Isobutyl Acetate: The Story, Science, and Scope of a Versatile Chemical
Historical Development
Long before big factories and complex supply chains, isobutyl acetate found its place in early chemical enterprises. Chemists in the late 19th and early 20th centuries began noticing the pleasant, fruity aroma from its distillation and extraction. It didn't take long for perfumers and flavorists to follow suit, latching onto isobutyl acetate for fragrances and flavoring. Its roots stretch back to research that chased after ways to mimic and enhance natural scents in an age where both everyday living and industry began depending less on pure extracts and more on reproducible molecules. Over decades, advances in esterification, distillation, and purification shaped how we produce and understand this versatile compound, making it easy to spot in both simple flavoring bottles and high-purity solvents used in advanced labs.
Product Overview
Isobutyl acetate steps into many roles, straddling the fine line between industrial necessity and consumer desire. Industry leans on it for its role as a fast-evaporating solvent in coatings, inks, and adhesives. It shows up in apple and pear flavorings, adding character to candies and beverages. Everyday products like nail polish remover and paint thinners highlight its ability to dissolve a broad range of resins and pigments. This balance between technical performance and sensory appeal keeps isobutyl acetate in strong demand, often eclipsing other esters in sectors where odor quality and solvent power both matter.
Physical & Chemical Properties
Isobutyl acetate has a sweet, fruity scent reminiscent of pears, but under the pleasant aroma sits a colorless, flammable liquid, boiling at 118°C and freezing at -99°C. Its moderate volatility means it evaporates faster than water but slower than lighter solvents like acetone. It blends easily with alcohols, ethers, and most organic solvents, yet separates from water. This behavior in mixtures matters in manufacturing, whether one is optimizing the drying of paints or developing stable flavors in food. With a molecular formula of C6H12O2 and a density hovering just under that of water, it stores and handles best under cool, well-ventilated conditions, away from sources of ignition.
Technical Specifications & Labeling
Regulators don’t cut corners when it comes to labeling and testing isobutyl acetate. Documents need to show purity—routinely over 99% in most applications—as well as checks for residual acids, moisture, and degradation products like isobutanol. Safety labels list its UN number (1220) along with flammable liquid pictograms, the signal word "Danger," and hazard statements. Technical sheets also reveal flash point data, vapor pressure, and handling precautions. In the world of exports and imports, keeping these numbers current can mean the difference between a shipment sailing through customs or hitting a regulatory wall.
Preparation Method
Factories make isobutyl acetate mainly by reacting isobutanol with acetic acid, a straightforward esterification catalyzed by acids like sulfuric acid. After distillation removes the ester from the reaction mixture, further purification tunes the product to meet tight industry standards. Some facilities recycle both starting materials and byproducts, aiming for higher yield and lower waste. This approach keeps production more sustainable and often reduces costs, which customers in competitive markets appreciate.
Chemical Reactions & Modifications
Isobutyl acetate doesn't just act as a solvent or scent enhancer. Its structure borrows stability from the ester bond linking isobutanol and acetic acid, but under heat or acid, it can break back down to its original components. In the lab, chemists sometimes hydrolyze it deliberately to isolate isobutanol. More rarely, they modify it further to produce complex molecules for specialty flavors or solvents. In modern polymer science, it's occasionally used as a building block for resins that demand specific flexibility or adhesion properties. In my own bench work, ester interchange reactions using isobutyl acetate often deliver rapid results due to its predictable behavior and volatility, helping to speed up synthesis with minimal fuss.
Synonyms & Product Names
Few chemicals carry as many alternate names as isobutyl acetate. Chemical suppliers catalog it under titles like 2-methylpropyl acetate, isobutyl ethanoate, and even the less formal isobutanol acetate. Some flavor and fragrance companies list it by its FEMA number (2043), while industrial suppliers sometimes bundle it under generic solvent categories for logistics and purchasing. No matter the alias, a smart buyer checks the CAS number (110-19-0) for complete certainty. Sloppy notation or mixed-up synonyms in the warehouse or order sheets quickly cause confusion, particularly when switching between suppliers from different regions.
Safety & Operational Standards
Working with isobutyl acetate, I’ve seen just how much safety procedures matter—particularly in poorly ventilated or crowded environments. Its low flash point and rapid evaporation can lead to air concentrations that approach nausea or, in bad cases, unconsciousness. Industry standards require airtight storage drums, flameproof fittings, and well-marked safety zones for both workers and emergency teams. Workplace monitoring keeps vapor concentrations below occupational exposure limits (typically 150 ppm in many jurisdictions). Simple steps—gloves, goggles, proper spill kits—keep minor incidents from turning into accidents. Responsible facilities also enforce strict documentation, training, and readiness drills, keeping both regulators and staff satisfied that high-volume operations won’t flirt with disaster.
Application Area
From the aroma of hard candies to the fresh smack of lacquer on new furniture, isobutyl acetate travels across surprising zones. Its biggest industrial demand springs from paints and coatings, especially where a consistent dry time and appealing smell seal the deal. The printing industry finds it indispensable in specialty inks. Even tiny doses find their way into food and beverage flavorings, while perfumers add it to blends that demand juicy pear, plum, or cherry overtones. Laboratory workers depend on it for extractions, mobile phases in chromatography, and even cleaning delicate equipment where residue-free finishes matter. In daily life, its distinctive scent often signals professional handiwork—fresh car interiors, glossy book covers, or a dab of high-end nail polish.
Research & Development
University labs and corporate R&D teams keep searching for greener synthesis routes and new applications. Green chemistry researchers chase catalyst upgrades that lower energy costs or tap into renewable acetic acid sources. Some teams have explored using enzymes rather than mineral acids to generate isobutyl acetate in milder reaction conditions. On the analytical side, gas chromatography and mass spectrometry push detection limits ever lower, essential for rigorous consumer safety and product quality. Other groups turn attention to bio-based feedstocks, squeezing isobutyl acetate from fermentation broths to reduce fossil resource use. Getting consistent yields from bio-route processes remains a challenge, but every advance in strain selection, bioreactor design, or purification makes this ester more sustainable for future generations.
Toxicity Research
Toxicity studies over decades support most regulatory conclusions: isobutyl acetate’s acute toxicity is low, but it’s no benign substance in careless hands. Inhalation at high concentrations quickly irritates mucous membranes and, with sustained exposure, could affect the central nervous system. Early animal testing mapped out safe exposure limits, and follow-up research in workplaces confirmed that headache, dizziness, and mild narcosis could occur above occupational thresholds. The compound doesn’t accumulate in the body, breaking down fast in animal models. Regulatory reviews often focus on its potential to enhance absorption of other chemicals or its interactions with vulnerable groups like pregnant workers. Studies rarely flag severe chronic effects, but long-term vigilance still makes sense, as chemical industries keep racing to update safety guidance based on fresh data.
Future Prospects
Isobutyl acetate’s future looks busy. Regulatory demands shape production, especially as companies seek to squeeze out lower emissions, higher recycling, and better worker safety. Green manufacturing projects and biotechnology seem poised to shake up how this compound comes to market, not just in developed economies but in rapidly industrializing regions hungry for lower-cost, higher-performance solvents. As flavor and fragrance design gets more precise, tailored variants of isobutyl acetate—modified for stability or sensory punch—may capture new niches in processed foods, cosmetics, and pharmaceuticals. Paint, printing, and electronics will keep needing purer and more consistent batches, which pushes both chemical engineers and plant operators to tweak old methods and experiment with new. There’s no sign the world’s ready to set this versatile ester aside, and with so much research energy behind it, isobutyl acetate promises to remain a mainstay in both labs and factories for years in ways that blend utility, safety, and even a dash of sensory delight.
What is Isobutyl Acetate used for?
The Smell Behind the Scenes
Step into a factory where workers fill perfume bottles, and the first thing you’ll notice isn’t the chaos of machines—it’s the sweet, fruity tang in the air. That’s isobutyl acetate. This compound gives many perfumes and consumer products their crisp, juicy punch. Years ago, I worked at a food flavoring company. Our team would mix concentrates for candies and baked goods, and isobutyl acetate’s profile—something between pears and raspberries—always stood out. It’s found in raspberries, so even that whiff of “natural” berry scent likely involves this compound.
More Than Just a Pleasant Scent
Perfumers count on isobutyl acetate for its strong aroma, but the list doesn’t stop there. Paint companies rely on it as a solvent. It dissolves tough resins and leaves paint smooth, streak-free. Most nail polishes also lean on this chemical, thinning the thick goop into something spreadable. Back in college, I worked in a hardware store, and I watched customers debate over wall colors, unaware that the solvent in the paint can affected how the color went up and how strong the odor lingered.
Role in Industry
Factories producing printing inks use isobutyl acetate to mix pigments evenly into fluid ink. Printers demand quick-drying results and vivid colors that don’t smudge. This solvent helps deliver. In factories, efficiency often means lowering drying time without compromising safety, and this is where isobutyl acetate fits in—it evaporates fast and doesn’t leave traces behind. This matters in mass production, since speed plus quality gives a business its edge.
Everyday Products
The compound pops up in adhesives too. Strong, clear glue in school crafts and home repairs often lists isobutyl acetate among its solvents. It keeps glue workable, then vanishes as the adhesive dries. Some companies in the cleaning business depend on it for its grease-cutting power. If you’ve ever scrubbed a stubborn mark off a window, you may have picked up a cleaner made punchier—sometimes unknowingly—by this solvent.
Food and Beverage Use
Though most uses are industrial, food companies use isobutyl acetate in tiny doses. In flavor enhancers and some fruit drinks, it rounds out flavors with a gentle pear-like note. The FDA approved its use in small, controlled amounts for foods. Strict limits exist because, just like salt or sugar in overabundance, it transforms from pleasant to overwhelming. Manufacturers must follow these regulations closely to keep customers safe and satisfied.
Sustainability and Safety
Some people worry about exposure. Studies show that low levels found in food and cosmetics pose little risk, but workers in factories still need protection. Ventilation, gloves, and proper storage become essential because inhaling pure isobutyl acetate can cause headaches and nausea. From experience, those responsible for workplace safety audits—like me, when I volunteered at a chemical warehouse—learn firsthand how enforcing rules keeps everyone healthy.
Where We Go From Here
The market leans toward eco-friendlier alternatives as industries rethink chemicals that end up in waterways or the air. Biobased sources for solvents gain traction, and companies start shifting from petroleum-driven production. Growing up, I saw changes in local plants as community health concerns forced updates in chemical handling. This keeps isobutyl acetate relevant and safer as society balances usefulness with sustainability.
Is Isobutyl Acetate safe to handle?
Practical Experience and Real-World Use
Ask anyone who’s spent time in a paint shop, printing facility, or even in a nail salon—chances are, they’ve already encountered isobutyl acetate. This clear liquid, famous for its fruity aroma, helps dissolve lacquers, inks, and aids in fragrances. Big industry uses it far more frequently than many realize. My own time working in a printing warehouse proved that isobutyl acetate isn’t some rare or exotic chemical stashed in research labs; it’s a regular fixture, handled by busy hands every single shift.
Safety concerns inevitably pop up. Folks hear "chemicals" and imagine danger. Truth is, safety comes down to knowing what you’re dealing with and respecting how it interacts with the body and the environment. Isobutyl acetate won’t leap off the shelf and bite you. It takes improper storage, bad ventilation, or careless contact to cause most problems.
The Facts Behind the Name
Regulators from the U.S. Occupational Safety and Health Administration (OSHA) and Europe’s ECHA recognize that isobutyl acetate can irritate skin, eyes, and the lungs. Those agencies don’t ring alarm bells without good reason. If you breathe in too much, headaches and dizziness might creep up. Prolonged exposure indoors—imagine a print worker touching the stuff bare-handed or working where fumes linger—can ramp up irritations or nausea.
It’s flammable. That means no open flames or sparks near open drums. Contractors and shop workers use gloves, eye protection, and proper air-flow as standard gear—not just for this one solvent, but for most of the stuff on storage shelves.
Why Attention Matters
One major aspect rarely mentioned outside training sessions: people lose focus during a long shift, especially if no problems show up after years of handling the same substance. This sense of routine can lull anyone into skipping simple safety steps. In settings with high exposure, personal protective equipment really makes a difference. Proper gloves stand between irritated skin and chemicals that wear on you over months or years. Goggles cut in as cheap insurance against splashes, which happen more often than anyone likes to admit.
Ventilation takes center stage. A cramped workshop without fans or hoods invites trouble. Fumes build up faster than expected. Regulations exist for a reason; they come from years of tracking accidents, illnesses, and near misses.
Supporting Safe Practices with Real Solutions
Staying safe comes down to basic habits. Read the label and the material safety data sheet before pouring or mixing. Store the chemical away from heat and sparks. Employees should take breaks out of fume-heavy rooms, whether the odor seems strong or not. This isn’t just for isobutyl acetate, but the advice carries extra weight because the solvent can hit hard if ignored.
Training plays a big role, and companies shouldn’t treat it as a tick-box exercise. Safety drills and quick refreshers remind everyone where the eyewash is and what to do if a spill happens. Responsibility starts with the individual, but management decisions matter too—ventilation upgrades and non-leaky containers keep trouble at bay.
Long story short, isobutyl acetate rewards respect and punishes negligence. Handle it with care, gear up, and stay mindful. Years of safe handling come from not getting lazy or complacent.
What are the storage and handling requirements for Isobutyl Acetate?
What Makes Isobutyl Acetate a Safety Priority
Some chemicals demand a bit more respect than others. From my time on the factory floor and in the lab, isobutyl acetate always grabbed my attention. That sharp, fruity scent seems harmless until headaches take over or a spill raises eyebrows. In short, it's flammable, creates strong vapors, and can turn careless habits into emergencies.
Flammability: Don’t Give It a Spark
Storing isobutyl acetate safely comes down to understanding its flammable nature. This liquid has a flash point around 22°C—think about a warm room in summer. Any open flame, spontaneously sparking tool, or faulty outlet in the wrong spot could start trouble. For that reason, keep it in well-ventilated, flame-proof spaces, preferably in containers designed for volatile solvents. From my days helping manage chemical storerooms, metal drums with tight seals kept us out of trouble. Don’t trust old, brittle plastic either; leaks and fumes can escape before you know it.
Ventilation and Vapor Management
Vapors from isobutyl acetate can build up quickly. Without airflow, fumes collect in low spots, ready to ignite. Nobody likes dizzy spells or sore throats after a routine task, so ventilation can’t be an afterthought. I look for rooms with mechanical exhaust and windows wherever possible. If it smells strong, ventilation isn't doing its job. Installing proper fume hoods or local exhaust systems reduces risk and keeps air healthy for workers.
Containers and Labeling
Each time I see a mystery drum with a faded label, I think back to near-misses and wasted hours trying to identify forgotten leftovers. Proper labels, clear hazard statements, and expiration dates make all the difference. Isobutyl acetate should always be kept in containers with clear chemical labels—no skipping this step or relying on memory. Choose ones rated for organic solvents, as some plastics soften after contact.
Avoiding Cross-Contamination
Never mix this solvent with oxidizing agents—peroxides, acids, and strong alkalis. Incompatibility leads to dangerous reactions, and I've seen what happens if lines aren’t flushed out properly or lids go astray. Dedicated storage spaces, far away from oxidizers or acids, prevent these mishaps. Use secondary containment like trays or tubs, so if something tips over, the spill stays small.
Personal Safety Steps
Handling isobutyl acetate safely begins before opening any container. Wear gloves that resist organic solvents, goggles, and a lab coat or chemical apron. Splash-proof face shields add another layer. I always had an emergency eye wash station within ten feet—no exceptions. It’s not just about personal protection; it keeps small mistakes from turning into big problems.
Disposal and Spill Response
Disposing of isobutyl acetate or its containers shouldn’t end up as a last-minute call. Workplaces need dedicated solvent waste bins, not general trash cans. Spills happen, especially if containers travel across busy floors. Absorbent pads, sand, or vermiculite control the spread. From past experience, acting fast with the right gear keeps everyone safe. Training staff on chemical spill procedures beats scrambling for instructions in a crisis.
Trust the Basics and Train the Team
Proper storage and handling keep isobutyl acetate from moving from useful to risky. Using the right containers, ventilation, labeling, and protective equipment cuts danger down to size. Sharing hands-on training and keeping emergency gear handy means nobody guesses in stressful moments. Mixing real-world experience with good practice makes all the difference for safety and peace of mind.
What is the chemical formula of Isobutyl Acetate?
Understanding the Formula: C6H12O2
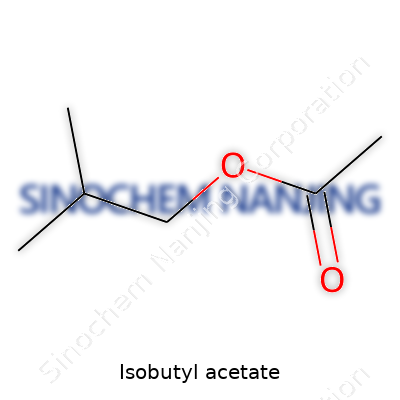
Isobutyl acetate gives off a fruity scent that calls up memories of pear drops or a bushel of fresh apples. You’ll find its chemical formula written as C6H12O2. To someone outside the lab, those characters look about as exciting as a phone number. But this formula actually tells a story about both structure and use. I remember my first encounter with this compound was during a project to identify unknown esters by smell. The distinct aroma landed it in a category I still associate with science and snack breaks.
Real-World Uses Rooted in Chemistry
This ester doesn’t just hang out in a test tube. Manufacturers draw on isobutyl acetate as a solvent in coatings, inks, and fragrances. The C6H12O2 arrangement makes it less polar than water, so it blends well with other organic chemicals. On the production line, this matters. Items like lacquers and adhesives dry faster and smoother because of that balance between volatility and solvency.
Back in high school chemistry, we’d mix acetic acid with isobutanol to make isobutyl acetate in small batches, always within range of a fume hood. Each compound in the mix, represented in the formula, does a job — carbon chains adding punchy notes, oxygen atoms bridging and adding reactivity, hydrogen filling in the structure. I realized that those six carbons, twelve hydrogens, and two oxygens shape everything from smell to performance.
Health, Safety, and Environmental Notes
Wearing gloves and goggles isn’t just classroom protocol. Industrial workers deal with isobutyl acetate daily, and exposure at high concentrations leads to headaches or nausea. OSHA sets clear limits, and ventilation is the rule, not the exception. Facts back this up; short-term exposure levels above 150 ppm bring out symptoms nobody wants. Using the right PPE, testing air quality, and substituting with less volatile solvents where possible keeps risk in check.
The formula also has implications for environmental responsibility. The ester biodegrades more easily than halogenated solvents, sparing the environment some havoc. I’ve seen labs switch to isobutyl acetate from harsher ethers, cutting back on hazardous waste. Still, spills pose problems for aquatic life, especially in concentrated amounts. Good chemical knowledge leads to better spill training, proper disposal, and sensible storage.
A Nod to the Formula’s Value
I’ve worked with many chemicals where complexity creates headaches. Isobutyl acetate stands out because C6H12O2 gives a clear map for both handling and benefit. Its popularity in paints and cleaning agents traces right back to the properties spelled out in those six carbons, twelve hydrogens, two oxygens. On top of practicalities, this compound pops up in everyday flavors and fragrances, linking chemistry to daily experience.
Building better product safety means checking formulas, understanding reactivity, and staying on top of research. Replacing more toxic choices and managing exposure keeps operations smooth. And remembering what each number and letter means reinforces why attention to fine chemical detail pays off for everyone — from student to scientist to factory floor.
What are the main hazards associated with Isobutyl Acetate?
Toxic Fumes in Everyday Workspaces
Isobutyl acetate turns up often in factories, labs, and paint shops. This clear liquid comes with a fruity smell, but breathing in its vapors can leave workers dizzy, give them headaches, or bring on sore throats. Prolonged exposure piles on stress to the nervous system, making it harder for folks to keep up a clear head. Over the years, I’ve watched people forget how common and sneaky chemical vapors can be—safety goggles and open windows aren’t enough. Spills or open containers let vapors gather, especially in small or poorly ventilated rooms, and even a few whiffs might leave someone feeling off their game.
Risk of Fire and Explosion
Isobutyl acetate lights up fast. Its low flash point—around 17°C—means it evaporates and catches fire more easily than water boils. Factories using isobutyl acetate need to take this risk seriously. Everyday routines like handling rags or empty barrels quickly turn risky, since static electricity or a random spark can set off a fire. Years spent around shop floors taught me that some workers let their guard down after repeating the same jobs for ages—forgetting to ground containers or allowing buildup of vapor in corners almost invites accidents. Government records from OSHA and the EPA both underline the need for strong venting and strict storage rules; these systems keep fires from spreading out of control.
Irritation and Short-Term Health Harm
Once isobutyl acetate hits skin, it starts to sap oils and burn. Anyone handling this solvent without gloves faces cracked hands, red patches, and, in worse cases, chemical burns. Vapors sting the eyes fast—the kind of sting you remember. In one year working around coatings, I saw a friend lose vision for days after a splash. He followed advice after the fact, flushing his eyes with water and seeking medical help, but that pain stuck around. Eyes, nose, and skin matter every bit as much as lungs—the lesson sticks if you learn it too late.
Long-Term Effects and Environmental Spillover
There’s not enough solid proof to tie isobutyl acetate to chronic illness or cancer, but nobody should assume time alone makes things safe. People working with solvents day after day risk losing their sense of smell, feeling tired, and developing liver trouble. I’ve also seen how chemical spills cause headaches for entire communities. Runoff or careless dumping can leak into water supplies and damage plant and fish life. While regulations try to keep a lid on large-scale leaks, everyday choices—like double-checking storage or fixing old seals on barrels—stop the small slip-ups that add up over months and years.
Simple Fixes That Make a Difference
Every job demands proper gear: gloves that stand up to chemicals, safety glasses, and masks that block out vapors save more trouble than they cost. On top of gear, real training—learning what signs to watch for, keeping workspaces aired out, and storing barrels in cool, out-of-the-way spots—keeps daily risks low. Building a solid culture of reporting near-misses, fixing leaky equipment, and shutting down sloppy shortcuts helps staff trust each other and the process. A good manager leads by example and knows that a safe shop doesn’t put production above people.
| Names | |
| Preferred IUPAC name | 2-methylpropyl ethanoate |
| Other names |
Acetic acid isobutyl ester Isobutyl ethanoate 2-Methylpropyl acetate |
| Pronunciation | /ˌaɪsoʊˈbjuːtɪl əˈsiːteɪt/ |
| Identifiers | |
| CAS Number | 110-19-0 |
| Beilstein Reference | 1718737 |
| ChEBI | CHEBI:31241 |
| ChEMBL | CHEMBL510893 |
| ChemSpider | 7076 |
| DrugBank | DB14193 |
| ECHA InfoCard | 01b9c0b6-9e4e-46bc-a6a5-5c62f9856a6a |
| EC Number | EC 203-745-1 |
| Gmelin Reference | 846 |
| KEGG | C11239 |
| MeSH | D009899 |
| PubChem CID | 8021 |
| RTECS number | NF5775000 |
| UNII | J3G15O17D6 |
| UN number | UN1123 |
| CompTox Dashboard (EPA) | DTXSID1039236 |
| Properties | |
| Chemical formula | C6H12O2 |
| Molar mass | 116.16 g/mol |
| Appearance | Colorless liquid with a fruity odor |
| Odor | Fruity |
| Density | 0.868 g/cm³ |
| Solubility in water | 0.7 g/100 mL (25 °C) |
| log P | 1.78 |
| Vapor pressure | 10 mmHg (20°C) |
| Acidity (pKa) | pKa ≈ 25 |
| Basicity (pKb) | pKb: 11.23 |
| Magnetic susceptibility (χ) | -59.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.394 |
| Viscosity | 0.68 mPa·s |
| Dipole moment | 1.62 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 285.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -425.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4640.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P280, P301+P312, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 1-3-0 |
| Flash point | 22°C |
| Autoignition temperature | 425 °C |
| Explosive limits | 1.3–7.5% (in air) |
| Lethal dose or concentration | LD50 oral rat 13,400 mg/kg |
| LD50 (median dose) | LD50 (median dose): 13,400 mg/kg (oral, rat) |
| NIOSH | NA0885 |
| PEL (Permissible) | PEL: 150 ppm |
| REL (Recommended) | 150 ppm |
| IDLH (Immediate danger) | 1,500 ppm |
| Related compounds | |
| Related compounds |
Acetic acid Isobutanol Butyl acetate Isopropyl acetate |

