Isobutyl Formate: An Editorial Look at a Quietly Essential Chemical
Tracing the Story of Isobutyl Formate
My first exposure to isobutyl formate came in a perfumery lab, with its fruity, almost pear-like aroma wafting up as soon as the vial uncapped. Most folks rarely hear about it outside chemistry circles, but its story winds back over a century. Researchers tinkering with esters realized early on that isobutyl formate brought a certain crisp, sweet note impossible to replicate with simpler mixtures. Even as the catalogue of organic chemistry swelled, this compound caught the interest of those chasing new flavors and solvents—reasons tied directly to its unique structure. Chemistry textbooks usually skirt past its quiet role during the expansion of ester production in the last century, but looking beneath the radar, it keeps turning up whenever someone explores how chemical engineering shapes daily life.
Product Overview in Practical Terms
Isobutyl formate is an organic ester from isobutanol and formic acid. Not much to look at, just a clear, colorless liquid with a scent that can trick someone into thinking they're handling a basket of ripe pears or apples. Its simple profile belies a versatility that makes it a go-to for synthetic flavors and specialty solvents. You’ll spot it in industry as a solution for tough dissolving jobs and as a middle note in natural-smelling perfumes. Hobbyists working on essential oil blends and professionals in coatings share an appreciation for how a little isobutyl formate can make big changes in the end result.
Physical and Chemical Properties People Notice
Chemists pay attention to boiling point, volatility, flammability, and solubility, as these turn theory into practical know-how. Isobutyl formate boils around 107°C, lighter than water and floats a low density just above 0.86 g/cm³. A whiff reveals a strong aroma, hinting at its volatile nature—careless handling fills a room fast. It dissolves easily in organic solvents, resists mixing with water, and lights up quickly if exposed to flame. In a lab with tight temperature control or an industrial setup handling multiple esters, these features come together to make it reliable but demanding of respect. More than one technician has learned to keep their workspace aired out, as fumes stick around and invade the senses.
Technical Specifications and Labeling—A Closer Look
Walking through a chemical store, I look closely at the labels on isobutyl formate containers. Standard practice calls for both CAS numbers and clear hazard pictograms—flammability can’t be taken lightly. Reputable suppliers specify purity, with the most practical batches reaching well over 98%. Moisture content becomes a sticking point for users in coatings or pharmaceuticals, as trace water can ruin an application. So, suppliers run inline analytics and print specs plainly, avoiding misunderstandings and helping users comply with safety codes. Users in regulated environments look for UN numbers relating to flammable liquids, which helps everyone coordinate handling and emergency response.
How Isobutyl Formate Comes Together
The classic route to isobutyl formate runs like a chemistry classroom staple. Mix isobutanol and formic acid, tip in an acid catalyst—often sulfuric acid—and heat gently. The reaction spits out water as a byproduct, which gets separated off, and the ester takes shape. Purification usually means washing, drying with salts, and distilling. Variations on this method keep surfacing, from greener, enzyme-based approaches to continuous-flow reactors aimed at saving time and energy. The basic method holds up, though, offering factories a way to churn out enough for both small shops and full-scale industrial users. Any lab serious about scalable ester synthesis keeps this approach on the tool belt, balancing efficiency against cost and purity.
Chemical Reactions and How Chemists Manipulate It
For all its simplicity, isobutyl formate stands up to a surprising range of chemical modifications. Its ester bond can break back down to isobutanol and formic acid under acidic or basic conditions—a trick used in analytical chemistry and industrial cleanup. In the presence of strong nucleophiles, it can undergo transesterification, forming other esters better suited to specific needs. Adding selective hydrogenation cracks the molecule open, giving routes to specialty alcohols or acids. Labs keep a close watch on how this compound interacts with stronger bases and oxidizers, steering clear of unintended exotherms or degradation. Knowledge of its reaction profile forms the backbone of safe, useful application across several industries.
Synonyms and Naming Confusion
Talk to different professionals and you quickly learn isobutyl formate wears several names—2-methylpropyl formate being most common in scientific registers. Among flavor chemists, it’s sometimes called “pear essence” for obvious reasons, though that can blur distinctions between natural oils and synthetically produced esters. Looking at ingredient lines on food additives or fragrance bases, trade designations sometimes obscure the simple underlying molecule. This tangled web of synonyms challenges those new to chemical sourcing or regulatory compliance—accuracy in naming prevents confusion and avoids costly mistakes.
Managing Safety and Working Standards
Standards for operations with isobutyl formate grow from experience. Anyone who’s spent a day in a poorly ventilated lab knows headaches and nausea can come fast if fumes build up. The compound’s flammability puts special pressure on storage, pushing firms to use explosion-proof equipment and anti-static measures in transfer systems. Handling calls for gloves and goggles, minimizing skin contact and eye exposure. Emergency showers and spill kits settle as background requirements, not add-ons. Experienced teams build thorough safety routines into daily work, not just to follow rules but to protect staff and keep accident rates low. Regulatory agencies shape many practices, with familiar GHS labeling and strict limits on worker exposure. Labs and factories joining the push for “green chemistry” work to keep emissions down and replace older, hazardous processing aids.
Where Isobutyl Formate Finds Its Niche
Ask someone in the world of artificial flavors why they keep coming back to isobutyl formate, and the answer is always scent and taste. Few other molecules match its balance in fruity notes for candies or sweet syrups. Solvent users appreciate its power to dissolve tough resins and lacquers, playing a part in specialty coatings and inks. The perfume industry leans on its volatility and odor profile for blending top and middle notes, helping products stand out in a crowded field. Research into fine chemicals and pharmaceutical intermediates keeps it on the shelf for those probing new reactions or developing greener synthetic pathways. Each application carries unique needs, but all users share respect for its punch and adaptability—which keep it relevant even as newer substances arrive on the market.
Continuous Research and Small Adaptations
The research community keeps circling back to isobutyl formate, looking for safer, quicker, or less wasteful ways to make it. Academic labs explore alternative catalysts, hoping to cut energy consumption and scale up with fewer emissions. For those on the applied side, tweaks to purification and transport technologies often pull the biggest returns, reducing both costs and risks. A growing green chemistry movement searches for raw materials drawn from renewables—a push that could transform how all formates enter the market. These advances rarely roar in with fanfare but accumulate steadily, quietly shifting best practices and inspiring better safeguards.
What We Know about Toxicity and Health
Toxicity research tells a story echoed by other low-molecular-weight esters: acute effects often limited to irritation of eyes, skin, or lungs, but real risks build from repeated exposure. Certain animal studies note impacts on respiratory rates and minor central nervous system depression at high doses; regulatory bodies set conservative thresholds for occupational handling to account for this. There’s ongoing interest in chronic toxicity, with recent focus on metabolic breakdown and potential trace contaminants that might sneak in from poorly purified batches. Compared with older, riskier solvents, isobutyl formate often fares better but that doesn’t mean it’s harmless—the best-performing workplaces build sharp physical safeguards and exposure protocols into daily life, not just relying on personal protective equipment.
Future Directions for Isobutyl Formate
Looking to the future, isobutyl formate stands on the threshold of several potential shifts. Process intensification and digitized quality control both promise tighter batch-to-batch consistency—key for food, fragrance, and industrial users. If the push for biobased chemicals persists, expect a migration toward feedstocks drawn from fermentation or even engineered microbes, cutting overall lifecycle carbon. Safety improvements won’t come as optional extras—users demand safer production lines, automated air monitoring, and real-time spill detection as routine protections. New downstream chemistries might draw out novel uses, especially as researchers hunt for solvents that dodge the harsh environmental impact of traditional formulations. Whether someone works in an industrial plant or a test kitchen, these small advances will change how they experience this quiet-but-potent ester, bringing new potential—well-grounded in science—to everyday formulations.
What is Isobutyl Formate used for?
What Is Isobutyl Formate?
Isobutyl formate sounds like a niche chemical, but anyone paying attention to flavors and scents has probably encountered its handiwork. When I walk through the produce aisle and catch the sharp-sweet scent of a pear, some of that comes from natural esters. Isobutyl formate puts the “pear” in so many products, both food and fragrance, despite not being squeezed from fruit itself.
Role in Flavor and Fragrance
The food industry reaches for isobutyl formate thanks to its ability to create that sharp, fresh, and somewhere-between-pear-and-raspberry flavor. This compound turns up in everything from candies to chewing gums. Bakers and confectioners want flavors that pop and linger; this ester pulls its weight by brightening up artificial fruit flavors at low concentrations. I remember reading a study from the Flavour and Extract Manufacturers Association that highlighted how small doses of isobutyl formate can give a noticeable, pleasant fruitiness. It’s a clear example of a molecule that punches above its weight.
Perfumers look for molecules that stick around, hold up against evaporation, and play well with others. Isobutyl formate meets these criteria. It fills out the edges in floral and orchard fragrances, blending the sweetness without overpowering the senses. You’ll find it listed on perfume ingredient panels, sometimes tucked away with a host of other fruit esters, each bringing their own character to the blend.
Manufacturing and Industry Use
Industrial applications give this ester another stage to perform. Solvents play a quiet but essential role in cleaning, degreasing, and extracting. Isobutyl formate acts as a fast-evaporating solvent. Factories use it for its quick-drying properties, whether it’s cleaning electronics or prepping metal surfaces. Its use is regulated, and workers follow safety protocols to avoid inhaling or directly contacting concentrated forms, as studies by the National Institute for Occupational Safety and Health show that exposure to esters at high levels can irritate eyes and lungs.
Paints, lacquers, and coatings also benefit from the solvent qualities of isobutyl formate. Smooth application and fast drying means less downtime in production lines, which appeals to manufacturers aiming to cut costs and increase output. From a practical standpoint, I’ve seen shop floors where switching from slower-drying solvents to options like isobutyl formate led to faster turnaround times.
Safety, Regulation, and Room for Improvement
Safety guidelines matter, especially when a chemical finds its way into both foodstuffs and machinery. Regulatory agencies such as the U.S. FDA review these substances, approving isobutyl formate for flavor use at specific concentrations. There’s a line between beneficial and harmful; scientists and health officials work to keep products on the right side of that line. In manufacturing, strong ventilation systems and protective gear make a difference in keeping factory workers healthy.
Research keeps moving. People are exploring greener synthetic routes and bio-based methods. Sustainable production offers a solution: reduce waste, minimize emissions, keep the chemical as effective as ever. Community health and industry growth move best together, not at each other's expense.
Final Thoughts
Isobutyl formate sits at the crossroads of sensorial pleasure and industrial function. It’s small but significant, a reminder that chemicals aren’t all just numbers and warnings—sometimes, they’re the secret behind something as simple as the taste of a fruit candy or the finish on a new metal part.
What are the safety precautions when handling Isobutyl Formate?
Learning From the Lab: Why Safety Steps Matter
Anyone who’s ever worked in a lab knows a sweet smell doesn’t always mean safety. Isobutyl formate smells like fruit and can make you think of flavoring for candy or soft drinks. Behind that scent sits a chemical with serious risks. I remember handling solvents with similar scents back in college. We trusted gloves and hoods, not our noses, and for good reason. There’s no shortcut for safety—dependable habits save skin, lungs, and careers every day.
What Isobutyl Formate Can Do
This stuff evaporates quickly. Just opening a bottle creates a low, invisible cloud in the air. Vapors irritate the nose, throat, and lungs right out of the gate. People sometimes develop headaches or dizziness if they spend too much time near an open container. Part of the risk comes from the way this chemical targets your central nervous system as it builds up. On top of that, any splash on the skin leads to dryness or, with enough contact, red, irritated patches that take days to heal. Eyes take the biggest hit. Even a small drop stings like crazy and may cause long-lasting problems.
It doesn’t take much effort for things to go sideways with a fast-moving solvent like this. A clumsy pour spreads fumes where you least expect them. An unplanned reaction with oxidizers or acids throws sparks. Since Isobutyl formate is flammable, sparks or static electricity can turn a spill into a fire hazard. No one wants to scramble for an extinguisher in a cloud of fumes. My years handling different solvents always taught me: stay ready, never assume you can wing it.
Real-World Safety: Gear and Habits that Work
For personal protection, I never cut corners. I’ll always reach for nitrile gloves and splash-resistant goggles. Lab coats or aprons with sleeves block splashes that would soak right through ordinary clothes. I’ve seen people rely on latex gloves and regret it—solvents tear right through latex within minutes. Ventilation means everything. A chemical fume hood pulls heavy vapors up and away; a fan in a window barely makes a dent.
Every bottle of this solvent should have its cap screwed on tight between uses—no exceptions. Even quick transfers call for smaller squeeze bottles designed for chemical use, not just random glassware. In storage, keep the containers far from any heat source, flame, or spot where static might build up. Fire extinguishers rated for chemical fires earn their place in any area where solvents show up.
What Solid Safety Means on the Job
Proper training beats any warning label. A team that respects a chemical’s risks and watches out for each other rarely ends up in trouble. Knowing what to do if a spill hits your skin matters more than memorizing hazard codes. I've spent enough time mentoring interns to see the difference good habits make: rinsing right away after a splash, heading to fresh air when vapors creep up, using spill kits instead of paper towels.
The real fix comes down to respect. Isobutyl formate isn’t evil—handled with care, it’s just a tool. Shortcuts, overconfidence, or shrugging off small accidents can turn an afternoon in the lab into a medical report. We owe it to each other—whether in research or in industry—to keep our hands, eyes, and lungs in the clear, every shift, every bottle, every time.
What is the chemical formula and structure of Isobutyl Formate?
Getting to Know Isobutyl Formate
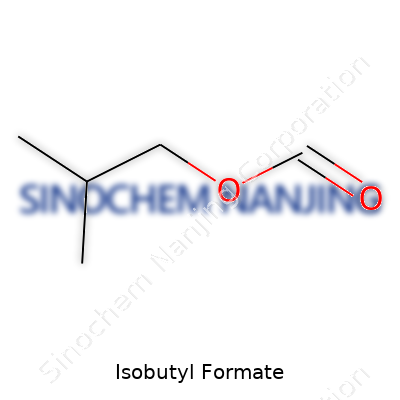
Step into any flavor laboratory or perfume workshop and you might catch a whiff of something fruity that masks its chemical origins. That distinct aroma owes a lot to esters, and one sweet contributor goes by the name of isobutyl formate. In plain chemistry, its formula reads C5H10O2. The structure lines up as HCOOCH2CH(CH3)2, laying out the formate group attached to an isobutyl backbone. You wouldn’t spot it on your grocery shelf, but its presence shapes everyday experiences, from food scents to solvents.
Why Structure Matters
Formulas only tell part of the story. The structure, with its branched isobutyl group and the simple formate, makes all the difference. This specific arrangement sets isobutyl formate apart from similar esters, impacting how it smells, reacts, and persists in the environment. Having tinkered with flavor chemicals myself, it’s clear that branching matters. A straight-chain butyl formate won’t remind you of pears in the same way. The branched chain tweaks its volatility and shifts its scent towards crisp, green fruit.
A Role in Fragrance and Flavor
The food industry relies on aroma molecules to shape experiences. Isobutyl formate appears in realistic apple, pear, and raspberry flavors. While it comes from synthetic recipes, nature offers it up in traces too. The perfume world puts it to use for brightening the top notes in fruit-heavy compositions. Chemists take this for granted, but for artisans, the right ester can make or break a formula.
Assessing Safety and Impact
Safety stands as a core concern. Food chemists and regulatory bodies like the FDA and EFSA have set their eyes on isobutyl formate for decades. Decades of review show small amounts in flavors pose little risk. Larger exposures—think factory handling, not taste tests—call for gloves, goggles, and respect for its volatility. It evaporates quickly and carries a flammable tag. Knowing proper storage and handling remains key for safer labs and manufacturing spaces.
Environmental Considerations
That quick evaporation means atmospheric release runs high. The ester breaks down in the air before long, mostly thanks to natural sunlight and ozone. Still, a push for greener chemicals now asks all producers to mind their waste streams and look for less impactful solvent systems where possible. Chemists searching for more sustainable solvents often look for those that don’t compromise on performance or safety.
Bringing Science and Industry Together
Aromatic chemicals like isobutyl formate show real chemistry at work in our daily lives. Having worked closely with food technologists, the meticulous blend of tradition and science gets impressive results while meeting regulations. Education matters in this space. Teams that understand both raw chemistry and safety regulations deliver the best results for consumers, making sure flavors enchant without crossing health boundaries.
Responsible Use Points to Solutions
Anyone working in the sector can see that knowledge sharing works better than rules alone. Connecting researchers, regulators, and manufacturers leads to safer practices and pushes for eco-friendlier approaches. Safer alternatives, waste minimization, and smart chemical engineering all form part of the toolkit for moving the industry forward without losing sight of quality and safety.
How should Isobutyl Formate be stored?
A Look at the Practical Side of Chemical Storage
Isobutyl formate plays a useful role in flavors and perfumes, yet most folks don’t hear about what happens behind the scenes. Anyone who spends time with chemicals understands the importance of keeping them under control. Sure, safety data will point out the fire risk, or how breathing too much vapor could quickly irritate your throat, but good storage goes deeper than just following the rules. I’ve seen bad storage ruin product, set off alarms, and interrupt business. Simple steps at the start save headaches later.
Why Isobutyl Formate Needs Special Attention
This compound gives off a strong, fruity smell and reacts quickly with air. It evaporates fast, and that vapor catches fire with just a spark. I once watched a worker grip a bottle without realizing a tiny leak could slowly fill an enclosed cabinet with enough fumes to turn a flipped light switch into a disaster. Storing it away from ignition sources is not just a lab rule—it should be second nature. An open flame, even static from synthetic clothing, can trigger trouble.
Heat speeds up evaporation and raises pressure inside the container, which means hot storage rooms risk swelling, leaks, or—worse yet—explosions. From what I’ve seen, keeping it in a cool, ventilated place isn’t just about ticking boxes; it brings peace of mind when you walk into the workspace, knowing the air won’t sting your nose or put everyone on edge.
The Right Containers Matter
You don’t need a PhD to know glass keeps out more air than old plastic jugs. The acids in this compound eat away at some plastics, but glass or sealed metal cans stand up against the vapors. Tightly sealing lids stops air from sneaking in and prevents leaks. Some will say that new containers cost too much, but the price of replacing lost material or cleaning up spills always turns out higher.
Labeling and Organization: Not Just Busywork
I’ve watched teams shuffle through cabinets stuffed shoulder-to-shoulder with bottles, not noticing labels peeling or missing. One quick mix-up and you can’t trust what’s inside, risking both safety and wasted product. Clear, dated labels with hazard warnings keep everyone sharp—no one likes guessing what’s in a bottle after the print fades. Simple color coding helps prevent mix-ups, especially during busy shifts.
Separate from Food and Water
This sounds obvious, but in shared storage spaces, bottles end up in the oddest places. A clear division between chemicals and anything meant for consumption—coffee, snacks, even cleaning gear—makes a real difference. I remember a misplaced bottle finding its way to a breakroom fridge; quick thinking prevented a health scare. A single shelf or locked cabinet designed only for chemicals solves this problem easily.
Guidelines: Written and Followed
Rules don’t work unless people understand why they’re there. It’s easy to write a policy, tougher to build a work culture where those guidelines stick. Regular training, honest communication, and easy-to-follow checklists turn good intentions into real habits. It helps if supervisors and workers talk openly about near-misses and slip-ups. Mistakes become lessons, not just paperwork.
Reduce Risks: Make It Routine
Treating chemical storage as a regular part of the work—not a box-checking chore—protects health, product, and peace of mind. Simple habits like routine checks, real labeling, solid containers, and firm separation from food prevent most problems before they start. Storing isobutyl formate safely doesn’t call for complex systems or fancy gear, just steady care and real respect for the risks.
What are the physical and chemical properties of Isobutyl Formate?
Down-to-Earth Look at Isobutyl Formate
Isobutyl formate tends to show up wherever folks work with flavors, fragrances, or solvents. With a molecular formula of C5H10O2, this clear liquid has a strong, somewhat fruity odor, not so different from rum or certain kinds of pear drops. Take a sniff in a lab setting and the sharp scent sticks with you longer than other formates. Some call it pleasant, others say it lingers too much, but that’s the character you get from this ester. Properties like these matter because they shape how companies handle, store, and use the chemical.
Key Physical Characteristics
This liquid boils at around 107°C (225°F), which sets it apart from some heavier esters. It evaporates faster than water but not so fast you can’t keep it in a sealed flask at room temperature. It comes in at a density of about 0.87 g/cm³, pretty similar to other small organic solvents. Pour a bit onto your skin (which I don’t recommend), and you’ll notice it evaporating quickly, leaving a cooling sensation. This volatility draws both attention and caution, since spills vaporize into the air fast and can catch sensitive noses unprepared.
Another thing worth noting: it hardly dissolves in water. You might see it float in a beaker as a distinct layer, but mix it into ethanol or diethyl ether and it blends well. That solubility profile determines how companies use isobutyl formate to extract flavors or craft perfume bases. It also decides how the chemical behaves during a spill—water won’t do much, so folks rely on absorbent materials or proper ventilation instead. At room temperature, watch for flammability. Vapors catch fire boldly above 22°C (72°F), right in the range of a spring day, so labels with bold “flammable” warnings aren’t just bureaucracy.
Chemical Behavior and Its Usefulness
Esters like isobutyl formate show a predictable set of chemical reactions. In the presence of acids or bases, the molecule breaks down to formic acid and isobutanol, both of which bring their own hazards. I’ve seen students overheat the mixture and kick off this reaction prematurely, creating fumes and chemical messes. Storage away from strong acids, strong bases, and oxidizers becomes routine in any chem lab or production facility. People rely on isobutyl formate for its ability to dissolve oils and resins, especially in applications involving varnishes or specialty cleaners. Its distinctive breakdown into harmless components—after controlled use—reduces the potential for lasting pollution, as long as disposal follows environmental standards.
Risks, Precautions, and Improvements
The quick-burning vapor makes fire suppression a top priority anywhere it’s handled. Good ventilation, spark-free tools, and regular safety reviews keep workplace incidents rare. Folks sensitive to chemicals need to know that lingering smell signals more than just a flavor ingredient—it alerts to exposure risk, so eye protection and gloves become non-negotiable. For large-scale industry, replacing isobutyl formate with safer alternatives or designing systems to capture and reuse vapors will cut down on emissions and waste. Robust training and spill response tools (not just warning labels) keep operations smooth and people safe. Innovation in closed-loop production and solvent recovery can drive the next leaps in both safety and sustainability for those relying on this quirky, useful ester.
| Names | |
| Preferred IUPAC name | 3-methylbutyl methanoate |
| Other names |
Formic acid isobutyl ester Isobutyl methanoate Isobutylformic acid Isobutanol formate |
| Pronunciation | /ˌaɪsəˈbjuːtɪl ˈfɔːrmeɪt/ |
| Identifiers | |
| CAS Number | 547-63-7 |
| 3D model (JSmol) | `Isobutyl Formate` JSmol 3D model (as string): ``` Isobutyl Formate COC(=O)CC(C)C ``` This is the SMILES string that JSmol uses to render the 3D model of Isobutyl Formate. |
| Beilstein Reference | 1741597 |
| ChEBI | CHEBI:87144 |
| ChEMBL | CHEMBL2050799 |
| ChemSpider | 69401 |
| DrugBank | DB14096 |
| ECHA InfoCard | 03b527102f7d-46b7-4d50-abe2-7e03be0994ea |
| EC Number | 203-695-2 |
| Gmelin Reference | 8225 |
| KEGG | C19600 |
| MeSH | D016624 |
| PubChem CID | 8029 |
| RTECS number | NT8225000 |
| UNII | 7B1QPC0089 |
| UN number | UN2287 |
| CompTox Dashboard (EPA) | DTXSID0021691 |
| Properties | |
| Chemical formula | C5H10O2 |
| Molar mass | **88.11 g/mol** |
| Appearance | Colourless liquid with a pleasant odour |
| Odor | fruity |
| Density | 0.861 g/mL at 25 °C |
| Solubility in water | 6.3 g/100 mL (25 °C) |
| log P | 1.48 |
| Vapor pressure | 11.7 mmHg (20 °C) |
| Acidity (pKa) | 3.75 |
| Basicity (pKb) | 13.17 |
| Magnetic susceptibility (χ) | -7.18×10⁻⁶ |
| Refractive index (nD) | 1.3680 |
| Viscosity | 0.543 mPa·s (at 25 °C) |
| Dipole moment | 1.73 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 269.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -460.15 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2907.7 kJ/mol |
| Pharmacology | |
| ATC code | '' |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H319, H336 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P271, P280, P303+P361+P353, P304+P340, P312, P305+P351+P338, P337+P313, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 1-3-2-Release |
| Flash point | 15 °C (closed cup) |
| Autoignition temperature | 415 °C |
| Explosive limits | 1.4–10.0% (in air) |
| Lethal dose or concentration | LDLo (oral, rat): 3,200 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 3,100 mg/kg |
| NIOSH | NA8500000 |
| PEL (Permissible) | PEL = "Isobutyl Formate: 100 ppm (425 mg/m3) as an 8-hour TWA (OSHA) |
| REL (Recommended) | 50 ppm |
| IDLH (Immediate danger) | IDLH: 1,600 ppm |
| Related compounds | |
| Related compounds |
Formic acid Isobutanol Methyl formate Ethyl formate Propyl formate |

