Isobutylbenzene: A Real-World Look at Its Journey, Uses, and Future
Historical Development
Back in the early 20th century, chemists looking for new building blocks in synthetic organic chemistry landed on isobutylbenzene. This compound, featuring a benzene ring with an isobutyl side chain, helped expand the production toolkit for pharmaceuticals and fine chemicals. Early industrial players in Europe and the US studied and commercialized isobutylbenzene alongside its straight-chain cousin, n-butylbenzene, seeing its promise in both research and large-scale operations. By the mid-1900s, isobutylbenzene played a central role in the synthesis of key painkillers, most famously ibuprofen. Decades later, companies still depend on efficient, scalable access to isobutylbenzene, underscoring how the small discoveries of one era can shape medicine and chemical manufacturing for generations.
Product Overview
Isobutylbenzene stands out for more than its structure. It takes the form of a colorless, oily liquid with a mild aromatic aroma, available as both a reagent and as a material for downstream synthesis. Chemists and manufacturers buy it for purity, stability, and its place in the supply chain for active pharmaceutical ingredients, surfactants, and agrochemicals. No matter where you source it, the drive for quality and consistency shapes its value in practical applications, whether in bench research or tons-at-a-time output.
Physical & Chemical Properties
Experience with bulk chemicals teaches you that every compound comes with tradeoffs. Isobutylbenzene melts around −30°C and boils near 170°C, making it a liquid in all but the most extreme conditions a lab might face. It carries a density a touch below water—about 0.86 g/cm³—so it floats and pours easily. The molecule itself, C10H14, gives off a faint aromatic odor, hinting at its structure and behavior. It dissolves into organic solvents and resists water, giving chemists precise control over where it goes in a synthetic route. Its moderate volatility and stability let plant operators store and transport the bulk chemical without constant headaches.
Technical Specifications & Labeling
Chemical producers do not mess around with specifications. Most offer isobutylbenzene with a purity above 99%, checked by gas chromatography and specified for water content, acidity, and residue. Drums and bottles carry batch numbers, hazard labels, and safety markings because regulators take flammability and exposure risks seriously. Material Safety Data Sheets (MSDS) get stapled to shipments, spelling out the rules for use, transport, and storage. Labels in the EU and US show the same warning—flammable liquid and vapor, don’t breathe fumes, store cool and away from ignition. Whether you work in a pharma lab or a distribution warehouse, these details make the difference between smooth operations and compliance trouble.
Preparation Method
Manufacturers typically turn to Friedel-Crafts alkylation for this compound, mixing benzene and isobutylene under acidic conditions, often with aluminum chloride as the catalyst. This approach, around since the 19th century, delivers high yields but calls for tight control to avoid side reactions and environmental problems. Modern operations lean toward greener variants: solid acid catalysts, closed-loop solvent recovery, and better waste management. Every improvement comes from decades of trial, error, and tightening regulations on industrial emissions and worker exposure.
Chemical Reactions & Modifications
Chemists see isobutylbenzene as both a starting block and a bridge to bigger molecules. One of the key reactions involves oxidation, which transforms it into p-isobutylacetophenone and then to ibuprofen. Other groups modify the side chain, install functional groups, or oxidize specific positions to tailor new intermediates. The molecule responds well to stepwise transformations, and the aromatic ring resists harsh conditions, letting skilled hands push synthesis further without breaking the framework. Every innovation around this compound opens new avenues for medicines, polymer additives, and specialty chemicals.
Synonyms & Product Names
In the catalogues, isobutylbenzene sometimes goes by 2-Phenylbutane, or 1-Phenyl-2-methylpropane, or CAS number 538-93-2. Depending on region and tradition, suppliers may use bulk trade names or shorthand like IBB. These aliases help logistics teams navigate customs and regulations, but chemists stick with clear identifiers to avoid mix-ups, especially with isomers like n-butylbenzene. Such naming details play a surprisingly big role during procurement and in global trade, where even a minor labeling slip can delay shipments or spark misunderstandings.
Safety & Operational Standards
Anyone working with organic chemicals like isobutylbenzene takes safety seriously, not just out of habit but from real-life lessons. Flammable vapors mean you keep open flames and sparks far from the lab bench or drum storage. Prolonged exposure irritates skin, eyes, and lungs, so gloves, goggles, and fume hoods become daily gear. Spills get absorbed and contained quickly—no shortcuts. Industrial users rely on detailed SOPs, fire suppression systems, and incident logs to protect people and property. Aging facilities that once overlooked best practices now upgrade to meet today’s OSHA and EU-REACH standards, knowing the true cost of shortcuts over time.
Application Area
Isobutylbenzene leaves its mark in several industries. In pharmaceuticals, it lays the groundwork for the anti-inflammatory drug ibuprofen, supplying a global market that treats headaches and arthritis on a daily basis. Chemical makers draw on it for surfactants and synthetic lubricants, where performance and reliability matter. Agrochemical companies use it as an intermediate for crop protection compounds, most of which get nowhere near public attention but quietly increase yield or reduce pests. The paint and coatings field taps it for specialty solvents, counting on its balance of volatility and solvency. Each sector faces competitive pressure and regulatory scrutiny, so the quality and supply of isobutylbenzene end up influencing more people than most realize.
Research & Development
R&D groups at universities and companies still push the frontiers of what isobutylbenzene can do. Efforts focus on new syntheses that cut environmental impact, both to lower waste and meet rising green-chemistry targets. Teams explore new derivatives, chasing better-performing pharmaceuticals and agrochemicals. Safety researchers profile exposure risks in new contexts, aiming to limit health effects in both workplace and end-use settings. Collaborations between academic labs and industry bring out cross-disciplinary innovations, whether that means a new catalytic system or entirely different uses for structural relatives of isobutylbenzene. The pressure to stay competitive drives constant test-and-learn cycles—nobody stays ahead by relying on last decade’s methods.
Toxicity Research
Any chemist who has worked with organic solvents knows there’s no such thing as a harmless chemical—only controlled risk. Acute exposure to isobutylbenzene in high doses can cause respiratory distress, irritation, and CNS symptoms. Chronic effects remain less understood, which pushes regulators and scientists to investigate metabolism, breakdown products, and long-term occupational exposures. Toxicology studies, often with animal models and cellular assays, point to moderate concern at high doses but suggest low persistence in the environment. Still, industry groups know the risk of underestimating unknowns, so responsible users invest in air monitoring, biological exposure indices, and ongoing medical surveillance for staff. Making this a regular part of operations helps avoid nasty surprises and regulatory action down the road.
Future Prospects
Looking ahead, the story of isobutylbenzene probably mirrors broader trends in specialty chemicals: cleaner production, improved worker safety, and expanded end uses. Efforts to catalyze the Friedel–Crafts process more efficiently, recycle solvents, and streamline waste management promise to cut environmental impact and save costs. Pharmaceutical demand shows no sign of dropping, and new crop protection products will keep drawing on isobutylbenzene’s structure and reactivity. As regulations worldwide clamp down on emissions and toxic releases, businesses that get out in front with greener methods and transparency will have a head start. Startups and academic groups may shake up the market with bio-based alternatives or new reaction pathways. Experience teaches that the essentials of production and stewardship can’t be taken for granted, especially for a workhorse chemical like this one.
What is Isobutylbenzene used for?
Understanding Isobutylbenzene
Isobutylbenzene doesn’t make headlines, but its impact lands on medicine cabinets everywhere. Most people walk by it in a chemical store, never realizing it helps bring pain relief to millions. Chemists use it as a building block for something people know: ibuprofen. When scientists first cracked ibuprofen’s structure, they needed a stable starting ingredient, and isobutylbenzene fit the bill. The way it behaves during reactions lets factories churn out painkillers at industrial scale, turning a clear liquid into relief for headaches, sore muscles, and stubborn fevers.
Stepping Into the Synthesis Process
Big pharmaceutical companies keep a steady demand for isobutylbenzene thanks to its key part in making common nonsteroidal anti-inflammatory drugs (NSAIDs). Chemists react it with other chemicals—mainly acetic anhydride and catalysts—to kick off a series of steps that lead to ibuprofen. Without this ingredient, production slows and costs rise. Pairing that with robust quality controls, manufacturers turn out consistent product batches safe for public use.
Beyond Ibuprofen: Lesser-Known Applications
Some factories use isobutylbenzene as a solvent because its structure lets it dissolve oily substances. Workers in labs and plants rely on these solvents in routine tests, separating specific compounds from mixtures. It also finds its way into making other fine chemicals. These might not turn into household names, but they play small roles in the background of modern industry.
Health, Safety, and Environmental Impact
Handling chemicals in bulk comes with hazards. Working around isobutylbenzene demands careful safety standards. If someone breathes in its vapors or gets it on their skin, irritation follows. In my stint at a lab, we handled similar solvents with gloves and fume hoods, cutting any risk of accidental exposure. Factories double-check ventilation and monitor air to meet health regulations. The goal? Keep workers safe and neighborhoods free from accidental releases.
On the environmental side, worry builds if isobutylbenzene leaks into water supplies or soil. It breaks down more slowly than some chemicals, making containment and clean-up crucial. Plant operators use closed systems, and local agencies monitor surrounding areas. Investing in strong storage tanks and real-time leak detectors pays off, and it protects both people and the planet.
Balancing Access and Responsibility
Without isobutylbenzene, the supply of basic painkillers would be cut off or get much more expensive. At the same time, the chemical industry bears a burden to produce and handle it responsibly. Rigorous licensing and transparent reporting help reassure the public. From my experience working with teams supervising chemical stockpiles, the best results come when ethics and science share the steering wheel. Chemical-makers and regulators talk openly about risks, and quick reporting keeps communities informed.
Next time someone reaches for a pack of ibuprofen, it’s worth remembering what happens before the pills hit the shelf. The journey starts with isobutylbenzene, a small but crucial player that connects chemistry’s puzzle pieces to real-world relief. Responsible production and awareness keep this ingredient in the right hands, turning quiet chemical know-how into widespread wellbeing.
Is Isobutylbenzene hazardous or toxic?
The Stuff Behind Isobutylbenzene
Isobutylbenzene has become a familiar name in the world of chemicals. Digging into its use, you’ll find it threaded through the production of several pharmaceuticals, like ibuprofen, where it acts as a building block. Factories, labs, and even some classes in college chemistry deal with the substance, so it’s hardly an unknown entity for those who work around chemicals. Exploration about what this substance means for health and safety isn’t just worthwhile—it’s necessary.
The Risks We Can’t Ignore
Isobutylbenzene looks harmless as a colorless liquid, but real problems can start if the stuff isn’t handled properly. Breathing in fumes or letting it touch skin isn’t just a matter of discomfort. People report headaches, dizziness, and nausea after exposure. That makes sense, since animal studies link inhalation to central nervous system issues. Touch it, and you risk skin irritation. In a place with poor airflow, fumes could build up, leading to stronger symptoms and danger.
Any chemical that can affect the nervous system deserves respect, not fear—but not complacency, either. Long-term exposure studies are thin, but the body doesn’t seem to like having even small doses for too long. Factory safety data sheets put plenty of warnings alongside this one.
Numbers, Not Just Warnings
The Occupational Safety and Health Administration (OSHA) and the National Institute for Occupational Safety and Health (NIOSH) set guidelines for workplace exposure to chemicals. Isobutylbenzene isn’t on everyone's hazard radar, but that doesn’t mean it’s risk-free. General hydrocarbon limits apply. When researchers measured how much gets absorbed, the results showed it can build up in tissues. Authorities in Europe and the United States both ask for good ventilation, skin protection, and thoughtful handling. The fire risk stays on the table too—fumes can ignite with the right spark.
Is It Toxic?
Some will ask point-blank if isobutylbenzene kills. If a worker drinks, inhales, or bathes in it, the results could be ugly, but it doesn’t rank with the world’s deadliest poisons. Still, it’s toxic enough to worry labs and plant operators. The US Environmental Protection Agency flags isobutylbenzene as an irritant and possible environmental hazard. Spills hurt fish and water quality. I remember, during a summer research stint, how panicked everyone got during a small lab spill. Fume hoods, gloves, goggles—everyone doubled up. People didn’t just use tools because the manual demanded it; nobody wanted to risk feeling out of sorts, getting a rash, or having to report to the medical office over sloppy handling.
Common-Sense Steps Forward
Simple moves can keep places safe. Proper gloves and goggles go a long way. Good ventilation and sealed containers matter. Training helps more than most people think. I’ve seen inexperienced lab techs make mistakes, but a five-minute talk from an older hand about what can go wrong tends to stick more than a checklist ever does. In my own lab life, routine care was more than a chore—it meant missing out on headaches, drama, and emergency showers.
Sensible regulation, regular safety refreshers, and respect for the substance work far better than scare tactics or bravado. That’s the kind of thinking that gets folks home healthy—and keeps Isobutylbenzene from being a bigger problem than it has to be.
What is the chemical formula of Isobutylbenzene?
What’s Behind the Formula
Isobutylbenzene shows up in a lot more corners of daily life than most would expect. From personal experience working in the lab, it’s not unusual to see a technician searching the shelves for this compound mid-experiment. The formula for isobutylbenzene is C10H14. If you picture the structure, it’s essentially a benzene ring connected to an isobutyl group. The simplicity of those numbers doesn’t do justice to how much chemistry fits inside.
Applications Make It Relevant
A lot of cold medicine relies on this compound. Isobutylbenzene acts as a building block during the creation of ibuprofen, one of the most trusted pain relievers in households worldwide. The role it plays in that synthesis gives it an outsize impact; simple changes in the feedstock or formula influence years’ worth of production globally. Chemists and process managers keep a sharp eye on its purity because mistakes can lead to batch failures or, worse, public recalls.
The answers people look for about C10H14 stretch beyond pill bottles. This compound sees use in fragrances, too. Its structure lets it carry specific volatile scents, making it an option in certain perfumery blends. From my time scent-testing at a small-scale essential oil lab, there’s real craft involved in getting just the right blend—too much or too little isobutylbenzene changes the final note, sometimes ruining a batch. The chemistry sets the foundation for innovation, not just industrial processes.
Why Accuracy Matters
Listing the correct chemical formula isn’t about passing chemistry quizzes. Formula accuracy keeps supply chains honest and science reliable. A wrong C11H16 label means another chemical entirely, and real-world outcomes can become dangerous. Toxicity data, safety precautions, and legal compliance all depend on the right numbers. For any producer or researcher, trusting a formula influences how confidently they work. My background in chemical safety drills this home: even a single error with a seemingly simple structure means paperwork, wasted resources, and sometimes even evacuation drills for safety’s sake.
Challenges and Solutions in Manufacturing
Producing high-quality isobutylbenzene brings challenges. Market demand for ibuprofen puts pressure on upstream suppliers to deliver at scale, but shortcuts usually show up in the product—either as impurities or unreliable deliveries. Investing in sustainable, traceable supply helps. Bio-based approaches gain traction, especially in places with strict regulations on pollution. By working directly with reputable suppliers, labs steer clear of quality drops and shady intermediaries. Digital inventory tracking, once a nuisance, now helps catch mix-ups early. Teams no longer rely on memory or penciled notes, but instead track every container and batch digitally, reducing costly mixing errors.
Looking Beyond the Formula
People often overlook how a single line of chemical symbols, such as C10H14, influences so much downstream. Each step—from raw ingredient, to warehouse, to medicine cabinet—rests on small details handled by people who know their craft. Regulations, market expectations, and ethical supply chains guide progress, but at the core, real humans make it work. They double-check numbers, ask for purity certificates, and share tips with each other in the break room after a successful run. That’s the nature of science: formulas pave the way, but it’s all about the people who use them wisely.
How should Isobutylbenzene be stored?
Why Proper Handling Gets Real
Isobutylbenzene might not be as headline-grabbing as some industrial chemicals, but anyone who’s spent time in a lab or on a factory floor knows that overlooking safety basics can turn a routine shift into a crisis. I once watched a coworker learn this the hard way when he stacked some solvent drums too close to an old heat register. The smell hit us before we saw the problem. No sirens went off, but the lesson stuck—no chemical rewards a lazy approach, especially flammables like isobutylbenzene.
This stuff brings a distinctive odor and flammable vapors. Misstore it, and you risk not just property but people. Turn on the news, and chemical fires always seem like freak accidents. From experience, sloppy storage habits often play a starring role. For something with a flash point below the temperature of some summer afternoons, it’s not hard to imagine what one careless spark can do.
Key Realities: Not Just a “Lab Thing”
A locked closet or a labeled shelf does not cut it for isobutylbenzene. The National Institute for Occupational Safety and Health flags it as a substance where airborne limits matter. Once, I worked at a facility that skipped regular ventilation checks to save costs. I saw headaches, nausea, and more than a few mysterious work absences. Only after an air quality audit did the link become obvious. Ventilation makes a real difference—store isobutylbenzene in a cool, well-aired spot, away from sunlight and hot pipes. No hotplates nearby, either. Heat and vapors never make good company.
Fire Risks Demand Respect
If you keep isobutylbenzene near oxidizing agents or anything sparky, you ignore the essential rule of flammable liquids. I remember advice from a grizzled chemical plant manager: “Treat flammables with the respect you want from your neighbors.” Ground your storage containers. Use spark-proof tools. Swap open shelving for sturdy, closed cabinets made for flammables. Fire-resistant containers go a long way; metal safety cans with tight lids remain the gold standard in facilities that can’t afford to gamble with safety. Check grounding wires regularly—static can build up faster than you realize, especially in dry weather.
Label Everything, Review Often
Hazmat teams train on clear labeling and updated logs for a reason. Sudden leaks have a way of finding the least prepared worker. The difference between an urgent cleanup and an emergency can be a well-placed warning sign. In my early days, I saw a coworker reach for the wrong bottle simply because the label had faded from sunlight over years. Luckily, it wasn’t the toxic kind. Never let paint or ink fade; replace labels as soon as you spot wear.
Better Storage, Better Outcomes
OSHA and EPA guidance draws on real-world mistakes—every line written after a close call. Cut corners and you pay, sometimes in lives lost. Plenty of companies have wised up by adopting checklists and routine audits, catching problems before they snowball. I’ve seen firsthand how low-tech solutions—such as buddy checks before the end of every shift—pick up misplaced cans and slow leaks. Sometimes the simplest ideas save the most headaches.
Chemicals like isobutylbenzene reward a thoughtful approach. Give it the right space, label it, lock it down, and audit your system. Those steps keep people safe and businesses running. I’d take visible vigilance over half-hearted compliance any day.
What are the safety precautions when handling Isobutylbenzene?
The Risks You Can’t Ignore
Most folks outside the world of chemistry or pharmaceuticals rarely cross paths with isobutylbenzene, but anyone who mixes chemicals in labs or works with industrial solvents knows this one can cause trouble when handled carelessly. Working in a small research lab years ago, I saw how easy it feels to get comfortable around regular solvents, then suddenly realize a headache or a skin rash caught you off guard. Isobutylbenzene is no pushover—short-term exposure brings its share of issues, from irritation to worse, if spilled or inhaled in high amounts.
Why Personal Protection Isn’t Up for Debate
Putting on gloves and goggles before handling any chemical was drilled into me from day one, and isobutylbenzene makes no exceptions. Splash-resistant lab coats, chemical goggles, and nitrile gloves shield skin and eyes from accidental splatter. It’s not paranoia—one bad splash near your eye, and you spend hours in the wash station, hoping permanent damage hasn’t set in.
Gloves do more than save your hands from discomfort. Prolonged skin contact can lead to sensitization—skin grows more reactive over time, so even a little exposure later triggers stronger symptoms. Given how easy it is to spill or splash liquid while transferring between containers, gloves aren’t just formality—they’re insurance.
Fresh Air Matters—A Lot
Fume hoods often sound like overkill to folks working outside the lab, but anyone who’s ever caught a whiff of isobutylbenzene vapor knows strong ventilation truly makes a difference. At my old workplace, our ventilation system once broke, and headaches swept through the lab after just half an hour. Eyes stung, and air felt hard to breathe. Even quick exposure adds up, as the vapors irritate lungs and mucous membranes.
A well-maintained fume hood runs quietly in the background, pulling off vapors before air quality dips. Investing in good ventilation pays off long before things take a dangerous turn. For places without built-in hoods, at least crack a window or set up a portable exhaust fan. The aim is always to push the air outside, not circulate toxic fumes.
Fire Hazards—A Real Problem
The label says “flammable,” and it isn’t kidding. One missed spark near open containers spells trouble. I’ve witnessed colleagues get complacent and leave bottles open too close to heat sources. Luckily, nothing exploded, but the potential is there. Static electricity, open flames, or even faulty wiring can all ignite vapors.
Storing isobutylbenzene in flammable safety cabinets creates that line of defense between chemical and accidental ignition. Never forget where the nearest extinguisher sits, and never pour the solvent near sources of sparks or static.
How Good Habits Save the Day
Regular training on spill clean-ups and emergency procedures saves precious seconds in a crisis. One energized coworker knocked a flask over during a busy shift. Panic can make things worse, but because the team drilled spill response, cleanup went smoothly, and no one got hurt. A dry chemical spill kit, proper absorbents, and a clear exit path can fix a bad situation before it spirals into a disaster.
Labeling every bottle and keeping containers tightly sealed once finished also prevents accidents. Habitual neatness might feel fussy, but I’ve seen mistakes happen because labels peeled, or bottles went unsealed.
Embracing a Culture of Precaution
Safety comes down to respecting the chemical and the space you work in. Being prepared with proper PPE, paying constant attention to ventilation, treating fire hazards seriously, and building muscle memory for handling spills turns routine work into safer work. By sharing real experiences and sticking to the practices proven to keep people safe, we show care for everyone stepping into a lab after us.
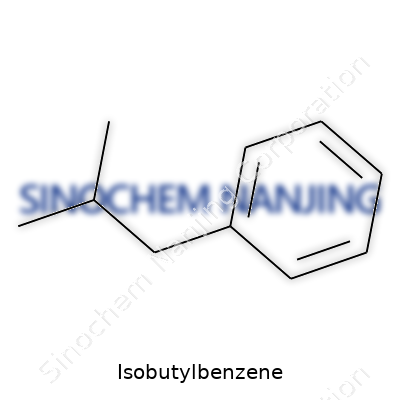
| Names | |
| Preferred IUPAC name | 1-Phenylbutane |
| Other names |
1-Phenylpropane 1-Phenylpropane Isobutylbenzol Benzene, isobutyl- 2-Methyl-1-phenylpropane Isobutylbenzen |
| Pronunciation | /ˌaɪsoʊˌbjuːtɪlˈbɛnziːn/ |
| Identifiers | |
| CAS Number | 538-93-2 |
| 3D model (JSmol) | Isobutylbenzene JSmol 3D model string: ``` Isobutylbenzene CC(C)CC1=CC=CC=C1 ``` |
| Beilstein Reference | 1209247 |
| ChEBI | CHEBI:45986 |
| ChEMBL | CHEMBL14238 |
| ChemSpider | 54686 |
| DrugBank | DB08702 |
| ECHA InfoCard | 100.088.461 |
| EC Number | 202-542-8 |
| Gmelin Reference | 80821 |
| KEGG | C06587 |
| MeSH | D007017 |
| PubChem CID | 7705 |
| RTECS number | CU1400000 |
| UNII | YFN7J0G0RO |
| UN number | UN1993 |
| Properties | |
| Chemical formula | C10H14 |
| Molar mass | 120.19 g/mol |
| Appearance | Colorless liquid |
| Odor | Aromatic |
| Density | 0.861 g/cm³ |
| Solubility in water | Insoluble |
| log P | 2.9 |
| Vapor pressure | 0.9 mmHg (20°C) |
| Acidity (pKa) | 15.0 |
| Basicity (pKb) | NaN |
| Magnetic susceptibility (χ) | -81.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.494 |
| Viscosity | 2.70 mPa·s (25 °C) |
| Dipole moment | 0.83 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | S⦵298 = 336.24 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -24.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -6610 kJ/mol |
| Pharmacology | |
| ATC code | N02CX06 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P301+P310, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P312, P321, P331, P332+P313, P333+P313, P337+P313, P362+P364, P370+P378, P403+P235, P405, P501 |
| Flash point | 142 °F (61 °C) |
| Autoignition temperature | 436 °C |
| Explosive limits | 1.1–6.0% |
| Lethal dose or concentration | LD50 (oral, rat): 1,400 mg/kg |
| LD50 (median dose) | LD50 (median dose) of Isobutylbenzene: "3,400 mg/kg (rat, oral) |
| NIOSH | SN1650000 |
| PEL (Permissible) | PEL: 50 ppm (240 mg/m³) |
| REL (Recommended) | 50 mg/m³ |
| IDLH (Immediate danger) | IDLH: 900 ppm |
| Related compounds | |
| Related compounds |
Benzene Cumene n-Propylbenzene sec-Butylbenzene tert-Butylbenzene Styrene |

