Isobutyl Chloroformate: More Than Just a Reagent
A Glimpse at the Journey
Chemistry as a field has always fascinated me for its blend of precision and creativity, and isobutyl chloroformate strikes me as one of those silent workhorses shaping both industry and research. Looking back, this compound didn’t just spring up overnight; its roots trace deep into the development of organic chemistry. During the 20th century, labs needed more reliable tools for activating carboxylic acids, and this prompted the search for better reactive intermediates. Isobutyl chloroformate carved out a niche, making itself indispensable wherever chemists looked for efficient coupling reactions. Over the decades, it gradually replaced rougher, less manageable reagents, slipping into synthetic protocols and becoming a regular guest in academic and industrial labs alike.
Understanding the Compound
Imagine handling a clear, colorless liquid that oozes a strong odor, easily identifiable to those with even a little time spent at a fume hood. That’s isobutyl chloroformate for you, with its molecular structure combining the isobutyl group with a reactive chloroformate ester. It boils at a modest temperature, making it somewhat volatile, but solvates easily in common organic solvents. Water spells trouble for it, provoking hydrolysis and a sharp release of hydrochloric acid. It’s not explosive, but it keeps its chemistry reined in until provoked by nucleophiles willing to break its fragile carbon-chlorine bond.
How It Enters Chemistry's Toolbox
The stories I remember hearing from veteran organic chemists involve many an early morning prepping isobutyl chloroformate to create acid chlorides or to step up peptide synthesis without raising too many side reactions. In standard labeling, the caution phrases practically roll off the tongue after a few years—flammable, harmful upon inhalation, and always stored in corrosion-resistant containers under a fume hood. I’d underline this: anyone who’s handled it knows the unmistakable tickle in the nose that signals its presence. Reading a label or a safety sheet, you don't just see regulatory boxes checked, you recognize the collective wisdom of chemists who learned a thing or two about what this compound can do, both good and bad.
Making the Stuff
Preparing isobutyl chloroformate isn’t alchemy, but it does demand respect. The go-to method involves reacting phosgene—a notorious compound itself—with isobutanol. This process doesn’t suffer fools; personal experience tells me that phosgene, even in small leaks, can scare even the bravest chemists. The outcome is straightforward, but only when you keep your ventilation systems humming and stay alert for HCl gas byproducts. Production always leaves bystanders with an appreciation for proper training and tight operational discipline.
Getting Down to Chemistry
Take isobutyl chloroformate into the lab, and you see a world of possibilities open up. One of its headline tricks is activating carboxylic acids so that they jump more willingly into reactions with amines, making amides swiftly and with less fuss. It takes over from tougher agents in peptide synthesis because it helps suppress unwanted side products, which matters a lot both for research and making actual pharmaceuticals. One thing I noticed? The reaction with nucleophiles may be quick, but so is the potential for accident; a splash or whiff, and you remember the importance of planning and reliable gear.
What’s in a Name?
Walk through any lab and you’ll hear isobutyl chloroformate traded by many synonyms: isobutyl carbonochloridate, carbonochloridic acid isobutyl ester, and even IBICF. These names reflect different corners of chemical literature and commercial references, but they all point to the same reactive backbone—the telltale chloroformate moiety ready to make a mark across reactions. This collection of names sometimes causes confusion for students, so it's always worth double-checking your bottle before a big experiment.
Watching for Hazards
Years of handling chemicals taught me not to be casual about safety. Isobutyl chloroformate doesn’t demand the panic reserved for strong acids or pressured gases, but it does command respect. Inhalation irritates lungs and eyes, even at modest concentrations. Splashing on skin feels worse than a sunburn. Working in a fume hood and using gloves aren’t academic rules; they're daily habits. Most regulations around storage and transport mandate tight seals, cool environments, and plans to neutralize accidental releases. Laxity breeds regret—no exceptions.
The Reach of Applications
This chemical doesn’t just sit idle on storage shelves. In the pharma industry, it plays a part in getting bioactive molecules ready for action, stepping in during the assembly of peptide drugs and specialty esters. Labs racing to build new organic compounds, or trying to modify biomolecules for research, lean on its reliable reactivity. Even analytical labs use it to prepare samples for detection, especially in mass spectrometry and chromatography workflows. Its touch brings chemical groups together that resist union by gentler means, which accelerates the pace of discovery and development.
Chasing the Next Breakthrough
Research keeps evolving, and the story of isobutyl chloroformate speaks to that drive. Projects aimed at making synthesis cleaner, safer, and more efficient revisit this compound’s reactivity, often seeking gentler reaction conditions or looking for new ways to activate compounds without creating a mess of byproducts. A few ambitious researchers examine greener solvent systems or substitute reagents as a response to environmental and regulatory pressures. These efforts reflect growing concerns in the community regarding both sustainability and health. Developing alternatives to phosgene-based processes draws its share of funding, reflecting worries over toxic intermediates.
The Shadow of Toxicity
No serious discussion of isobutyl chloroformate brushes aside the question of toxicity. Researchers have run their animals and cell lines through exposure studies, and there’s no ignoring the bite in acute exposures. Its vapor, once inhaled, hits the respiratory system hard—red eyes, coughing, and even chest pain aren’t rare if you don’t watch your ventilation. Chronic studies remain less developed, but most occupational health agencies err on the side of caution, setting strict exposure limits. Decades of practice show that training, detection systems, and a strong culture of safety make all the difference. Every near-miss with harmful vapors offers a reminder that safety isn’t optional, but life-critical.
Looking Down the Road
Industries and labs will likely rethink how they use isobutyl chloroformate as new safety, environmental, and regulatory demands take shape. Green chemistry is no longer just a buzzword; it’s a necessity. Synthetic chemists look at each new project with an eye to reducing risk and environmental load, which may mean smarter uses of this compound alongside developments of less toxic alternatives. I expect that both process engineers and academic researchers will keep searching for ways to limit byproducts, manage effluents, and possibly circumnavigate the hazards bound to old-school synthesis. The story of isobutyl chloroformate remains open, pushed forward by pragmatic science and the persistent need to simplify difficult chemical unions without sacrificing safety or the environment. From my experience, every chapter written by hands-on chemists matters as much as anything in the academic literature.
What is Isobutyl Chloroformate used for?
Why Chemists Reach for Isobutyl Chloroformate
Isobutyl chloroformate often shows up in labs where chemists need a strong, reliable reagent. This isn’t something you'd see sold at a hardware store or sitting under the kitchen sink. In research and the pharma world, this colorless liquid plays a role that’s both precise and crucial. People use it mainly to kickstart reactions—especially ones that make molecules stick together in a very controlled way.
A Key Player in Drug and Compound Synthesis
Drug makers use isobutyl chloroformate to put chemical “handles” or groups onto molecules. These tweaks can take something basic and turn it into a new antibiotic, an antiviral, or even fuel for future cancer treatments. Take peptide synthesis, for example. Every biology or chemistry student learns that proteins form through amide bonds, and linking those amino acids just right makes all the difference. Isobutyl chloroformate activates the acid part, so the new bond forms cleanly, with fewer leftovers to clean up. Science journals back this up—papers in Organic Process Research & Development describe its role in making sure new molecular combinations go as planned.
Industry, Safety, and the Everyday Impact
High-end coatings, perfumes, and even agricultural chemicals benefit from reactions powered by isobutyl chloroformate. Alongside the breakthroughs, there’s always a messier side. This stuff doesn’t just dissolve harmlessly if someone spills it. It reacts with water, letting out corrosive fumes. Anyone handling it needs goggles, gloves, and trained habits. Factory workers and lab staff rely on safety protocols, local regulations, and a watchful eye to make sure it goes exactly where scientists intend. According to the Centers for Disease Control and Prevention (CDC), eye and respiratory injuries from mishandling do turn up, mostly when people skip the basics of chemical safety or when ventilation fails.
The Environmental Angle
Waste remains a bigger topic each year. Once the work with isobutyl chloroformate wraps up, what’s left can’t get rinsed down the drain. Most labs partner with hazardous waste handlers, breaking down or neutralizing the leftovers. Companies look for ways to swap in greener reagents when performance allows. Pressure comes from environmental agencies and committed scientists who want the next chapter in chemistry to be safer, inside and out.
Where Responsibility Steps In
Access to powerful chemicals comes with trust. Labs need training and oversight. Academic and government reviews track every gram. This helps keep isobutyl chloroformate out of the wrong hands, discourages shortcuts, and encourages innovation in safety and technique. In my own college lab days, this approach helped prevent close calls and shaped a respect for chemicals—appreciation mixed with a little healthy fear of mistakes.
Finding Better Solutions
Researchers don’t rest on long-standing recipes. Smarter synthesis tools are always under review, and teams run trials with alternative reagents whenever possible. Courses on green chemistry are now common in universities, equipping new generations to reduce risk—not just in the lab, but out in the world, once these compounds hit the commercial pipeline. This drive matters. Chemical progress should raise standards for health and the environment, even when it means changing how business has always worked.
What are the safety precautions for handling Isobutyl Chloroformate?
A Closer Look at the Risks
I once stepped into a chemical storage room and caught that harsh, sharp smell of something volatile. Experience teaches caution; some compounds do more than sting your nose. Isobutyl chloroformate joins a long list of chemicals that demand careful respect. It’s not just an irritant—this stuff can lash out at eyes, skin, lungs, and it reacts fast with water or many chemicals. Even seasoned chemists occasionally get caught off guard by its fumes or splash, especially during transfers and mixing.
Understanding Real Exposure Hazards
Isobutyl chloroformate releases toxic vapors, which can knock you off balance if inhaled in a closed space. Short exposure burns the eyes and throat, and longer exposure might send someone to the hospital. I’ve seen gloves dissolve from chemical splashes that seemed minor at first. Prolonged inhalation or contact with this reagent causes long-term health problems, so working without proper safeguards invites real trouble.
Personal Protective Equipment Matters
Safety always starts with what you wear. Thick nitrile or butyl rubber gloves hold up much better than those thin latex ones. Lab coats, goggles, and face shields keep splashes off your skin and out of your eyes. Rely on a sturdy fume hood to manage vapors—not just an open window. Respirators with the right filters step in for big spills and high concentrations, but nothing beats keeping vapors from building up in the first place. It's tempting to take shortcuts when you’re in a rush, but every rushed step risks a chemical burn or worse.
Handling and Storage Says It All
All bottles and containers need a clear label. I remember a colleague who ignored that step; a mix-up led to some near misses and plenty of paperwork. Store isobutyl chloroformate in a cool, dry place, away from acids, bases, moisture, and ignition sources—never in a regular storeroom or anywhere it might leak into shared space. Keep containers tightly sealed. Solvents like this creep into cracks you don’t even notice. A secure chemical storage cabinet with exhaust—protected from direct sunlight—makes a big difference. Spills spread easier than people think. Spill containment kits and adsorbent materials should sit within arm’s reach any time this chemical is open.
Emergency Action: Fast, Clear Steps
Everyone working with isobutyl chloroformate ought to know what to do if there’s a splash or spill. An eyewash station and safety shower should stay clear, checked, and easy to reach. Get any splashed chemical washed off skin or eyes with lots of water right away—no hesitation. Call for help as soon as possible if symptoms like coughing or irritation pop up. Reporting spills and exposures immediately can mean the difference between an incident and a disaster. I’ve watched lab teams lock up after a small spill and come back to lingering odor—always clean up and ventilate as soon as it’s safe.
Building a Culture of Accountability
Before anyone handles this chemical, regular safety training pays off. Updates on new standards or emergency drills keep skills sharp. Supervisors and peers share responsibility—if you see someone handling isobutyl chloroformate without the right gear, call it out. It’s better to repeat instructions than call an ambulance. Confidence around chemicals grows from good habits and clear communication, not just written procedures gathering dust. Trust grows from watching out for each other and spotting risky shortcuts before they become problems.
What is the chemical formula and molecular weight of Isobutyl Chloroformate?
The Chemical Structure Explained
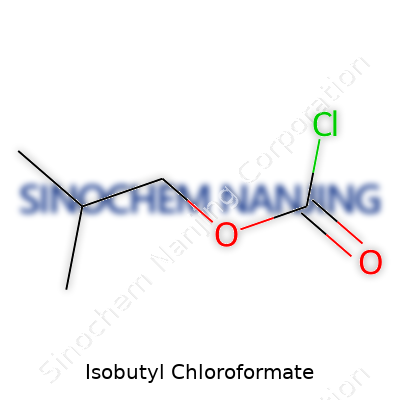
Isobutyl chloroformate goes by the formula C5H9ClO2. Each molecule contains five carbon atoms, nine hydrogen atoms, one chlorine atom, and two oxygens. Not many folks outside chemistry circles stop to wonder about those details, but understanding the structure helps grasp its uses and manage its risks. By calculating the molecular weight—adding up each atom—we arrive at 136.58 grams per mole. That comes from Carbon’s atomic weight (12.01), Hydrogen’s (1.01), Chlorine’s (35.45), and Oxygen’s (16.00). Getting these numbers straight keeps the calculations right, whether you’re ordering lab supplies or scaling up an industrial process.
Why Knowing the Formula Matters
Recognizing and recalling chemical formulas means more than passing an exam. Take isobutyl chloroformate, for example. It belongs to a family of compounds well-known for their reactions during the synthesis of pharmaceuticals and fine chemicals. This particular molecule acts as a go-to reagent for making carbamates and urethanes, intermediates found in everything from drug development to material science. Risk creeps in when people overlook the specifics. I’ve seen folks confuse similar-sounding names or swap formulas by mistake, which can end badly in a synthesis or in safety protocols. Mistakes in shipping or storage come down to labels and clear understanding—knowing a single digit in the formula can mean the difference between a safe shipment and a disaster.
Molecular Weight in Real-World Practice
Molecular weight isn’t just a textbook figure. It determines how much of a chemical you’ll use for a reaction, how it’ll behave in the air, and what regulations kick in for transporting it. Isobutyl chloroformate, at 136.58 grams per mole, tilts toward volatility. It evaporates more quickly than hefty molecules. A lower molecular weight often means tighter control in the workplace, stronger ventilation, and special training for everyone who might handle the bottle. In some formulations, a miscalculation leads to dangerous volatility or unwanted byproducts. Most labs adopt digital safety sheets, but I still see older ones relying on paper charts where a single typo might throw off the entire process.
Tying Formula and Function Together
The story of isobutyl chloroformate—formula and molecular weight included—plays out in quality control and research every day. Scientists reference these numbers not to sound smart, but because small details change whole outcomes. During my years in the lab, a chemical’s proper identification set the foundation for safety, regulatory compliance, and success in research. Regulations around transportation or waste disposal pull their triggers based on formula and weight. For new hires, drilling the details—double-checking molecular weights, cross-referencing formulas—builds habits of care that extend to the entire team.
Looking Ahead: Solutions for Common Challenges
Mix-ups happen less often when databases stay updated and training focuses on these fundamental details. Teams who share experience, check each other’s work, and cultivate a culture of accuracy run into fewer surprises with their reagents. Digital labeling, regular audits, and ongoing hands-on training handle most gaps before they widen. Respect for the humble molecular formula and weight doesn’t just keep the paperwork tidy; it keeps projects on schedule and people safe.
How should Isobutyl Chloroformate be stored?
Look Past the Label, Think Safety
Isobutyl chloroformate sits in countless chemical supply rooms across factories, schools, and startups. The label calls for “cool, dry, well-ventilated” storage, but those words lose meaning unless we know what happens if corners get cut. Early in my career, I once found a half-full drum sweating in July heat, leaking fumes strong enough to set off the lab’s gas detector. Turns out, some rules really are written in pain.
Heat and Humidity: The Enemy
Leaving this chemical near a radiator or in direct sunlight risks more than just faster spoilage. Isobutyl chloroformate decomposes easily with moisture or heat. That reaction sends out hydrochloric acid gas and phosgene, which can burn skin and attack lungs. Numbers from OSHA point out that phosgene exposure, even at low levels, can send workers to the emergency room. So, my take: Find storage spaces cut off from steam pipes, hot equipment, and open windows in stormy weather.
The Metal Drum Complication
Factories and labs often receive isobutyl chloroformate in metal drums. Metal can corrode on contact with acid fumes, especially under humid conditions. I've seen a corroded drum bottom out and leak into the floor. Use secondary containment—plastic spill trays or a chemical safety cabinet built for acids and bases. They’re standard gear for good reason.
Labeling Isn’t Enough
I once visited a workshop where all the containers, including hazardous ones, looked the same. If a janitor mistakes this chemical for less harmful solvents, accidents become much more likely. Clear hazard signs, dated labels, and an inventory log make a difference—especially when students or new team members handle storage. According to the Chemical Safety Board, most critical incidents start with missing labels or outdated inventories.
Ventilation and the Human Factor
No one relishes opening a cabinet to a wall of invisible fumes. Isobutyl chloroformate releases toxic vapors. Storing it in well-ventilated rooms—not the dusty broom closet—helps break down vapors before someone gets a noseful. Fume hoods or cabinets ducted outdoors make life much safer. For small labs where this might feel like overkill, cracked windows and exhaust fans still help.
Know Emergency Steps
Accidents ignore timetables. Safety showers, eyewash stations, and spill kits with absorbent pads belong within arm’s reach of wherever this chemical is stored. I learned the hard way that a paper towel and running tap water won’t cut it if something splashes. Familiarity with the SDS (Safety Data Sheet) makes a big difference; don’t wait for an emergency to skim it.
Tight Controls and Access
Hazardous storage works best if everyone knows who has the key. Limit access to trained staff. Store isobutyl chloroformate away from acids, bases, and water-reactive materials—never let chance decide if a spill turns into a chemical reaction.
Final Word
Promises and policies don’t prevent injuries; habits do. Isobutyl chloroformate rewards those who treat it with control and respect. That cool, dry, ventilated closet might not look like much, but someday it could save a life. In chemical storage, every careful choice matters.
What are the potential health hazards of Isobutyl Chloroformate exposure?
Real World Risks Involved
Not everybody has heard of isobutyl chloroformate, but if you walk into a chemical plant, work in a lab, or know anyone in certain manufacturing jobs, it’s something worth knowing about. From my time working summers at an industrial supplier, I saw the importance of taking chemical safety seriously. Isobutyl chloroformate, a reagent used in pharmaceuticals and plastics, comes with hazards that stick with you longer than just a whiff in the air.
Ways Exposure Happens
Spills, leaks, or even just opening a bottle in a room with bad ventilation turn routine work into a serious health risk. This stuff evaporates fast and creates fumes that fill enclosed spaces before you realize it. It doesn’t just go away if you open a window.
Breathing Problems Are Just the Start
Inhaling the fumes sits at the top of my concerns. Acute exposure can kick off coughing, shortness of breath, and burning in the nose and throat. The chemical reacts with moisture, so it goes after the lining in your airways. Coughing isn’t just uncomfortable; in more serious cases, it turns into chemical pneumonia, which needs real medical treatment. I’ve watched a coworker struggle after forgetting to check his mask. Those coughs and labored breaths hit everyone in earshot with a reality check. Safety data from the Centers for Disease Control and Prevention says rapid removal to fresh air is critical, as effects can sneak up fast.
Skin and Eye Damage Hits Fast
Broken bottles or splashes create another problem. Isobutyl chloroformate loves to burn any skin it touches. Even a quick splash leaves red, itchy welts, and longer contact breaks down tissue. Eyes need extra care—just a droplet brings swelling, pain, and vision problems that don’t always heal cleanly. I learned to always double-check my goggles, because one careless moment with chemicals like this can mean a trip to the emergency room.
Why We Can’t Shrug This Off
Chronic effects don’t get enough attention. Repeated exposure, even at levels too low to cause a headline-grabbing accident, brings headaches, dizziness, and deep fatigue. It’s easy to ignore until you realize your body just isn’t bouncing back anymore. I’ve seen colleagues lose time at work over recurring symptoms that doctors traced back, eventually, to chemicals like this.
Solutions That Matter
Controlling these risks isn’t rocket science, but it takes commitment. Good ventilation, regular equipment checks, and clear safety protocols do more than tick off boxes on a checklist. Wearing gloves, face shields, and proper respirators kept me and my team out of the clinic more than once. Training matters—a lot of injuries I witnessed came from shortcuts or not speaking up when something seemed off. Companies owe it to their workers to build a culture where safety isn’t just a slogan. Regular air quality testing and spill drills aren’t wasted time. Anyone handling isobutyl chloroformate—whether on a big production floor or in a research lab—has to know exactly what to do when danger strikes.
Looking Ahead
Chemists and safety officers constantly learn from new research, improving standards every year. Solving the risks linked to chemicals like isobutyl chloroformate means treating worker health as something as important as output or deadlines. If you ask anyone in the field, the consensus is clear: putting the right precautions in place saves lives and keeps jobs safer for everyone.
| Names | |
| Preferred IUPAC name | 2-Methylpropyl carbonochloridate |
| Other names |
Isobutyl chlorocarbonate Chloroformic acid isobutyl ester Isobutyl chloroformiate Isobutyl carbonochloridate |
| Pronunciation | /ˌaɪ.səˈbjuː.tɪl klɔːˈrɒfərˌeɪt/ |
| Identifiers | |
| CAS Number | 543-27-1 |
| Beilstein Reference | 626220 |
| ChEBI | CHEBI:53031 |
| ChEMBL | CHEMBL1431185 |
| ChemSpider | 57100 |
| DrugBank | DB11107 |
| ECHA InfoCard | 100.008.668 |
| EC Number | 209-570-0 |
| Gmelin Reference | 1335 |
| KEGG | C14133 |
| MeSH | D002222 |
| PubChem CID | 6570 |
| RTECS number | NT8225000 |
| UNII | SJ10Q32P12 |
| UN number | UN1987 |
| CompTox Dashboard (EPA) | DTXSID4020731 |
| Properties | |
| Chemical formula | C5H9ClO2 |
| Molar mass | 120.56 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Pungent |
| Density | 1.116 g/mL at 25 °C |
| Solubility in water | Decomposes |
| log P | 1.93 |
| Vapor pressure | 10 mmHg (20 °C) |
| Acidity (pKa) | 1.70 |
| Basicity (pKb) | 1.73 |
| Magnetic susceptibility (χ) | -60.4e-6 cm³/mol |
| Refractive index (nD) | 1.393 |
| Viscosity | 2.26 mPa·s (at 25 °C) |
| Dipole moment | 2.15 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 364.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -358.2 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -648.6 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS02,GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H302, H311, H314, H331 |
| Precautionary statements | P210, P261, P271, P273, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P311, P312, P370+P378, P403+P233, P501 |
| NFPA 704 (fire diamond) | 2-3-1-W |
| Flash point | 50 °F (10 °C) |
| Autoignition temperature | 451°F (233°C) |
| Explosive limits | Explosive limits: 2.3–12% |
| Lethal dose or concentration | LD50 oral rat 1,800 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1,525 mg/kg (rat, oral) |
| NIOSH | NA8485000 |
| PEL (Permissible) | PEL: 0.005 ppm |
| REL (Recommended) | 0.005 ppm |
| IDLH (Immediate danger) | IDLH: 50 ppm |
| Related compounds | |
| Related compounds |
Methyl chloroformate Ethyl chloroformate Propyl chloroformate Isopropyl chloroformate Butyl chloroformate Sec-butyl chloroformate |

