Rethinking Indole-3-Ethanol: From Historical Curiosity to Modern Utility
Unpacking the Past: The Journey of Indole-3-Ethanol
Decades of research in organic chemistry bring certain compounds from obscurity into sharper focus. Indole-3-ethanol fits that bill, with roots tracing back to curiosity-driven studies on plant metabolites during the early twentieth century. Researchers found this molecule hiding in the biosynthetic pathways of indole alkaloids, woven into the complex tapestry of plant growth and human health. While less celebrated than its cousin indole-3-acetic acid, indole-3-ethanol seeped quietly into fields as diverse as regulatory biology, plant signaling, and even aroma chemistry. Echoes of early isolation experiments still linger in the protocols today, a testament to the dogged work of scientists who mapped out every reaction step long before modern analytical tools landed on benchtops. The historical arc of indole-3-ethanol reveals more than a simple chemical transformation; it tells a story about how close observation and persistence can elevate a single compound into broader relevance.
Physical and Chemical Reality: Understanding the Material
Face-to-face with a vial of pure indole-3-ethanol, one recognizes that chemistry isn’t just theory. With its faint floral aroma, the white-to-off-white powder suggests something soft, but the reality packs more punch. C10H11NO stands for a structure featuring a condensed indole ring and a two-carbon ethyl side-chain crowned with a hydroxyl group. The molecular weight sits near 161.2 g/mol, and a melting range hovering between 70 to 74°C puts it within reach of many practical applications. Solubility stretches comfortably into most organic solvents, from ethanol to DMSO, which means handling and blending seldom hold up a well-organized lab. Stability under standard storage keeps costs down and waste low, an underrated advantage in scaled processing. Indole-3-ethanol does not ask for exotic atmospheres or convoluted handling, a reminder that some compounds deliver their best without fuss.
Guiding Hands: Synthesis, Modification, and Labeling
Organic labs see indole-3-ethanol surface as a frequent guest. Most routes stem from indole or tryptophan, depending on resource availability and downstream goals. Classic Friedel–Crafts alkylation or hydrogenation techniques remain popular for their accessibility and yield reliability. Some chemists favor the reduction of indole-3-acetaldehyde, exploiting mild conditions and minimal byproducts. On the bench, purity and batch consistency matter because downstream reactions can magnify even faint impurities. Technically speaking, labeling should never stray from IUPAC conventions—3-(2-Hydroxyethyl)indole and Indole-3-ethanol stay clear, facilitating clear reporting and supply chain traceability. Synonyms often enter the conversation: β-(3-Indolyl)ethanol, 3-Indolylethanol, and IET call attention to the web of nomenclature spun by different research communities and regulatory frameworks over the decades. Respect for these names and the historical decisions behind them helps avoid confusion and keeps records straight as compounds travel between research groups.
Safety and Standards: Navigating Regulatory Terrain
Safety concerns seldom take a backseat in modern chemistry. Indole-3-ethanol stands on the lower-toxicity end compared to its aromatic siblings, but standard operating procedures deserve vigilance. Skin and eye protection make sense on the bench since even mild irritants can cloud a day’s work. Waste management grows more complicated as environmental regulations tighten, so supervision over solvent and byproduct streams becomes everyday business. Many labs rely on regularly updated safety data sheets from trusted suppliers, but hands-on experience still matters most when troubleshooting accidents. Regulatory standards shaping pharmaceutical or food research tightly control allowable impurities and trace metals, with annual audits ensuring that nothing slips through the cracks. Although not flagged by global agencies as a top health concern, indole-3-ethanol calls for measured handling and attention to evolving guidance, especially in consumer-facing applications. Good manufacturing practice—once regarded as a bureaucratic hurdle—now opens the door to broader research and commercial partnerships.
Application Arena: From Laboratory Curiosity to Broader Horizons
Indole-3-ethanol is not just a laboratory talking point. Its real value comes through daily applications. In plant science, it attracts attention as a biomarker for auxin-related processes, shedding light on how roots respond to stress or signaling molecules. Aroma chemists pluck it from natural extracts, considering its subtle floral notes as key building blocks in fragrances and flavor enhancers. In biomedical fields, metabolic pathways involving indole-3-ethanol open up questions about neuroprotection, hormone modulation, and gut-brain signaling. Its ability to act as a precursor or intermediate keeps it relevant in synthetic schemes for bioactive compounds. Researchers use it to probe enzyme activities or disrupt normal pathways, hoping to unlock new modes of action around inflammation, cell growth, or defense responses. Whenever a project asks for a flexible indolic intermediate with low background activity and straightforward handling, indole-3-ethanol lands on the short list—a statement that grows even truer as interdisciplinary research blurs old boundaries between chemistry and biology.
Deepening the Science: Research, Development, and Toxicity Insights
A close look at published data makes it clear: Indole-3-ethanol inspires curiosity from basic form to advanced function. Decades of biosynthetic mapping verified its formation from tryptophan in plants, linking it to growth regulation and pathogen defense. Analytical labs around the globe use it as a reference compound for tracking natural product transformations, while synthetic teams leverage it as a scaffold for more ambitious molecular modifications. On the toxicity front, acute exposure studies suggest a low hazard profile, at least in low concentrations and under standard lab exposures. Chronic effects haven’t flagged major warning signs, but the modern push to move past "absence of evidence" means longer-term studies with animal models and cellular assays remain vital. Researchers seek to clarify whether subtle effects on hormone signaling or neurotransmission could appear after repeated or high-level exposure, especially with demand rising for biotechnological applications. Every data point now adds value because regulatory frameworks move faster than before.
Paving the Way Forward: Future Prospects
Looking to tomorrow, indole-3-ethanol won’t fade quietly into chemical history. Agricultural tech companies eye its role in developing new biostimulants and plant growth regulators tailored for climate resilience. Pharmaceutical groups explore its structural motifs to inform the next round of neuroactive drug candidates or metabolic enzyme inhibitors. Green chemistry advocates appreciate its bio-based production potential, with fermentation techniques promising to trim energy use and lower environmental footprints. Forward-thinking labs devote resources to high-throughput screening, betting on indole-3-ethanol as a bridge toward complex alkaloids with therapeutic promise. In flavor and fragrance sectors, the push to identify "natural" aroma ingredients only fuels more attention. Whether in the hands of a solo researcher searching for regulatory clues or a large team mapping biosynthetic pathways, indole-3-ethanol proves its worth through versatility, reliability, and the simple fact that science always benefits from a few quiet workhorses just waiting to take center stage.
What is Indole-3-Ethanol used for?
Digging into the Role of Indole-3-Ethanol
Indole-3-ethanol pulls a lot of attention from researchers and the agricultural world, but its use doesn't end on the lab bench. As a plant growth regulator, this compound finds its way into discussions about sustainability, crop yields, and plant biotechnology. Anyone who spends time around greenhouses or crop research programs knows these regulators can change the game for growers looking to boost both quantity and quality.
What Makes Indole-3-Ethanol Useful?
Indole-3-ethanol comes from the indole family, which means it shares its backbone with a number of other plant hormones and signaling compounds. One key trait: it often serves as a precursor for indole-3-acetic acid, a well-known natural auxin. Auxins control how plants grow, adapt, and respond to their surroundings. Using this compound, researchers run trials on everything from root architecture to fruit ripening. It’s not just a molecule; it’s a practical tool for anyone working to refine how plants develop and how agricultural outputs adapt to changing climates.
Outside the Field: Broader Biological Research
Beyond agriculture, indole-3-ethanol appears in studies focused on cellular communication and stress responses. Scientists have found its metabolic pathways useful for tracing how plants detoxify, defend themselves, or interact with microbes. Anyone who spent time in a plant physiology lab might have come across this compound during projects targeting disease resistance or bioengineering crops. Universities point to it as a building block for making new plant varieties better suited to fluctuating weather or pest pressures.
Supporting Healthier Crop Cycles
Food security depends on healthy plants, and the science behind growth regulators impacts more than yield per acre. With indole-3-ethanol, growers sometimes see improved rooting, more even fruit set, or stretches of reliable flowering. Each of these outcomes means more predictable harvests and steadier income for farmers, especially in regions facing weather extremes or shifting seasons. The compound can play into integrated management systems where plants get precise nudges at key development stages, opening doors to reduced chemical fertilizer use and smarter irrigation patterns.
Room for Improvement and Looking Ahead
Indole-3-ethanol holds obvious promise, but scaling its use responsibly takes work. Not every crop responds the same way, and a solution in one climate may cause issues in another. More research on its interactions with native soils and local pests could yield safer recommendations for growers. Collaborations between university labs and the ag industry keep advancing our grasp of these regulators. Public funding for these studies, plus openness around data, could drive better guidelines and weed out trial-by-error guesswork.
Conclusion Is Not Needed
Indole-3-ethanol isn’t just another chemical on a shelf. Its ability to influence plant growth sets the stage for new strategies in agriculture and environmental science. Progress here promises benefits not only for scientists but also for farmers and consumers everywhere.
Is Indole-3-Ethanol safe for human consumption?
What Science Knows About Indole-3-Ethanol
Indole-3-ethanol comes from the breakdown of tryptophan, which is a natural amino acid found in everyday foods like chicken, eggs, and cheese. Scientists have found this compound in small quantities in some fruits and vegetables. The basic building blocks of this chemical seem safe enough – after all, nobody worries about eating broccoli or bananas. The real question pops up when we look at indole-3-ethanol by itself, used as a supplement or additive, beyond the trace amounts in food.
Human Research Isn’t There Yet
Plenty of compounds made by our bodies or found in our foods can behave differently in concentrated or purified forms. I’ve seen a lot of research on tryptophan and its relatives, but not much on putting indole-3-ethanol in pills or powders for people to eat. The FDA hasn’t approved this molecule for dietary supplement use, and there aren’t big clinical trials showing what happens when someone actually consumes larger doses regularly. Without controlled studies, the long-term effects remain a big question mark.
Safety Concerns Start With the Basics
It sounds responsible to ask, “Is it safe?” before swallowing any new compound, even one that occurs naturally. Our bodies often process and clear out small quantities of chemicals from plants and protein-rich foods, but bigger doses can overwhelm those systems. Indole compounds sometimes react with liver enzymes, changing the way they’re handled inside the body. With other amino acid breakdown products, high amounts can trigger problems such as headaches, stomach upset, or worse. No reason exists to treat indole-3-ethanol as an exception until the data says otherwise.
Why Transparency Matters
Safety isn’t only about whether someone ends up in the hospital. Allergies, interactions with prescription drugs, and hidden contaminants all come into play, especially with unregulated supplements sold online. If a label lists indole-3-ethanol, there needs to be clarity about the actual amount and its source. Healthcare professionals rely on documented cases and scientific studies; right now, those are missing for this compound. It makes sense for researchers and companies to take a cautious approach and put transparency first, before marketing claims step ahead of the evidence.
What Smart Consumers Can Do Now
Nobody wants to end up as a guinea pig. Reading ingredient labels, double-checking health claims, and asking a doctor about risks pays off. Nutrition experts always stress that most people get enough bioactive compounds from a balanced diet full of fruits, vegetables, and healthy proteins. If indole-3-ethanol proves useful in the future – say, for gut health or cell signaling – any benefits would need clear proof. Until then, skepticism stays healthy, and the safe bet sounds a lot like “stick with foods your body already knows.”
The Way Forward
Science builds trust by showing its work, running experiments, and sharing real outcomes. Indole-3-ethanol still sits in the maybe column when it comes to supplements. As researchers dig up more facts, the rest of us do well to keep an eye on updates and hold brands and policymakers to the highest standards for safety and proof. Until we know more, it’s wise to put curiosity ahead of hype and keep healthy habits grounded in what’s already known.
What is the recommended dosage of Indole-3-Ethanol?
Understanding the Compound Before Using It
Indole-3-ethanol isn’t something you find on a grocery shelf. It pops up in niche plant research, some fermentation work, and, rarely, nutritional supplements or fine chemicals. Most people hear about it because of plant experiments aiming to understand how compounds affect growth. There’s so much information flying around about plant supplements and potential health products. That makes it easy for folks to think natural always means harmless.
Sticking With Facts, Not Guesswork
Digging into scientific databases like PubMed, I find next to nothing on recommended dosages for humans. Clinical trials don’t focus on this compound. Most research ties it to plant pathways or microorganism metabolism. Plant physiologists track trace amounts, often in micromolar concentrations, when running their greenhouse trials. Sometimes, rodent studies tinker with milligram doses, but translating those numbers to people skips over a truckload of safety concerns.
Chasing an exact figure for dosage becomes a shot in the dark without robust human data. There’s a risk in assuming anything used in a test tube or a plant pot behaves the same way once it meets a human digestive system. Our bodies break down chemicals in ways labs can’t always predict. I learned that lesson working in clinical research—human trials uncover risks no petri dish or greenhouse ever hints at.
No FDA Guidance Means Caution
The U.S. Food and Drug Administration doesn’t list Indole-3-ethanol as an evaluated food additive or supplement. No government health body sets a daily limit. Supplements on the market seldom use this specific compound. Instead, most health claims float around relatives like indole-3-carbinol or diindolylmethane, usually found in cruciferous vegetables.
When a compound lacks regulatory oversight, the gap leaves users unprotected. A safe amount for one person may cause problems for another, particularly if a product interacts with prescription drugs or if someone has liver problems. I’ve seen supplement claims outpace clinical data many times. Nearly every time, the best bet was relying on established safety records from well-run trials or, even better, sticking to real food sources when hunting for trace bioactives.
Safety Over Hype
Some companies or online forums throw out numbers—milligrams per kilogram, vague microgram ranges—but nobody should treat anonymous advice as gospel. Real risks sneak in when self-dosing on chemical isolates. As much as people chase novelty in health, skipping the doctor or a registered dietitian can backfire fast. One study in drug safety published by the NIH found dozens of adverse effects from compounds labeled “natural” but poorly tested.
Better Approaches and Potential Solutions
Instead of taking a gamble, consumers can press supplement producers and health stores for detailed documentation. Certified labs that follow Good Manufacturing Practices may offer certificates of analysis. I’ve seen consumers get peace of mind when third-party verification steps in. Until clinical evidence gives clear answers, nobody should use Indole-3-ethanol as a regular supplement without professional consultation.
As interest grows in natural compounds, researchers and regulators can lean into stronger, publicly accessible safety registries. Crowdsourced adverse-event databases and transparent reporting move the industry away from the Wild West and toward evidence-based guidance. I have learned from the “next big thing” headlines—the best health decisions come from patience, wise skepticism, and solid science, not rushed hype.
Are there any side effects of using Indole-3-Ethanol?
Understanding Indole-3-Ethanol
Indole-3-ethanol pops up sometimes in conversations about supplements, plant biology, and niche chemical research. You see this compound more often in labs than on store shelves. It has ties to natural plant hormones and attracts attention from some scientists thinking about new ways to tackle health issues or enhance plant growth. Whenever people start talking about using a compound like this for human benefit, one of the first common-sense questions always comes down to safety—and possible side effects.
Possible Health Risks
There’s not a lot of clinical research on Indole-3-ethanol’s impact on humans. No official safety profile exists, and you won’t find it listed as a supplement on most pharmacy shelves. Because of that, researchers and healthcare professionals feel cautious about its use. Indole-related compounds don’t get the same level of regulation or scrutiny as approved medicines. Nobody wants to be the test case for an unproven chemical, especially when so many side effects could go unreported or even unnoticed.
A compound in the same family, indole-3-carbinol, sometimes pops up in health circles as part of cruciferous vegetables and has some studies examining safety. But indole-3-ethanol itself doesn’t fit exactly in this category. Combining a lack of published data with some anecdotal speculation just isn’t enough. In science and medicine, an absence of evidence doesn’t mean safety. It means we don’t know the risks—a fact that should keep most people wary.
Learning From Similar Substances
Many chemicals that affect plant growth, like auxins derived from indole, can interfere with hormone systems. Even low-level exposure to certain derivatives might cause stomach upset, dizziness, allergic reactions, or more serious effects, depending on dosage and delivery. Since indole-3-ethanol is closely related, experts pay close attention to these possible problems. Animal studies sometimes highlight changes in metabolism, liver function, and skin irritation, especially with repeated exposure or in larger quantities.
Some research hints at oxidative stress or disruption of normal enzyme activity. That means that cells could end up functioning less efficiently and organs take on extra burden. It’s rare to feel the impact from one-off exposure, but repeated or unchecked use heightens risk. The body works best when it only breaks down familiar, well-studied compounds. Anything outside this group brings the potential for negative or unintended effects.
Practical Advice and Moving Forward
If someone finds themselves tempted to try something like Indole-3-ethanol, it makes more sense to pause and talk to a medical professional first. Health decisions should always start with reliable evidence, not just curiosity or word of mouth. Lack of regulation and missing long-term studies means your best safeguard is skepticism and research. Most reputable doctors and scientists advise against experimenting with unproven substances, especially those with little hard data on safety.
People who work in labs or industries handling chemicals like Indole-3-ethanol already know the value of protective gloves, ventilation, and proper disposal. Safety data sheets recommend staying strict with safety gear and handling processes, since even a little skin exposure might cause rashes or irritation. Documented side effects often come from these workplace exposures rather than clinical use, but they show the importance of caution all the same.
At the end of the day, trusting tried-and-true pathways in health makes a lot more sense than gambling with an untested compound. Science makes progress through careful trials and observation, not shortcuts. As far as Indole-3-ethanol goes, the door stays open for more research, but nobody wins by skipping the careful steps that protect people’s health.
How should Indole-3-Ethanol be stored?
The Realities of Chemical Storage
People rarely talk about chemical storage until something goes wrong. Some assume a shelf in a cool room will do. I’ve worked in labs where a lack of attention turned useful chemicals into useless or even dangerous waste. Nowhere does this ring truer than with organic compounds like Indole-3-Ethanol (I3E). A handful of careless choices can spoil a sample, which costs money and wastes time.
What Makes Indole-3-Ethanol Unique?
I3E holds value in research and agriculture. Its structure features both an indole ring and an alcohol group, which gives it sensitivity to air, light, and moisture. From experience, even tiny traces of water or UV light can cause degradation, changing color and possibly triggering side reactions that make the compound nearly useless for experiments.
Learning From Mistakes: Common Problems
Years ago, I found myself working with a batch that went yellow from improper storage. This happened because the sample sat in a clear vial exposed to lab lights and humidity. The entire week was wasted, and the lesson stuck with me: chemistry is unforgiving to shortcuts. Stories like mine float around the research world, reminding us why extra steps matter.
Rules That Work in the Real World
Use Airtight Containers: Glass vials work best, provided they seal tightly. Avoid plastics that breathe or let through air and vapors. Laboratory-grade glass, with a PTFE-lined cap, stands up to most solvents and blocks unwanted air and moisture.
Fight Moisture: Water in the air works its way into containers. I recommend storing vials with small desiccant packs in the same cabinet. If possible, purge with an inert gas (like dry nitrogen or argon) before closing up, especially if you’re not sure how often the cabinet gets opened.
Keep It in the Dark: Regular lighting degrades I3E over time. Amber or brown glass bottles prevent UV rays from sneaking in. In my experience, an opaque box tucked at the back of a closed cabinet provides extra insurance. Never leave a vial out under bright lights “just for a minute.” Those minutes add up quickly.
Temperature Matters: I3E responds best to cool storage. Not every lab has a dedicated cold room, but standard refrigerators work well. Keep samples in a box or bag to protect from accidental spills or that annoying frost buildup. Always label with both the chemical name and date stored—future-you will appreciate it.
Separate From Reactive Chemicals: Never store I3E near acids, bases, or oxidizers. Even the vapors can cause silent reactions that you won’t spot until it’s too late. Organize cabinets by chemical type to reduce risk. Clear labeling and an easy-to-read inventory help keep mistakes rare.
Backing Up Advice With Research
Studies show indole derivatives break down faster in light, warmth, and moisture. Major suppliers like Sigma-Aldrich and TCI guide users to keep I3E dry, dark, and below room temperature. These aren’t just suggestions made up for paperwork; you see the results in longer shelf life and consistent experimental results.
Better Habits, Stronger Results
Some see proper storage as overkill, but the reality is clear—lab time and research budgets don’t stretch to cover shortcuts. Everything from keeping the lights off storage vials to purging air before sealing adds up to purity and reliability. Investing two minutes to store things right pays dividends in the long run. No piece of lab equipment or clever shortcut will ever make up for careful, practical storage methods.
| Names | |
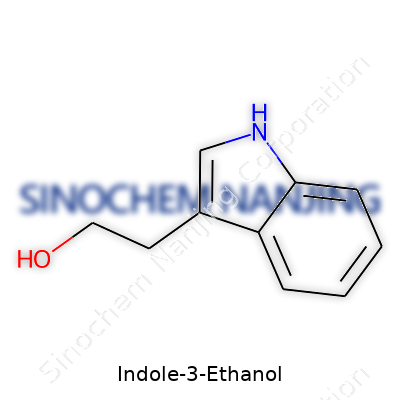
| Preferred IUPAC name | 2-(1H-indol-3-yl)ethan-1-ol |
| Other names |
Tryptophol 3-Indoleethanol 3-(2-Hydroxyethyl)indole |
| Pronunciation | /ˈɪn.doʊl θri ˈɛθ.ə.nɒl/ |
| Identifiers | |
| CAS Number | 487-89-8 |
| Beilstein Reference | 1208572 |
| ChEBI | CHEBI:28605 |
| ChEMBL | CHEMBL47405 |
| ChemSpider | 72095 |
| DrugBank | DB04296 |
| ECHA InfoCard | 100.037.514 |
| EC Number | 1.1.1.1 |
| Gmelin Reference | 72238 |
| KEGG | C01743 |
| MeSH | D007248 |
| PubChem CID | 7019 |
| RTECS number | NL0490000 |
| UNII | 1M8L715F4B |
| UN number | Not dangerous |
| CompTox Dashboard (EPA) | DTXSID7051474 |
| Properties | |
| Chemical formula | C10H11NO |
| Molar mass | 161.20 g/mol |
| Appearance | White to almost white crystalline powder |
| Odor | slight, floral |
| Density | 1.22 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 1.49 |
| Vapor pressure | 4.4 x 10^-4 mm Hg (@ 25 °C) |
| Acidity (pKa) | 15.4 |
| Basicity (pKb) | 7.89 |
| Magnetic susceptibility (χ) | -60.0e-6 cm³/mol |
| Refractive index (nD) | 1.639 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.33 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 357.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -196.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3852 kJ/mol |
| Pharmacology | |
| ATC code | N06AX13 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes serious eye irritation |
| GHS labelling | GHS07 Warning |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | Causes serious eye irritation. |
| Precautionary statements | P264, P270, P273, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | 110°C |
| Autoignition temperature | 455 °C |
| Lethal dose or concentration | Lethal dose or concentration (LD50, Oral, Mouse): 750 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral >5000 mg/kg |
| NIOSH | Not established |
| PEL (Permissible) | Not established |
| REL (Recommended) | 50 mg |
| Related compounds | |
| Related compounds |
Tryptophol Indole-3-acetic acid Indole-3-acetaldehyde Indole-3-carbinol Indole-3-butyric acid |

