Iodine Pentafluoride: Depth Beyond the Formula
Historical Development
Looking back, the story of iodine pentafluoride traces a fascinating path through the world of industrial chemistry. Chemists explored new fluorine-based compounds in the early 20th century, always looking for compounds that could serve the swelling needs of science and technology. Iodine pentafluoride first emerged out of that race, produced by reacting iodine with elemental fluorine. It quickly stood out thanks to its strong oxidizing power. This wasn’t just a lab curiosity—the compound found its way into the annals of inorganic fluorine chemistry, opening doors for broader halogen studies. I still remember my own first encounter with IF5 in a research setting, surprised by how straightforward its preparation could be compared to its notorious neighboring compounds, like chlorine pentafluoride or bromine pentafluoride.
Product Overview
Iodine pentafluoride comes across as a pale yellow liquid with a pungent odor. Unlike so many other industrial chemicals that have faded from the stage, it has stuck around for a reason. People call it IF5 or iodine fluoride, but behind these simple names stands a complex agent used in both academic research and industrial processes. Handling this substance always calls for respect, given the ease with which it reacts, and its long history proves that value often springs from sheer reliability and uncompromising chemical potential.
Physical and Chemical Properties
At room temperature, IF5 holds its liquid state, with a melting point resting at around 9°C and boiling climbing to roughly 97°C under atmospheric pressure. Anyone who has used this material learns quickly about its density—close to two times that of water. Water invites an aggressive, violent reaction from it, spewing out hydrogen fluoride gas and liberating heat. Nothing beats seeing a small drop fizz and hiss once in contact with moisture during a controlled experiment; it’s a hands-on reminder that even experienced scientists need to pay attention. IF5 displays strong oxidizing properties, and its reactivity spans a wide array of organic and inorganic substrates. The molecule itself, with its trigonal bipyramidal shape, helps explain its behavior: five fluorine atoms surround a central iodine, eager to part with electrons and tear into less stable molecules.
Technical Specifications and Labeling
Every drum, cylinder, or ampule of IF5 comes clearly labeled with essential warnings—corrosive, water-reactive, toxic. Industrial suppliers provide IF5 at concentrations above 99%, with impurities like moisture and other halides kept to extremely low levels. Material safety data sheets spell out emergency handling, leak procedures, and storage needs, and regulatory IDs such as the UN number (UN2496) and CAS number (7783-66-6) sit front and center. In many jurisdictions, the product falls under strict hazardous materials regulation, so manufacturers ensure containers are made of compatible metals like nickel or Monel. My own observation: even a pinprick exposure can corrode less-resistant metals, which can easily catch the unprepared off-guard.
Preparation Method
The path to pure IF5 starts with elemental iodine and fluorine, two substances that command respect on their own. Industrial plants feed dry iodine and a steady stream of fluorine into a reaction chamber—often lined with nickel due to the strong corrosive nature of the product and reagents. The direct reaction releases a fair bit of energy, so cooling and careful control keep things in check. The balanced equation reads: I2 + 5 F2 → 2 IF5. To clean up the crude product, distillation removes lower-boiling impurities, leaving liquid IF5 behind. Operators must rely on specialized gear and trusted protocols, since even tiny leaks can become emergencies due to hydrogen fluoride vapor. Seeing an automated plant in action—robot arms, thick shields, vacuum lines—reminds everyone just how far industrial chemistry has progressed since the early days of bench-top synthesis.
Chemical Reactions and Modifications
In any lab or plant, IF5 acts as a brutal fluorinating agent, adding fluorine to organic molecules with a kind of aggressive persistence. Chemists looking to convert alcohols, ketones, or sulfur compounds often turn to IF5 because it sidesteps more cumbersome approaches. It doesn’t shy away from oxidizing metals, nonmetals, or complex organic molecules, and its appetite for reaction produces everything from SF6 to iodine heptafluoride if you press the reaction to the extreme. Chemical engineers trust it to strip silicon from electronics parts or to create specialty materials used in niche battery research. In the right company, IF5 can seed the creation of exotic perfluorinated solvents and surface-active agents, always offering a blend of danger and opportunity.
Synonyms and Product Names
Over the years, IF5 has collected a string of synonyms in scientific literature and trade—iodine fluoride, iodine(V) fluoride, and at times the less commonly seen pentafluoro-λ5-iodane. For purchasing, suppliers stick to “iodine pentafluoride” or “IF5,” rarely straying from the basics, which might explain its staying power in so many catalogs. Some older chemistry texts refer to it in the context of its role as a fluorinating agent, but the standard nomenclature holds steady across decades and languages. That reliability helps researchers and industrial buyers avoid confusion, cutting through the jargon and getting straight to business.
Safety and Operational Standards
No one calls iodine pentafluoride a gentle chemical. It fumes in moist air, corrodes skin and metal, and demolishes lungs if inhaled. OSHA and the European Chemicals Agency draw hard lines with maximum exposure limits and mandatory protective standards. Full-face respirators, acid-resistant gloves, and reinforced clothing form the first defense in any lab or plant. Emergency medical kits must stay within arm’s reach. Fire departments and HAZMAT teams learn to treat IF5 spills as top-priority hazards, since any contact with water yields hydrogen fluoride—a poison and corrosive agent of the worst kind. Anyone who has worked with fluorides remembers the drills: calcium gluconate gel ready for skin exposures, dry sand for containment, and strict procedures for neutralization and cleanup.
Application Area
Industry and research rely on IF5 in select roles where its strong fluorinating power serves a unique purpose. Microelectronics manufacturers use it to etch advanced circuit pathways. Synthetic chemists depend on it to create complex fluorinated organic molecules needed for specialty oils, blood substitutes, and high-performance polymers. In the nuclear fuel industry, IF5 helps in processing uranium, separating isotopes in demanding environments. Analytical labs sometimes use it to break apart mineral samples for trace analysis. My time consulting with specialty labs introduced me to its less-publicized uses—sometimes just a few grams can unlock a synthesis that has stalled for months.
Research and Development
Fresh research seeks safer handling protocols, new derivatives, and innovative applications for IF5. Teams at top chemical institutes pursue ways to control its reactions with increasing precision, using microfluidic chips and computer modeling to predict outcomes before ever opening a valve. Some studies focus on blending IF5 into fluoropolymer development, while others chase next-generation battery chemistries that take advantage of unusual fluorinated intermediates. Academic labs continue probing its structure, turning to spectroscopy and high-level quantum calculations. Innovation doesn’t slow down. It builds on over a century of practical experience and hard-won lessons.
Toxicity Research
The health hazards of IF5 cannot be overstated. Toxicological studies report both acute and chronic risks, often linked to the hydrogen fluoride given off in contact with moisture. Even small doses can produce severe burns and respiratory depression in humans and animals. Recent animal research continues to map out safe exposure windows, tissue uptake rates, and antidotes. Lab incidents still occur, making training essential for anyone who handles even tiny volumes. Environmental studies chart the fate of IF5 spills, showing that soil and water react violently and release toxic byproducts. Industrial sites keep extensive accident logs to help public health officials respond quickly when issues arise. Ongoing developments in protective coatings and neutralization chemistry promise to bring better safety in the years ahead.
Future Prospects
Big changes are ahead for iodine pentafluoride. Research into sustainable alternatives to traditional fluorination agents still keeps IF5 in the picture, thanks to its unique properties. Advanced microchips, greener solvents, specialized polymers, and evolving medical imaging agents will fuel demand. Regulatory agencies are pushing for smarter containment strategies, lowering the risk of accidents and environmental harm. Meanwhile, research efforts look to harness IF5 with leaner, automated systems that limit worker exposure, streamline waste management, and open new possibilities in chemical manufacturing. The next generation of chemists and engineers faces the challenge of balancing power and responsibility—something that IF5’s long history makes impossible to ignore.
What is iodine pentafluoride used for?
Not Your Everyday Chemical
Iodine pentafluoride rarely pops up in regular conversation, yet it does some heavy lifting in chemistry labs and industry. You won’t find it under your kitchen sink or in your favorite snack, but in certain circles, especially where advanced chemistry happens, it holds real weight. This compound, made by reacting iodine with fluorine, carries a pungent smell and a strong bite—definitely not something you’d want to spill on your shoes. That said, it has a clear role in shaping modern science, especially in making complex molecules easier to build or study.
Helping Chemists Make Sense of Fluorine
Fluorine sits high on lots of chemists’ wish lists because it can turn ordinary molecules into powerful drugs, specialized materials, or even stronger rocket fuel. Getting fluorine into molecules isn’t easy, though. It tends to be wild and uncooperative. Here’s where iodine pentafluoride steps in. At the lab bench, it gives chemists a safer, more controlled way to stick fluorine onto carbon-based compounds. This process, called fluorination, lets researchers create designer molecules with new abilities, such as longer shelf life or better disease-fighting properties.
Beyond the Lab—Industrial Applications
Big business uses iodine pentafluoride more quietly but just as meaningfully. Manufacturers sometimes lean on it for etching and cleaning during microchip production, banking on its chemical muscle to strip away unwanted layers on silicon wafers. A smooth, clean surface helps companies build better electronic parts, which trickles down into the devices we use every day—think phones, cars, even medical equipment.
Not All Chemistry Happens Without Risk
I worked around hazardous chemicals early in my career, and understanding the danger shapes your respect. Just like other fluorine-based compounds, iodine pentafluoride brings environmental and health concerns. Breathing in fumes or spilling this stuff can quickly turn a routine day sideways, injuring workers or damaging equipment. Responsible companies stay sharp with strict safety gear, ventilation systems, and training that leaves nothing to chance. I remember drills where we practiced evacuations and suiting up just in case something spilled, because mistakes in these settings hurt more than just pride.
Weighing Progress and Precaution
Any chemical, especially one as reactive as this, sparks questions about long-term impacts. Disposal lingers as a key issue. Factories ought to neutralize every drop instead of sending waste downstream. Regulations ask for full reporting and monitoring, but enforcement can lag. Better oversight and zero-excuse safety culture protect both workers and neighbors. Some organizations also hunt for replacement technologies—safer fluorinating agents or physical etching methods—to reduce reliance on substances that could poison air or water if left unchecked.
The Push for Greener Chemistry
Green chemistry isn’t a buzz phrase—it’s how industry keeps moving forward without leaving scars. Academic and corporate teams are testing new reactions that get the same results without making things so risky. Some reactions use milder chemicals that pose less hazard to workers and nature while still spreading fluorine where it counts. Investing in smarter solutions pays off for everyone. Healthy workers, cleaner air, and responsible innovation build stronger companies in the long run.
Why It Matters
People rarely meet iodine pentafluoride in daily life, yet it quietly props up entire industries. Chemists, manufacturers, and safety pros work together to pull value out of such a sharp-edged tool while staying watchful for the risks. Safe handling, better alternatives, and sound regulation offer the best path forward, making cutting-edge technology possible without cutting corners on health or the environment.
What are the safety precautions when handling iodine pentafluoride?
What Makes Iodine Pentafluoride Risky?
Handling chemicals brings back memories from my early career, back when the thought of a poorly sealed lid made my heart skip a beat. Iodine pentafluoride stands out on the hazard list. This yellowish liquid doesn’t need much encouragement to react. It attacks skin, eyes, and lungs in a flash. I remember the acrid smell in the lab air after a tiny spill. You can’t mistake it.
The danger comes from more than its ability to burn on contact. It releases fumes that can damage tissue before you realize what happened. Even glassware can fall victim, since this substance eats away at common lab materials. What that taught me: never underestimate the speed of chemical reactions, especially with powerful oxidizers.
What Good Safety Looks Like
One lesson stands firm: preparation beats improvisation. Before touching any bottle, know the plan and the route out if something goes wrong. Setups with secure ventilation give you breathing room. Fume hoods are not a formality—they keep those invisible vapors from winding up in your lungs. I’ve seen a quick test in an open area turn into a week-long cleanup.
Gloves matter, but not the blue latex from the first-aid kit. Heavy-duty, chemical-resistant gloves create a better barrier, since iodine pentafluoride chews through common materials. Face shields and splash-proof safety goggles help when even a tiny droplet would make a trip to the hospital. Lab coats, closed shoes, and full-length sleeves should be standard. During a university training, we once watched an unprotected wrist blister from just a spot of spilled chemical—the memory sticks with you.
Storage brings its own set of worries. Keeping iodine pentafluoride in a tightly sealed container saves a lot of grief. Dry, cool spots away from water or anything organic helps cut down risk. Mixing with water or common solvents creates aggressive reactions, producing hydrogen fluoride gas, among the nastiest you’ll run into in a lab. Clear labels and isolated shelves save lives. My former supervisor locked up oxidizers and acids separately, a rule I stick to now without compromise.
What to Do If Something Goes Wrong
Accidents happen. Fast action matters. Skin contact? Wash instantly with lots of water—no dilly-dallying or waiting for a supervisor’s OK. For inhaled fumes, fresh air and emergency medical help top the list. Fire from this compound burns hot, and not every extinguisher helps. Dry chemical fire extinguishers, not water, have a fighting chance.
Everyone in the lab should practice emergency drills. A team that knows where the showers and exits are will always react quicker than panicked individuals. Clear instructions posted in visible spots make a real difference, especially for new people or visitors.
Learning and Moving Forward
These lessons came from mistakes as well as careful training. Over the years, I’ve seen complacency surprise both newcomers and veterans, especially during routine work. Review of material safety data sheets makes sense before each use, not just once during onboarding. Regular gear checks and short reminders at the start of the week help keep the team sharp.
Improving safety culture takes time. Open conversations about near-misses and better reporting systems do more than check boxes—they give everyone a stake in staying safe. Iodine pentafluoride, with all its hazards, doesn’t forgive shortcuts. In every lab I’ve worked in, people only stay safe by taking those risks seriously, planning ahead, and watching out for each other.
What is the chemical formula and structure of iodine pentafluoride?
Chemical Formula of Iodine Pentafluoride
Iodine pentafluoride, known to chemists as IF5, brings together one atom of iodine and five atoms of fluorine into a single molecule. The construction looks simple, but there’s a lot packed into that small frame. As someone who well remembers the first time a drop hit a classroom bench and left a burn mark, IF5 doesn’t mess around. It comes as a colorless to pale yellow liquid, and just a whiff warns you to treat it with care.
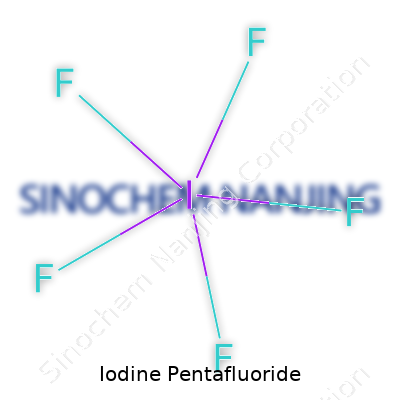
Structure and Molecular Geometry
The molecule carries a shape that chemists call square pyramidal. Picture a pyramid with a square base – that’s most of what IF5 looks like, with the iodine atom planted at the center of the base and fluorine atoms at each of the corners and one at the top. The lone pair of electrons on the iodine takes up the position underneath the square, nudging the five fluorines into formation.
Drawing it out on paper in a lab, the structure always reminded me of molecular geometry lessons, where you see how lone electron pairs kick out the symmetry. These squabbles between electrons have real consequences. The way the atoms arrange themselves gives IF5 its physical and chemical punch, allowing it to interact with other substances in ways that push boundaries—sometimes, quite literally, setting things on fire.
Importance in Industry and Research
Anyone working in chemical manufacturing often runs across iodine pentafluoride’s fingerprint. Halogenation, etching, and the creation of specialty compounds rely on its strong fluorinating power. The molecule rips hydrogen out of organic compounds and slaps in fluorine, transforming the building blocks of medicines, pesticides, and high-performance plastics.
One job I tackled in a specialty gases lab introduced me to its sheer reactivity. Protective gear felt like armor rather than an option. The bottle arrived with stern warnings: “Corrosive,” “Toxic.” Handling it safely demanded tight protocols, fume hoods, and steady nerves. One slip could send you running for the safety shower.
A teacher once pointed out, “Every new molecule we create shapes the future of electronics, energy, and health.” IF5 impressed me as a force multiplier—a substance that, in small amounts, makes possible new discoveries and technologies.
Navigating Safety and Environmental Impact
The value of iodine pentafluoride comes bundled with risk. Exposed to air and moisture, it produces toxic vapors of hydrogen fluoride and iodine vapors. These byproducts corrode skin, lungs, and metal. So, responsible use takes priority. Regulations and handling guidelines require professionals to inspect their equipment, track every gram, and neutralize waste before disposal. Stories circulate of people sickened when a seal leaked or a bottle cracked at the wrong moment.
Companies continue developing containment systems and cleanup methods that reduce accidental releases. Academic groups research alternatives for certain fluorination processes so dependence on hazardous chemicals like IF5 can shrink over time. Teaching respect for such substances in early chemistry classes pays large dividends, both for personal safety and environmental stewardship.
Choosing the right materials and double-checking procedures transforms IF5 from a hazard into a tool that unlocks progress. Chemical expertise and respect for the risks allow this molecule to play its part in shaping new technologies, medicines, and solutions to modern problems.
How should iodine pentafluoride be stored?
Why Proper Storage Really Matters
Iodine pentafluoride makes most of us who deal with chemicals pause for good reason. I’ve seen what happens when storage gets sloppy: corrosion on metal shelves, leaky containers, and more than a few close calls with fumes. Even one small mistake turns this chemical from a useful tool into a hospital trip waiting to happen. Iodine pentafluoride reacts fiercely with water, and if moisture gets in, expect nasty gases and a risk of burns few will forget. Proper storage isn’t just about following rules—it’s about keeping people and property out of harm’s way.
Physical Storage Approach
No one wants a surprise chemical reaction. Storing iodine pentafluoride starts with choosing the right container. Glass or certain plastics handle the stuff well, since metals corrode or break down. Seals must be tight, and container labels should stand out to everyone. Keep it away from acids, alcohol, and any hint of water. I learned early on that even humid air gets inside basic containers, so a cool, dry chemical cabinet with reliable ventilation works best. Chemical safety isn’t glamorous, just practical—dobasics right and toxic gas leaks won’t become tomorrow’s emergency drill.
The Human Factor: Training and Awareness
Engineers and lab techs can reel off chemical formulas, but knowledge doesn’t matter much unless people use it. I’ve lost count of the number of workplace accidents that trace back to someone not reading the label or not remembering a safety poster. It’s easy to set protocols, but everyone from the seasoned chemist to the part-time intern needs to know what iodine pentafluoride looks like, smells like, and how it behaves if something goes wrong. That takes hands-on safety training—eye protection on, gloves up, and a drill for spill containment. We’re all human. Mistakes happen. Regular practice and reminders make those mistakes less likely.
Emergency Preparedness
If storage slips, so does safety. Corrosive fumes, fire—none of it gives warning. That means glass containers need secondary containment trays beneath them, and those should handle spills. Emergency showers and eye-wash stations earn their space near the storage room. I’ve walked through places that cut corners here, and it’s never worth it. Safety data sheets stay on hand, so even the newest staff can react fast. Chemical fume hoods should never double as storage, since airflow isn’t threat-proof.
Addressing Systemic Issues
It’s not just about putting a lid on a bottle. I see big gaps where oversight falls short: old cabinets, missing training logs, worn labels. Strong policies and audits fill those gaps. Regular reviews, inventory checks, and staff refreshers on chemical hazards make a difference. In my experience, putting safety culture up front protects everyone, and that matters far more than any bottom line.
Solutions and Moving Forward
Facilities need sturdy, chemical-resistant cabinets with clear signage showing what’s inside. Regular staff retraining—especially before new chemicals arrive—keeps skills sharp. Invest in leak-proof containers and spills kits nearby. No shortcut comes close to real, lasting safety with iodine pentafluoride, and that means attention to detail every day.
What are the physical and chemical properties of iodine pentafluoride?
What This Compound Looks and Acts Like
Iodine pentafluoride grabs attention the moment you see it, mostly due to its pale yellow appearance. Pouring it brings an obvious liquid splash, but don’t expect a regular water-like flow; its viscosity reminds you this isn’t an everyday chemical. It sits just below 100°C before boiling off, and it doesn’t appreciate room temperature for long. Its fumes bite at the nose in a lab setting, and those who’ve opened a fresh bottle know the sharp, reactive scent that fills the air. Handling this liquid, you learn respect quickly, since it reacts so dramatically with water and organic matter.
The Chemistry Behind the Bite
At its core, iodine pentafluoride isn’t just reactive—it feels almost eager. Toss a drop into water, and you get an immediate hiss, releasing hydrofluoric and iodic acids. These aren’t gentle compounds—either can cause burns, and the fumes alone promise trouble. In the lab, we learned that glass can get etched and compromised if any residue sits for too long. The strength of the I–F bonds keeps the molecule together, but under the right conditions, it willingly donates fluorine atoms to other elements. That’s the edge it brings to the table; its oxidizing power means it can convert metals or nonmetals into higher oxidation states.
Why Fluorinating Agents Stand Out
Iodine pentafluoride goes beyond its color and vapor to serve a real purpose, especially where fluorine chemistry matters. Turning hydrocarbons or even certain metals into their fluorinated versions means industries don’t need elemental fluorine, which poses even more hazards. This makes life easier for chemists, yet every handling event reminds you of personal protective equipment’s value. Even seasoned researchers run through strict safety checks whenever IF5 comes out of storage.
Risks Keeping You On Your Toes
One misstep—like a wet glove or a sweaty palm—invites trouble. Skin contact with its hydrolysis products brings serious harm, and just a few breaths of vapor in an unventilated space causes coughing and eye discomfort. Accidents with IF5 don’t just clear out a lab; they take time, money, and health with them. That’s why modern storage leans on high-grade fluoropolymer containers, and every transport includes secondary containment.
Handling Hazards and Finding Better Practices
Mistakes in the past taught the hard lesson that dry conditions, adequate ventilation, and chemical-resistant gear must all line up before working with iodine pentafluoride. Automated pipetting and fume hoods add a helpful layer of safety. Emergency showers and eyewash stations close by aren’t optional; they are lifelines in chemical work. Companies now put more effort into training, and regulators enforce clear labeling and rigorous protocols. Knowledge and respect for this chemical’s properties have grown from stories and scars—the best teachers in any lab.
Looking for Safer Substitutes
Across academic and industrial research, the push to find alternative fluorinating agents stays strong. Compounds offering similar reactivity, but with less aggressive byproducts or easier containment, appeal to anyone tired of iodine pentafluoride’s risks. Green chemistry approaches try to minimize toxicity and find reagents that generate simple, manageable waste instead of acid-laden water vapor. Still, when demanding fluorination jobs pop up, IF5 maintains a spot in the chemical arsenal.
| Names | |
| Preferred IUPAC name | Iodine(V) fluoride |
| Other names |
Iodine fluoride (IF5) Pentafluoroiodine |
| Pronunciation | /ˈaɪəˌdiːn ˌpɛntəˈfluːraɪd/ |
| Identifiers | |
| CAS Number | 7783-66-6 |
| Beilstein Reference | 358873 |
| ChEBI | CHEBI:53083 |
| ChEMBL | CHEMBL108298 |
| ChemSpider | 21517 |
| DrugBank | DB01638 |
| ECHA InfoCard | 100.035.253 |
| EC Number | 231-556-4 |
| Gmelin Reference | 7257 |
| KEGG | C14315 |
| MeSH | D007471 |
| PubChem CID | 24816 |
| RTECS number | NN0175000 |
| UNII | 9GHU85V74V |
| UN number | UN1745 |
| CompTox Dashboard (EPA) | DTXSID0021798 |
| Properties | |
| Chemical formula | IF5 |
| Molar mass | 221.89 g/mol |
| Appearance | Colorless or yellow liquid |
| Odor | pungent |
| Density | 3.25 g/cm³ |
| Solubility in water | Reacts |
| log P | 4.3 |
| Vapor pressure | 26 mmHg (21 °C) |
| Acidity (pKa) | -2 |
| Basicity (pKb) | pKb ≈ 15 |
| Magnetic susceptibility (χ) | -57.8e-6 cm³/mol |
| Refractive index (nD) | 1.546 |
| Viscosity | 2.763 mPa·s (25 °C) |
| Dipole moment | 2.60 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 250.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −840.29 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –763.2 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Toxic if inhaled, causes severe burns to skin and eyes, reacts violently with water, releases corrosive and toxic fumes. |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | Hazard statements: Fatal if inhaled. Causes severe skin burns and eye damage. Causes serious eye damage. |
| Precautionary statements | P260, P262, P280, P301+P330+P331, P304+P340, P305+P351+P338, P310, P320 |
| NFPA 704 (fire diamond) | 4-0-2-OX |
| Autoignition temperature | 200 °C (392 °F; 473 K) |
| LD50 (median dose) | LD50 (median dose): 100 mg/kg (oral, rat) |
| NIOSH | SF7700000 |
| PEL (Permissible) | 0.1 ppm (as F) |
| REL (Recommended) | 0.01 ppm |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
IF3 ICl ICl3 IBr IBr3 I2 Iodine heptafluoride (IF7) Bromine pentafluoride (BrF5) Chlorine pentafluoride (ClF5) |

