Iron Arsenate: A Deep Dive Into a Key Inorganic Compound
Historical Development
Iron arsenate earned its place in mineralogy and industry long before modern labs mapped its structure. My first brush with this compound came digging through old mining records, tracing its use in early pigment production. In the 1800s, miners stumbled across the mineral scorodite, a hydrated iron arsenate, and chemists isolated its basic formula not long after. Factories in Europe blended it into pigments and wood preservatives, frequently without strict rules on handling toxic materials. The compound also found its way into some early agricultural products used to control insects and fungi, which laid down both scientific and regulatory roots that shape its story today.
Product Overview
Iron arsenate usually shows up as a powder or crystalline solid, with colors ranging from greenish-blues to browns, depending on its water content and impurities. If you’ve ever looked through a catalog of laboratory chemicals or industrial products, you probably noticed iron(III) arsenate bulk jars. The material pops up in metallurgy, research, and—although less common now—certain ceramic glazes. Factories label the compound by trade names tied to its hydrate level and source, but the actual stuff inside acts predictably, with a reputation for both stability and, because of the arsenic, caution.
Physical & Chemical Properties
Iron arsenate doesn’t dissolve much in water, which changes its risk profile. In the lab, it resists heat up to several hundred degrees Celsius. The structure features iron cations paired with arsenate anions, building a lattice that blocks easy breakdown but will decompose with strong acids or bases. You’ll find that its density and color reveal clues about purity and hydration—hydrated forms show up lighter and bulkier, while anhydrous samples carry more heft. Handling it in the open, you notice the fine dust, which can become airborne and is best kept far from your face or food.
Technical Specifications & Labeling
Suppliers list technical specs with numbers that matter most to regulators and engineers: purity often runs above 99%, with iron and arsenate percentages checked by titration or spectrophotometry. Packaging features hazard labels for both toxic and environmental harm, plus batch numbers and MSDS sheets. I remember one shipment stamped with red “TOXIC” diamond labels—a reminder that even though the substance sits quietly in a jar, its risks call for respect. Each bottle usually includes solubility data, melting points, and warnings about environmental release.
Preparation Method
Preparing iron arsenate in the lab doesn’t involve exotic tricks. The classic method blends a soluble iron salt, like iron(III) chloride, with a soluble arsenate such as sodium arsenate or arsenic acid. The reaction kicks out a precipitate as soon as the two touch in aqueous solution. Filtering, washing, and drying the product generates either the hydrated or anhydrous form, depending on temperature and treatments. On the industrial scale, controlling pH and concentration ensures yields that meet the required standard, while any waste solution gets neutralized and checked for traces of arsenic before disposal.
Chemical Reactions & Modifications
What stands out about iron arsenate is its ability to trap arsenic. In environmental labs, engineers add iron compounds to polluted water so arsenate ions hook up with iron and drop out of solution. The product stays pretty stable until strong acids enter the picture, breaking the lattice and freeing arsenic. You can exchange some of the water in hydrated forms or replace iron with other metals. With the right conditions, iron arsenate can also serve as a source of arsenic in specialty syntheses, though most labs favor more controlled reagents for such steps. Heating or mixing with other metal salts lets researchers tweak its structure for different purposes.
Synonyms & Product Names
Depending on the source or end use, iron arsenate goes by several names. You may see it listed as ferric arsenate, iron(III) arsenate, or, in mineral form, as scorodite. Some older literature uses terms like iron orthoarsenate, reflecting knowledge at the time. Industry catalogues reference it by hydrate level—such as iron arsenate pentahydrate—and sometimes tie it to brand names when used as part of a formulation. Customs records and shipping manifests usually list it under harmonized system codes linked with toxic inorganic chemicals.
Safety & Operational Standards
Lab veterans know iron arsenate demands gloves, eye protection, and a well-ventilated workspace. Even brief, careless handling increases exposure risk, so most processes use fume hoods and sealed containers. The main concern revolves around the arsenic content; inhalation or accidental ingestion leads to serious health issues, including acute toxicity and possible carcinogenic effects. Workplaces follow OSHA and EPA rules for handling arsenic compounds, and spills trigger well-rehearsed decontamination routines with specialized waste collection. Regular training keeps everyone aware of dangers, and disposal contracts include detailed documentation to show compliance at every stage.
Application Area
Though its use shrank over the decades, iron arsenate still finds niche roles. In environmental remediation, it helps trap arsenic in soil and water, forming a stable product that can be removed or contained. Researchers use it as a reference standard, especially in arsenic speciation studies. A handful of pigment producers continue to use iron arsenate to make deep green or blue glazes, favored for reproduction of antique ceramics. In metallurgy, it acts as both a waste management tool and a recovery agent. Each use carries strict oversight because regulators keep a close watch on any application that could lead to arsenic entering the water supply or food chain.
Research & Development
Most recent studies branch into environmental engineering, where scientists work out new ways to convert soluble arsenic into iron arsenate forms that resist leaching. I’ve watched graduate students run column tests with iron oxides, showing how layers of iron trap passing arsenic and form solid precipitates. Researchers also study iron arsenate’s crystal structure to understand long-term stability in waste repositories. Emerging applications in battery technology and catalysis pop up now and then, but health and safety questions usually hold back widespread use. Teams working in these areas keep refining processes to minimize risk and maximize containment.
Toxicity Research
No one in the field overlooks the hazards tied to iron arsenate. Lab animal studies point to acute and chronic toxicity from even modest doses, with arsenic at the root of the problem. Regular monitoring of workplace air and surfaces helps catch contamination early. Researchers test new containment and disposal technologies, comparing their effectiveness at locking away arsenic. The lingering health effects of past industrial use provide a warning—cases of groundwater pollution and worker illnesses still trickle out in environmental and medical studies. Efforts now focus on early detection, strict exposure limits, and new detoxification strategies.
Future Prospects
Iron arsenate’s story won’t disappear from industry or science soon. Calls for better arsenic remediation keep it in the toolkit for cleaning up mines and polluted water. Scientists look for safer ways to form, handle, and break down iron arsenate, often hoping to apply what they learn to other toxic materials. If new regulations push for tighter arsenic limits in food, water, or materials, demand for stable sequestration compounds will likely climb. The hunt for innovative uses—such as in batteries or catalysis—continues, balanced by hard lessons from decades of occupational and environmental health research.
What is Iron Arsenate used for?
Looking Beyond the Periodic Table
Iron arsenate doesn’t have a role that pops up on the average person's radar. Most folks aren’t sprinkling it in their gardens or adding it to their morning cereal. But this substance, made by combining iron with arsenic and oxygen, has a long backstory—one that stretches from mining to museum collections. My first encounter with iron arsenate came as a chemistry student, grappling with mineral samples and field notes, not quite understanding why such an obscure name kept turning up in industrial reports.
A Historical Fix for Pests
Farmers once relied on iron arsenate as a pesticide. In the early twentieth century, chemical pest control was new, and crops suffered from bugs that outsmarted older methods. Iron arsenate powders showed up in apple orchards and cotton fields across North America. It worked; bugs hated it, plants survived. People at the time trusted what science offered, but new problems always emerge when you wrap poison into public health.
A Shift in Perspective
By the 1950s, scientists raised alarms about arsenic buildup in the environment. My own memories come from reading decades-old journals in the university library, pages yellowed and scented with musty warnings. People realized kids playing in treated yards could get exposed. Rivers near farmland tested above safe arsenic levels. Iron arsenate became another line item in the long list of pesticides phased out over safety concerns. Governments clamped down, and application in agriculture faded away.
Still Hanging Around in Ore Processing
Just because a chemical loses favor in one industry doesn’t mean it disappears. Iron arsenate still shows up in ore treatment at mining sites. Mining can pull heavy metals and arsenic from the earth. Smelters and refiners work to limit toxic waste. Precipitating iron arsenate from process water helps capture arsenic before it escapes into rivers or soil. Mining trade publications count iron arsenate formation as a necessary step in environmental compliance. Some mines add iron salts to reaction tanks on purpose to form iron arsenate, then settle it out with the rest of the solid waste.
Science and Safety
Researchers today use iron arsenate for scientific studies. In environmental chemistry labs, it serves as a test material to model how arsenic moves in soil and water. University students and PhDs poke at these reactions because communities downstream from mines want safer drinking water. The World Health Organization and U.S. EPA set arsenic limits in public water systems for a reason—chronic exposure links to cancer and skin lesions.
What's Next?
Communities count on clear information and safety rules. Mining companies can avoid shortcuts, keep filtration updated, and follow the best available science for waste treatment. Pesticide regulations need constant review; old stockpiles should be disposed of with oversight—not abandoned where future generations can stumble over them. Local governments must support water testing and communication, so families know what comes out of their taps.
Lessons Learned
Iron arsenate reminds us that progress sometimes creates unintended consequences. Chemical tools solve problems until they introduce new ones. Balancing agriculture, industry, and public safety calls for careful attention, clear labels, and a willingness to review decisions as new research surfaces. As someone who’s seen both the lab bench and the community meeting, I trust a process powered by evidence, transparency, and honesty.
Is Iron Arsenate toxic to humans or animals?
The Real Story Behind Iron Arsenate
Iron arsenate, a gray-green powdery compound made from iron and arsenic, sometimes pops up in soil treatment, pigments, or as a lab chemical. It doesn’t end up in store-bought vitamins or breakfast cereal. I’ve met folks who assume anything with iron must be healthy. That’s not true here. Arsenic has a dark reputation: it causes health problems, and iron arsenate is no different.
What Science Says About Safety
Let’s cut to the chase. Studies have shown exposure to iron arsenate can hurt both people and pets. The problem comes straight from arsenic. The World Health Organization reports that arsenic compounds, including those mixed with iron, can trigger coughing, stomach pain, vomiting, and diarrhea after just a small amount. Higher doses, especially if swallowed, damage vital organs. In the U.S., the Agency for Toxic Substances and Disease Registry lists arsenic among the most dangerous environmental substances.
I once worked at a community garden near a site treated with historic pesticides—many of which relied on arsenic. The anxiety in the neighborhood ran high. Blood tests showed trace arsenic among some frequent volunteers, with symptoms like fatigue and skin trouble. Pets digging in contaminated dirt, especially dogs, risk similar symptoms or worse. Tiny particles cling to fur or paws and end up in their mouths. This isn’t just alarmist talk. Research backs it up.
Questions About Chronic Risk
Some ask if iron arsenate, locked in soil or paint, stays dangerous. It does. Over time, rain and weather break down compounds, letting arsenic leach into groundwater. Runoff affects crops, drinking water, and animal forage—even if nobody realizes it at first. Long-term exposure leads to cancer, nerve disease, and lasting heart problems. Countries that struggle with groundwater arsenic contamination show higher rates of such issues. Iron arsenate goes through similar breakdown processes, never truly leaving an area unless cleaned out.
Managing Risks and Finding Safer Practices
So what can people do about it? My experience with older properties taught me to watch out for unknown powders stored in sheds or used around old orchards. Communities with a farming past often face soil tests for arsenic. Where possible, experts recommend swapping out tainted soil, testing wells, and planting non-edible ground covers rather than fruit or vegetables. Anyone working with possible contamination should wear gloves, keep pets away, and wash up thoroughly—even on days dirt seems harmless.
Schools, playgrounds, and public landscapes sometimes cover contaminated ground with clean soil or mulch. That’s not a cure, but it reduces contact. Regulatory agencies track arsenic in public land and monitor drinking water standards closely. Some cities provide home testing kits at low cost. It’s easier to deal with this chemical before it spreads than to fix the problem later. If you suspect iron arsenate, never handle it unprotected or pour it down drains—special collection programs take care of safe disposal.
The bottom line: iron arsenate isn’t something to treat lightly. Knowing how it affects people and animals, respecting the warnings, and focusing on clean-up keeps families and communities safer. The facts from research, real-life cases, and medical records tell one story—there’s no safe role for this stuff outside of controlled lab settings.
What are the safety precautions when handling Iron Arsenate?
The Basics of Dealing With Iron Arsenate
Iron arsenate brings a sense of real concern, even in small doses, because arsenic compounds have a long reputation for being risky. Handling this chemical isn’t just about wearing gloves and running a fan. Over the years, I’ve worked around chemicals in labs where mistakes don’t always give second chances. Iron arsenate is no exception.
Understanding Its Hazards
The biggest trouble with iron arsenate comes from its arsenic content. Arsenic slips into the body through inhalation and skin exposure, and it accumulates over time. Even short-term handling leaves its mark if you don’t take precautions. The dust finds its way onto surfaces, into small scratches on your skin, and into your lungs if you breathe too freely. This stuff isn’t like table salt. It’s linked to lung problems, nerve damage, and cancer.
Many safety data sheets warn about symptoms like vomiting, headaches, confusion, or numbness. Those aren’t easy to ignore. I’ve seen a coworker develop unusual symptoms after a careless spill. It gets frightening when a cough lingers for days or hands tingle for no obvious reason. Reporting and tracking even small exposures keeps people honest about these risks.
Personal Protective Equipment Is Non-Negotiable
There’s no shortcut here. Labs with good track records rely on proper gear every time someone opens a container. Gloves mean nitrile or neoprene, never those thin plastic ones people sometimes grab for quick jobs. Face shields or safety goggles block dust particles from drifting into your eyes. Disposable coveralls or lab coats catch anything that lands on clothing.
In my lab days, nobody skipped the respirator—an NIOSH-approved P100 mask for arsenic compounds—when handling powders or mixing solutions. I learned quickly that dust masks from the hardware store don’t count. The right filter keeps those invisible particles out of your airways.
Ventilation and Cleanliness
You want iron arsenate under a fume hood, not at an open bench. Good exhaust ventilation stops fumes and dust from escaping into shared spaces. Anyone who has cleaned up after a spill sees how far those tiny particles can travel. Frequent cleaning of work areas with wet methods, never dry sweeping, helps avoid stirring up leftover dust.
Trash that’s been contaminated with iron arsenate (gloves, wipes, containers) gets sealed in labeled bags for hazardous waste pickup. Regular trash can’t handle heavy metals. Failing to separate the waste risks tainting the workplace and any environment downstream.
Medical Monitoring and Prompt Action
People handling arsenic compounds get baseline blood and urine tests. Repeat those tests at regular intervals or after any spill. This isn’t overkill; some effects creep up slowly and only show up on a lab report. If anything feels off—fatigue, rash, cough—report it to a supervisor and get checked out.
Training and Respect for the Rules
Nobody picks up proper arsenic handling from a manual alone. Training involves hands-on instruction, watching for shortcuts, and correcting mistakes before they cause harm. There’s always that urge to skip a step when rushing, but in my experience, every serious incident traces back to missing or broken routines.
Taking these steps turns iron arsenate from a clear danger into a manageable workplace hazard. Each layer—personal gear, ventilation, careful cleanup, and medical checks—reinforces the next, and together, they protect everyone who works around this chemical. Real respect for the risks comes through practice, not just paperwork.
How should Iron Arsenate be stored and disposed of?
What Iron Arsenate Really Means for Safety
Iron arsenate turns up in a range of industries, from pigment production to lab research. No matter where you find it, this is not a compound made for casual handling. For those who have spent any time around chemicals, the word “arsenic” jumps out, setting off alarm bells about chronic exposure, long-term health risk, and the headaches that come from store-room mishaps.
Storing Iron Arsenate: Simple Habits Make a Big Difference
Glass bottles and tightly sealed containers bring a sense of relief to anyone stashing iron arsenate. This isn’t a material to leave near heat sources or sunlight. I’ve seen rusty containers and blown lids when corners get cut, and those small mistakes turn storage areas into hazards. Cool, dry spaces away from acids and bases give you a better shot at keeping things safe. Labels shouldn’t wear off. Clear labeling saves lives—no one wants a distracted worker pouring acids on what they thought was sand, only to trigger a reaction.
Even in well-kept labs, locked cabinets separate these compounds from other supplies. Signs keep nosey hands away, and secondary containment—plastic trays, spill-proof bins—help in case a container tips. These sound like simple moves, but every chemist or maintenance worker knows the pain of finding out someone took a shortcut. Airflow matters, too. Ventilated rooms keep dust in check, which limits what ends up in someone’s lungs.
Moving to Disposal: Why Rigorous Steps Count
Where storage is about peace of mind, disposal becomes a legal and ethical crossroads. Once iron arsenate overstays its welcome, thinking ‘this can just go down the drain’ is a recipe for groundwater disaster. Arsenic contaminates water systems, threatens crops, and climbs the food chain faster than people realize.
Trusted companies cart off hazardous waste for a reason. Laws require this, but it also stands as common sense. Incineration doesn’t break down arsenic, so landfills designed to contain toxics take over. Sometimes, consolidation and chemical reduction methods pop up in specialized facilities, but these belong in trained hands, not do-it-yourself projects.
Training and Honesty: How to Protect Workers and Neighbors
I learned early on that regular safety drills—the kind most workers roll their eyes over—save headaches when spills happen. Written plans help, but real muscle memory comes from practicing what to do if a bottle falls or dust escapes. PPE, including gloves and tight-fitting masks, should never gather dust on a shelf.
Regular inspections keep old bottles from corroding through or leaking unnoticed. Logging every use, every disposal action, creates accountability easily lost in busy workplaces. Quick access to material safety data sheets lets people know what they’re dealing with and how much danger lurks in a spill.
Potential Improvements and Community Trust
Regulations get tighter as science uncovers more about chronic arsenic exposure. Companies and schools can take that as a call to invest in better storage rooms, stronger containers, and training sessions that stick. Some firms look to alternatives, swapping out older chemicals for safer ones when possible.
Transparency with surrounding neighborhoods often pays off, especially after high-profile mishaps. Sharing what steps a facility uses for storage and disposal, while opening the door to site tours or Q&A sessions, builds trust that regulations alone can’t buy.
Storing and disposing of iron arsenate remains a heavy responsibility. Simple, consistent safety habits, supported by clear policies and honest communication, can keep workers and communities out of harm’s way.
What are the chemical properties and formula of Iron Arsenate?
Digging Into Iron Arsenate's Make-Up
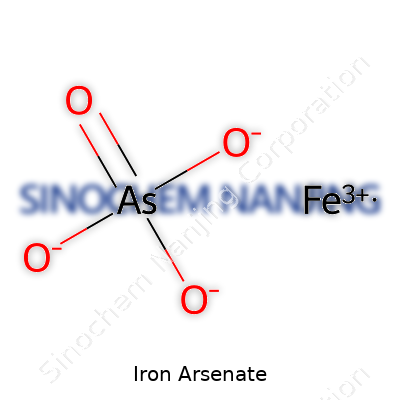
Iron arsenate turns up in soil, rivers, and even some manufactured products. At its core, iron arsenate comes with two common formulas: FeAsO4 and Fe3(AsO4)2. The first, ferric arsenate, includes iron in its trivalent state (Fe3+), while the second holds two iron atoms for every three arsenate ions. Each formula stands apart by the arrangement of atoms and its behavior under real-world conditions.
Chemical Personality
With its formula, iron arsenate’s key feature is the bond between iron and the arsenate ion (AsO43–). This bond locks away arsenic, a notorious environmental pollutant, at least temporarily. In nature, you spot iron arsenate as the mineral scorodite—a greenish crystal often surfacing in mining zones rich in both iron and arsenic. Exposure to air gives scorodite a rusty touch as the iron oxidizes.
Test tubes show how iron arsenate defies solubility. Drop it in water and it barely budges. That’s a safety advantage, since free arsenic in groundwater raises public health alarms. The arsenate part comes from arsenic acid, while the iron side relies on ferric salts. Mix these two in labs or in acid mine drainage. The solid falls out—a vivid reminder that chemistry shapes our daily environment.
Touching on Toxicity and the Environment
Iron arsenate often pops up in places where mining or smelting goes unchecked. In the past, it even landed in some pesticides before scientists realized arsenic’s nasty health effects. Kids playing near contaminated soil risk ingesting bits of iron arsenate, and while this stuff doesn’t dissolve easily, any shift in pH or chemistry—from acid rain or fertilizer runoff—may release arsenic ions. Where health and safety matter, monitoring soil chemistry makes sense.
Growing up in a place where abandoned mines dotted the hills, I remember folks worrying about old waste piles seeping into wells. The EPA found out that iron arsenate could immobilize arsenic for a while, but not forever. Changing weather patterns and soil acidity could break down iron arsenate, dumping arsenic into streams.
Facing the Challenge
Addressing iron arsenate’s environmental role doesn’t mean banning it outright. Instead, practical strategies protect communities. Regular groundwater checks near former industrial sites catch problems early. Soil amendments such as adding lime help stabilize pH, keeping arsenate locked in place. Planting deep-rooted grasses can slow the movement of contaminated dust—a trick borrowed from phytoremediation research.
Iron arsenate provides a valuable lesson in chemistry’s impact beyond the laboratory. The way iron and arsenate stick together shapes both risk and opportunity. Communities can’t afford to ignore these lessons, especially as development pushes into areas with a legacy of heavy metal use. Getting the chemistry right keeps water cleaner, kids safer, and ecosystems in better shape—one formula at a time.
| Names | |
| Preferred IUPAC name | iron(III) arsenate |
| Other names |
Ferric arsenate Iron(III) arsenate Ferric orthoarsenate Iron arsenate(V) |
| Pronunciation | /ˌaɪ.ərn ɑːˈsɛ.nɛt/ |
| Identifiers | |
| CAS Number | 10103-61-2 |
| Beilstein Reference | 22312 |
| ChEBI | CHEBI:86355 |
| ChEMBL | CHEMBL1201647 |
| ChemSpider | 20533 |
| DrugBank | DB14626 |
| ECHA InfoCard | 100.930.302 |
| EC Number | 234-001-8 |
| Gmelin Reference | 82121 |
| KEGG | C01697 |
| MeSH | D017806 |
| PubChem CID | 24556 |
| RTECS number | CB9625000 |
| UNII | 1C8U6U56V2 |
| UN number | UN1588 |
| Properties | |
| Chemical formula | FeAsO4 |
| Molar mass | 242.68 g/mol |
| Appearance | White or pale green powder |
| Odor | Odorless |
| Density | 4.38 g/cm³ |
| Solubility in water | Insoluble |
| log P | -36.45 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 13.0 |
| Basicity (pKb) | 11.82 |
| Magnetic susceptibility (χ) | +5200.0e-6 cm³/mol |
| Refractive index (nD) | 1.720 |
| Dipole moment | 0 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 99.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -822 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1230.2 kJ/mol |
| Pharmacology | |
| ATC code | B03AB05 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; causes damage to organs; may cause cancer; very toxic to aquatic life. |
| GHS labelling | GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H302: Harmful if swallowed. H332: Harmful if inhaled. H350: May cause cancer. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P201, P202, P260, P264, P270, P272, P273, P301+P312, P301+P330+P331, P302+P352, P304+P340, P305+P351+P338, P308+P313, P314, P321, P330, P362+P364, P391, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Lethal dose or concentration | LD₅₀ oral (rat): 20 mg/kg |
| LD50 (median dose) | LD50 (median dose): 20 mg/kg |
| NIOSH | NA9100 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Iron Arsenate: "0.01 mg/m³ (as As) |
| REL (Recommended) | 0.01 mgAs/m³ |
| IDLH (Immediate danger) | 5 mg/m3 |
| Related compounds | |
| Related compounds |
Aluminium arsenate Silver arsenate Lead(II) arsenate Calcium arsenate |

