Indoline-2-Carboxylic Acid: A Commentary on Its Role, Development, and Prospects
Historical Development
My work with organic molecules leads me toward stories wrapped up in both trial and ambition. Indoline-2-carboxylic acid is no stranger to either. Chemists noticed the indoline backbone many decades ago, tracing interest back to early investigations of nitrogen-containing heterocycles. Through the mid-20th century, synthetic chemists started isolating indoline derivatives, sketching out their reactivity with the reliable patience of glassware and heat. Interest blossomed once folks saw connections between the structure and possible routes into complex bioactive compounds. Years of hands-on lab work—hot-plate reactions, solvent swaps, constant reflux—pushed this compound from a sketch in a German patent to a named entry in training manuals and industry supply lists. Progress never looked linear, with many routes explored until efficient methods became preferred for both research and scaled-up production.
Product Overview
Indoline-2-carboxylic acid is a small organic molecule. It comes as a white crystalline solid if it’s pure enough, with slight off-white tones if not. The structure features a fused benzene and pyrrolidine ring, dressed with a carboxylic acid group on the two position. Its uses run from chemical building blocks for peptides to feedstock for more complex small molecules, bridging the gap between basic organic synthesis and advanced pharmaceutical research. Many labs will list it as a multipurpose intermediate, especially when exploring enzyme inhibitors, imaging agents, or cardiovascular lead compounds. Its presence says a lot about the curiosity and persistence of chemists, aiming to pull valuable new traits out of the ordinary.
Physical & Chemical Properties
Indoline-2-carboxylic acid comes parked with a melting point around 196-200°C—good thermal stability, not fussy about ordinary bench conditions. It dissolves in water just enough for most practical work, thriving much more in polar organic solvents like ethanol, methanol, or dimethylformamide. Under a sharp nose, it lacks any strong or offensive aroma, a blessing in a world full of chemicals that scream their presence through scent. Its molecular weight hovers near 163 g/mol. On a chemical level, the carboxyl group reacts as expected: acids-neutralize, amides-form, esters-synthesize. The aromatic indoline ring provides rigidity and surprises chemists by shielding parts of the molecule from unwanted reactions, while still offering room for modification at various sites without breaking the core structure.
Technical Specifications & Labeling
My experience reviewing product labels for fine chemicals teaches me to look for detailed disclosures: purity, form (hydrate or anhydrous), lot number, production date, and the supplier’s address and contact information. For indoline-2-carboxylic acid, reputable vendors fix purity at 98% or higher, sometimes adding “HPLC grade” or “analytical standard” for specialty shipment. Packaging runs from small amber bottles—handy for research—to kilo-bags for pilot plants. Hazard information, storage requirements (cool, dry, away from incompatible oxidizers), and suggestions for safe handling all appear on the technical data sheets. The best suppliers anticipate international trade constraints or compliance with REACH in Europe or TSCA in North America by listing all relevant regulatory information with the shipment. Folks on the lab floor value this clarity as much as the compound itself, since bad labeling or incomplete documentation blocks research and wastes resources.
Preparation Method
Most synthetic chemists choose between the Fischer indole synthesis or a modified Pictet-Spengler route depending on availability of starting materials and the desired quantity. I’ve found that reactions starting with 2-nitrobenzaldehyde or related substrates often give reasonable yields, especially when coupled to catalytic hydrogenation to bring about the indoline core. Acidification then liberates the carboxylic acid group, no longer hidden as an ester or masked as a protected group. For large-scale work, folks often lean toward more environmentally friendly solvents or route modifications that minimize extraction steps. The process isn’t set in stone; in recent years, enzymatic or microwave-assisted chemistries lowered costs and trimmed reaction time while maintaining high purity. Recrystallization and proper drying nudge the product to a reliable, stable form.
Chemical Reactions & Modifications
Indoline-2-carboxylic acid plays nicely in reactions typical for both aromatic compounds and carboxylic acids. It supports amide coupling to give way to peptidomimetics, often target scaffolds in pharmaceutical screening. It tolerates reductions or oxidations at the indoline ring system, sometimes opening the door to derivatives like indoles and hydroxyindolines. Direct halogenation or nitration can occur on the benzene ring, giving chemists levers to tune solubility or bioactivity. Esterification and decarboxylation add to the mix, and in some hands the compound morphs into whole new skeletal structures after careful transformation. Ongoing research pushes bioconjugation and metal-catalyzed cross-coupling as modern moves for this acid, with every change in protocol offering a shift in function and potential use case.
Synonyms & Product Names
This molecule slips through the literature under many labels: 2-Carboxyindoline, 1,2,3,4-tetrahydroindole-2-carboxylic acid, and sometimes the abbreviation ITICA. You’ll also spot international synonyms, reflecting legacy catalogs or supplier house brands. Thoughtful product listings match the CAS number (1652-44-2) against chemical nomenclature to ensure nobody grabs the wrong variant. Old lab notebooks and product orders sometimes just collect dust with “indolinecarboxylic acid” on the cover. What matters to researchers and industry buyers is clarity in the trade names and accuracy in the labeling—a slip here means wasted weeks or lost opportunity for new insights.
Safety & Operational Standards
Lab workers adapt quickly to safety requirements. Handling indoline-2-carboxylic acid demands the usual gear: gloves, goggles, solid airflow over the bench, and no food or drink anywhere nearby. While acute toxicity data is less notorious than for some other aromatic amines, ingestion or inhalation is never taken lightly. Dust forms during open handling, especially on dry days, so careful transfer and cleanup keep respiratory exposure low. Safety data sheets point out mild skin and eye irritation risk, with the potential for respiratory discomfort after extended or careless exposure. Waste disposal tracks standard organic acid protocol—neutralize, bundle for incineration through licensed handlers. Employees at production or handling facilities train regularly against spill scenarios and accidental exposure, bringing compliance with OSHA or equivalent global standards. That diligence reflects more than just box-checking; it grounds laboratory culture in responsibility.
Application Area
Indoline-2-carboxylic acid shows up most in advanced pharmaceutical research. Medicinal chemists scavenge its core to build novel peptidomimtics, probing how new substitutions affect physiological activity and drug-candidate stability. Some researchers fix its skeleton into enzyme inhibitors, anti-inflammatory prototypes, and imaging tools for cancer detection. Biochemical supply houses market it for specialty synthesis in peptide chemistry, looping it into libraries for automated combinatorial discovery. Beyond pharma, organic electronic developers test it in new materials’ synthesis, betting on the stability and predictable reactivity of the indoline unit. Universities teaching organic chemistry value its manageable reactivity in student experiments, showing how a single molecule turns into many other valuable tools with a slight tweak. The real value gets measured in discoveries made and hurdles crossed using this acid as the starting point.
Research & Development
New research on this molecule takes two broad directions. The first travels deeper into making analogs that hit novel therapeutic targets or sharpen the selectivity of existing leads. Chemical modification strategies chase higher yield, fewer side-products, and shorter synthetic routes. Some researchers study its behavior under enzymatic conditions, drawing lessons from natural biosynthetic pathways in animals and plants. The second embraces new application spaces: solar cell research, advanced coatings, metal complexation, and polymers all see the indoline backbone as a possible advantage. Collaboration between labs, especially through global partnerships, brings new screening results and physical property data back into the public domain. Peer-reviewed papers document breakthroughs, with transparent detailing of methods and experimental challenges, so others can pick up where one group leaves off.
Toxicity Research
Toxicologists dig for long-term effects, drawing lines between acute symptoms and slow-onset risks. Indoline-2-carboxylic acid doesn’t show the alarming mutagenicity of some related compounds, but studies always call for caution and follow-up. Standard animal models confirm low acute oral and dermal toxicity, but they also highlight mild skin irritation and the uncertainty of prolonged inhalation. Researchers keep watch for metabolites, checking if the compound or its downstream products interact with liver enzymes or trigger oxidative stress. Workers in industry settings respect that even routine exposure accumulates risk, leading management to install monitoring equipment and require periodic health screenings. Any new data from animal or cell studies gets logged and weighed against exposure protocols, since the learning never stops.
Future Prospects
People working on future paths for indoline-2-carboxylic acid see promise in broadening both the chemistry and the application field. Improved synthetic processes—possibly running under greener, less energy-intensive conditions—offer both economic benefit and a lighter environmental footprint. The tail ends of pharmaceutical research keep circling back, as new disease targets appear suited for modifications of the indoline core. Some companies place bets on specialty materials, such as organic frameworks for sensing or selectively binding environmental toxins, using the acid group as a point of entry. The next breakthroughs will not fall from the sky but from labs where chemists, engineers, and toxicologists work side by side, pooling practical know-how and curiosity in equal measure. For anyone following fine chemicals, the story of indoline-2-carboxylic acid serves as a reminder that progress builds from daily attention to small details, clear records, and an unbroken chain of questions.
What is Indoline-2-Carboxylic Acid used for?
Stepping Beyond the Science Lab
Some chemicals stay tucked away in obscure textbooks, but indoline-2-carboxylic acid keeps making appearances where real discovery happens. Walk into a medicinal chemistry lab, and chances are, you’ll spot it among the core building blocks. Over years of conversations with researchers and hands-on experience in small-scale synthesis, I’ve noticed its reputation holding strong for good reason: it forms the backbone of many pharmaceutical and agrochemical products.
The Building Block for Treatments and Techniques
Drug discovery moves fast, and chemists often feel pressure to find lead compounds that show promise early in the process. Indoline-2-carboxylic acid provides a springboard for creating molecules that interact with proteins inside the body in very specific ways. Sometimes, a minor tweak on the indoline ring changes everything, helping the compound stay stable longer or bind tighter to a biological target. This not only saves time, but sometimes saves entire projects from dead ends.
Working with indoline-2-carboxylic acid led to breakthroughs in areas like anti-inflammatory drugs and antibiotics. Journal articles and patents back up these claims, highlighting successful modifications of the molecule to produce active agents for pain relief or improved antibacterial properties. Chemists don’t stumble upon those advances by accident; they choose starting points that have shown potential in earlier trials, and indoline-2-carboxylic acid has the track record that counts.
Fine Chemicals and Material Science
Indoline-2-carboxylic acid sits at the crossroads of fine chemical production, too. In dye and pigment manufacturing, it plays an essential role in creating compounds that resist fading. Paint experts and textile chemists both value color stability. From fabric that holds its blue edges through summer after summer, to coatings for electronics, this chemical walks the line between utility and innovation.
In material science, attention shifts from formulas to function. Indoline derivatives, used in certain conductive polymers, can boost battery life or improve solar cell efficiency. Development teams often turn to these derivatives while testing out new devices, aiming for materials that last longer or charge faster.
Navigating the Challenges
Production scale-up and safe handling never lose their importance. Chemists who spend their days in the lab know handling indoline-2-carboxylic acid requires reliable equipment and protective measures. Accidental spills or dust clouds pose risks, not just to the person, but to the whole batch. Training, clear labeling, and regular safety audits limit hazards. It’s a lesson learned early and remembered often.
On the ethics side, companies need to vet sources and production methods to minimize environmental impact. Waste byproducts, left unchecked, can damage waterways or air quality. Responsible producers invest in efficient systems that recycle solvents or capture emissions, supporting both community health and long-term business stability.
Bridging Science and Society
Years spent talking with students and industry veterans taught me the same lesson: important chemicals don’t exist in a bubble. Indoline-2-carboxylic acid underpins therapies, agricultural products, and new materials that shape lives beyond the lab. Smart investment in research, balanced with strong regulations and thoughtful environmental care, sets the stage for safe and steady progress. Every vial, every experiment—each part counts toward the whole, proving that practical chemistry makes a difference you can see and feel.
What is the molecular formula of Indoline-2-Carboxylic Acid?
The Structure Behind the Name
Organic chemistry has a way of throwing long, complicated words at you. Indoline-2-carboxylic acid sounds like one of those intimidating terms you’d find in a graduate-level textbook, but dig into its structure and you start to see a familiar form. This compound is made up of fused rings and a carboxylic acid group, essential in many biological and synthetic pathways.
The molecular formula for indoline-2-carboxylic acid is C9H9NO2. You’ve got a nine-carbon backbone, one nitrogen, and two oxygens. For anyone who has spent time struggling through spectrometry data or laboring over mole conversions in the lab, recognizing these counts means you’re halfway to understanding reactivity and use. Locations of the atoms shape everything. The carboxyl group hangs off the second carbon—right on the fusion of the benzene and five-membered ring. It’s not ornamentation. This structure lets the acid behave differently compared to its positional isomers.
Molecular Formulas: Why They Matter
Some might think a formula is just a static string, but nine carbons, nine hydrogens, a nitrogen, two oxygens—this configuration means everything in the organic lab. Chemists rely on these numbers to navigate unexpected outcomes. In pharmaceutical research, I’ve seen molecular formulas become a sort of shorthand for how reactive a material acts, what solvents it prefers, and how easy it is to purify. Use the wrong formula, and you end up repeating work, wasting valuable reagents.
Indoline-2-carboxylic acid sits in the limelight for a few reasons. Its backbone resembles structures you’d see in pharmacology—precursors to drugs used in mental health, antiviral medications, and more. The specific arrangement contributes to functional behavior in synthesis. One example: its ease of forming amide bonds makes it a key building block. Lab work can become expensive, especially if sourcing materials with rare or ill-defined formulas. Using precise formulas keep things verifiable.
Quality and Verification: Trust Built on Experience
Lab routines have trained me not to take these formulas lightly. Skepticism serves well. Products labeled with the wrong molecular formula ruin results, and trust in chemical suppliers matters as much as the substance in the vial. I’ve seen teams waste days due to a subtle typo in a product description. Many researchers use NMR, mass spectrometry, and other analytical tools to match real-world samples to these formulas, double-checking every time. Peer-reviewed studies on indoline derivatives point to this cautious, detail-driven approach as critical to reproducibility and progress. The more precise the information, the more valuable the result.
Improving Accuracy and Reliability
Mistakes happen, so the chemical community benefits from open databases, shared protocols, and routine analytical verification. Educational efforts that go beyond memorizing structures help researchers ask better questions—honing in on how formulas influence real-world outcomes. Encouraging cross-lab checks and providing open-access data prevents tiny errors from snowballing. Building on the established molecular recipe of indoline-2-carboxylic acid, chemists open doors for new catalyst designs and safer drugs.
Moving Forward with Better Chemistry
Knowing the formula C9H9NO2 forms the baseline for chemistry work. Years of lab frustrations and breakthroughs have taught me that accuracy at this level leads to larger wins. Science moves faster—and safer—when teams use the right molecular information from the start.
How should Indoline-2-Carboxylic Acid be stored?
Understanding the Risks and Rewards
Few folks outside chemical labs spend much time thinking about indoline-2-carboxylic acid. But for those who work with it—pharmaceutical manufacturers, academic chemists, quality managers—the details of storage have real weight. This compound plays a role in research, especially in synthetic pathways leading to drugs or advanced materials. Mess up the storage, and you risk contamination, loss of product, or even safety incidents. That isn’t just a nuisance, it’s a hit to budgets and research timelines. Watching a valuable batch degrade because of a preventable mistake always stings.
Keep the Container Right
I remember supervisors drilling into us in the lab: the container matters. Polyethylene screw-cap bottles usually stand up well. Glass bottles with sturdy, airtight lids also do the trick. Avoiding metal containers cuts down on the chance of unwanted reactions. Clean, dry, and dedicated bottles for indoline-2-carboxylic acid help prevent cross-contamination, and labeling each with the date and batch keeps recalls and mishaps to a minimum.
Temperature and Light: Two Big Factors
Most chemical suppliers recommend room temperature storage, around 20-25°C. Too much heat, and there’s a risk of breakdown or unwanted side reactions. Turning the storeroom into a sauna spells trouble. Refrigeration isn’t standard unless a supplier specifically calls for it, but make sure to avoid direct sunlight. Light, especially ultraviolet rays, can start breaking molecular bonds—fast. Shelves away from windows or in a cabinet with solid doors go a long way.
Humidity: The Silent Saboteur
Even with solid containers, humidity creeps in. Water vapor works on chemicals in ways that cause caking, clumping, or unwanted hydrolysis. Keeping a desiccator handy—those sealed jars or cabinets filled with drying agents—helps extract stray moisture. Not every lab invests in fancy climate control systems, but even a basic silica gel pack tucked inside the storage cabinet makes a measurable difference. In places with sticky, humid summers, this simple trick keeps quality up and losses down.
Handling Residues and Spills
Sloppy handling has sunk more than one research project. Traces left in the wrong place can contaminate other compounds, so using separate scoops and avoiding “double-dipping” matters. Spills aren’t just wasteful—they put staff at risk. Indoline-2-carboxylic acid doesn’t have a reputation for high toxicity, but no one wants even low-level exposure. Standard gloves, lab coats, and goggles keep the work safe, and staff training—both formal and on-the-job—nips bad habits before they spread.
Documentation and Supplier Communication
Suppliers provide material safety data sheets for indoline-2-carboxylic acid. Reading those documents before opening the bottle saves headaches. Regulations sometimes change, and keeping an updated copy—digital or printed—close to hand helps resolve arguments or confusion fast. Staying in touch with suppliers goes beyond just buying—it opens a channel for updates or tips if they change recommended storage.
Room for Improvement
For larger operations, a digital inventory system reduces overlooked expiry dates and overstock. Barcode scanners, basic spreadsheets, or more advanced lab software help reduce waste. Smaller labs can match this by setting up regular checks and keeping a simple log. Storing compounds like indoline-2-carboxylic acid right isn’t glamorous, but it supports both everyday lab safety and breakthroughs in the field.
Is Indoline-2-Carboxylic Acid hazardous or toxic?
Looking Past the Lab Bench
Indoline-2-carboxylic acid appears in a lot of chemical discussions, especially in pharmaceutical R&D, but most people outside the lab probably haven’t heard of it. The name sounds intimidating, so it’s easy for concerns to bubble up about health or environmental harm. The truth requires context, caution, common sense, and respect for proper handling.
Direct Health Risks: Fact Over Fear
Not every chemical with a complicated name is inherently dangerous, but every substance deserves respect. Indoline-2-carboxylic acid exists as a white or off-white solid. Currently, there isn’t much mainstream toxicological data from major health agencies like the CDC or WHO that details specific effects on human health. Published material safety data sheets suggest that this compound can irritate skin, eyes, or respiratory tract. Swallowing or inhaling dust could trigger similar irritation. These symptoms echo what’s seen with many simple organic chemicals.
Working with bulk materials in closed spaces can increase risks through airborne dust. Using proper gloves, goggles, and dust masks makes a real difference. Out of habit, most chemists quickly learn to avoid careless contact or inhalation of powdery chemicals—not because every substance is deadly, but because consistent caution pays off over a career.
Chronic Exposure and Carcinogenicity
Chemicals move from curiosity to concern when chronic exposure leads to long-term health problems. Cancer scares tend to lead the headlines, usually with industrial solvents or pesticides. There’s no strong evidence or classification from institutions like IARC that puts indoline-2-carboxylic acid on a cancer risk list. This lack of data can mean two things: either there hasn’t been enough study, or there’s not much cause for alarm. In cases like this, treating the compound with respect makes sense. Until science learns more, nobody should treat it casually.
Environmental Impact
After personal safety, chemists pay attention to waste and environmental effects. Some compounds persist, degrade slowly, or harm aquatic life. Indoline-2-carboxylic acid doesn’t feature in environmental watchlists or hazardous waste registers—but that’s not a green light for careless disposal. Pouring chemicals down the drain, especially if working in volume, can have ripple effects. Following local regulations around chemical disposal protects water systems and wildlife, which doesn't just help the planet but avoids the headaches of regulatory fines or contamination claims.
Better Practices, Safer Outcomes
Knowledge changes the conversation. While there’s no evidence that indoline-2-carboxylic acid ranks high on toxicity charts, routine chemicals can still bring trouble if someone lets their guard down. The best approach is to keep using standard protective gear, ventilate workspaces, label containers clearly, and keep powders off skin or out of lungs. Treating waste responsibly moves safety from theory to practice.
Chemistry is never about shortcuts—it’s about building habits and getting details right, every time. Respecting risks, even when they seem minor, keeps work safe and reputations solid. That’s what turns a list of warnings into real protection, both for people and the places where they work and live.
What are the common applications of Indoline-2-Carboxylic Acid in research or industry?
Tapping into Molecular Diversity
At the bench in a research lab, a chemist often craves molecular building blocks that unlock new structures. Indoline-2-carboxylic acid is one of those unsung heroes of synthetic chemistry. Its skeleton, made up of a bicyclic structure fused to a carboxylic acid, feeds innovation. It isn’t a shelf product you see in drugstore aisles, yet its impact touches pharmaceuticals, agrochemicals, and advanced materials.
Pharmaceutical Research: Laying Foundations for Medicine
Drug discovery sometimes feels like running through an endless maze, each turn a potential dead end. What keeps the process moving are compounds like indoline-2-carboxylic acid. Medicinal chemists use it to build up molecules that target disease. Its backbone appears in research aimed at developing treatments for nervous system disorders and cancer. I’ve seen colleagues in the lab crafting new analogs using indoline-2-carboxylic acid as a starting point because its unique shape creates possibilities for locking into biological targets. It acts as a scaffold, offering a solid anchor for designing ligands and enzyme inhibitors. Across studies, researchers explore its role in structures meant to inhibit kinases or proteases, two protein types central to several health conditions.
Peptide Synthesis: Building Chains That Matter
Peptides—small chains of amino acids—bring hope for new therapies. Indoline-2-carboxylic acid lets chemists insert rigidity into these chains. This matters, since flexible chains can flop around and fail to interact as needed. I’ve worked with teams incorporating this acid to make cyclic or constrained peptides, forcing their three-dimensional shape into just the right fold. Following this path, they boost the chance these peptides resist enzymatic breakdown and survive in the body long enough to do their work. This application is catching attention among those designing drugs for metabolic and infectious diseases.
Material Science and Sensors
Polymer scientists see promise in every new building block. With indoline-2-carboxylic acid, they tap into a structure that can enhance polymer backbones or embed new functional groups. This tweaks properties like solubility, charge, or response to light. Some teams drive this research towards organic semiconductors or advanced coatings. Thin films that sense chemical changes or store energy often rely on such carefully chosen ingredients. Several studies highlight the value of indoline derivatives in organic electronics and fluorescent sensors, where their unique electronic features come into play.
Fine Chemicals and Specialty Synthesis
Scale-up chemists in industry watch costs and complexity but also chase novelty. Indoline-2-carboxylic acid finds its way into processes that generate dyes, pigments, and optical brighteners. I’ve heard chatter from fine chemical producers about using it to develop products for textile and imaging markets. Its reactive sites allow for the attachment of various substituents, opening the door for colorful, light-responsive materials.
Taking the Next Steps
The big challenge comes down to sourcing: maintaining purity, controlling costs, and optimizing yields. This isn’t just a matter of finding a supplier but managing waste and solvent use in the lab or plant. Researchers constantly adjust protocols, searching for greener routes. Teams working in universities collaborate with specialty chemical companies, aiming for smarter, safer, and more sustainable synthesis. Open dialogue between these worlds helps push boundaries and promotes responsible use of powerful molecules like indoline-2-carboxylic acid.
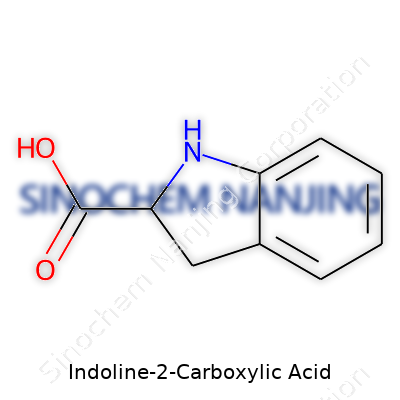
| Names | |
| Preferred IUPAC name | 2,3-Dihydro-1H-indole-2-carboxylic acid |
| Other names |
2-Indolinecarboxylic acid 2-Carboxyindoline 1,2-Dihydroindole-2-carboxylic acid Indolin-2-ylcarboxylic acid Indolin-2-carboxylic acid 2-Carboxy-2,3-dihydro-1H-indole |
| Pronunciation | /ˌɪn.dəˈliːn tuː kɑːrˈbɒk.sɪl.ɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 451-77-4 |
| Beilstein Reference | 136512 |
| ChEBI | CHEBI:35497 |
| ChEMBL | CHEMBL507400 |
| ChemSpider | 20539 |
| DrugBank | DB08375 |
| ECHA InfoCard | 03b9ec94-f616-44fc-b6b2-1b5b6a9de020 |
| EC Number | 872-43-3 |
| Gmelin Reference | 82256 |
| KEGG | C07532 |
| MeSH | D000972 |
| PubChem CID | 73576 |
| RTECS number | NL2975000 |
| UNII | 0A6T0X44OE |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DHBB3TJ7E7 |
| Properties | |
| Chemical formula | C9H9NO2 |
| Molar mass | 165.18 g/mol |
| Appearance | White to off-white powder |
| Odor | Odorless |
| Density | 1.282 g/cm³ |
| Solubility in water | Slightly soluble in water |
| log P | 0.6 |
| Vapor pressure | 0.0000137 mmHg at 25°C |
| Acidity (pKa) | 5.0 |
| Basicity (pKb) | 5.02 |
| Magnetic susceptibility (χ) | -47.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.622 |
| Viscosity | 0.894 cP (25°C, water) |
| Dipole moment | 3.28 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 221.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -292.6 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -5198.7 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin irritation, causes serious eye irritation. |
| GHS labelling | GHS07, Warning, H315, H319, H335 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| Precautionary statements | Precautionary statements: P261, P264, P271, P280, P302+P352, P305+P351+P338, P312, P332+P313, P337+P313, P362+P364 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | 110.6 °C |
| Autoignition temperature | The autoignition temperature of Indoline-2-Carboxylic Acid is 410°C. |
| LD50 (median dose) | LD50 (median dose): >5000 mg/kg (Rat, oral) |
| NIOSH | SE1750000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 50 mg |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Indoline Indole-2-carboxylic acid Indoline-2-carboxamide Indoline-2-carboxylate Indoline-2-sulfonic acid |

