When working with volatile solvents in a lab, that extra volatility means extra care: the compound evaporates more quickly, so glassware needs tighter lids, and fume hoods matter more during experiments or production runs. I remember a time during a group synthesis project. A colleague underestimated how quickly isobutyraldehyde would blaze off at room temperature. Within minutes, the smell – that sharp, paint-like scent – lingered for the rest of the afternoon. The lesson stuck with me: a structure like this isn’t just trivia, it affects every step from transport to storage.
Industrial Importance and Safety
Manufacturers use isobutyraldehyde as a building block. It’s a starting point for vitamins (like Vitamin B5, pantothenic acid), flavors, and fragrances. By shifting just one branch in the carbon chain, you get a feedstock that helps produce plasticizers, resins, and agricultural chemicals. Crops fed by these chemical precursors grow bigger or resist pests better. In the fragrance world, small molecules like isobutyraldehyde add punch to high-end perfumes, letting designers hit just the right sharp note in the blend. Upstream in the chemical world, factories churn out thousands of tons each year, showing that this is more than a curiosity for chem students.
Health and Environmental Concerns
Isobutyraldehyde comes with its own risks. Exposure can irritate the eyes and skin, and the low boiling point makes vapor inhalation a concern. Factories fix this with proper ventilation and gloves. But smaller businesses and classrooms may not always have top-notch protection. I learned early, after a splash in the lab, to double up on goggles and never assume a “common” chemical is harmless. On the environmental side, isobutyraldehyde can break down under sunlight, forming smog-producing molecules. This isn’t just theory—cities with chemical plants must monitor air quality closely to avoid spikes in pollution.
Pushing Toward Solution
Better labeling, training, and access to safety data sheets help cut incidents in small labs and factories. Green chemistry practices look for less volatile alternatives where possible, or tweak ventilation systems to keep air clean. Manufacturers keep investing in closed systems for production and transfer so that less vapor slips out. As schools and workplaces get more informed, fewer people stumble into accidents that come from underestimating things that look “simple” on paper.
How should Isobutyraldehyde be stored?
Understanding What We’re Dealing With
Isobutyraldehyde doesn’t make headlines like more common chemicals, but anyone who’s worked in labs or plants knows it packs a punch. It gives off a strong, unpleasant odor — believe me, you’ll recognize it if a cap gets left loose. More importantly, breathing in its vapors doesn’t do lungs any favors, and its low flash point means any carelessness could set off a serious fire. If a drum leaks, not only will people complain about the stench, but you’re also facing some heavy regulatory trouble.
Keeping Things Cool (Literally)
Heat and isobutyraldehyde don’t mix. Even mild temperatures nudge it toward evaporation, so inside storage beats any outdoor shed. From experience, sealed steel drums in a well-ventilated storeroom keep vapors at bay. Don’t let the thermometer tick above room temperature; a cool, shaded spot slows down evaporation and limits risk. With its flash point sitting around –6°C (21°F), even a simple lightbulb too close can spell trouble.
Don’t Slack on Ventilation
Skipping on proper air flow leads to a bad time. It might seem easier to keep windows closed to trap fumes inside, but every time someone opens a drum, invisible clouds build up. Walk into a closed-off room and you’ll taste it in the air. Fans fitted above storage racks make a big difference, and a ducted exhaust draws out lingering fumes. Not just for comfort — a vapor-rich space can lead to explosions if someone walks in with static on their clothes.
Keep the Water Out
Isobutyraldehyde reacts with moisture. Rusty ceilings and leaky pipes cost more than patch jobs when chemicals start acting up. Any sign of water in storage drums brings headaches, and I’ve seen a shipment turned into hazardous waste from a simple roof leak. Make regular roof and pipe inspections a habit, not an afterthought.
Spill Containment Isn’t Optional
Even the best drum caps wear out or get left loose during a long shift. Pouring anything into open trays or buckets only invites accidents. Store every container on a spill pallet. If a drum tips or leaks, that sump buys time to fix the problem before it spreads to aisles or drains. Chemical-resistant gloves and goggles belong next to storage areas, not tucked in a locked office, so anyone can reach them in a pinch.
Training Beats Labels Any Day
Labeling a drum “flammable” matters, but training trumps a hundred warning stickers. Make sure everyone — from seasoned crew to newcomers — knows how to spot pressure build-up, leaks, or sour smells. I’ve witnessed emergencies avoided thanks to eagle-eyed staff spotting vapor haze or a bulging drum lid. People, not policies, keep a warehouse safe.
Regulations Keep Us Honest
OSHA and EPA rules don’t exist just to fill binders. Regular audits and up-to-date Material Safety Data Sheets protect both workers and the public. Skipping required paperwork or shortcuts in inspections can rack up serious fines and endanger everyone around.
Bottom Line: Safety Starts with Simple Steps
Every time isobutyraldehyde sits on a shelf, there’s an obligation to keep things safe. Store it smart, control the risks, and treat every batch like it could go wrong—the peace of mind is worth it.
What are the potential health effects of exposure to Isobutyraldehyde?
Walking Into the World of Chemicals
Many chemicals drift under the radar in daily conversations. Isobutyraldehyde lands in that group, even though folks working in chemical plants, labs, or some manufacturing lines may meet it more than they’d like. It’s a colorless liquid with a sharp smell—something you won’t mistake if it hits your nose. While the average person rarely runs into it, shipping, spills, or poor storage can bring it closer to home or workplace than you might expect.
How Isobutyraldehyde Delivers Its Punch
Breathing in vapors usually delivers the quickest hit. Even brief whiffs irritate eyes, nose, and throat. Imagine a scratchy, stinging feeling taking over after a single breath. Apart from fumes, skin absorbs this chemical if it splashes, bringing rashes and itching—sometimes even deep skin burns if left sitting too long. Eyes can redden or tear up within seconds of contact.
The Environmental Protection Agency underscores how repeated exposure, even at lower doses, chips away at health. Coughing, chest tightness, and headaches often follow. Over time, long-term inhalation runs the risk of damaging the lungs and liver. Workers in factories using isobutyraldehyde face higher odds—especially if ventilation or personal protective gear falls short.
Digging Into Deeper Health Risks
Chronic health effects paint a more worrying picture. Once isobutyraldehyde makes its way into the bloodstream, it may affect organs beyond the lungs. Studies, mostly from animal models, raise concerns about liver and kidney strain. There’s also the never-ending question hanging over chemicals like this: could it raise cancer risk? No clear evidence confirms it for humans yet, but agencies like the World Health Organization still ask for caution and keep watching new data.
My Own Dive Into Occupational Hazards
Before writing became my main gig, I spent summers in a small paint factory, mixing formulas and loading barrels. Isobutyraldehyde wasn’t a household word, but warning signs about “volatile organic compounds” were taped everywhere. A few days into the job left my eyes watering from fumes I could barely see. My supervisor handed out masks and reminded us not to skip them, even if we thought our shift would be “just a couple hours.” Cutting corners meant headaches nights later, and one coworker ended up with a nasty rash from a single afternoon handling spills with bare arms.
What Companies and Workers Can Do Right Now
No protective policy in the world helps if no one follows it. OSHA sets strict limits for workplace air concentrations. Good ventilation, real-time air quality monitors, and chemical-resistant gloves and masks help workers push back against exposure. Spills need immediate cleanup—and no one should ignore broken equipment or blocked safety showers.
Education helps, too. When people understand more about the chemicals around them, they take warnings seriously. Mandatory training and regular health checkups can spot problems early before they spiral out of control. Unions and worker groups often act as watchdogs, pressing for safer standards when management drags its feet.
Turning Concern Into Action
Many basic protections cost less than the fallout from serious health incidents or lawsuits. Regulators and health professionals can push for regular updates in standards, better recordkeeping on long-term health, and stronger whistleblower protections. Community groups near chemical plants deserve real-time updates and transparency. Folks living or working near storage sites ought to know what’s in the air and how to react in case alarms sound.
The story of isobutyraldehyde echoes the lesson that our health ties directly to the chemicals we handle—each one carrying its own risks and rules. Real change only happens when companies, workers, and communities all pull together so health doesn’t take a back seat to convenience.
| Names | |
| Preferred IUPAC name | 2-Methylpropanal |
| Other names |
2-Methylpropanal Isobutanal Isobutylaldehyde 2-Methylpropionaldehyde |
| Pronunciation | /ˌaɪ.soʊˌbjuː.tɪrˈæl.dɪ.haɪd/ |
| Identifiers | |
| CAS Number | 78-84-2 |
| Beilstein Reference | 603932 |
| ChEBI | CHEBI:17274 |
| ChEMBL | CHEMBL16438 |
| ChemSpider | 6085 |
| DrugBank | DB13872 |
| ECHA InfoCard | 03a9d457-89b6-4ad0-a0ef-5793a36d8d57 |
| EC Number | 200-837-3 |
| Gmelin Reference | 878 |
| KEGG | C05925 |
| MeSH | D007538 |
| PubChem CID | 6575 |
| RTECS number | NI5600000 |
| UNII | 46Q844R1K3 |
| UN number | UN1212 |
| Properties | |
| Chemical formula | C4H8O |
| Molar mass | 72.106 g/mol |
| Appearance | Colorless liquid with a pungent odor |
| Odor | Pungent |
| Density | 0.802 g/cm3 |
| Solubility in water | Moderately soluble |
| log P | 0.83 |
| Vapor pressure | 18.4 kPa (20 °C) |
| Acidity (pKa) | 17.2 |
| Magnetic susceptibility (χ) | -44.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.3761 |
| Viscosity | Viscosity: 0.418 mPa·s (20 °C) |
| Dipole moment | 2.72 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 229.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -233.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2325 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P312, P331, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 5 °C (closed cup) |
| Autoignition temperature | 415 °C |
| Explosive limits | 1.7% - 10.6% |
| Lethal dose or concentration | LD50 oral rat 2300 mg/kg |
| LD50 (median dose) | LD50 (median dose): 790 mg/kg (rat, oral) |
| NIOSH | PUA38000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Isobutyraldehyde is 50 ppm (150 mg/m3) as an 8-hour TWA (OSHA) |
| REL (Recommended) | 200 ppm |
| IDLH (Immediate danger) | 200 ppm |
| Related compounds | |
| Related compounds |
Propanal Butyraldehyde n-Butyraldehyde Isobutanol Isobutyric acid |

