Understanding Iron Hydroxyoxide: Past, Present, and Where It’s Headed
Historical Development of Iron Hydroxyoxide
Iron hydroxyoxide shows up throughout history, woven into everything from ancient pigments to modern industrial processes. Ancient Egyptians used it in ochre paints. By the 18th and 19th centuries, chemists unraveled its formation in rust, and its role as a base for many corrosion-resistant coatings. Iron hydroxyoxide doesn’t carry the same allure as gold or platinum, but it shapes our infrastructure, influences environmental science, and helps in medicine. Generations of researchers and metallurgists have refined ways to control it—initially as an annoyance to be removed from iron, now as a valuable material in its own right. Over time, its image shifted from an industrial nuisance to a resource with a steady spot in technology, catalysis, and even water treatment.
Product Overview
Iron hydroxyoxide falls somewhere between simple rust and advanced engineered materials. Its powdery or granular appearance hides a versatility that serves chemists and manufacturers alike. I’ve seen it packaged as yellow or reddish-brown powder, shipped in drums, and used without fanfare in processes ranging from pigment blending to contaminant removal. Some know it by other names—it isn’t always labeled “iron hydroxyoxide”—yet it remains a regular fixture in labs and plants. Its understated role in everything from agriculture to environmental remediation keeps it close to the foundation of industrial chemistry.
Physical & Chemical Properties
Iron hydroxyoxide looks unremarkable—a yellow or brown powder, often hydrophilic, sometimes gritty, sometimes smooth. It comes in various crystalline forms, but goethite (α-FeO(OH)) or lepidocrocite (γ-FeO(OH)) represent the most familiar faces. It doesn’t dissolve in water but suspends easily, offering a tacky surface that grabs onto heavy metals. Under a microscope, the particles show needle-like or plate structures. At room temperature, its thermal stability holds up until 250–300°C, beyond which it converts to iron oxide. Its amphoteric nature lets it react with acids to form iron salts and with bases to yield complex ions. These features don’t stand out on glossy marketing sheets, but they underlie most of its applications—especially when efficiency hinges on how a substance grabs, holds, and changes other compounds.
Technical Specifications & Labeling
Buyers in the industry expect labels that name the relevant phase—goethite, lepidocrocite, or even “hydrated iron oxide.” Product datasheets typically specify iron content above 55%, moisture content under 15%, and trace heavy metals kept tightly controlled. Granule size matters, since filtration systems require consistent particle diameter. I’ve reviewed many MSDS and shipment documents highlighting pH, bulk density, and purity checks. Detailed labeling has only grown in importance, especially as regulations climb and customers demand tighter consistency for safety and performance. Manufacturers who cut corners here often end up losing trust or facing complications in product recalls.
Preparation Method
Lab synthesis of iron hydroxyoxide tends to follow a path of dissolving iron(III) salts in water—like ferric chloride or ferric sulfate—and adjusting the pH past 7 with sodium hydroxide or ammonium hydroxide. The moment the pH hits that sweet spot, a yellow-brown precipitate appears. Industrial setups scale this approach, introducing controlled agitation and precise dosing to yield consistent crystals. Methods for improved purity often include repeated washing and filtration, while tweaks to temperature and pH unlock different crystal habits. I’ve watched factory setups cycle batch after batch through reactors, then dry the precipitate to a powder before packaging or milling to spec. Strong attention to water quality, tight environmental controls, and careful waste management helps control the downstream environmental impact—no small detail considering regulatory scrutiny.
Chemical Reactions & Modifications
Iron hydroxyoxide finds itself at the center of some fascinating chemistry. Expose it to acids and it shifts into iron (III) salts—think ferric chloride or ferric sulfate. Mix it with strong bases and you’ll find complex ions, all depending on the anion present. It doesn’t stay inert. Researchers clay-coat the surface, dope it with other metals, or calcine it to enhance magnetic and catalytic properties. In water treatment, functionalization with organic molecules helps tailor it to snatch undesirable metals. Its ability to lose or gain water molecules leads to forms like hydrated ferric oxide or transforms it, on heating, to pure iron oxide—hematite. Selective reduction opens up magnetite. These aren’t textbook reactions—industry routinely tunes properties like surface area, porosity, and reactivity for specific needs.
Synonyms & Product Names
Iron hydroxyoxide is rarely called the same thing twice. Some labels say ferric oxyhydroxide, others mention hydrated iron oxide, goethite, or yellow iron oxide. Trade names can sound cryptic, focusing more on color or origin than structure. If you browse pigment suppliers or environmental remediation catalogs, you’ll find “ferrihydrite” or references tied to mining regions. These aliases reflect both the chemical structure and centuries of shifting discovery. Buyers should dig through the technical data sheets, since two products with similar names may act very differently in real-world use.
Safety & Operational Standards
Handling iron hydroxyoxide feels routine but deserves real caution, especially in bulk. I’ve spent time on sites where airborne dust results in respiratory irritation during pouring or mixing. Workers must suit up with N95 masks, gloves, and splash-protective eyewear, all mandated by OSHA and European REACH. Keeping bulk storage dry prevents caking and maintains flow during feedstock delivery. Spills can stain equipment but, handled right, pose little long-term risk outside dust inhalation. Most suppliers mandate secure labeling and clear documentation for every shipment. Environmental standards require scrubbing wastewater and filtering solids before disposal. Compliance audits go beyond paperwork, with environmental managers double-checking that operations don’t let particles escape air filters or storm drains.
Application Area
Iron hydroxyoxide powers more industries than most realize. Its use as a filtering agent in drinking water treatment gives it a starring role in removing arsenic, phosphate, or heavy metals. Pigment factories rely on its color stability for art supplies and industrial paints. Soil remediation projects bank on its adsorption properties. Sewer plants apply it for phosphorus capture, cutting down algae blooms downstream. Chemists use it as a starting point for catalysts that drive important reactions. Even electronics manufacturers sometimes seek it for battery electrode precursors. Each application demands variations in purity, surface area, and physical form. The need for clean water and safe soil shows no signs of fading, giving iron hydroxyoxide a lasting job description.
Research & Development
Research around iron hydroxyoxide pivots on finding better ways to manipulate its form and function. Scientists hunt for increased surface area, tuning synthesis methods for nanoparticles or porous granulates. Environmental researchers optimize functionalization for selective adsorption of toxins. Battery engineers target new uses in energy storage, testing modified compounds against lithium or sodium ions. Universities keep pushing for green syntheses and recycling of waste iron into valuable products. Journals now fill with papers on plant-based or bacterial synthesis, seeking ways to lower costs and improve control. Collaborations between chemists, material scientists, and engineers underline just how many hands and minds contribute to every new breakthrough.
Toxicity Research
Consensus draws a line between minor irritation from inhaling dust and serious risk. Acute toxicity remains low for humans, which led regulators to focus on inhalation limits. In lab animals, high-dose studies suggest limited systemic absorption but raise questions for workers exposed for decades. Research rounds out with environmental assessments—iron hydroxyoxide stays inert in soils and does not seem to bioaccumulate, according to EPA studies, but its surface can absorb toxins that may later leach into groundwater if poorly managed. As regulatory bodies update their guidance, it falls on producers and users to stay vigilant, documenting every change in formulation and highlighting safety data to regulators and health professionals.
Future Prospects
Iron hydroxyoxide faces a future shaped by evolving needs. Water scarcity and pollution sharpen focus on better adsorbents for clean water. Battery technologies may call for iron-based compounds, not only to cut costs but to reduce environmental liabilities of rare metals. Bio-inspired synthesis and waste recovery open doors for green chemistry, cutting energy costs, and sidestepping toxic inputs. Climate change and stricter regulation fuel innovation in site remediation and resource recycling. As new applications demand more tailored products, those who dig deep into surface chemistry and engineering stand ready to shape the next era of iron hydroxyoxide.
What is Iron Hydroxyoxide used for?
Foundations in Pigments and Paints
Iron hydroxyoxide stands out in the world of color. Known to many as yellow ochre or a core component in earthy pigment blends, this mineral shapes the tone and durability of paints people use on their homes or studios. Long before synthetic colors flooded the market, natural iron compounds gave artists the tools for lasting murals and frescos. Even now, paint companies rely on this mineral for its lightfast nature and non-toxic profile. Children’s art supplies, for example, often use these pigments because they’re safe and don’t fade fast under sunlight. In road markings and protective industrial coatings, iron hydroxyoxide provides corrosion resistance and visibility.
Environmental Clean-up and Water Treatment
Water treatment plants turn to iron hydroxyoxide for help with filtration. Heavy metals can linger in drinking water, and this mineral gets used to grab arsenic, lead, or phosphate out of the solution. The science works through adsorption—pollutants stick to the iron compound, making it easier to separate clean water from dangerous particles. People living in areas with older water infrastructure depend on these systems to keep their taps safe. Environmental engineers have found it effective for treating mine runoff and tackling soil that’s been contaminated by industry or agriculture. So much of public health work happens behind the scenes, and minerals like this one make a direct difference in the water people trust.
Pharmaceuticals and Personal Care Products
Iron hydroxyoxide doesn’t just work outside the body. Pharmaceutical companies use it as an active ingredient in some antacids and iron supplements. Doctors often recommend these products to help patients build up iron stores, especially folks with anemia. Because the body can absorb the right forms of this mineral, it brings a safe, reliable way to boost iron levels. Personal care products—such as certain toothpastes and skin creams—also use tiny amounts. These compounds give color, stability, or mild abrasive qualities. On ingredient lists, it might appear under different names, but the science comes down to the same reliable chemistry.
Catalysis and Industrial Processing
Factories rely on iron hydroxyoxide for its ability to speed up chemical reactions. Refining petrochemicals or making ammonia for fertilizer both need catalysts to run efficiently. The mineral provides a surface that encourages reactants to meet and combine—saving time, energy, and raw materials. Some processes also harness its filtering properties, pulling out unwanted byproducts before they can escape into the air or water. Efficiency and waste reduction matter in high-volume settings, and these practical uses keep costs and pollution under better control. My own visits to manufacturing plants always highlight how these seemingly boring minerals have a real impact on global supply chains.
Finding Better Solutions
As research grows, the uses for iron hydroxyoxide seem to expand with it. Scientists explore new ways to recycle the mineral after it collects contaminants, aiming for less landfill waste. In renewable energy sectors, researchers test its properties for storage or conversion of solar power, offering potential improvements that reach beyond its historic uses. Staying open to these innovations gives society a way forward, harnessing both tradition and science to solve the next set of challenges.
Is Iron Hydroxyoxide safe for human consumption?
Looking Closer at Iron Hydroxyoxide
Iron stands out as a mineral everyone needs. It builds red blood cells, carries oxygen, and powers energy. The conversation about iron hydroxyoxide lands squarely in the space between chemistry class and the dinner table. Food grade iron compounds often sound more like lab ingredients than nutrients, but iron hydroxyoxide pops up in discussions because of its relationship to popular iron supplements and fortification strategies.
What Is Iron Hydroxyoxide?
All iron compounds aren’t created with the same goal. Iron hydroxyoxide occurs naturally, usually as a rust-colored mineral. Common names include ferric hydroxide or iron(III) hydroxide. Technically, it’s a form of oxidized iron you can spot on rusty pipes or sediments in water systems. Food manufacturers pick their iron compounds for bioavailability and absorption, not for industrial rust.
Food Use and Approval
People often ask about the safety of eating iron hydroxyoxide, especially when it comes to supplements or fortifying flours and cereals. So far, regulatory agencies in the US, Europe, and Japan stick to iron compounds like ferrous sulfate, ferrous fumarate, or ferrous gluconate. These compounds work better in the body. Iron hydroxyoxide never earned the same track record. Scientific reviews suggest it doesn’t absorb well from the gut, so it fails as a primary supplement source.
The US Food and Drug Administration keeps a list of compounds considered Generally Recognized As Safe (GRAS). Iron hydroxyoxide does not appear on this list for food supplementation. That should always remind people to check before trusting any supplement label. The European Food Safety Authority and the Japanese Food Safety Commission line up with this approach. They put more familiar iron salts on the allowed list and leave iron hydroxyoxide to the chemists.
What Happens If You Eat Iron Hydroxyoxide?
No strong poison risk comes from small accidental ingestion. The body passes much of it right through. My own experience with iron supplements taught me two lessons: absorption makes the difference, and any iron that fails to absorb can upset the stomach. Medical research shows most iron hydroxyoxide sits around, unabsorbed, and may contribute to constipation or discomfort. At high doses, undissolved iron gums up the works, occasionally turning stool dark or affecting gut bacteria.
Iron overdoses in kids nearly always come from flavored chewable tablets left lying around, packed with highly absorbable forms. Iron hydroxyoxide turns up more in water filters or as a side effect of rusty plumbing than on any nutritional label. If you see it on an ingredient list, it’s smart to take a second look; better absorbed forms actually help with iron deficiency and have decades of safety data behind them.
Better Options and Best Practices
Iron deficiency affects millions, and dealing with it means choosing forms that the body can use. Ferrous sulfate and ferrous fumarate remain gold standards—they show strong absorption, cost little, and have massive bodies of safety research. People with sensitive stomachs sometimes switch to ferrous gluconate. For pediatric and pregnancy iron needs, doctors often pick tried-and-true formulations and ignore uncommon compounds to keep things simple and safe.
Supplements should always match what’s proven and well-studied. Anyone concerned about iron levels or ingredient lists should look for the supplement facts panel, research allowed forms, or talk with a doctor or registered dietitian. Cheap, unfamiliar compounds like iron hydroxyoxide, usually found in industrial rather than nutritional products, never replaced the better choices available.
Takeaway: Stick with What Works
Iron hydroxyoxide doesn’t pose an acute danger. At the same time, it doesn’t feed your cells or solve dietary gaps. Iron supplementation works best with compounds the body recognizes and soaks up without confusion. Quality matters. If your energy wanes or your doctor talks iron, pick formulas with solid safety and absorption history, not just an exotic scientific name.
What are the physical properties of Iron Hydroxyoxide?
What You See: Color and Texture
Iron hydroxyoxide usually grabs attention with its earthy hues. You’ll spot it by its deep reds, rusty oranges, and sometimes ochre-yellow shades. These colors show up thanks to small differences in its structure and trace elements in the rocks or soils where it forms. The mineral often appears as a fine-grained, powdery deposit or it can take a more solid, massive form. If you take some between your fingers, it feels almost chalky. It smears easily and leaves colored streaks behind—just ask anyone who’s ever handled a rusted garden tool.
Feel and Form
This material often occurs in two main shapes: goethite and lepidocrocite. Goethite tends to be harder, a bit fibrous, and tougher under pressure. Lepidocrocite shows off a softer, more velvety surface. On the Mohs scale of mineral hardness, iron hydroxyoxide usually falls between 3.5 and 5.5. For comparison, a steel knife lands around 5.5, so the mineral won’t scratch steel but can mark softer rocks and surfaces. This makes removal and processing less energy-intensive when mining or cleaning water wells full of these deposits.
Density and Porosity
Iron hydroxyoxide doesn’t weigh down the pockets. It’s denser than many silicate minerals—goethite shows about 4.2 grams per cubic centimeter. Still, loose deposits or weathered forms collect water and air in their gaps. The porosity of these deposits makes them handy for trapping impurities in treatment plants and natural wetlands. High surface area gives them a leg up when it comes to filtering out heavy metals, arsenic, or phosphate from drinking water. Studies and real-life use in rural wells prove their value.
Thermal and Magnetic Behavior
As for heat, this mineral holds steady up to moderate temperatures. Heat it past about 200°C and you’ll spot changes—it can lose water and shift into hematite, another iron oxide, which carries different magnetic traits and color. Put a magnet near iron hydroxyoxide and you’ll see weak attraction, especially if you test a pure sample. This property means scrap yards and recovery sites can pull iron-rich dust out with magnetic separators. It’s basic, but it works.
Why These Traits Matter
These simple traits matter for real reasons. Anyone using iron hydroxyoxide in soil cleanup, paint pigments, or as drinking water filters actually counts on its colors and porosity. Kids spot it in playground soil, farmers track rust in their equipment, and families in parts of the world use it to make water safe for drinking. Pure science meets daily life. With more research, iron hydroxyoxide stands to solve even bigger environmental problems—helping folks keep water clean without high-tech solutions. If the mineral gets processed more efficiently, supply lines for eco-friendly paints or water purifiers could stretch much further. Down the road, simple tools using this mineral might bring cleaner water to people everywhere, not just to those with big budgets and advanced labs.
Possible Paths to Better Use
A good next step would be finding smarter ways to gather and clean iron hydroxyoxide from natural sources. Community groups in parts of Africa and Asia already use it to trap contaminants, but more local processing could stretch budgets and reach more homes. Universities and startups are teaming up to blend ancient know-how with new tech, testing better filters and even low-impact paints. Small changes to how the mineral gets used or combined can open new doors for villages and cities wrestling with pollution. Success means people get a practical, low-cost tool to tackle real problems—one handful at a time.
How should Iron Hydroxyoxide be stored?
Iron Hydroxyoxide Isn’t Just Rust
Folks might recognize iron hydroxyoxide from schooldays, maybe during lessons on rust or pigments. But this compound doesn’t behave like a chunk of old rebar in the backyard. It exists in everything from water treatment facilities to artists’ studios. Storing it right prevents bigger problems—think spoiled product, headaches for staff, or even risks to health and the environment.
Moisture and Air: The Usual Suspects
Leaving iron hydroxyoxide open to humidity almost guarantees clumping, which changes how it mixes and spreads. In one lab, a colleague forgot to seal a jar during a muggy spell. Clumps formed overnight, tough to break apart even with stubborn grinding. That ruined a paint batch and cost extra money. Dry conditions, sealed containers, and cool shelves kept the next batch sound. That simple change saved time and stress.
Oxygen in the air spells trouble too. Oxygen exposure can shift the compound into different forms, ones that lose their properties fast. Some suppliers pack it under nitrogen or in vacuum-sealed bags. This isn’t overkill—just smart chemistry. Any tricks that limit oxygen can stretch shelf life by months, sometimes years.
Container Choices Make a Difference
Folks working with iron hydroxyoxide soon realize not all containers do the trick. Paper sacks draw in water and let the powder spill everywhere. Plastic works, but only if it seals tight and resists punctures. Glass keeps the powder dry, but larger volumes turn clumsy, and glass shatters under rough handling. I trust thick-walled plastic or steel drums lined with moisture barriers. Those containers travel well and hold up during long storage. Labels matter—missing a “do not expose to moisture” warning caused one supply room to flood with orange dust after HVAC broke down. Always check that every drum or jar gets a bright warning label about air and water.
Avoid Sparks and Heat
Iron hydroxyoxide seldom draws complaints about fire. Still, concentrated powders need care near electrical panels or equipment with a hot surface. Once, I watched a storeroom worker dump a half-bag close to a welding bay. Sparks landed nearby; nothing happened, this time. But fine powder plus careless spark invites disaster eventually. Store the compound away from sources of heat and static electricity, just like flour or powdered sugar. Good procedures mean fewer close calls.
Spills Aren’t Harmless
Wet iron hydroxyoxide stains concrete, skin, and clothing deep orange. Cleanup isn’t pleasant—scrubbing and solvents don’t always fix the mess. Vacuums with HEPA filters collect dry spills fast. The right mats and trays under drums catch accidental leaks, too. Gloves and dust masks protect staff. Maintenance crews remember the time someone tracked red dust all over HQ. Now, a double-door policy and disposable boot covers stop repeat problems.
Regulation Isn’t Just Red Tape
Government rules around industrial chemicals read like a hassle, until something goes wrong. Iron hydroxyoxide sits on several safety and reporting lists, especially for facilities near rivers. In one business, regulators dropped in just after a shipment arrived. Staff only dodged a fine by showing airtight logs detailing storage temperatures, humidity readings, and leak checks. Smart managers keep a logbook and run regular checks so inspectors—and more importantly, staff—know the compound isn’t putting anyone in harm’s way.
Solutions for Smoother Storage
Start with dry, airtight containers. Store them on shelves, not floors. Mark labels in bold. Keep bags and drums capped right after dipping or weighing out product. Train all staff on spill drills and personal protective gear. Review local laws, don’t just rely on what a supplier says is “standard.” Small habits add up and stop big headaches. Every batch that stays powdery and pure saves money and keeps everyone safer on the job.
What is the difference between Iron Hydroxyoxide and Iron Oxide?
The Basics of Iron Compounds
Iron sits at the core of a lot of industries and everyday products. Some folks might only think of rust or steel, but in the chemical world, iron compounds have real personalities. The most common you’ll come across are iron oxides and iron hydroxyoxides. On the surface, both sound similar, but their chemistry and how they behave reveal important differences.
How Science Puts Them Apart
Iron oxide forms when iron meets oxygen. The most familiar form—rust—turns up everywhere from old bike chains to red rocks in the American Southwest. The formula, Fe2O3 or FeO, tells you there’s only iron and oxygen holding things together. Iron oxide pigments help color bricks, cosmetics, and even artist paints.
Now, iron hydroxyoxide adds another twist. It brings hydrogen into the mix, usually showing up as FeO(OH) or Fe(OH)3. That single “hydroxy” group means water plays a role in its formation, often from iron’s reaction to both water and oxygen. The yellowish-brown streak of limonite, or the ochre you find in cave paintings, points to hydroxyoxide’s presence.
The Role Each Plays
Iron oxide proves tough and stable. This trait works well in pigments that care about not fading in sunlight or washing away in the rain. Iron oxide’s resistance is one of the reasons folks trust it in sunscreen and tattoo ink.
Iron hydroxyoxide tells a different story. It reacts easy with acids and dissolves in water under certain conditions. Water treatment plants count on this feature when removing arsenic from drinking water. That means iron hydroxyoxide isn’t just a pigment or leftover from metal rusting—it's a reactive helper in cleaning some of the dirtiest water on the planet.
Health, Environment, and Everyday Effects
No one wants extra iron floating around in groundwater or house paint. Iron oxide doesn’t dissolve in water or create much worry unless someone breathes in fine particles day after day. Miners and factory workers might risk “siderosis,” a lung condition, but it doesn’t turn toxic inside most homes.
On the other hand, iron hydroxyoxide plays a part in cleaning up heavy metals and poisons in soil and water. Take Bangladesh, where natural arsenic in groundwater causes health troubles for millions. Simple sand filters packed with iron hydroxyoxide catch that arsenic before anyone drinks it. That sort of direct health link makes understanding differences more than just book learning. Access to safe water means communities thrive.
Smarter Use and Future Choices
Getting the right chemical for the job can reduce waste and save money. Iron oxide lasts long in bricks and paints. Iron hydroxyoxide, with its knack for grabbing toxins, helps with sustainable water treatment. Parents, builders, and policy makers do better when they recognize what each iron form does.
Scientists keep finding new uses. Modified hydroxyoxides now scrub wastewater of dyes or pharmaceuticals that once flowed back into rivers. Iron oxide nanoparticles help clean oil spills and deliver cancer drugs. It surprises people how simple minerals can solve complex problems, especially when ordinary folks work alongside researchers to put them to work in real communities.
| Names | |
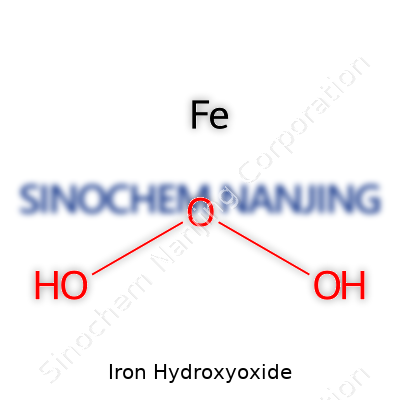
| Preferred IUPAC name | iron(III) oxyhydroxide |
| Other names |
Ferric oxyhydroxide Iron(III) oxide-hydroxide Ferric hydroxide Rust |
| Pronunciation | /ˈaɪərən haɪˌdrɒk.siˈɒk.saɪd/ |
| Identifiers | |
| CAS Number | 1310-14-1 |
| Beilstein Reference | 3527489 |
| ChEBI | CHEBI:31514 |
| ChEMBL | CHEMBL1201881 |
| ChemSpider | 22599348 |
| DrugBank | DB14148 |
| ECHA InfoCard | ECHA InfoCard: 100.031.863 |
| EC Number | 215-687-4 |
| Gmelin Reference | 85300 |
| KEGG | C18798 |
| MeSH | D017981 |
| PubChem CID | 16211982 |
| RTECS number | NO8560000 |
| UNII | E1UOL152H7 |
| UN number | UN3178 |
| Properties | |
| Chemical formula | FeO(OH) |
| Molar mass | 106.87 g/mol |
| Appearance | Yellowish-brown powder |
| Odor | Odorless |
| Density | D = 3.4 g/cm³ |
| Solubility in water | insoluble |
| log P | -37.568 |
| Vapor pressure | Negligible |
| Acidity (pKa) | ~6.0 |
| Basicity (pKb) | 15.3 |
| Magnetic susceptibility (χ) | +1800·10⁻⁶ |
| Refractive index (nD) | 1.897 |
| Dipole moment | 0.00 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 83.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -490.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -624 kJ/mol |
| Pharmacology | |
| ATC code | B03AB05 |
| Hazards | |
| Main hazards | May cause irritation to eyes, skin, and respiratory tract. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS07,GHS08 |
| Signal word | Warning |
| Hazard statements | Hazard statements: Not a hazardous substance or mixture according to Regulation (EC) No. 1272/2008. |
| Precautionary statements | Keep out of reach of children. Avoid breathing dust. Wash hands thoroughly after handling. Use only outdoors or in a well-ventilated area. Wear protective gloves/protective clothing/eye protection/face protection. |
| NFPA 704 (fire diamond) | 1-0-0 |
| Lethal dose or concentration | LD50 (Oral, Rat) > 5,000 mg/kg |
| LD50 (median dose) | > 5000 mg/kg (rat, oral) |
| NIOSH | Iron Hydroxyoxide: "NO4680000 |
| PEL (Permissible) | 5 mg/m3 |
| REL (Recommended) | 25 mg as iron |
| Related compounds | |
| Related compounds |
Iron(II) oxide Iron(III) oxide Iron(II) hydroxide Iron(III) hydroxide Iron oxyhydroxide |

