Iodic Acid: More Than Just a Laboratory Staple
Historical Development: An Element’s Journey
Anyone who spent time in a chemistry lab probably recalls the sharp bitterness of Iodic Acid on the tip of the tongue—or at least the way it settles quietly at the back of the reagent shelf. Discovery isn’t often glamorous; Iodic Acid crept its way into chemistry books in the early 19th century after Bernard Courtois first isolated elemental iodine from seaweed ash. Chemists quickly realized that combining iodine with strong oxidizers led to several distinct acids. Iodic Acid, stable and crystalline, became a key object of study. Over the decades, it stood at crossroads between analytical chemistry and a handful of industrial processes. Without the splashy fame of sulfuric or nitric acid, it still carved a unique niche thanks to its oxidizing strength and reliable behavior. The major push came in the late 19th and early 20th centuries when industries ramped up iodine extraction for medicine and photography, pulling iodic acid along with it. By the time World War I rolled around, Iodic Acid had already found several homes in research and manufacture.
Product Overview: What Sits in That Bottle
Iodic Acid feels unassuming at a glance—white, crystalline, easy to handle. Chemically, it’s better known as HIO₃ and some old-timers might refer to it as hydrogen iodate. Open a bottle, and the solid flows like sugar through your fingers. Odorless, yet distinct in its tendency to cling to glassware, Iodic Acid rarely calls attention unless called upon for heavy-duty oxidation or analytical chemistry. Its role sprawls from analine dye intermediates to nutritional fortification in a handful of animal feeds. Iodic Acid offers predictability, vital for chemists who depend on results that track from week to week. In the grander landscape of acids, it lacks theatrics and wins instead through its straightforward nature and calculable results.
Physical & Chemical Properties: The Grit Behind the Name
Take a scoop out of a humidity-controlled bin and Iodic Acid presents itself as a clean, white, crystalline compound. Its melting point sits high, around 110°C, and it dissolves readily in water—making it easy to formulate or dilute as needed. As an oxidizer, HIO₃ commands respect. Drop it into an organic mixture and it chews through iodides and certain metals, liberating free iodine or complexes in the process. Controlling the reaction takes patience and protective equipment, since its contact with combustibles or strong reducers signals trouble. Purity levels usually top 99.5 percent in scientific supply chains; trace amounts of lead or arsenic earn quick rejection from regulatory agencies, especially in pharmaceutical or food supplement use. Chemists in fields like forensic science rely on these properties to push reactions forward or tease out minute traces of other iodine species for verification work.
Technical Standards & Labeling: Trust, But Always Verify
The bottle on the lab bench wears regulatory stickers: oxidizing agent, health hazard, and a UN shipping code for international transport. No lab can sidestep these marks. In regions where food or pharma production uses Iodic Acid for fortification, technical standards tighten further. Every batch comes with spectral and gravimetric data, confirming identity and purity down to tenths of a percent. Labels invoke national and international norms like the European Pharmacopoeia or US FCC. Without these, risk creeps in—not just for chemical handling, but in regularity and traceability audits. Documentation, thorough as it can be, keeps both the end-users and overseers confident that each scoop or solution brings exactly what it claims.
Preparation Methods: From Rough Rocks to Pure Reagent
No bottle of reagent-grade Iodic Acid appears in isolation. Most commercial supplies start with elemental iodine, often extracted from subterranean brines in Japan, Chile, and the US. Chemists oxidize iodine—with chlorine or nitric acid—forming intermediate iodine compounds before hitting an acidification step. After reacting with concentrated nitric acid or chlorine in an aqueous environment, crystalline Iodic Acid precipitates out as the solution cools or gets evaporated. The resulting cake goes through a gauntlet of purification steps, usually repeated crystallization and washing, to knock out trace impurities before packaging. Some labs on tighter budgets or with specific isotope requirements follow old-school routes: electrochemical oxidation of iodate salts or conversion from iodic anhydride, tuning the protocol for scale. What matters most is reproducibility. Whether for industrial-scale fortification or milligram-sensitive analytical work, supply chains have learned to favor those sources that document each step, batch by batch.
Chemical Reactions & Modifications: Power and Precision
Iodic Acid rarely rests idle on the shelf. The compound acts as a moderate oxidizer, snatching electrons from lower valence species with a reliability that makes it a favorite among educators and researchers. In classic redox titrations, Iodic Acid fills the role of electron acceptor—oxidizing sulfides, ferrous ions, and even certain organic compounds. Its interaction with potassium iodide generates iodine, a reaction that underpins iodine standardization tests worldwide. It combines with bases to yield iodate salts, which themselves play crucial roles in analytical chemistry and food fortification. Although researchers rarely push Iodic Acid much further—its most robust chemistry keeps to reliable, clean electron transfers—derivatives like periodic acid reveal the family’s range and thirst for electrons. By controlling the reaction milieu (acidity, temperature, concentration), chemists wring out selective results, either using Iodic Acid as a direct reagent or driving it toward more complex iodine-based oxidizers.
Synonyms & Product Names: The Many Faces on the Label
Nothing creates more confusion in a reagents storeroom than labels fighting for meaning. Iodic Acid pops up as hydrogen iodate, acidum iodatum, or simply HIO₃. Some suppliers rely on European traditions, others on US nomenclature. Catalogs may hide it under orthoperiodic acid or present it with translation quirks—none fundamentally changing the crystal underneath, but sometimes causing double-takes before the bottle finds a new home in the right chemical drawer. The common thread: every chemist recognizes the “IO₃” core, anchoring the substance to both its history and use.
Safety & Operational Standards: Respect Drives the Routine
Anyone who has handled Iodic Acid develops a healthy respect for its dangers and quirks. Contact with skin or mucous membranes stings. Inhalation by mistake signals immediate attention, as exposure can irritate respiratory tracts or, in larger doses, push thyroid function out of balance. Personal experience reinforces these written warnings—once caught with a cracked glove or careless flask pour, you remember the crisp burn or cloud of drawn breath for your whole career. Standard operating procedures demand gloves, goggles, and fume hoods. Older labs remember open beakers and casual handling, but regulatory bodies and safety consciousness now demand tight control. Disposal isn’t a toss-and-forget affair; neutralization and containment turn up in university training sessions and industrial manuals alike. Safety sheets flag the risk of fire if the powder contacts organic matter. Inconsistent use or forgotten precautions can result in serious harm to individuals and compromised experiment results.
Application Areas: Beyond Titrations and the Lab
The reach of Iodic Acid goes outside glass beakers and burettes. In animal feed production, this compound serves as a regulated additive to address iodine deficiency—a low-profile intervention with real effects on dairy and egg production in regions where soils run short of native iodine. Agrochemical companies keep iodic acid on their radar for selenium-iodine blends, stimulating animal growth and reproductive output. Analytical chemists, especially those designing environmental or food safety protocols, trust Iodic Acid’s chemistry for the clean and repeatable liberation of iodine in comparison tests. In some pharmaceutical syntheses, the compound bridges the gap between raw iodine and finished iodine compounds, providing a clean intermediate with manageable risks. Power stations and water-treatment specialists sometimes deploy Iodic Acid for sterilization or algae control—appreciating the way the molecule delivers oxidation without the lasting persistence of heavier halogens. It rarely draws headlines, but thousands of processes in dozens of trades quietly benefit every year from its predictability and mild footprint.
Research & Development: The Quiet Activity Behind Progress
Research into Iodic Acid hasn’t cracked the popularity charts in academic journals, but steady progress tells another story. Analytical teams develop new titration methods and detection techniques, leaning on the clean reactions Iodic Acid provides. In bioinorganic chemistry, researchers track the fate of iodine species using Iodic Acid to establish baseline measurements. Food scientists and nutritionists partner to review whether iodic acid fortification remains the safest way to reverse endemic goiter in deficient populations. Engineers working in advanced battery chemistry ask whether certain iodine compounds—born from simple molecules like Iodic Acid—could stabilize next-generation energy storage. Toxicologists keep reviewing data, tuning the compound’s risk profile with new evidence and more careful case studies. Progress rarely gets a splashy headline, yet real advances almost always build on foundations set in labs decades ago.
Toxicity Research: Complication and Clarity
Iodine, in most of its forms, sits on a razor’s edge. On one side, it’s life-saving in the right dose; on the other, it’s a toxin. Iodic Acid doesn’t escape this double-edged sword. Animal studies confirm that high or prolonged exposure triggers thyroid disruption and organ distress. At moderate levels, ingestion can cause chronic symptoms—dryness, skin irritation, joint pain. Handling accidents shine a light on rapid toxicity, especially for those allergic or sensitive to iodine compounds. Workplaces staffed by experienced chemists drive home proper handling and immediate response to spills or skin contact, as recovery from exposure involves more than a quick rinse. Research pushes for lower workplace exposure limits, improved detection methods, and anti-exposure protocols tailored for industrial users. Both the medical and regulatory communities keep reviewing and updating guidelines, relying on two centuries of accumulated reports, animal data, and the occasional human mishap.
Future Prospects: A Substance on the Upward Curve
The world doesn’t usually stop to notice the products that make everything else work better, yet under-the-radar chemicals like Iodic Acid power incremental progress. Scientists looking for safer food fortification in developing nations watch for evidence supporting slow-release iodic acid pellets or blended animal feed additives. Environmental chemists consider Iodic Acid’s ability to sanitize water or reduce organic pollutants without creating persistent halogenated waste. Battery manufacturers investigating iodine-based redox flow systems weigh upfront toxicity against high energy density—searching for evidence that the old-school chemistry can withstand modern demand. Meanwhile, the world’s population grows, and the pressure mounts for reliable, safe, and controllable sources of essential elements like iodine. As new research emerges, expect policies and practical usage methods to adapt the lessons of the past to the challenges of tomorrow, with Iodic Acid maintaining its steady, unglamorous but vital role.
What is Iodic Acid used for?
Getting Down to the Basics
Iodic acid might sound like one more chemical in a long list, but I keep it at the top of my mind when thinking about science, nutrition, and even water safety. In labs, it’s a strong oxidizer. If you ever checked out high school chemistry, you saw reactions light up with iodic acid driving things forward. It’s one ingredient that pulls its weight—whether you’re creating iodine compounds for medicine, or helping students understand chemical reactions.
Protecting Health Through Nutrition
Countries with healthy populations often have something in common: enough iodine in the diet. I grew up hearing about “iodized salt,” but never knew how the industry made sure iodine stayed put in the shaker. Here’s where iodic acid steps in. In salt production, it stabilizes iodine so it doesn’t just disappear before it even hits the table. Iodic acid provides a form of iodine that’s less likely to volatilize or get washed away during storage and shipping—something salt producers struggle with in hot or humid climates.
Without enough iodine, thyroid problems sneak in and steal people’s energy and focus. More than two billion people live in regions where the natural world doesn’t hand out much iodine. I witnessed firsthand the impact in rural health clinics, where simple goiters remain common when diets fall short. The World Health Organization points out that using chemicals like iodic acid for fortification cuts back on these problems—a simple solution that works.
Beyond Nutrition: Testing and Industry
With iodic acid sitting on lab shelves, analysts use it to standardize chemical solutions. Its predictable behavior means analysts can trust their results. Testing for vitamin C in orange juice at the bottling plant, or checking the purity of pharmaceuticals before they reach patients, both use iodic acid for accuracy. I could never overstate the importance of this kind of confidence in chemistry—small errors here ripple out far beyond the lab.
Industries that make specialty glass lean on iodic acid too. Even minor tweaks to the chemistry change the way glass looks and works, which matters for high-end optics or safe laboratory containers. Battery makers sometimes rely on iodic acid to get the behavior they want from their products as new technologies call for better power sources.
Challenges and Possible Paths Forward
Working with strong oxidizers always demands respect. Mishandling brings risk. Factories and laboratories that use iodic acid keep protocols tight, because no one wants an accident spilling into local water supplies or putting workers in danger. Regulators call for careful tracking of hazardous materials. I see a growing push for safer alternatives or better containment methods—especially as manufacturing expands in less regulated parts of the world.
On a bigger scale, education makes a difference. Teaching workers, engineers, and even farmers how these chemicals behave cuts down on mistakes. Companies that spend time on safety and transparency build public trust. Public health authorities and researchers can help, too, by keeping up studies on the long-term environmental impact and encouraging better recycling or neutralization.
Why It Matters
Iodic acid doesn’t get the spotlight, but it underpins stability in areas that touch millions of lives, from nutrition programs to testing in high-tech labs. Treating these chemicals with respect, improving oversight, and staying curious about safer practices set everyone up for fewer headaches. Real progress rests on steady, practical use—something iodic acid has quietly delivered for decades.
Is Iodic Acid hazardous to health?
The Nature of Iodic Acid
Iodic acid turns up in a few specialized fields—mostly labs, chemical synthesis shops, and sometimes in water treatment spaces. In high school chemistry class, someone once spilled a tiny bit of it during an experiment, and the whole room tensed up as the teacher rushed for the materials safety data sheet. That response stuck with me because it showed how easily caution can turn to panic, even among people used to handling chemicals.
Health Hazards Are Real—But Manageable
Anyone asking about the risks of iodic acid probably wants clarity, not alarm bells. The stuff is corrosive. On skin, it burns. If it splashes in your eyes, it may cause permanent damage. Inhaling dust or mist can irritate the lungs and throat. Swallowing it—never a good idea—often leads to severe internal harm. The main risk follows from contact: the acid breaks down living tissue just like it corrodes lab surfaces or metals.
There’s another layer to consider. Iodic acid contains iodine. Workers exposed to airborne particles day after day might absorb enough iodine to throw off their thyroid function, since the body only needs a trace for regular hormone production. Thyroid problems can mean anything from tiredness and weight fluctuations to more serious cardiovascular effects.
Managing Exposure in the Real World
Chemical safety always comes down to respect for the substance and respect for the people who use it. People who work with iodic acid daily wear gloves, goggles, and coats, and use fume hoods to catch vapors and dust. Cleaning up spills means neutralizing the acid with something alkaline—usually baking soda, in a pinch, then scooping everything with tools, not hands.
A mistake I’ve seen over and over in schools or hobby labs: chemicals left open or mislabeled. Iodic acid looks harmless in a clear bottle, so someone unaware may treat it like water. Proper labeling and locked cabinets go further in keeping a space safe than any warning label.
Fact-Checking the Risk
According to the CDC and NIOSH, iodic acid sits in the same camp as other strong acids (like sulfuric), needing good ventilation and prompt cleanup of spills. Poison control centers advise immediate medical help if it’s swallowed or splashed in the eyes. Long-term exposure data is less common—probably because this acid doesn’t get used often outside specialized workplaces. The World Health Organization marks it as hazardous, but less concerning than industrial toxins like mercury or asbestos, so fears can be balanced with context.
Reducing Hazards Starts With Education
Turning chemistry class into a safety culture made a huge difference for me. We practiced mock emergencies, which felt excessive at the time, but years later, those drills kept my hands out of harm’s way, even during routine tasks. Sharing these simple safety basics keeps everyone protected—never underestimate the value of habit when it comes to dangerous chemicals.
Companies using iodic acid often put strict safety measures in place: regular training, signs in plain language, checklists next to storage cabinets. If the goal is to lower risk, consistency trumps everything. Even small workplaces can post clear guidelines near sinks and hoods, so the rules never become background noise.
Practical Solutions Matter More Than Fear
Iodic acid requires respect—and it’s that respect, not fear, that keeps people safe. If schools, labs, and factories make sure only trained folks handle it, and nobody cuts corners with protective gear or storage, the risks shrink fast. It’s a harsh substance, but with the right handling, it rarely bites back.
What is the chemical formula of Iodic Acid?
Looking Beyond the Letters: Why HIO3 Matters
Staring at the formula HIO3 for iodic acid, it’s easy to forget how much goes on behind those three letters and numbers. This compound brings together hydrogen, iodine, and oxygen in a way that has real-world impact. High school chemistry reminds us that H stands for hydrogen, I for iodine, and O for oxygen. Combining them creates a molecule that people working in medicine, research, and industry have relied on for years.
Digging Into Iodic Acid’s Properties
HIO3 doesn’t just show up as a name in a textbook. It acts as a strong acid and a powerful oxidizing agent. In the lab, the solid form appears white and crystalline. In water, it dissolves cleanly, ready to release its protons and tip the pH scale. Scientists pick it for analytical chemistry because of its stable reactivity. In my university years, every student had to know the difference between iodic acid and its cousins like iodous acid or hydroiodic acid. Only HIO3 packs the right punch for certain chemical reactions, particularly where controlled oxidation matters.
Iodic Acid in Public Health
Dive deeper, and HIO3 plays a role in protecting communities. Public health teams need iodine in safe, effective forms to fight iodine deficiency, which causes thyroid problems around the world. People often discuss table salt and iodine supplements, but the journey starts with compounds like iodic acid. It offers a source of iodine that doesn’t quickly evaporate, even in humid regions. UNICEF and the World Health Organization highlight ways to iodize salt, and HIO3 always makes the shortlist because of its stability.
Environmental Impact: Where Chemistry Meets Nature
Every chemical, even ones with obvious benefits, leaves an imprint on the world. Factories producing large volumes of HIO3 need to handle waste and exposure risks. Iodic acid itself doesn’t build up in the environment the way some industrial byproducts do, but mishandling can still strain water sources. In my short stint working in waste management, we always paid close attention to strong acids, knowing their impact didn’t always vanish after neutralization. Smart facilities reuse and treat every drop, not just to meet regulations but because every community downstream deserves protection.
Thinking About Safer Practices and Broader Access
Iodic acid’s story reflects bigger issues in chemical safety and public health. More research helps find uses with lower risks, such as new techniques in iodine supplementation or less hazardous ways to dispose of acid waste. Teams from universities and private labs work together, often sharing best practices. I've seen smaller water treatment outfits benefit just from open-source guides about equipment and handling protocols. Solutions also extend to education—young chemists need practical training, not just theory, to keep both themselves and their environment safe.
The Core Value of a Simple Formula
HIO3 may look simple, but its value runs wide and deep. Whether used in testing, health supplements, industry, or environmental protection, its management calls for care and respect. Chemistry isn’t only about mixing powders or memorizing equations. It’s about the choices people make—how they use the knowledge behind these formulas to shape safer, healthier lives.
How should Iodic Acid be stored?
Why Proper Storage of Iodic Acid Deserves Attention
People tend to overlook chemical storage until something goes wrong. I recall chemistry labs in high school where a small mistake—like placing a reagent on the wrong shelf—could create a mess or, worse, pose danger. Iodic acid isn’t a compound you want to treat lightly. It stands out for being a strong oxidizing agent, and with that comes some risk if left unchecked.
Understanding Iodic Acid’s Temperament
Iodic acid doesn’t explode on sight, but it reacts in unpredictable ways when it meets organic materials, flammable liquids, or reducing agents. Leaving it out on a counter or near incompatible substances adds real hazards to any workplace. Safety authorities and chemical suppliers, including Sigma-Aldrich and Fisher Scientific, underline this. Ignoring these warnings brings unnecessary risk to people nearby and the environment beyond those walls.
Storage Conditions For Health and Safety
A few non-negotiables stand out. Store iodic acid in a tightly closed container to keep moisture out and avoid dust formation. I’ve seen people transfer chemicals to makeshift bottles—a practice born out of convenience, not safety. Stick with the original containers provided by chemical manufacturers. Polyethylene and glass offer solid resistance against leaks and accidents, while old soda bottles or unmarked jars just invite trouble.
Temperature matters as much as the container. Keep it in a cool, dry, well-ventilated spot—away from sunlight, heat sources, and sparks. Chemical storerooms shouldn’t feel like a sauna or a greenhouse. Any spike in temperature, even on a hot summer day, may raise the risk of unexpected chemical reactions.
Segregation Stops Trouble Before It Starts
It’s tempting to group everything together on a shelf to save space, but mixing oxidizers like iodic acid with organic chemicals or reducing agents can trigger fires. I once toured a lab that separated substances by risk factor, each section marked with bright labels and simple pictograms. Keeping iodic acid apart from combustibles, acids, bases, and reducing agents cuts down on accidental contact. Safety cabinets rated for oxidizers don’t cost much compared to the damages an unmanaged reaction can bring.
Ventilation Protects Workers, Not Just the Compound
Working near stored iodic acid means breathing becomes a concern if the container leaks or breaks. Good ventilation disperses any fumes or accidental dust, protecting air quality. Relying on a simple fan won’t cut it here—fume hoods or dedicated chemical exhaust systems deliver stronger protection.
Emergency Preparedness Isn’t Optional
Storing iodic acid isn’t just about right shelving. Have spill kits, eye wash stations, and fire extinguishers close by. Employees should actually know how to use them. Clear labels on storage containers don’t just satisfy regulations—they let everyone know exactly what they’re dealing with, including first responders.
Training makes all the difference. People need reminders on what goes where, how to recognize signs of chemical deterioration, and what to do if they spot a leak. Regular safety checks help catch problems long before they start.
Putting Safety Above Convenience
All this effort might feel excessive on a calm day, but the consequences of neglecting basic storage rules can ripple well beyond the four walls of a storeroom. Respecting iodic acid, reading the labels, and using the right storage strategies turns a risky chemical into a manageable one—responsibility is far better than regret.
What are the safety precautions when handling Iodic Acid?
Respecting a Strong Oxidizer
Iodic acid pops up in some chemical labs, especially in analytical work, and it comes with a reputation for being a powerful oxidizer. My early days in the lab taught me that strong acids need respect, but something stronger like iodic acid ups the stakes. It’s not like handling table salt—every little shortcut raises real risks, both for people and the workspace. Working with it, I always felt a quiet pressure to double-check myself, because accidents have a way of stacking up fast if you let your guard down.
Staying Protected
Putting on gloves and safety goggles seems obvious, but with iodic acid, I go for more than basic gear. Nitrile gloves stop the acid getting through; latex doesn’t cut it, and I’ve seen folks learn the hard way that regular work clothes don’t block splashes. A proper lab coat, closed shoes, and eye protection are non-negotiable. If you ever get even a drop on your skin, it starts to burn and itch almost straight away. With eye contact, you’re not just looking at discomfort; there’s a real chance of permanent injury.
Keeping the Air Clean
Iodic acid powder can throw up dust that’s easy to inhale. I always do the weighing and transfer in a fume hood. Otherwise, tiny crystals drifting into the air end up in your nose or lungs. One labmate of mine didn’t think twice and worked outside the hood—he ended up coughing and wheezing for days. That fume hood isn’t just another piece of equipment; it’s the barrier between you and the stuff you absolutely don’t want in your body.
Mixing Without Mishaps
Mixing iodic acid never means rushing. Pour too fast, add to the wrong solvent, or bring it near combustible materials, and things go sideways quick. I read about a researcher who mixed it with a bit of organic solvent without thinking; the reaction ruined their bench and nearly set off a fire. It wasn’t a freak one-in-a-million event, either. Storing this acid near paper, oil, or anything flammable multiplies the risk. I double up on labeling and never let acids or organics share the same cabinet.
Getting Rid of Waste
It’s tempting to treat any acid waste as just another bottle for disposal. With iodic acid, I take less for granted. Mixing waste streams causes reactions that even seasoned chemists misjudge. Every local guidelines sheet I’ve seen stresses separate acid waste bottles, and I always alert the safety officer if I’m not sure. Even a dilute solution can still act as an oxidizer. There’s no shame in asking; plenty of minor spills started with someone hoping to save a few minutes by skipping official procedures.
What Steps Help?
More regular training makes a difference. Newcomers don’t always know what a “strong oxidizer” can do until it’s too late. I keep an SDS (Safety Data Sheet) close—these aren’t just for audits, they answer what to do if something goes wrong. Clear signage, ready access to eyewash stations, and making spill kits prominent keep everyone a bit sharper. Peer checks during risky procedures cut down on mistakes. We talk through the steps together, even if it slows things down. In all my years, I’ve never regretted taking that extra moment.
| Names | |
| Preferred IUPAC name | Iodic acid |
| Other names |
Hydrogen iodate Iodic(V) acid Iodine pentoxide hydrate |
| Pronunciation | /ˌaɪ.əˈdɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 7782-68-5 |
| Beilstein Reference | 3587151 |
| ChEBI | CHEBI:24859 |
| ChEMBL | CHEMBL1366 |
| ChemSpider | 57907 |
| DrugBank | DB01811 |
| ECHA InfoCard | 100.029.070 |
| EC Number | 231-839-9 |
| Gmelin Reference | 7315 |
| KEGG | C16535 |
| MeSH | D007470 |
| PubChem CID | 24847 |
| RTECS number | NN1575000 |
| UNII | 88X2Z36974 |
| UN number | UN1479 |
| Properties | |
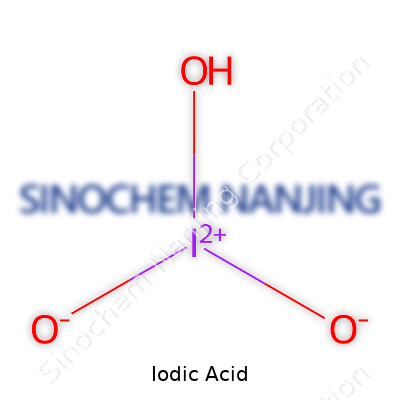
| Chemical formula | HIO3 |
| Molar mass | 207.89 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 4.62 g/cm³ |
| Solubility in water | Very soluble |
| log P | -2.0 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 0.80 |
| Basicity (pKb) | 11.1 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 1.867 |
| Viscosity | 1.734 cP (25 °C) |
| Dipole moment | 1.82 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 146.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -295.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -295 kJ/mol |
| Pharmacology | |
| ATC code | V09IA05 |
| Hazards | |
| Main hazards | Oxidizer, corrosive, harmful if swallowed, causes severe skin burns and eye damage |
| GHS labelling | Warning; H272, H314, P210, P220, P260, P280, P305+P351+P338, P310, P501, GHS03, GHS05 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H314: Causes severe skin burns and eye damage. |
| Precautionary statements | P210, P220, P221, P264, P273, P280, P301+P312, P305+P351+P338, P330, P370+P378, P403+P233, P501 |
| NFPA 704 (fire diamond) | 3-0-2-OX |
| Lethal dose or concentration | LD50 oral rat 1100 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1,420 mg/kg (oral, rat) |
| NIOSH | KW3850000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 0.1 – 0.3 ppm |
| IDLH (Immediate danger) | IDLH: 50 mg/m³ |
| Related compounds | |
| Related compounds |
Chloric acid Bromic acid Periodic acid Hypoiodous acid Iodous acid |

