Octafluoroisobutylene: An Editorial Commentary
Historical Development
Synthetic chemistry has chased after compounds that promise industrial advantage and reliable performance. Octafluoroisobutylene (OFIB) steps out of that drive for progress. Chemists got serious about OFIB nearly a century ago, back when the world began to see the power of fluorinated molecules. Advances in polymer science and refrigerant development pushed research in this direction. Work in labs chasing stronger, safer, and more durable chemical building blocks brought about the development of OFIB. The post-war chemical boom in the United States and Western Europe turned attention to organofluorine chemistry, including molecules built for specific reactions and sturdy end-products. This historical arc matters because it shaped the values behind OFIB’s applications: stability, reliability, and the promise of new properties that aren’t found in less exotic chemistry.
Product Overview
OFIB comes across as a specialist’s tool. Unlike the more famous cousins like Teflon or Freon, OFIB rarely shows up in everyday consumer conversations. Its chemical backbone, loaded with eight fluorine atoms, marks it as a compound for serious applications. In some corners of industry, OFIB has a reputation as both a builder and a challenge. Its strong carbon–fluorine bonds put it on a short list of compounds that can outlast, outperform, or outmaneuver organic materials in harsh environments. People often recognize it by its sharp chemical scent and its role in making specialty fluoropolymers or advanced coatings, where less robust molecules just can’t deliver.
Physical & Chemical Properties
OFIB stands as a colorless, reactive gas with a pungent odor—those acquainted with it know to keep their distance. That strong, pungent smell isn’t just a warning; it’s proof of its reactivity. High density and volatility combine with an ability to push back against corrosive conditions. Its thermal stability catches the attention of engineers looking for reliability in sealed electronic systems or chemical reactors. Many chemical professionals lean on OFIB’s nonflammable nature, especially in high-risk settings where safety sits right alongside productivity. That combination of durability and reactivity is rare. Everything about the molecule—right down to its low solubility in water and high molecular weight—reminds one that this compound runs on a different set of rules compared to the more familiar hydrocarbons.
Technical Specifications & Labeling
OFIB never gets administered lightly. Handling, transport, and labeling standards for OFIB sit at the tough end of the regulatory spectrum, and for good reason. Regulations pin down storage requirements to airtight, corrosion-resistant containers, away from heat and light. Container labels display hazard symbols that speak plainly: toxicity and reactivity take center stage. Engineering controls in facilities running OFIB involve full ventilation, leak detection equipment, and strict protocols for cylinder handling. There’s no casual approach to managing this chemistry, and that’s in line with long-standing Occupational Safety and Health Administration guidelines, as well as similar international frameworks. Every step from receiving to disposal carries strict oversight, and failure to comply brings real risk.
Preparation Method
Synthesis of OFIB usually comes from specialized fluorination of a suitable hydrocarbon precursor. Often, industrial preparation involves treating isobutylene or a structurally-related alkene with elemental fluorine or a fluorination agent in the presence of controlled conditions. This isn’t kitchen chemistry. Controlling the reaction temperature, pressure, and feed rate keeps it both efficient and safe. Any deviation in those parameters risks side reactions, lower yields, or safety incidents. In practice, only facilities with high-grade chemical engineering controls and trained personnel attempt large-scale OFIB preparations. This chain of custody reflects a broader industry truth: skill and precision still matter most.
Chemical Reactions & Modifications
OFIB’s chemical backbone offers opportunities and risks in equal measure. Its reactive double bond, heavily shielded by electron-withdrawing fluorine atoms, sets the stage for select addition reactions. Many synthetic chemists use OFIB to create specialized fluoropolymers by controlled polymerization. At the same time, its electrophilic nature challenges established reaction approaches, putting a premium on specialized catalysts and carefully chosen reaction partners. Attempts to modify OFIB, whether by nucleophilic attack or free-radical methods, call for solid training and patience. Some research groups keep searching for new ways to tame or transform OFIB’s unique chemistry, hoping to unlock new structural motifs or functional materials that nobody’s managed before.
Synonyms & Product Names
OFIB rarely travels under a single name. Some call it Perfluoroisobutylene, while others refer to it by its systematic designation, Octafluoro-2-methylpropene. Chemical catalogs may list it under abbreviations. The proliferation of names stands as a reminder of both the compound’s complex history and the global nature of chemical commerce. Synonyms can create confusion, but in most regulated facilities, safety data sheets and product codes keep everyone aligned. The importance of clear, consistent identification cannot be overstated, especially in cross-border shipments or collaborative research where language and standards can shift from one region to another.
Safety & Operational Standards
OFIB is not forgiving. Exposure causes severe irritation, and even small leaks demand an immediate response. Chemical operators, engineers, and emergency responders draw on robust safety protocols: respirators, chemical suits, and air monitoring come into play. Additional layers of protocol shut down processes at the first sign of trouble. Inhalation risks drive much of the regulatory guidance, with workers rotating out of exposure zones and medical teams monitoring for acute symptoms. Regulatory bodies from the United States Environmental Protection Agency to the European Chemicals Agency categorize OFIB as a substance of very high concern, and for good reason. Operational standards stress lock-out procedures, dedicated ventilation, and rapid decontamination capacity. My own experience in chemical labs taught me early that working with compounds like OFIB is never just technical; it demands a cultural commitment to risk awareness and constant vigilance.
Application Area
Very few industrial arenas find space for OFIB. Its most prominent role lands in the manufacture of high-performance fluoropolymers and advanced coatings. These materials ride on OFIB’s chemical resilience, offering exceptional resistance to heat, acids, and aggressive solvents. The electronics sector also relies on materials made with OFIB: circuit board manufacturers, wire insulation engineers, and semiconductor fabricators use OFIB-based polymers to protect sensitive hardware. There’s also limited use as a chemical intermediate for producing specialty surfactants and lubricants where breakdown equals disaster. Some defense applications linger on the margins, where environmental endurance meets reliability needs. My work intersected with OFIB only briefly, but the professionals around it always spoke of the molecule’s contribution to pushing the edge of what’s possible in harsh service environments.
Research & Development
OFIB continues to draw research interest from academic labs and industrial innovation programs. Investigation focuses on safer production, improved polymerization controls, and new chemical modifications that might extend utility beyond existing fluoropolymer fields. A growing wave of researchers targets less hazardous, more environmentally-friendly routes for fluorinated product synthesis. The constant challenge is clear: preserve the performance benefit without amplifying risk or environmental impact. Patents keep rolling in on specialized derivatives and functionalized fluorinated polymers, signaling steady investment in this space. Interdisciplinary research—where synthetic chemists work with environmental scientists, toxicologists, and engineers—bolsters the scientific rigor behind every new development. This collaborative approach aligns with the ongoing push from regulatory bodies and global watchdogs for a more responsible fluorochemical industry.
Toxicity Research
TOXICITY commands respect in every discussion around OFIB. Inhalation reports echo serious risks, including respiratory distress and systemic toxicity at exposures lower than many other industrial gases. Animal studies and case histories point to lung damage and neurological symptoms at low concentrations. Much of the toxicity research now explores mechanisms behind these effects. Molecular toxicologists look at OFIB’s reactivity with biological macromolecules, trying to map out exactly how damage unfolds in tissues. Occupational health guidelines reflect conservative limits—if any uncertainty exists, safety gets the benefit of the doubt. Ongoing studies chase better medical countermeasures and preventative protocols, since field incidents can evolve quickly from exposure to crisis. There’s no shortcut; rigorous research and honest reporting hold the line for worker safety and emergency preparedness.
Future Prospects
Change always pushes up against tradition, especially in chemistry. OFIB faces an uncertain future, shaped by both its indispensable performance and its tough reputation for risk. Manufacturers and researchers keep searching for safer alternatives, greener synthetic avenues, and process improvements that reduce both hazard and waste. Regulatory agencies worldwide lean hard on persistence, bioaccumulation, and toxicity data, so any process involving OFIB must answer to ever-tighter standards. Some predict a shift away from compounds like OFIB as new fluorine-free polymers offer similar durability without the legacy risks. Others argue that for some extreme conditions, nothing beats the chemistry OFIB delivers. What’s clear across the industry: no one can afford to ignore growing demand for sustainability, transparency, and accountability. The next decade could deliver fresh breakthroughs or new regulations that redraw the boundaries for OFIB’s use, but every change will stand on a mix of technical achievement and long-term responsibility.
What is Octafluoroisobutylene used for?
Understanding Its Place in Industry
Octafluoroisobutylene isn’t a term you'll overhear chatting at the grocery store, but it holds a key spot in the world of specialty chemicals. This colorless, highly reactive gas gets most of its attention from folks in chemical manufacturing. It has eight fluorine atoms. That may not sound exciting, but every fluorine atom brings serious chemical muscle. The bonds are tough, so materials made from this gas tend to resist heat, acids, and electricity better than a lot of other options.
Where It’s Used and Why It Matters
Manufacturers mainly use octafluoroisobutylene to create specialty polymers and fluorinated surfactants. Teflon might ring a bell when you think of slick frying pans, but it’s not just about slippery cookware. Chemical companies use octafluoroisobutylene to build blocks for copolymers that wind up in aerospace parts, fuel-resistant seals, and high-performance gaskets. Those seals hold steady where other materials would melt or crack, especially around rocket fuel or aggressive solvents.
During my time working in a custom plastics shop, I saw how manufacturing teams respect these specialty polymers. We’d receive huge blocks of raw copolymer. Engineers wanted it because it wouldn’t deform in hot or caustic environments. People trust these kinds of plastics not because someone said, “It’s high-tech,” but because they’ve seen other options fail in tough real-life tests.
Risk and Safety Concerns
Octafluoroisobutylene isn’t something you’d want in your lungs. Breathing even a little bit causes severe harm, especially to the lungs and nervous system. Fast-acting medical attention is the only way anyone walks away from acute exposure. I’ve heard from professionals in the safety field who say handling this gas requires elaborate equipment and airtight procedures. Leaks aren’t just bad for people close by; they can cause trouble for neighborhoods around chemical plants if things go sideways.
Regulators have strict guidelines for storing and transporting this material. Employees wear suits that keep them fully sealed. Factories use sensors to catch even the tiniest leak. Building this infrastructure costs a lot, but experience shows that doctors can’t fix the worst damage. There are no shortcuts – elaborate safety gear and careful planning keep folks from tragic mistakes.
Environmental Impact
Fluorinated chemicals linger in the environment far longer than most pollutants. Some of them break down into stubborn forms that drift through soil and water, sticking around for years. It’s the same pattern seen with PFAS—“forever chemicals”—which build up in people, animals, and whole ecosystems. Once these chemicals escape, cleanup takes a massive effort and a big financial toll. Scientists continue digging into long-term health effects that might not show for decades.
Looking For Solutions
Companies need safer alternatives and new protective gear for workers. Stronger chemical bonding or less hazardous substitutes could lower risks. I’ve met researchers trying to break down persistent fluorinated compounds with advanced filtration and chemical treatments—but real progress comes slow and leaves a trail of expenses. Community groups near chemical plants often push for open communication and more transparent safety audits. Stories from neighbors remind us that trust doesn’t come from press releases. Instead, it grows when people see that companies act before accidents happen.
Octafluoroisobutylene carves out its role in the industry thanks to its unique chemistry, but long-lasting safety and environmental challenges require greater investment, rigorous oversight, and honest conversations among workers, neighbors, regulators, and manufacturers.
What are the safety precautions when handling Octafluoroisobutylene?
Understanding the Risks
Octafluoroisobutylene sits in chemical storage rooms with a reputation most folks wouldn’t want on their property. Breathing in just a bit of this gas can send someone from feeling just fine to suffering from coughing fits, breathing trouble, even worse. There’s a reason many chemists and plant workers respect this substance before they get anywhere near it. The warnings about its toxicity don’t come from corporate policy—they come from years of on-the-job experience and, sometimes, close calls.
Contact with skin or eyes leads to irritation. Inhalation proves even more dangerous. The lungs can react badly. The first signs might go unnoticed, but once symptoms show up, the chance for harm is real. I learned early that even so-called minor exposure ends poorly. There is no casual handling with this gas.
Personal Protective Equipment: The Real Shield
Down on the plant floor or in the lab, gloves and goggles only begin to scratch the surface. Full-face respirators become part of the uniform. Rubber gloves, chemical-resistant clothing, and boots help form a barrier. I’ve seen what happens without them—blistering, rashes, and scrambles to emergency showers. Respirator cartridges need to match the specific gas, not just any filter. Companies issue fit tests for good reason; a mask must create a reliable seal, or the protection just doesn’t work.
Ventilation and Engineering
Everyone hears about keeping doors open for fresh air, but the real defense involves flowing air out and away from the workspace. Fume hoods or well-designed extraction systems remain must-haves. I worked in a facility where the fan broke, and even with open doors, the vapor left us coughing. No gas escapes routine checks here—engineers walk the area, check monitors, and verify airflow every shift.
Handling and Storage: Vigilance Pays Off
Storage involves much more than sticking a cylinder on a shelf. Octafluoroisobutylene can leak at threads and valves, so double-checks with soapy water help spot trouble. No one ever eats or drinks near storage to avoid accidental exposure. Each bottle sits far from acids, bases, and anything reactive. Labels stay visible; date checks and regular inspections help remind everyone what’s in each container and how old it is.
Transport means short trips with secure straps and upright cylinders. No shortcuts. Training new team members, I always started with this lesson: never skip the pressure check on a cylinder, and never assume someone else already did it.
Planning for Spills and Emergencies
Accidents still happen. Every good lab or plant keeps a spill kit, not just tucked away but clearly visible and ready. Emergency showers and eyewash stations line the routes out. Colleagues drill together, and every new hire walks the escape plan on day one. My old supervisor repeated every month: “The first minutes in a spill mean the difference between walking away and a hospital bed.”
Local codes call for alarms and monitors. The smartest plants go further. Real-time gas monitors hang from jackets. People carry radios, not just for convenience but to call for help fast. Medical response teams know the agent involved before someone calls for backup.
Building a Culture of Safety
No safety manual can replace good training and a healthy respect for what octafluoroisobutylene can do. Chemistry has given humanity many tools, but some require caution every step of the way. Trust in good habits, modern gear, and open conversation; lives and livelihoods count on it.
What is the chemical formula of Octafluoroisobutylene?
Getting to Know Octafluoroisobutylene
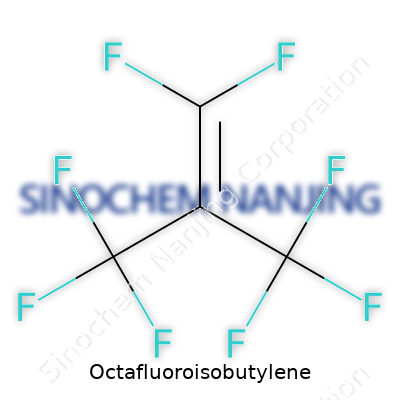
Octafluoroisobutylene, or OFIB, stands out in the world of industrial chemicals because of its unique combination of carbon and fluorine. The chemical formula is C4F8. You’re looking at a molecule made up of four carbon atoms, each surrounded by eight fluorine atoms. It’s simple in how it’s put together, but its properties pack a punch. Fluorine atoms change the way molecules behave, often making them resistant to heat, water, and solvents.
The Chemical Formula Makes a Difference
Every atom in this formula matters. Those eight fluorine atoms bring a level of stability that’s tough to match. It creates a barrier against breakdown, so the compound sticks around longer than most. That’s attractive for industries making synthetic rubbers, certain polymers, or specialized coatings. Once I worked alongside a team looking for something tough enough to survive harsh chemical environments, and OFIB provided a level of endurance that other compounds couldn’t touch. It’s not just tough; it’s also exceptionally reactive in some settings, which can help with making fluorinated materials.
Risks and Public Health
No one can talk about this formula without thinking about the risks. Just a few years ago, I attended a chemical safety talk where the dangers of inhaling fluorinated gases were front and center. OFIB fits into that category. Short-term exposure brings sharp respiratory problems. According to the Centers for Disease Control and Prevention (CDC), exposure can cause throat and lung irritation—sometimes severe enough to affect breathing. People working around it face more danger if they don’t use proper protective equipment or handle it inside ventilation systems designed for the job.
Industrial Use and Environmental Impact
Industries don’t choose OFIB by accident. Its chemical resistance helps produce high-performance materials, like fluoroelastomers. The downside comes from its stability. If it escapes into the environment, it can stick around, and breakdown happens slowly, if at all. Even trace leaks can affect air quality over time. That’s where real responsibility comes in—companies need to track emissions, capture leaks early, and invest in safe handling practices. Modern regulations, like those from the Environmental Protection Agency (EPA), encourage safer storage and disposal. Still, accidental releases do happen, so strong protocols can’t just sit on the shelf—they need to be followed every day.
Working Toward Safer Solutions
Solving risks with OFIB involves more than just better masks or tighter seals. Facility operators benefit from continuous air monitoring. Workers need updated training—not just once, but regularly—because complacency can creep in over years on the job. On the research side, chemists hunt for substitutions that offer similar durability but with less environmental baggage. Even small gains in breakdown or safer byproducts can add up across an industry. Open reporting of near-misses or accidental exposures helps develop smarter strategies for everyone. Sharing these lessons, instead of hiding them, pushes the whole field forward.
Learning from the Chemistry
Every formula tells a bigger story. Octafluoroisobutylene’s C4F8 structure reminds us that real safety comes from understanding both the promise and the pitfalls of a compound. Decades of industrial experience show that respect for such powerful chemistry never goes out of style. Whether you’re handling OFIB or writing a new safety protocol, knowing the risks shapes better choices and keeps people out of harm’s way.
Is Octafluoroisobutylene hazardous to human health?
What Makes This Chemical Stand Out
Octafluoroisobutylene shows up in niche industrial settings. It’s a colorless gas and ranks high among the reactive fluorinated compounds. Chemical plants and specialty manufacturers rely on it for specific purposes, but most people have never heard of it. That’s part of the problem. If you don’t know about something, you might underestimate it.
Potential Health Dangers Are No Small Matter
People working around octafluoroisobutylene often see warnings and need real personal protective equipment. Short exposure can cause irritation of eyes, nose, and throat. Most modern safety data makes it clear that inhaling this chemical can trigger coughing and breathing problems even at low levels. In larger doses, it hits harder—think fluid buildup in the lungs, known as pulmonary edema. That outcome can be fatal without fast medical attention.
I once worked in an emergency facility near a chemical plant. We’d sometimes get calls from hazmat teams about accidental exposures. You can’t just run a fan and walk it off. When someone has inhaled a reactive gas, you see their distress fast—difficulty drawing breath and a panic in their eyes. Not all gases produce an immediate smell or taste, and perfluorinated chemicals especially have this deceitful way of seeming harmless at first.
Why This Chemical Deserves More Attention
Chemical safety rules in the U.S. and Europe treat octafluoroisobutylene as a substance needing careful control. Animal testing points to several alarming trends. Even if the full impact on humans needs more study, the animal data steers policies for good reason. Chronic exposure can mess with lung function and may even cause damage to other organs. We know some perfluorinated compounds stick around inside our bodies longer than most other chemicals. That raises concerns about long-term effects beyond what’s obvious.
Workers face the highest risk, but communities near plants or waste sites could also face exposure—especially after spills, leaks, or fires. The Environmental Protection Agency groups it with other fluorinated greenhouse gases and tracks releases. Governments set exposure limits, but enforcement and accident response varies widely. I’ve seen towns left out of the loop about what’s moving through pipelines under their feet. A one-off leak can put hundreds at risk before anyone realizes what happened.
What Steps Build Better Safety
Effective safety practices don’t leave things to chance. Chemical companies already require hazard awareness training, air monitoring, and full respiratory protection. Quick reporting systems help spot small leaks before they turn into disasters. Community right-to-know laws give neighbors some control and reassurance. I’ve learned that public information campaigns work best when they empower ordinary people to ask hard questions. Real transparency encourages prevention, not just response after the fact.
Safer storage, better emergency drills, and clear labeling prevent most mistakes. Technology can catch what people miss. Sensors now flag contamination in real time, prompting faster evacuations or repairs. Regulators need enough resources to keep up with new chemicals and to hold polluters accountable.
Nobody wants an industrial process shut down by fear, but we can’t gamble with silent, invisible hazards. I’ve seen firsthand how trust between plant workers, emergency responders, and communities pays off. When everyone speaks up about what makes them nervous, accidents drop, and lives get saved. The dangerous legacy of chemicals like octafluoroisobutylene isn’t inevitable—especially if we refuse to look away or hope for the best.
How should Octafluoroisobutylene be stored and transported?
Knowing What You’re Working With
Octafluoroisobutylene grabs little attention outside of chemical circles, but those who use or ship this gas understand the weight of responsibility. This isn’t the kind of material where a label and a locked cabinet do the job. A colorless, highly toxic gas, Octafluoroisobutylene isn’t something you want loose in a workspace, much less a truck or rail car. Breathing it leads to severe lung irritation or worse, and any leak can spell a major emergency. My own near-miss stories stay vivid, sometimes triggered by the hiss of a cylinder valve or a drifting memory from a chemical plant floor.
Hazard Recognition Shapes the Rules
People often underestimate how ruthlessly unforgiving toxic gases become when things go sideways. For Octafluoroisobutylene, every facility and shipping lane depends on steel cylinders built for high pressure. Engineers run stress tests and regulators call for periodic inspections, and there’s no room for neglect. Even minor valves or regulators need materials tough enough to resist corrosion. A cheap fitting turns into a weak spot, and somewhere along the line, experience has shown that repairs with the wrong parts raise the odds of a dangerous mistake.
Temperature and Ventilation: Constant Vigilance
Safe storage rarely happens by accident. Cylinder racks in cool, shaded spaces keep temperatures stable and lower pressure inside the tanks. Direct sunlight isn’t just an inconvenience — it could lead to cylinders venting or rupturing. Facilities with real-world safety records ensure their storage zones include strong ventilation or robust automated detection. I remember stories of operators relying on their noses for leaks, but with a gas like this, human senses give false comfort. Continuous monitors offer better odds, and a good system buys time to respond to an alarm before a small leak becomes a catastrophe.
Transport Creates New Risks
Moving cylinders means putting faith in a chain of drivers, containers, and schedules. Secure strapping inside dedicated vehicles helps prevent rolling or shifting. I’ve seen drivers get lazy and sling tanks into pickup beds without proper supports, and a single jolt can shear off a valve. Regulations force clear hazard markings on every cylinder, and logs track not only what gets moved but where it ends up. Decision makers can strengthen safety by requiring double checks at every loading point and destination, making sure no container sits unaccounted for.
Emergency Preparedness Matters
In the event of a release, there are rarely second chances. Workers keep escape hoods within easy reach, and quick-draining showers and eyewash stations stand nearby. Up-to-date information feeds into quick-reference guides: spill containment, evacuation routes, and emergency numbers posted right on walls. Drills play a big part. My team ran through scenarios until muscle memory replaced panic, and that training made all the difference during a real incident. Facilities with strong track records encourage staff to flag gaps and suggest improvements, rather than hiding mistakes out of fear.
Better Storage and Transport, Fewer Headlines
Mistakes involving Octafluoroisobutylene rarely stay private. News of a leak draws a crowd, from neighbors to regulators. Companies staying ahead of problems review their protocols, adapt storage to changing weather, and refresh supply chains instead of falling back on “how it’s always been done.” Creating a culture built on responsibility rather than shortcuts does more for safety than any policy binder ever could.
| Names | |
| Preferred IUPAC name | 2,3,3,3-Tetrafluoro-2-(trifluoromethyl)prop-1-ene |
| Other names |
Perfluoroisobutene PFIB |
| Pronunciation | /ˌɒk.təˌflʊə.roʊˌaɪ.səˈbjuː.tɪliːn/ |
| Identifiers | |
| CAS Number | 307-32-4 |
| 3D model (JSmol) | `JSME 3D model string for Octafluoroisobutylene:` `CF2=C(CF3)2` |
| Beilstein Reference | 87328 |
| ChEBI | CHEBI:38160 |
| ChEMBL | CHEMBL371076 |
| ChemSpider | 12260 |
| DrugBank | DB08184 |
| ECHA InfoCard | 13b95b80-4177-4585-b8c5-116d71e20a67 |
| EC Number | 206-202-1 |
| Gmelin Reference | 303181 |
| KEGG | C18682 |
| MeSH | D010370 |
| PubChem CID | 6433 |
| RTECS number | YO8585000 |
| UNII | Q7J506278F |
| UN number | UN2419 |
| CompTox Dashboard (EPA) | DB11553 |
| Properties | |
| Chemical formula | C4F8 |
| Molar mass | 216.02 g/mol |
| Appearance | Colorless gas |
| Odor | Odorless |
| Density | 1.53 g/mL at 25 °C |
| Solubility in water | Insoluble |
| log P | 1.34 |
| Vapor pressure | 5515 mmHg (20 °C) |
| Acidity (pKa) | Approximately -5. |
| Basicity (pKb) | pKb: -6.2 |
| Magnetic susceptibility (χ) | -37.5×10^-6 cm³/mol |
| Refractive index (nD) | 1.252 |
| Viscosity | 0.41 mPa·s (25 °C) |
| Dipole moment | 0.53 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 256.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1431 kJ mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1285.3 kJ/mol |
| Hazards | |
| Main hazards | Toxic if inhaled, causes severe respiratory irritation, may cause delayed pulmonary edema. |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | Hazard statements: H301, H330 |
| Precautionary statements | P260, P262, P271, P280, P284, P304+P340, P320, P308+P311, P312, P403, P405, P501 |
| NFPA 704 (fire diamond) | 3-4-2-W |
| Flash point | -78 °C |
| Autoignition temperature | 180°C |
| Explosive limits | Explosive limits: 4.5–24% |
| Lethal dose or concentration | Lethal Concentration (LC50, rat, inhalation): 1 ppm/4 hr |
| LD50 (median dose) | LD50 (median dose): 270 mg/m3 (inhalation, rat) |
| NIOSH | NIOSH: TQ4550000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Octafluoroisobutylene: **0.01 ppm** |
| REL (Recommended) | 100 ppm |
| IDLH (Immediate danger) | IDLH: 10 ppm |
| Related compounds | |
| Related compounds |
Hexafluoroisobutylene Perfluoroisobutene |

