Isoamyl Nitrate: A Comprehensive Look at Its Past, Present, and Future
Historical Development
Isoamyl nitrate didn’t just show up in modern labs. Chemists in the nineteenth century uncovered this volatile compound during early experiments with organic nitrites. Steam-driven distilleries and new glassware fueled creative chemistry in Victorian Europe, where discoveries often happened through curious trial and error. By the twentieth century, isoamyl nitrate had picked up attention for both legitimate laboratory work and oddball roles as a quick fix for engine performance, thanks to its explosive decomposition and easy vaporization. Over time, stronger regulations and technological advances kept a closer eye on its use, but creative thinkers still found new niches for it in research settings and specialized industries.
Product Overview
Isoamyl nitrate falls into the realm of alkyl nitrites, known for their distinct fruity smell and clear, oily appearance. Unlike some chemicals that hide behind technicalities, this compound demands respect for both its quirky applications and its hazards. It hosts a five-carbon branched chain, making it part of the amyl nitrite family but with its own signature profile. Production isn’t huge compared to bigger industrial players, but it consistently pops up where specific performance–like rapid-pressure impulses or test-lab reactivity–matters more than large-scale supply.
Physical & Chemical Properties
This clear liquid comes with a flash point low enough to keep fire marshals on alert. It boils at about 98°C, and its density keeps it under water in any accidental spill. That sweet, almost banana-like odor? Pure molecular quirk. You’d spot its oily sheen, but it slips easily into organic solvents, making it a familiar friend to chemists who favor experimentation with esters and nitrites. Where oxygen runs thin, this nitrite turns from friend to foe faster than many would expect, a reminder of why personal safety never takes a back seat with volatile organics.
Technical Specifications & Labeling
Labels never cover every angle, but regulations push for eye-watering clarity. Isoamyl nitrate bottles wear hazard warnings for toxicity, flammability, and environmental threat. Lists show its purity—commonly over 98% for research purposes—plus note on any solvent or stabilizer present. Packaging gets checked for compatibility with nitrites to prevent leaks or reactions with metal closures. Anyone handling the stuff keeps a Material Safety Data Sheet close by, detailing first aid steps, protective equipment, and emergency handling. Proper labeling proves just as important as the chemistry inside.
Preparation Method
Making isoamyl nitrate comes down to a dance between isoamyl alcohol and concentrated nitric acid. Add sulfuric acid, and the reaction nudges toward completion. Good ventilation is crucial here—runoff of nitrogen oxides can put a dent in lab safety. Doing this on a larger scale, techs use glass or plastic reactors and chillers to tame side reactions and minimize exposure. The product gets washed, dried, and checked for leftover acid. Careless work ups always threaten contamination, so method and patience set apart the seasoned chemist from the careless.
Chemical Reactions & Modifications
Isoamyl nitrate does not shy away from chemistry. Its nitrite group opens doors for redox reactions, delivering a quick punch of nitric oxide that triggers physiological effects in medical or experimental use. Heated or shocked, it can decompose rapidly, which spells risk and opportunity. Common tweaks include alkyl substitutions, but some researchers push deeper, swapping out the base carbon chain or introducing aromatic groups. Even small swaps change reactivity. In my experience, minor impurities from preparation can throw off entire experiments—purity drives reliability here far more than in many everyday compounds.
Synonyms & Product Names
Names get thrown around: 3-methylbutyl nitrite, isoamyl nitrite, isopentyl nitrite. Street and trade labels pop up in some gray market corners, but legitimate suppliers favor the technical names. In catalogs and reference books, distinctions stay sharp to keep isoamyl straight from the regular amyl nitrite, which swims along a slightly different carbon skeleton. Spotting the right synonym proves essential for safe ordering and regulatory compliance, especially as international shipments invite confusion from mismatched labeling.
Safety & Operational Standards
Laboratories and storage rooms tolerate no guesswork with isoamyl nitrate. Any facility keeps it away from heat, light, and sources of static discharge. Access limits follow local chemical safety laws and insurance guidelines, and tightly sealed containers reduce risk of exposure. Safety goggles, gloves, and fume hoods come standard, not optional extras. Inhalation or skin absorption carries acute risks, and any spill kicks off evacuation and serious cleanup. Waste disposal means neutralization and a trip to hazardous collection points. Years in the lab have taught me most chemical “accidents” come down to shortcutting these hard-earned standards.
Application Area
Most focus lands on its role in research and niche industry work. Isoamyl nitrate supports chemical syntheses where selective nitrosation is key. Some specialty explosives use it as a rapid gas generator. Its physiological effect as an instant vasodilator means it once showed up in emergency medicine, though today’s health professionals steer toward safer and more predictable alternatives. Automotive and aviation engineers exploited its power in additives, but environmental and safety rules pulled back on these older tricks. Real progress in the last decade has come from its controlled use in organic synthesis and analytical chemistry, where no shortcut really does what isoamyl nitrate handles with ease.
Research & Development
Research teams chase better reaction yields, safer handling, and lower toxicity variants. A few groups focus on derivatives that keep the beneficial chemical reactivity but reduce health risks. Others look at its breakdown products to understand environmental impact and develop neutralization treatments before disposal. Analytical chemists work on more accurate ways to quantify tiny contamination, since old GC methods sometimes miss stray peaks from closely related nitrites. Collaborative projects across universities shed light on where future innovation lies—smaller doses in technology, cleaner processes, and smarter detection. In my experience, cross-disciplinary teams unlock better insights into both practical uses and safety enhancements.
Toxicity Research
The dangers of inhaling or ingesting isoamyl nitrate go beyond fainting or headaches. It can cause methemoglobinemia, a blood disorder that blocks cells from carrying oxygen, which gets serious within minutes at high doses. Animal studies have shown organ impacts and reproductive risks from long-term exposure. Researchers track metabolites to map out toxicity in different tissues. Poison control resources list isoamyl nitrate with other alkyl nitrites as hazardous, emphasizing swift response in exposure events. Ongoing health studies drive more transparent regulations and improved personal protective practices: a reminder that chemical curiosity never outpaces the need for health and safety vigilance.
Future Prospects
Isoamyl nitrate likely remains a pinpoint tool instead of a widespread staple. As science moves toward green chemistry, future research centers on safer synthesis and biodegradable analogs. Engineering advances push for automated handling using robotics and sensors to keep people at a distance. Regulations grow more sophisticated, focusing both on user safety and the impact of nitrosated waste streams. In my own years handling specialty chemicals, those labs that invest in training, regular equipment checks, and ongoing education stay ahead—no matter how hard the rules push or markets change. New applications may emerge as understanding of the molecule’s reactivity grows, but respect for its power will always shape how and where it serves the next generation of chemists.
What is isoamyl nitrate used for?
A Quick Chemistry Lesson
Isoamyl nitrate calls to mind the sharp, banana-like scent that lingers in a lab, but there’s more beneath the surface. Chemically, it combines isoamyl alcohol and nitric acid to form a clear, volatile liquid. It evaporates quickly, which is probably why it’s found its way into certain niches both in and out of the lab.
Medical Roots and Real-World Experience
Long before synthetic heart medications became household names, doctors reached for compounds like isoamyl nitrate for quick relief of chest pain. Its value came from the ability to relax smooth muscles, easing angina attacks within seconds. I’ve seen physicians reminisce about using such remedies—the trembling hands of ER staff, the tense faces of worried patients, and the sense of hope that came with even a few drops from a small brown vial.
As faster and safer options entered the scene, hospitals stopped relying on isoamyl nitrate. Still, old textbooks keep its stories alive for medical students learning how people once handled sudden bouts of heart pain. Like many remedies, the march of time replaced it with drugs like nitroglycerin, which brought fewer risks and more predictable results for most patients.
Beyond Medicine: How Industries Tap Its Power
People now associate isoamyl nitrate more with testing fuels and some obscure cleaning techniques than with health care. That sharp, sweet smell appears around rail yards and garages, where technicians use isoamyl nitrate as an ignition agent for diesel engines. Since certain engines depend on high temperatures to start, pouring a little of this compound into the system helps produce the kick needed to get metal pistons moving in deep cold.
Manufacturers sometimes choose it for calibration of gas detection equipment. Its fast evaporation makes it useful for short, controlled bursts of vapor, allowing engineers to set instruments properly for safety in confined spaces like mines and chemical plants. People who keep cities running behind the scenes cannot afford uncertainty during routine tests, so a reliable substance like isoamyl nitrate remains on the shelf, just in case.
The Other Side: Misuse and Public Health
The trouble comes in the form of recreational misuse. Labeled as "poppers" in some nightlife scenes, isoamyl nitrate can produce an intense, fleeting rush people chase at parties. While stories float around about its allure, it’s easy to gloss over fallout like headaches, low blood pressure, dizziness, or—in rare cases—serious complications like sudden drops in oxygen or loss of consciousness. As someone who’s lived in big cities and seen health trends come and go, the risk always outweighs the thrill, no matter what’s trending on social media.
Balancing Access and Safety
Access to isoamyl nitrate stays limited because of safety concerns, but it keeps popping up in markets that regulators struggle to police. Harm reduction calls for more than stern warnings. Outreach teams can help people make healthier choices by offering plain facts about side effects, handing out safer alternatives for pain relief, or even providing free health checks for those who’ve used it.
Strict enforcement helps, but public education and honest conversations between doctors, pharmacists, and young people make the biggest difference. Unpacking the real-world risks and offering safer medical solutions builds trust and encourages people to ask smart questions before reaching for a quick fix.
Looking Forward
Isoamyl nitrate’s story brings together history, chemistry, engineering, and social trends. Knowing its background helps guide choices today, from protecting engine crews on frozen train lines to preventing harm in nightlife settings. In a world flush with new chemicals and old temptations, understanding each compound’s journey reminds us how science, medicine, and personal experience remain deeply connected.
Is isoamyl nitrate safe to use?
What Isoamyl Nitrate Does
Isoamyl nitrate brings up a lot of questions about risk and safety. Workers in specific industries, chemists, and recreational drug users have all come across this compound. In my chemistry labs during college, I saw isoamyl nitrate used for legitimate testing and cleaning. Its strong smell and rapid evaporation always left an impression. Some people recognize it as one of the chemicals in a group known as “poppers,” a term that has been tied to recreational use.
Clear-Cut Dangers
Isoamyl nitrate expands blood vessels. That’s the characteristic behind the intense head rush users describe, but it also means stress on the heart. When someone inhales the vapors, they’re cutting off oxygen in exchange for a quick high. One mistake—too deep a breath, using with other drugs, or mixing with alcohol—and the risk spikes sharply. Even for otherwise healthy people, a big enough dose can spark dangerously low blood pressure, dizziness, fainting, and in rare cases, even heart failure. I remember reading about people in my city passing out in clubs after pushing the limits with these chemicals. The stories always followed a pattern: nobody expected it, and things escalated fast.
Direct contact with the skin brings its own set of hazards. In labs, safety protocols call for gloves and fume hoods, not only because the liquid irritates the skin, but also because the fumes can cause headaches or nausea. I’ve seen technicians struggle with splitting headaches after only a brief exposure. Even the Centers for Disease Control and Prevention warns against inhaling the substance and advises immediate medical help in case of overexposure.
Regulation and Medical Warnings
Doctors and toxicologists don’t mince words: isoamyl nitrate use should not be taken lightly. The U.S. Food and Drug Administration has cracked down on over-the-counter availability, warning that products labeled as “liquid incense” or “room odorizer” often serve as code for poppers. Over time, heavy use can destroy red blood cells, making it hard for oxygen to travel where it’s needed in the body. People with anemia, heart problems, or blood pressure issues carry much greater risk.
Pursuing Safer Choices
Rather than chasing short-term effects, health professionals encourage honest conversations about substance use. If someone is considering experimenting, talking with a doctor first offers a safer path. Everyone has the right to informed choices, and pharmacists, poison control centers, or support groups can help untangle myths from facts. For those using isoamyl nitrate in work or research, following lab rules and respecting the substance’s dangers minimizes harm. Proper training, good ventilation, and regular review of emergency procedures can literally save lives.
Building a Healthier Attitude
Conversations about chemicals like isoamyl nitrate shouldn’t hide behind stigma or fear alone. Education and clear-headed discussion provide much more protection than keeping quiet. By staying honest about the potential harm, people can shape better decisions and look out for each other’s well-being. Knowledge won’t eliminate risk, but it gives people the freedom to choose wisely—and sometimes, that’s what really counts.
What are the side effects of isoamyl nitrate?
What Isoamyl Nitrate Does to the Body
Isoamyl nitrate gets a lot of attention as a recreational drug, especially in nightlife scenes. Breathing in its vapors creates a fast rush, a brief sense of euphoria, and even feelings of relaxation. Here’s the problem: that quick head high masks some real risks. This chemical acts as a vasodilator. Blood vessels widen, blood pressure drops, and the body has to scramble to catch up. Some people say it feels like a warm flush sweeping over them, but that reaction signals a sharp drop in blood pressure. A steep dip like that causes lightheadedness, dizziness, and even fainting. Fainting in public or while alone opens the door to head injuries and other accidents.
Short-Term Side Effects That Shouldn’t Be Ignored
Anyone tempted to try isoamyl nitrate might shrug off talk about mild side effects. Most people mention headaches, nausea, and rapid heartbeat. Those are common, yes, but here’s what stands out after speaking with folks who’ve used poppers: they often feel confused, their vision blurs, and some get chest pain. These are warning signs, not just minor annoyances. Reports from poison control centers show that breathing in isoamyl nitrate can also cause short-term memory loss or feelings of agitation. If someone already has heart issues or low blood pressure, a single use loads on serious risk.
Long-Term Damage and Rare Reactions
Long-term use brings different troubles. Chronic users develop skin rashes around the nose or mouth, sometimes called "poppers dermatitis." Blood oxygen drops with repeated use, and that means organs aren't getting what they need. Methemoglobinemia is one of the scarier effects—the blood can't carry enough oxygen. People with this condition show up in emergency rooms blue around the lips and gasping for air. Health agencies in multiple countries warn that just a few exposures can trigger this, especially in people with existing breathing issues.
Mixing With Other Drugs and Alcohol
Plenty of folks mix isoamyl nitrate with other drugs or alcohol. That's asking for trouble. Combining it with erectile dysfunction medication, like sildenafil (Viagra), creates a perfect recipe for dangerously low blood pressure. A combination like that lands people in hospitals every year. Mixing with stimulants, such as cocaine or ecstasy, strains the heart and doubles the risk of fatal arrhythmias. As someone who’s known folks in urban queer nightlife, I’ve seen accidents happen—not because users wanted to harm themselves, but because it’s easy to misjudge the risks in the moment.
Solutions: Safer Choices and Honest Conversations
Addressing these dangers means sharing facts and not shaming people for what they do. Harm reduction programs work better than scare tactics. Outreach teams in places like San Francisco listen to what people need, hand out information, and encourage regular health checks. Medical staff in clinics keep an eye out for symptoms related to popper use and talk openly about safer alternatives for sexual and recreational experience. Legal regulation also plays a role. Some countries require clear labeling and restrict the sale to adults, sending a signal that this isn’t a harmless product for everyone. Open conversations about side effects lead to fewer emergency room visits and much wiser decision-making.
How should isoamyl nitrate be stored?
Understanding the Risks
Isoamyl nitrate barely gets mentioned outside of a few science labs and industry settings, but the chaos it can unleash if forgotten in the wrong conditions is worth a hard look. Clear memory here: In college, a glass bottle broke in the lab’s refrigerator. The sharp, sweet odor cut through everything. Panic hit, then we scrambled to clean things up, eyes watering. After that, the lesson stuck—take chemical storage seriously.
Straight Talk: Why Storage Matters
Isoamyl nitrate isn’t just another flammable liquid—it’s got some edge. With a low flash point, this compound easily catches on fire if stored carelessly. Even small leaks or exposures to air can spell big trouble. Health-wise, it’s no joke either; prolonged exposure might lead to headaches or even something worse if fumes build up.
Common Sense Practices
Lab safety manuals suggest common steps for a reason. Keep isoamyl nitrate in tightly sealed glass containers. Plastic can react, so glass wins this round. Avoid storing it anywhere near sources of heat like radiators, ovens, or sunlight-baked windowsills. The chemical likes the dark.
Personal story backs this up. Working in a university prep room, a colleague left a small vial near a sunny bench. Temperature climbed, pressure inside the vial built up, and we nearly lost some lab notes to a small fire. The memory makes me careful whenever handling anything even remotely similar.
Ventilation and Security
A locked, dedicated cabinet for flammable chemicals inside a ventilated storage area keeps the risk of fumes and accidents low. No point cutting corners here. Good ventilation whisks away any escaping vapors. It’s tough to forget the sting in your eyes or a tightness in your chest after just seconds of exposure, so extra air flow is worth every penny.
Labeling and Awareness
Every bottle or container gets a clear, easy-to-read label—no scribbles or fading ink. Expired supplies go straight to hazardous waste bins, not back onto the shelves. Too often, old stock builds up because of lazy habits, only to be rediscovered during a crisis. If co-workers or labmates don’t recognize or understand the risk, chances grow for a dangerous slip-up.
Emergency Prep and Training
Accidents don’t announce themselves. Every person who walks into a room with isoamyl nitrate should know where to find spill kits, fire extinguishers, and goggles. Training matters. After every scare, those drills feel less of a chore and more like life insurance.
Regulations and Taking Responsibility
OSHA, CDC, and local fire codes set strict rules for a reason. Facts show improperly stored chemicals cause hundreds of fire incidents each year in labs and industry, which cost millions. Old habits die hard, but making sure chemicals stay in the right place at the right temp, away from danger zones, isn’t negotiable. Remembering past mistakes, and hearing stories from older chemists, drives home the importance of taking these steps. Safety really can’t wait for a reminder from tragedy.
Is isoamyl nitrate legal to purchase and possess?
Understanding the Law on Isoamyl Nitrate
Isoamyl nitrate, commonly known as a “poppers” ingredient, runs into lots of debate and confusion. Some see it as a simple chemical with a small medical history. Others know it only as the thing people inhale at clubs. But legality doesn’t just come down to how folks use something — it depends on how governments classify a molecule and how authorities treat the people selling or carrying it.
Federal law in the United States doesn’t make isoamyl nitrate a controlled substance. It’s not on the main lists beside drugs like heroin or cocaine. That said, not being scheduled doesn’t offer a free pass. The Food and Drug Administration (FDA) banned alkyl nitrites sold for human consumption in 1988, using a part of drug law that covers anything people might take for a “high.” The FDA said these could cause serious health problems, such as blood pressure drops, dizziness, and damage to the eyes. If someone bought or sold isoamyl nitrate where it was labeled or marketed for recreational use, both seller and buyer risk trouble with the law. That’s where the gray area comes in: sellers often call these bottles “room deodorizers” or “leather cleaners” to dodge that FDA rule, even if everyone knows why they’re really on the shelf.
The Reality of Purchasing Isoamyl Nitrate
Walking into any smoke shop in certain cities makes it hard to miss tiny vials with barely-there labels. They rarely say anything about inhaling, but the message is clear enough. Stores carry them because enforcement often focuses more on how they’re sold than the substance itself. The patchwork of laws between states and cities leads to confusion. For example, California has cracked down on sales and made arrests, even raiding shops. Texas has stricter local laws in some places. In other states, police might look the other way unless the products appear near schools or come with open encouragement to use them as drugs.
Online stores sometimes make matters worse, sending such products across state lines in boxes that claim “not for human consumption.” Shipping might seem safe — until a package gets flagged, and now someone’s explaining to authorities why a dozen bottles of isoamyl nitrate are sitting in their mail.
Why This Matters for Everyday People
Whether someone is curious or simply worried about what’s in these bottles, the stakes run higher than many realize. Toxicology reports and poison control centers keep seeing calls about accidental overdoses or mixed use with other drugs. The CDC published papers on hospitalizations linked to alkyl nitrites, including isoamyl nitrate. Eye damage, shortness of breath, and heart issues top the list. In my own years helping on a volunteer medical team at music events, I watched panicked partiers burst in seeking help after passing out or bleeding from the nose. None expected big trouble from “something legal at the shop down the street.”
Blocking these dangers means pushing past myths about harmless highs. The best fix starts with honest labels and more conversations within communities. Young people especially could use straight, clear education before they ever run into a bottle. Doctors and first responders know what these chemicals can do, but word spreads slowly among those most likely to try them.
More consistent labeling laws, better retail enforcement, and real public health campaigns would clear up confusion. Everyone deserves to know both the legal risks and the health risks before making a decision at the register. Sometimes that means listening to the people who see the fallout up close.
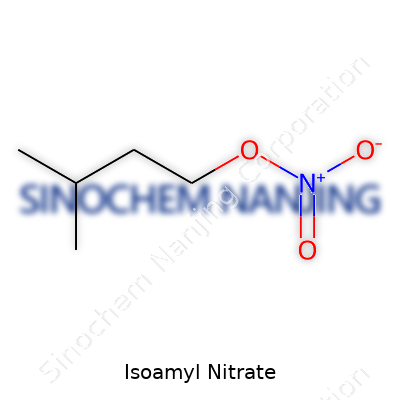
| Names | |
| Preferred IUPAC name | 3-methylbutyl nitrate |
| Other names |
Isopentyl nitrate 3-Methylbutyl nitrate Isoamyl nitrite |
| Pronunciation | /ˌaɪ.soʊˈæm.ɪl ˈnaɪ.treɪt/ |
| Identifiers | |
| CAS Number | 110-46-3 |
| Beilstein Reference | 1090828 |
| ChEBI | CHEBI:53093 |
| ChEMBL | CHEMBL133579 |
| ChemSpider | 8350 |
| DrugBank | DB01422 |
| ECHA InfoCard | ECHA InfoCard: 01-2119486972-18-0000 |
| EC Number | 208-779-0 |
| Gmelin Reference | 8144 |
| KEGG | C06502 |
| MeSH | D009907 |
| PubChem CID | 6277 |
| RTECS number | NT8050000 |
| UNII | 1F8VWT3BM6 |
| UN number | UN1205 |
| CompTox Dashboard (EPA) | DTXSID9020635 |
| Properties | |
| Chemical formula | C5H11NO3 |
| Molar mass | **117.15 g/mol** |
| Appearance | Yellowish liquid |
| Odor | fruity odor |
| Density | 0.872 g/mL at 25 °C |
| Solubility in water | slightly soluble |
| log P | 2.8 |
| Vapor pressure | 24 mmHg (20°C) |
| Acidity (pKa) | 15.1 |
| Basicity (pKb) | 12.96 |
| Magnetic susceptibility (χ) | -7.2×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.408 |
| Viscosity | 0.763 cP (20°C) |
| Dipole moment | 1.514 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 230.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -234.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1654 kJ/mol |
| Pharmacology | |
| ATC code | V03AB06 |
| Hazards | |
| GHS labelling | GHS02, GHS06 |
| Pictograms | GHS02,GHS06 |
| Signal word | Danger |
| Hazard statements | H226, H302, H332, H335, H351, H372 |
| Precautionary statements | P210, P261, P271, P304+P340, P312, P403+P233 |
| NFPA 704 (fire diamond) | 1-4-3-W |
| Flash point | 13°C |
| Autoignition temperature | 194 °C |
| Explosive limits | 1.1–8.0% |
| Lethal dose or concentration | LD50 (oral, rat): 505 mg/kg |
| LD50 (median dose) | LD50 (median dose): 5050 mg/kg (oral, rat) |
| NIOSH | SA9400000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Isoamyl Nitrate: "Amyl nitrite: OSHA PEL = 0.1 ppm (skin), 0.8 mg/m³ (skin) |
| REL (Recommended) | 100 ppm |
| IDLH (Immediate danger) | 300 ppm |
| Related compounds | |
| Related compounds |
Amyl nitrite Isopropyl nitrate Methyl nitrite Ethyl nitrite n-Butyl nitrite Isoamyl alcohol |

