Iron Arsenite: Story, Properties, and Prospects
Historical Development
Traces of iron arsenite date back to the earliest days of analytical chemistry, when researchers began exploring arsenic compounds for both their vivid colors and mysterious toxicity. Before the modern periodic table came together, chemists already used iron arsenite for pigments, and people mined arsenic minerals out of curiosity about their peculiar traits. Over the decades, laboratories and industry learned to treat the compound with respect. In the nineteenth century, iron arsenite captured attention for its green tint, turning up in paints and wallpapers—though the cost soon became clear when toxic arsenic dust leached out. Later, the expansion of environmental regulations forced everyone to reconsider how and where iron arsenite belongs.
Product Overview
Iron arsenite generally exists as a hydrated salt, FeAsO3, or sometimes in varying hydration states. You won’t find it on household shelves, nor does it pop up in consumer products. Labs and industrial sites ask for this compound in specific grades, depending on what they need—often for research, pigment analysis, or very narrow uses that tap into its unique mix of reactivity and toxicity. Iron arsenite brings forward properties from both iron and arsenic chemistry, a mix relevant not just to industrial users but also to anyone studying heavy metal pollution.
Physical & Chemical Properties
Iron arsenite crops up in the lab as a yellowish or pale green powder, sometimes as a crusty crystalline solid. It dissolves poorly in water, a trait that has both advantages and disadvantages. On one hand, limited solubility means runoff is slower, so less arsenic leaches into the environment in some situations. On the other hand, proper disposal becomes a challenge. This compound stays fairly stable at room temperature, though heating or strong acids break it down to release both iron and arsenic ions—neither of which you want loose in water supplies. Under the microscope, it often reveals a fairly nondescript crystal form, unless it gets mixed with other transition metals, which can alter color and texture.
Technical Specifications & Labeling
I’ve seen most iron arsenite come packaged in sealed, moisture-proof containers, often labeled with hazard warnings as required by REACH, OSHA, and other chemical safety agencies. Typical labeling cites a purity range based on the manufacturing process, usually above 98% for lab-grade materials, with trace metals, silica, or sulfates as common contaminants. Every batch ships with a Material Safety Data Sheet (MSDS) listing health risks, ecological hazards, recommended personal protective equipment, and emergency spill procedures. Labels do not sugarcoat things—the word “toxic” stands out, as it should, alongside “environmental hazard” and advice to keep away from acids and strong oxidizers.
Preparation Method
Most iron arsenite starts out as a reaction product between ferrous salts, like ferrous sulfate, and sodium arsenite or arsenic acid solutions. You pour the two solutions together under controlled conditions, usually in a fume hood, and out comes a precipitate of iron arsenite, which you filter, wash, and dry. Extra steps help to remove any leftover reactants or byproducts, since impurities can radically shift how the compound behaves. The whole process needs tight controls on temperature, mixing speed, and pH to get a useful yield; without those, the reaction products drift toward other arsenic-iron species or remain suspended in solution.
Chemical Reactions & Modifications
Iron arsenite doesn’t sit quietly in every setting. Start adding oxidizers, and the arsenite ion converts into arsenate, which has a whole different reactivity profile and greater environmental mobility. On the flip side, sulfur or phosphorous additions can tweak the compound, shifting its stability or making it less mobile in soils. Strong acids dissolve the solid, releasing iron and highly toxic arsenic(III) ions, which makes spills or improper storage incredibly dangerous around waterways. Some research groups have tried doping the structure with zinc or manganese to explore how such changes might trap arsenic or alter its toxicity, especially for applications in contamination remediation or pigment manufacturing.
Synonyms & Product Names
People don’t always use the same name in every context. You might find iron(II) arsenite, ferrous arsenite, or even just generic “iron arsenite” in catalogs and safety protocols. In mineralogy, scorodite sometimes stands in as the natural iron arsenite hydrate, though this mineral carries more water in its structure than the lab-made material. In pigment industries, old labels like “arsenite green” or “Scheele’s Green” referred to similar but distinct compounds, sometimes loaded with copper or mixed with other transition metals.
Safety & Operational Standards
Handling iron arsenite calls for maximum respect. I’ve seen compulsory use of gloves, goggles, fume hoods, and chemical-resistant aprons in every lab or plant that works with it. Wastewater treatment and safe disposal rules follow strict guidelines—incineration isn’t the answer, and landfill disposal requires secure encapsulation. Occupational health standards force ventilation and spill containment at every handling step, especially to protect workers from inhaling dust or risking skin absorption. Regular air and surface monitoring keeps accidental exposure in check, while extensive documentation trails give regulatory agencies confidence that contamination risks are under control.
Application Area
Most current uses of iron arsenite fall into research and niche remediation projects, not bulk manufacturing. The pigment industry has largely stepped back from arsenic-based compounds because of toxicity, though historical pigments still appear in art conservation studies and forensic analysis. Soil remediation researchers look at iron arsenite as a way to immobilize arsenic in polluted sites, creating less soluble forms that leak less into groundwater. Analytical chemists use it to investigate how arsenic migrates through soil or water, since it represents a form that shows up in certain mining or smelting waste sites.
Research & Development
Labs keep digging into iron arsenite’s crystal structure, reactivity, and interaction with biological systems. One focus looks at controlling how iron arsenite forms in abandoned mines and whether engineered bacteria or chemicals might turn mobile arsenic into this less soluble form—kind of locking up the poison underground. Materials scientists examine how iron arsenite interacts with clays or other geologic materials, searching for ways to stabilize contaminated sites. Some researchers tackle the challenge of breaking down arsenic-containing waste without creating new hazards; for iron arsenite, that means efficient conversion or immobilization, not just moving the problem somewhere else.
Toxicity Research
The toxicology of iron arsenite links closely to the infamous dangers of arsenic(III) species. Inhalation, skin contact, or accidental ingestion all bring significant risks—acute poisoning, cancer, and chronic organ damage lie on the table with enough exposure. Animal models show that the arsenite part hits enzymes directly, blocking sulfhydryl groups and disrupting metabolism. Even brief exposures at low levels cause trouble over time. Regulators set extremely low thresholds, and water quality standards treat any arsenite form as an urgent concern. The iron component adds its own issues at high doses, though the dominating worry always circles back to the arsenic content.
Future Prospects
Cleaner chemistry and stricter safety rules have pushed direct uses for iron arsenite to the background, but its relevance in environmental science only grows. People want solutions for legacy pollution, and iron arsenite’s ability to lock up mobile arsenic gives researchers hope for cost-effective cleanups. New synthetic approaches may improve stability and lower incidental risks, especially as engineered nanoparticles or composite materials get into the mix. The next generation of work will probably focus on environmental containment, real-time monitoring, and new ways to destroy or neutralize arsenic compounds once and for all—less about making pigments or chemicals, more about cleaning up old mistakes while guarding against new ones.
What is Iron Arsenite used for?
A Look at Iron Arsenite in the Real World
Iron arsenite often gets tucked away in textbooks or databases, but its real actions sit in some complex corners of industry and environmental science. With a reputation tied to both minerals and toxicity, iron arsenite isn’t something people stumble across at the hardware store. Still, it’s important to take a closer look at what it actually does and why we should pay attention.
Industrial Legacy and Use
In the past, iron arsenite mainly served a purpose in pigment production. Dark greens and browns, especially in 19th-century paints and wallpapers, used this and related arsenic compounds. Artists and manufacturers valued the staying power and unusual hues these minerals gave. It wasn’t uncommon to find traces in Victorian homes, a detail which still sends shivers down the spines of restoration experts today. Part of my own interest in chemistry grew out of hearing stories about those arsenic “poisoned” greens in old paintings.
Today, its role in artistic materials nearly vanished because of well-founded health concerns. Most modern uses involve tightly controlled environments, like certain lab-based syntheses that rely on its properties as a mineral source or reagent. Some rare mineral collectors and geologists still encounter iron arsenite in naturally occurring mineral forms, usually in mining regions or excavation sites.
Health and Environment Risks
Iron arsenite contains arsenic, which is toxic to humans and most animals. Arsenic can cause everything from gastrointestinal problems to severe organ damage, even cancer. I’ve worked in older science labs where warning labels around storage cabinets weren’t just for show. Handling iron arsenite requires strict personal protective equipment, ventilation, and disposal protocols for good reason. Researchers must treat any compound containing arsenic with real respect for its power and potential for harm.
Leaks and waste left behind by industries from decades ago don’t just disappear. Some areas still struggle with arsenic leaching into groundwater, much of it coming from long-closed processing plants. The compounds break down slowly, contaminating soil and water, and clean-up efforts can drag on for years. Public health data regularly links elevated arsenic in drinking water with higher rates of skin lesions and cancers, adding urgency to environmental monitoring.
Solutions and Safer Practices
Modern labs and manufacturers place strict limits on any process that could release iron arsenite or other arsenic compounds into the open environment. Instead of using iron arsenite directly, safer alternatives often replace it in pigments and industrial chemistry. Those who still need to work with it follow hazard communication rules, well-tested containment, and robust waste treatment procedures. In my experience, people in the field don’t take shortcuts: regular air and water testing, clear labeling, and documented safety training really make a difference.
Remediation of old arsenic sites has become a science all its own. Techniques like soil washing, phytoremediation with special plants, and chemical stabilization turn dangerous waste into something manageable. Successful projects depend on clear oversight, honest reporting, and community engagement. I’ve seen greater trust develop in towns where cleanup crews share their data and involve residents. That way, the lessons from iron arsenite’s past help inform a safer, more transparent future.
Is Iron Arsenite toxic or hazardous?
Looking Beyond the Chemistry
Iron arsenite sounds like a mouthful from chemistry class, and to some people, it’s just a compound on a periodic table chart. It’s actually a mix of iron and arsenic in a certain ratio. Right out of the gate, anytime you see “arsenic” in a chemical’s name, alarm bells ought to ring. From my experience teaching basic science safety, arsenic comes up a lot when we talk real dangers—this element earned its notoriety both as a slow-acting poison in crime novels and as a real public health issue.
An Unmistakable Danger
Iron arsenite isn’t one of those chemicals you want collecting dust anywhere people eat, sleep, or breathe. Arsenic compounds in general are toxic, and iron arsenite shares that trait. Breathing in the dust or getting it on your skin can spell trouble. The problem grows if fine particles become airborne, drifting into lungs or sticking to hands and faces. Ingesting even a small amount of arsenic can have a nasty effect, and regular exposure raises the risk for problems like skin lesions, cancer, and nerve issues. Chronic arsenic poisoning has cropped up in cases where well water or environment was tainted from natural sources or mining spoil.
No Room for Guesswork
Nobody wants to find themselves scrambling after realizing toxic exposure happened because folks skipped reading up. I’ve seen workplaces where people treat everything as safe if it isn’t smoking or bubbling, and that can get people in trouble fast. The World Health Organization, the CDC, and OSHA all agree: arsenic-based minerals, including iron arsenite, require respect and serious handling. Exposure limits are set low for a reason.
Real Risks, Real Responses
Iron arsenite’s dangers don’t stop at personal health. Dumping it out into the ground or water spreads the hazard. Runoff from extraction sites with arsenic minerals endangers wildlife, seeps into groundwater, and stays there for decades. These facts aren’t just regulatory red tape—they come from hard experience and tracking real-world disasters where contaminated sites cost millions to treat.
In the classroom, I always told my students that hazard labels on jars serve as a warning, not just a requirement. Those skull-and-crossbones stickers didn’t get slapped on by accident. The science behind iron arsenite’s toxicity rests on a clear foundation: arsenic interferes with how our cells generate energy, damages DNA, and messes with critical enzyme systems. Iron doesn’t make the compound “slightly safer”; its presence just changes the chemistry, not the risk.
Keeping People Safe
For those who ever need to handle iron arsenite—whether in a lab, old industrial site, or during a clean-up project—safety comes down to airtight procedures. Protective gear matters: gloves, tight-fitting masks, sealed containers, and good ventilation. Decontamination steps mean washing up thoroughly after any exposure.
Regulation and watchdog groups demand companies report, label, and secure arsenic minerals at every step. Environmental cleanup teams use special absorbents and filtering technologies to catch and lock up arsenic before it leaches out. There’s no quick fix, and no cutting corners when arsenic compounds are part of the picture. Community watchdogs, government inspectors, and industry scientists must rely on each other to keep standards straight and keep workers and neighbors out of harm’s way.
What are the safety precautions when handling Iron Arsenite?
Recognizing the Risks in the Lab
Iron arsenite doesn’t play around. The stuff is made from iron and arsenic, which should ring alarm bells for anyone who’s dealt with chemicals in the lab. Arsenic exposure, even at low doses, has been linked to all sorts of health problems, including skin cancer, lung problems, and neurological issues. A lot of us remember lessons on how arsenic affects cells, and handling anything with arsenic content can feel a bit like working with an invisible threat wound tightly into the powder.
Personal Protective Gear Matters
The first thing to grab is protection for skin, eyes, and lungs. Skip the shortcuts—this is not a chemical to trust with a splash of good luck. Wear a lab coat, gloves—nitrile or neoprene, never bare hands—and safety goggles. Dust from iron arsenite travels further than most expect, so a particulate respirator is not just helpful, it’s required. Don’t brush your hair back or rub your nose with contaminated gloves.
I once heard about a technician who ended up with a persistent cough after routine handling of arsenic compounds in a poorly ventilated area. Maintenance staff later found powder residue on doorknobs and work surfaces. The lesson stuck—small exposures over time add up, especially when people get casual and skip on gear.
Keeping Contamination Out of Everyday Life
Iron arsenite can turn up in places you wouldn’t expect if people aren’t careful. Wash hands thoroughly before eating or leaving the lab, and never reach for a piece of fruit or a coffee cup with the same gloves that touched this stuff. Contaminated clothing can be just as risky. Nobody wants to carry dangerous dust home to family or onto public transport, so labs need laundry protocols or disposal bins for lab coats and gloves.
Safe Storage Stops Accidents
Dry, cool, and locked away—these should guide how to store iron arsenite. Don’t keep it with acids or oxidizers; reactions could get out of control fast. Clearly label containers, and choose ones that seal tightly to keep out humidity and block any leaks or dust from escaping. Responsible chemists never store hazardous chemicals near break rooms or exits, so separating storage makes sense. Regular checks of inventory pinpoint leaks or broken lids before they turn into real problems.
Ventilation and Cleanup Plans Keep Everyone Protected
Every place using iron arsenite needs good ventilation. Fume hoods pull particles away before they ever reach a technician’s lungs. Wet mop floors; don’t sweep up powder. Dry sweeping can send the compound into the air, ready for breathing. Spills shouldn’t be left for “later.” If any powder makes it outside its container, clean it right away, using damp wipes or a vacuum designed for hazardous dust, followed by safe disposal in dedicated waste bins.
Training and Emergency Prep Save Lives
Anyone working around iron arsenite ought to understand its risks and emergency steps. That’s more than a sign taped to a cabinet. Hands-on training, regular drills, and a clear plan give people the confidence to act fast if anything goes wrong. Keep poison control information and Material Safety Data Sheets where staff can reach them, not buried in a forgotten binder. Regular reviews and feedback sessions help everyone stay sharp.
Facing Environmental Impact Honestly
Proper disposal means treating iron arsenite waste like the hazardous material it is. Dumping it down the drain, burning it, or tossing it with ordinary trash threatens not just the handler, but water supplies, soil, and wildlife. Use only approved hazardous waste containers and partner with licensed waste handlers who document every step of the process.
The biggest truth about handling iron arsenite: prevention beats any cleanup. Prepared people, the right gear, and good habits make the difference between a routine task and a dangerous mistake.
What is the chemical formula of Iron Arsenite?
A Closer Look at Chemistry’s Building Blocks
Many people ask simple questions about chemistry, but even the basics can turn complex fast. Take iron arsenite. Its name might stir up memories from high school labs, or bring to mind dusty bottles with faded labels. Yet, the curiosity behind the formula for iron arsenite takes us deeper—both into the world of compounds and into practical realities that touch environmental science and health.
The Formula That Unlocks the Story
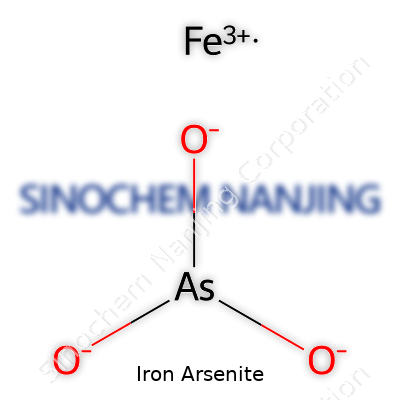
The chemical formula FeAsO3 captures iron arsenite in its simplest form. It contains iron (Fe), arsenic (As), and oxygen (O)—each offering properties that influence why this compound matters. People rarely see this combination outside of textbooks, but each part tells a story. Iron, carrying the +2 charge, links up with arsenite’s three oxygens and one arsenic to make the overall balance work. In real-world language, understanding FeAsO3 isn't just about memorizing symbols; it's about following a chain of chemical logic.
Iron Arsenite in the Wild—Or in the Lab
You probably won't see iron arsenite in household products. Arsenic compounds, as history shows, carry significant toxicity. Mining and mineral processing industries occasionally run into iron arsenites as byproducts. These industries must manage them well, since dissolving compounds like FeAsO3 in groundwater can threaten drinking supplies. I’ve seen environmental teams scramble to contain arsenic-laden runoff after heavy rains, rushing to shield local communities from long-term exposure risks.
Even a small lab using arsenite salts for experiments knows the risks: masks, gloves, and careful cleanup become standard. Nobody laughs off a spill when arsenic’s on the line—scientists learn to treat each compound with respect because the stakes reach beyond test tubes to public health.
Important Conversations: Health and Safety
Both the World Health Organization and the CDC have written at length about arsenic exposure and the cancer risks it creates. Chronic exposure to arsenites can damage organs and raise risks for skin and lung cancers. Drinking water in some parts of the world, particularly areas downstream from mining, still tests positive for unsafe arsenic levels. People working with iron arsenite, or living near such sites, face another layer of risk.
Regulation stands as the strongest barrier between industry practices and community health issues. Clear guidelines and good enforcement lower exposure. In my experience working with environmental remediation, strict handling and disposal rules go hand-in-hand with real results—less contamination and fewer sick days in affected communities. It’s not fancy technology; it’s simply enforcing what common sense and science connect.
Reducing Risk and Looking Forward
Iron arsenite will never become a household chemical. Far from making headlines, it remains buried in textbooks until someone accidentally releases it into soil or water. The lesson is straightforward: chemicals like FeAsO3 demand care and respect, both in how we study them and in how we handle them in the field. Strong regulations, wise disposal, prompt cleanup, and honest conversations about real-world hazards go a long way. Facts, not fear, drive safe choices—and that includes knowing the real story behind even the smallest formula.
How should Iron Arsenite be stored and disposed of?
Everyday Values Matter When Handling Hazards
Iron arsenite isn’t a household name, but the risks it brings show up in our daily lives more than we think. As someone who grew up in a small-town community where old mining waste still shapes local health stories, I’ve seen what happens when chemicals like iron arsenite get ignored or stored the wrong way. Families and towns pay the price, not distant corporations.
Storage: Why Simple Precautions Save Trouble
The way iron arsenite gets stored says a lot about a business or school’s commitment to safety. It shouldn’t touch the air and moisture outside. For long-term storage, chemical-safe cabinets with proper ventilation and corrosion-proof linings matter. Metal drums with tight lids or glass containers fitted with reliable seals block leaks and dust. Labels must stay bright and easy to read: faded stickers lead to mistakes.
I remember hearing from a mentor in the environmental field how just one crack in a storage drum once set off months of emergency cleanup at a university lab. Leaking arsenic-based compounds don’t just stay put—they find gaps in concrete, move with water, and end up in places no one expects. Storing containers in secondary containment trays, away from acids and bases, helps lower the risk of disastrous reactions or spills.
Disposal: Responsibility Doesn’t End at the Door
Throwing out iron arsenite isn’t something you hand off to the regular trash truck. Its arsenic content brings toxic risk for people and wildlife. Hazardous waste rules in most states require using certified chemical waste firms. These companies collect, track, and treat this material for safe destruction or storage in licensed hazardous landfills.
In one local case, workers poured spent chemical solutions down drains, thinking water systems would handle the problem. Water samples still show higher arsenic levels, proving risks stick around for years. Permanent solutions demand planning. Neutralization with chemicals like sulfide, under strict lab controls, can shift toxic forms of arsenic to less harmful ones, and solidification with cement-based methods can lock up iron arsenite, keeping it from dissolving into groundwater.
Community and Worker Awareness: A Missed Step
Even teachers or warehouse staff can end up managing dangerous chemicals without training. I’ve talked with custodians who discovered a leaking container by smell alone. Regular safety audits and honest conversation about what materials are on site goes further than layers of paperwork. Simple posters and annual safety briefings make a meaningful difference.
No one wants to think about the worst-case scenario, but spill kits for arsenic compounds should always be close by, with clear instructions for use. Local fire departments and emergency rooms stay safer when staff have the heads-up about chemical inventories nearby.
Solutions Start With Accountability and Transparency
Basic respect for health turns storage and disposal from a box-checking exercise into something that builds trust across generations. Real transparency means schools, labs, and plants post clear, public inventories and encourage local oversight groups to visit. This isn’t about bureaucracy—it’s about dignity for the people most likely to be affected. Keeping iron arsenite managed with care signals a culture that values prevention over quick fixes. Everyone benefits: workers, neighbors, and future generations who rely on clean water and safe workplaces.
| Names | |
| Preferred IUPAC name | iron(II) arsenite |
| Other names |
Ferrous arsenite Iron(II) arsenite |
| Pronunciation | /ˈaɪərn ɑːr.səˌnaɪt/ |
| Identifiers | |
| CAS Number | 1343-28-2 |
| Beilstein Reference | 4258896 |
| ChEBI | CHEBI:86158 |
| ChEMBL | CHEMBL1201562 |
| ChemSpider | 23045 |
| DrugBank | DB14606 |
| ECHA InfoCard | 100.012.597 |
| EC Number | 215-712-0 |
| Gmelin Reference | 8999 |
| KEGG | C18668 |
| MeSH | D017715 |
| PubChem CID | 166873 |
| UNII | 825F2O91HL |
| UN number | UN1512 |
| Properties | |
| Chemical formula | FeAsO3 |
| Molar mass | 229.775 g/mol |
| Appearance | White solid |
| Odor | Odorless |
| Density | 3.86 g/cm³ |
| Solubility in water | Insoluble |
| log P | -36.65 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 5.29 |
| Magnetic susceptibility (χ) | Paramagnetic |
| Refractive index (nD) | 1.924 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 147.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -924 kJ/mol |
| Pharmacology | |
| ATC code | B03AB01 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled or in contact with skin. |
| GHS labelling | GHS labelling: "Danger; H301, H332, H373, H400, H410; P261, P273, P301+P310, P304+P340, P405, P501; Skull and crossbones, Health hazard, Environment |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. |
| Precautionary statements | P260, P264, P270, P271, P272, P273, P280, P301+P310, P302+P352, P304+P340, P308+P311, P310, P314, P320, P330, P361+P364, P405, P501 |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 oral rat 20 mg/kg |
| LD50 (median dose) | 106 mg/kg |
| NIOSH | NIF6475000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Iron Arsenite: 0.01 mg/m³ |
| REL (Recommended) | 0.2 mg As/m3 |
| IDLH (Immediate danger) | IDLH: 5 mg As/m³ |
| Related compounds | |
| Related compounds |
Iron(II) arsenate Iron(III) arsenate Iron(III) arsenite |

