Isobutyl Isocyanate: More Than Just a Chemical Compound
Historical Threads: From Organic Experiments to Industrial Necessity
Isobutyl isocyanate didn’t just appear on a lab bench out of thin air. In the early half of the twentieth century, synthetic chemists poked, prodded, and pushed simple hydrocarbons like isobutane into new directions. German chemists turned their curiosity about alkyl isocyanates into practical knowledge by developing low-cost synthetic routes. The war-driven push for new polymers and agricultural chemicals drove isocyanates out of the chemistry journals and into industry. By the 1950s, isobutyl isocyanate had staked a claim in both pesticide and pharmaceutical product lines. Its utility shaped not only how we made carbamates and ureas, but also how safety protocols evolved for handling toxic chemicals.
Getting to Know Isobutyl Isocyanate
On paper, isobutyl isocyanate stands out as a clear, colorless liquid with a biting odor. Chemists remember its CAS number: 111-36-4, but out in the warehouse or on the factory floor, the strong, pungent smell sets it apart immediately. Formula-wise, this stuff means business—C5H9NO. Unlike its cousin methyl isocyanate, which stole headlines during the Bhopal disaster, isobutyl isocyanate operates more in quiet industry corners, but it demands respect all the same. Its boiling point hovers around 140°C, and its density at 0.874 g/cm3 brings handling choices like specialized glass lines or compatible polymers. The molecule’s isocyanate group enables it to react vigorously with nucleophiles, including water and alcohols. Folks working with it never take chances without reliable ventilation and gloves.
Physical and Chemical Traits
Isobutyl isocyanate doesn’t hide its volatility. The flash point sits uncomfortably low, and its vapor pressure reminds us of its readiness to disperse in air. People learning to store this chemical quickly figure out that even at room temperature, evaporation can pose both health risks and product loss. That’s not just a minor inconvenience; unchecked vapors spell real trouble in enclosed workspaces. In terms of reactivity, it interacts fiercely with acids, bases, and, above all, water. This isn’t the kind of substance you leave uncapped or unlabeled. Physical exposure—eyes, skin, or lungs—demands emergency attention, and chronic exposure is linked to significant health hazards. I remember more than a few anxious new techs reaching for goggles and respirators the first time they uncorked a fresh drum.
Specifications and Labeling That Matter
Labels keep the lab and manufacturing floor safe. Isobutyl isocyanate ships under hazardous material marks, with UN 2480 catching the eye. Strict purity standards—usually 98% or higher—show up in purchasing requests for pharmaceutical or crop-protection uses. Quality control pulls samples and runs gas chromatography to detect residual isobutanol or potential phosgene byproducts, which can spell product failure. Labels don’t just tell you the content—they warn about the volatility, the toxicity, and the need for personal protective equipment. Every responsible operator reads them before breaking any seal.
Preparation in Practice
Making isobutyl isocyanate calls for careful chemistry. Lab procedures often start with isobutylamine and phosgene, a combination as old as industrial organic synthesis. The reaction must stay dry since water anywhere in the line can kill the yield and release clouds of corrosive gas. Refluxing under controlled conditions, the process generates hydrogen chloride as a byproduct, which must be neutralized and scrubbed swiftly. Many producers now turn to phosgene substitutes or anchored carbonyl transfer agents, especially in academic settings, but full-scale plants still run tons of traditional chemistry. Waste handling proves just as important as product isolation. Nobody wants leftover phosgene or chloride ions lurking in the effluent stream.
Chemical Reactions That Define Its Role
Isobutyl isocyanate wears its reactivity on its sleeve. It bonds with alcohols to crank out carbamates—compounds valued for their use as insecticides, herbicides, and even some drugs. Nucleophiles, from water to amines, pounce on the isocyanate group, producing ureas or emitting carbon dioxide. In pharmaceutical synthesis, these reactions mean streamlined protocols for building molecular complexity. During scale-up, reaction exotherms require careful monitoring; unplanned heat generation can lead to runaway conditions. Over the years, my colleagues discovered that timing, temperature, and reagent purity play out on the bottom line. Mismanaged reactions lead directly to lost batches and, with this chemical, real danger.
What They Call It: Synonyms and Product Names
Not every supplier calls it by the same name. Buyers have seen bottles labeled as 2-Methylpropyl isocyanate or isocyanic acid, isobutyl ester. Some catalogs just print “IBIC” in bold, but no matter how it lands on the manifest, the red diamond hazard symbol remains. In the lab, shorthand grows contagious—“iso-butyl IC” slips easily into spoken orders or notebook scribbles. All the same, those three words—Isobutyl Isocyanate—carry weight. The international community harmonizes these names, but always with a nod toward hazard classification first.
Setting the Bar: Safety and Operational Standards
Nothing ruins a lab or a plant run quicker than a safety oversight with isobutyl isocyanate. The stuff chokes and burns if it escapes into breathing air. Storage recommendations never surprise anyone with chemical experience: cool, dry, well-ventilated, away from oxidizers and incompatible organics. Gloves—nitrile, not latex—plus splash goggles and face shields set the uniform. Respirators follow standardized cartridge specifications, chosen for isocyanate and organic vapor protection. Every operator knows the emergency eyewash and shower stations. Environmental controls matter as much as personal ones; regulatory agencies fine or shut down sites that ignore release prevention and waste management protocols. Isobutyl isocyanate does not go down the drain. Accident response squads drill for vapor leaks, chemical burns, and full decontamination—mistakes here leave no room for error.
Where It Goes: Application Area
Farm fields and pharmaceutical factories share more with chemical plants than outsiders might imagine. Isobutyl isocyanate turns up in synthesizing key pesticides, especially carbamate insecticides that protect crops against pests. Over the years, pesticide bans and revised toxicity thresholds forced companies to tweak formulations, but carbamate compounds stick around in high value sectors. The pharmaceutical industry leans on isobutyl isocyanate as a building block, producing ureas and related bioactive molecules. Research chemists still reach for it when mapping out custom syntheses. Foam production might tap isocyanates, but the low-volume, high-value world of isobutyl isocyanate lands mostly in specialty applications and research reagents.
Push for Innovation: Research and Development
Research keeps shifting toward safer, greener ways to work with—or around—this chemical. Green chemistry initiatives emphasize alternatives to phosgene, driving researchers to test new synthesis routes like carbonyl diimidazole-based transfer, or biocatalysis for specialty derivatives. Analytical chemists advance detection methods—GC-MS, FTIR, and portable real-time sensors—so on-site labs monitor trace vapors and product purity in record time. The pharmaceutical sector funds studies on selectivity, yield, and waste minimization, hoping for more sustainable pipelines. Each improvement reduces health risks, process costs, and environmental impact, which speaks clearly to both workers and neighbors.
What We Know: Toxicity Research
Toxicologists have been watching isobutyl isocyanate since it first hit the market. Acute exposure means respiratory irritation, lung edema, and skin burns. Long-term studies in animals warn of organ damage with regular contact. Laboratory tests suggest potential links to carcinogenicity, although regulatory bodies hesitate to classify it without more definitive human data. Any plant with isocyanate emissions hovers under both OSHA and EPA scrutiny. Workplace monitoring logs measure parts per billion, and thresholds for immediate evacuation take up prime space in training materials. No one in their right mind would ever treat this chemical lightly.
Where It’s Headed: Future Prospects
The market for isobutyl isocyanate faces both opportunities and headwinds. On one side, evolving pesticide regulations and demand for specialty pharmaceuticals could keep it profitable. Synthetic chemists seek new ways to tamp down emissions, limit worker exposure, and build greener supply chains. Some see promise in digital monitoring—IoT sensors, automated shut-off protocols, and machine learning models that predict leaks. Academics continue to study non-toxic alternatives for the isocyanate group, chasing after equally effective but safer reagents. In the next decade, tighter regulations and community pressure may force industry to reshape protocols for handling, storage, and substitution. The story of isobutyl isocyanate echoes the crossroads facing the broader chemical industry: can innovation and responsibility keep pace with utility and risk? From what I’ve seen, nobody bets on standing still.
What are the main uses of Isobutyl Isocyanate?
Key Role in Pharmaceutical Manufacturing
Stepping into a lab, you’ll spot isobutyl isocyanate on plenty of inventory sheets. This chemical works as a building block in the synthesis of pharmaceuticals, particularly drug molecules with carbamate or urea groups. Complex medicines often get their start from reactions with isocyanates like this one, which help link up tricky chemical chains. I’ve chatted with chemists who spend weeks perfecting reactions so drug candidates form the correct structures. The right raw materials can make the process smoother, cutting wasted resources.
In practice, you’re dealing with batches of painkillers, antivirals, or even medications for neurological conditions. Chemical safety matters here—the work is often high-risk because exposure to vapors can irritate skin and eyes, so strict controls stay in place. But without isobutyl isocyanate or similar reagents, drug pipelines would stall.
Pesticide and Agrochemical Production
Many commercial weed and pest killers start from urea and carbamate derivatives. Isobutyl isocyanate enters the scene as a key input for these compounds. Agricultural chemistry can seem far removed from city life, but for anyone who’s worked planting or maintaining fields, reliable pest management products are vital.
Getting the right herbicides and insecticides into the hands of farmers makes it possible to grow more food on less land. The fine-tuned molecules built up from chemicals like isobutyl isocyanate cut crop losses by controlling weeds and pests. Overuse or mishandling poses health and environmental hazards, so factories continuously refine recipes to reduce off-target effects and increase precision.
Resins and Polymer Applications
Industrial settings often turn to specialty resins that need specific isocyanates. Isobutyl isocyanate figures in the creation of polyurethane foams, adhesives, and coatings. Walking into a furniture factory or automotive plant, you’ll notice equipment lined with protective coatings that owe their resilience to polyurethane chemistry.
These polymers must handle hot summers, cold winters, and daily abrasion. Smaller manufacturers tell me they need flexible supply chains for essentials like isobutyl isocyanate—any hiccup can halt production for days. Environmental and workplace safety stories frequently crop up—people want to see better fume management and protective protocols.
Paths to Safer, More Sustainable Chemistry
One of the most significant issues involves how facilities store and handle isocyanates, including isobutyl isocyanate. Mistakes with storage or leaks can endanger both workers and the neighborhood. Local emergency planners sometimes run drills specifically for chemical spills at plants using this substance.
Green chemistry initiatives promote new synthetic methods to cut down hazardous waste and exposure. That shift often requires business investments in safer storage systems, real-time air monitors, and comprehensive worker training that empowers teams to call out problems before they escalate.
Transparency in the supply chain, pre-planned containment, upgrading ventilation—these steps support a safer future. The chemical industry continues to look for alternatives as well. People working toward less-toxic reagents and closed-loop production make a real difference, so that making medicines and materials improves without extra risk.
What are the safety precautions for handling Isobutyl Isocyanate?
Understanding the Dangers
Isobutyl Isocyanate earns a spot on any hazardous chemical list. Workers in labs or plants can feel how strong its fumes are, even with the smallest spill. This colorless liquid doesn’t spread a warning odor until it’s already in the air. Anyone caught unaware can quickly face eye irritation, coughing, nausea, or worse, swelling of the throat from a single breath in a poorly ventilated zone. Stories from chemical plants show how fast someone can go down with acute exposure. The threat isn’t just from inhalation. Even skin contact brings a risk of burns and blistering. On busy days, even experienced hands put themselves at risk by cutting corners on gloves or masks.
Building Safer Routines
In my years working with reactive compounds, shortcuts never paid off. Using proper gear always works out best. Splash goggles, a well-fitted respirator, long nitrile gloves, and a tight lab coat matter more than speed. Forgetting just one piece is like driving without brakes on a mountain road.
No chance for open windows or cheap fans either. Fume hoods equipped with chemical filters keep vapors out of your lungs. Any project with isocyanates pushes mechanical ventilation to the top of the checklist. In the plants I visited, you hear old-timers remind new hires: “Smell nothing, see nothing, still suit up.” It’s not paranoia — it’s experience talking.
Storing and Handling the Right Way
Isobutyl Isocyanate turns volatile if left near moisture or not closed up tight. Rusted valves and plastic jugs set anyone up for leaks. Steel, glass, or sealed original containers do their job much better. Clear labels in big letters keep confusion away. Accidents sneak in during transfer or sampling. I’ve seen someone caught out by a poorly rinsed beaker, ending up with a chemical burn in under two minutes. Double-check equipment, purge lines with dry nitrogen, and work slow. Fast hands usually slow the whole team with an emergency shower.
Emergency Actions and Training
Scary stories about chemical burns get retold for a reason. Immediate washing with water offers the only real escape from exposure on the skin or eyes. Forget homemade remedies. Inhalation means everyone leaves, alarms ring, and medical staff step in. Practice makes these responses sharp. Drills at labs and plants keep panic out of the picture. Workers trained to spot leaks or container wear keep problems tiny.
Records on exposure and training prove their value during audits and emergencies. People who track near-misses spot patterns before disasters. I’ve seen teams realize a loading dock needed new ventilation only after a close call showed up in the logbook.
Improving Safety By Paying Attention
Isobutyl Isocyanate brings out the value of real-world vigilance. Small habits, like reading a label twice or swapping out old gloves, keep hands and lungs in better shape. Nobody remembers all the times they “almost forgot” something, but no one forgets the day they end up in the ER. Bringing newer, less toxic replacements to the table helps too. Until those exist, sticking to sharp routines and open conversations stops mistakes in their tracks. Safety comes down to not getting comfortable around dangerous chemicals, no matter how many times you’ve handled them in the past.
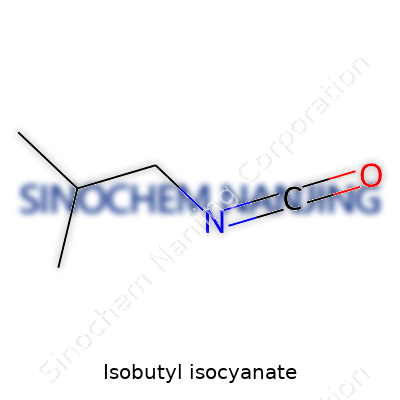
What is the chemical formula and structure of Isobutyl Isocyanate?
Chemical Identity
Isobutyl isocyanate, known in labs and industry for its reactive nature, carries the chemical formula C5H9NO. Unpacking its structure shows a straightforward chain: a central carbon backbone shaped like isobutane, with an isocyanate group (-N=C=O) clinging to the end. Lay it out on paper and you see the carbon atom linked with two methyl groups and a methylene, which lines up to connect with the reactive isocyanate moiety.
Understanding the Structure
The backbone follows a simple pattern: (CH3)2CHCH2NCO. Two methyl groups attach to a central carbon, joined by a single bond to a methylene group (CH2). This methylene throws its weight behind the isocyanate group, one nitrogen double-bonded to a carbon, which then double-bonds to an oxygen atom.
I first encountered isobutyl isocyanate flipping through lab catalogs in grad school, searching for building blocks in organic synthesis. Its structure caught my eye—four carbons in a forked chain and a highly reactive end. No fancy rings, no tangled branches, just a clean line of atoms. That simple setup belies a punchy reactivity. The isocyanate group doesn’t play around; it reacts with amines, alcohols, and water, making it both useful and hazardous.
Why Is Structure Important?
Chemical structure shapes how molecules behave. In isobutyl isocyanate, that straight-shot backbone dictates its volatility and the speed of its reactions. It’s not as bulky or slow as aromatic isocyanates. Working in synthesis, speed and reliability matter. A straightforward chain means predictable reactivity. Chemists rely on this predictability when stringing together complex molecules for drugs or polymers.
Not all isocyanates act alike. Some, like toluene diisocyanate, get the headlines for polyurethane foams and insulation. Isobutyl isocyanate sticks to specialty uses, thriving in the corners of pharmaceutical research and coatings chemistry where the need for smaller, more nimble molecules stands out. Its comparatively low boiling point means extra vigilance once the bottle opens—fumes drift quickly, so I always grabbed the heavy-duty gloves and lined up my reactions in the fume hood.
Health Risks and Handling
People working with isobutyl isocyanate learn fast—it’s not forgiving. Inhalation, skin exposure, even minor spills can bring nasty symptoms. Reactions can lead to asthma, skin rashes, or worse, so regulations keep controls tight in well-run labs. In my own work, gloves, goggles, and solid ventilation turned from recommendation to routine. Studies show chronic exposure to isocyanates brings long-term health concerns. That reality makes proper protocols non-negotiable.
Safer Alternatives and Next Steps
The chemical world keeps pushing for greener routes, not just because of regulations but because safer processes save lives and costs. Researchers now seek lower-toxicity alternatives where possible and limit the use of isocyanates to settings with solid engineering solutions. In academic labs, educational campaigns and updated training materials connect new generations of chemists with real-world risk management.
Looking ahead, the push to minimize exposure doesn't just fall to industry watchdogs—lab managers, chemists, and policy makers all have skin in the game. Tighter controls, better substitutes, and honest communication can cut down on preventable injuries. My own take: any tool with power brings responsibility, and that includes isobutyl isocyanate. Know your molecule before you uncap the flask.
How should Isobutyl Isocyanate be stored and transported?
Understanding the Risks Up Front
Isobutyl isocyanate, a chemical with a sharp odor and powerful reactivity, rewards carelessness with serious consequences. If you’ve followed chemical safety stories, you probably remember disasters that started with poor storage. Not every fire or leak earns a headline, but small lapses add up. Handling this compound safely doesn’t demand perfection, but it does require respect and solid routines.
Why Ventilation and Temperature Matter
People working around isocyanates know fumes pack a punch, irritating eyes, skin, and lungs. Even small spills or leaks can make a workspace miserable or dangerous. I once saw a poorly ventilated storage area force a rushed evacuation—no one expects to sprint out on a regular Tuesday. Good ventilation isn’t optional. Exhaust fans, open airflow, and constant air monitoring build a safer buffer between workers and errant vapors.
Heat changes the game. Warmth speeds up vapor release, ramps up pressure inside containers, and sometimes even sparks unwanted reactions. This chemical stays in its comfort zone below 30°C. That means shaded storage, climate-controlled rooms, and smart placement—far from machine exhausts, boilers, or direct sunlight. Folks ignore this until the thermometer breaks a record and a drum starts hissing.
Picking the Right Containers
Fresh isocyanate keeps best in airtight steel drums or specialized glass bottles that resist corrosion and seal tightly. Leaky caps invite moisture, which can trigger pressure build-up or even explosions. Over years in chemical shipping, I saw more problems blamed on careless sealing than anything else. Incompatible containers—such as standard plastic ones—risk chemical reactions or degrade unexpectedly. The right gear saves money, time, and, most important, keeps everyone safe.
Segregation and Labeling Aren’t Just Boxes to Check
Keeping isobutyl isocyanate away from acids, water, and amines keeps accidents rare. Mixing these neighbors can kick off violent chain reactions. Bold labels and strict policies make all the difference, especially for new warehouse staff who might not know what “danger—moisture sensitive” means at a glance. Clear hazard signs matter. I met a warehouse supervisor who switched to oversize color-coded labels after a close call. Nobody missed a drum’s real identity again.
Preparation Trumps Panic During Transportation
Bringing isocyanate on the road puts stress on even the best packaging. Specialized hazmat carriers understand the law and the risks, and invest in drivers trained for chemical emergencies. Trucks need upright containers strapped down, leakproof spill kits, and a direct line to emergency response if the worst happens. My experience says a little more spent on a certified transporter costs less than the fallout from a highway spill—think evacuations, lawsuits, and environmental fines.
Proactive Mindset Benefits Everyone
Storing and moving isobutyl isocyanate doesn’t just check a regulatory box. It builds habits and teamwork. Regular inspections, up-to-date safety data sheets, and honest communication train people to spot problems early. Real trust grows in companies that put worker health ahead of shipping speed. A workplace that values vigilance doesn’t just avoid fines—it keeps families, communities, and the environment safe from accidents that shouldn’t happen in the first place.
Solid Steps Go a Long Way
Simple principles keep this chemical where it belongs: ventilate well, keep things cool and dry, choose trusted containers, use bold signs, and move with professionals. Isobutyl isocyanate won’t give second chances, but it doesn’t need to ruin anyone’s day if treated right. My experience says that’s a lesson worth repeating, in warehouses large and small.
What are the potential health hazards and first aid measures for Isobutyl Isocyanate exposure?
Recognizing the Risks
Breathing in isobutyl isocyanate feels nothing like a harmless whiff of everyday chemicals. It goes straight for your eyes, skin, and lungs. The first sign usually hits the nose and throat—a burning feeling that sticks. Exposed skin reacts almost right away, turning red, itching, or swelling up with blisters. I've seen lab workers pull off gloves and find angry red marks that take days to ease. Splash some in your eye, expect watering, pain, and in rough cases, possible vision trouble. If accidently inhaled during a leak, coughing and chest tightness begin within minutes, and that’s not hyperbole.
Why Exposure Isn’t Just an Annoyance
This chemical does more than irritate. Breathing in just a bit too much can spark asthma-like attacks, putting even healthy lungs under pressure. Doctors say repeated exposure turns this into a long-term risk. You won’t always see isobutyl isocyanate sneaking out, either. It doesn’t have a powerful odor—you can be in danger before your nose picks up a thing.
After reviewing safety reports and speaking with emergency responders in chemical plants, the overwhelming theme is this: Even a fast discovery might not keep someone out of harm’s way for long. Workers exposed to a strong cloud can spiral into trouble—wheezing, gasping for air, or feeling confused—within minutes.
Immediate First Aid: Acting Fast Saves Lives
Nobody should stand around debating what to do after a spill. If this chemical touches skin, get that patch under running water for at least 15 minutes, then strip any contaminated clothes without pulling them over the head. Eyes need care, too—flush with water, and keep those lids open. Emergency rooms expect phone calls about splashes that go untreated for too long, and it can make all the difference.
Inhaling vapors or dust from isobutyl isocyanate should never be taken lightly. Step outside or move to fresh air immediately. Sometimes, oxygen or breathing treatments become necessary, especially in people who start coughing up pink or frothy sputum, a common sign of chemical lung irritation. No one should try to induce vomiting if any gets swallowed—instead, rinse the mouth, and get medical help fast.
Solutions Start Before Accidents Happen
Keeping everyone safe means wearing gloves and goggles, using proper ventilation, and planning escape routes long before trouble shows up. Regular safety drills build habits, not just paperwork. It’s smart to stock eyewash stations and emergency showers where this chemical gets used. I’ve seen close calls where simple, well-marked equipment let someone act on instinct, limiting the severity of symptoms.
Employers and chemical handlers need honest conversations about risks and real-time monitoring. Education from voices with on-the-ground experience, not just slide decks, resonates with workers and sticks better than warning labels. There’s no substitute for team members who feel empowered to speak up about leaks, spills, or suspicious symptoms in their coworkers.
Final Thoughts on Staying Powerfully Prepared
The hazards linked to isobutyl isocyanate carry lasting effects, but solid training and fast first aid matter most. Communities benefit from a workforce that trusts not just protocols but one another. Straight talk, clear information, and genuine respect for the dangers at hand give everyone a better shot at going home healthy.
| Names | |
| Preferred IUPAC name | 2-methylpropyl isocyanate |
| Other names |
2-Isocyanatopropane 2-Methylpropyl isocyanate Isocyanic acid, isobutyl ester |
| Pronunciation | /ˌaɪ.səˈbjuː.tɪl ˌaɪ.səʊ.saɪˈə.neɪt/ |
| Identifiers | |
| CAS Number | 542-28-9 |
| Beilstein Reference | 873823 |
| ChEBI | CHEBI:141441 |
| ChEMBL | CHEMBL4298817 |
| ChemSpider | 64144 |
| DrugBank | DB08555 |
| ECHA InfoCard | 100.013.057 |
| EC Number | Isobutyl Isocyanate" EC Number: "208-750-2 |
| Gmelin Reference | Gmelin Reference: 9243 |
| KEGG | C19282 |
| MeSH | D016976 |
| PubChem CID | 8306 |
| RTECS number | NT9865000 |
| UNII | 8Y5ZT4GG8H |
| UN number | UN2480 |
| Properties | |
| Chemical formula | C5H9NO |
| Molar mass | 115.15 g/mol |
| Appearance | Colorless to yellowish liquid |
| Odor | pungent |
| Density | 0.864 g/mL at 25 °C (lit.) |
| Solubility in water | Reacts with water |
| log P | 1.97 |
| Vapor pressure | 10 mmHg (20°C) |
| Acidity (pKa) | 16.57 |
| Basicity (pKb) | 11.57 |
| Magnetic susceptibility (χ) | -6.13×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.393 |
| Viscosity | 1.01 mPa·s (20 °C) |
| Dipole moment | 1.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 277.8 J⋅mol⁻¹⋅K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -134.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3936.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS02,GHS06 |
| Signal word | Danger |
| Hazard statements | H226, H301, H311, H315, H317, H319, H330, H334, H335, H410 |
| Precautionary statements | P210, P260, P273, P280, P284, P301+P310, P304+P340, P305+P351+P338, P308+P311, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-3-2-W |
| Flash point | 34 °C |
| Autoignition temperature | 441 °C |
| Explosive limits | Explosive limits: 1.1–8.4% (in air) |
| Lethal dose or concentration | LD50 oral rat 1500 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 150 mg/kg |
| NIOSH | NIOSH: NI0175000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Isobutyl Isocyanate: "0.005 ppm (0.043 mg/m³) as an 8-hour TWA |
| REL (Recommended) | REL (Recommended Exposure Limit) of Isobutyl Isocyanate: "0.1 ppm (0.4 mg/m3) TWA |
| IDLH (Immediate danger) | IDLH: 50 ppm |
| Related compounds | |
| Related compounds |
n-Butyl isocyanate tert-Butyl isocyanate Methyl isocyanate Ethyl isocyanate Phenyl isocyanate |

