Indium Nitrate: A Closer Look From Laboratory to Industry
Historical Development
Indium nitrate stands as a result of both curiosity and persistence in the world of inorganic chemistry. Back in the early 20th century, folks worked their way up from isolating indium to finding unique combinations, including its nitrate. Mining operations in Germany unearthed indium around 1863, tucked away as a trace element in zinc ores. Researchers, determined to get more use out of this odd silver-white metal, mixed it with nitric acid. This simple reaction opened a whole new chapter for indium chemistry. Over decades, laboratories in Europe and North America gave the compound a proper shakedown, pushing it into the spotlight for scientists looking for rare metal nitrates with interesting traits. By the time electronics started their rapid growth in the second half of the 20th century, indium nitrate became a reliable tool in research kits, feeding experiments on thin film technology and high purity gas applications.
Product Overview
Indium nitrate, often delivered as a white to pale yellow crystalline solid or offered up dissolved in water, comes mainly from companies packaging chemicals for academic or industrial use. Its formula, In(NO3)3·xH2O, shows off how indium hooks up with three nitrate groups and, depending on conditions, drags along some water molecules. Mainline producers label it under trade names like "Indium(III) Nitrate Hydrate" or, more simply, "indium nitrate." Buyers rarely need much—grams to small kilograms—mostly since indium's cost and the niche uses keep volumes low. Mistakes in purity, heavy metal contamination, or even errors in hydration level, all show up fast if you’re working in fields like microelectronics or advanced ceramics, where quality can’t dip.
Physical & Chemical Properties
Indium nitrate tops the list for hygroscopic salts that catch moisture from the air almost without fail, turning clumpy when left in the open. It’s easily soluble in water, forming clear or slightly turbid solutions at room temperature. Chemists know it starts to break down at moderate heating, gradually giving off brown nitrogen oxides and leaving indium oxide as a stable residue. Under standard lab lights, it stays stable, but stack it too close to acids or bases and things can get messy fast. Unlike more famous nitrates like silver’s or potassium’s, indium nitrate’s color and low melting points make it pretty distinctive on the bench.
Technical Specifications & Labeling
Suppliers ship indium nitrate with purity levels ranging from 99.99% upward, generally reporting impurities below 10 parts per million. Labels never skip hazard warnings since the material irritates skin and eyes, and solutions can make metal surfaces pit or tarnish. Some certificates of analysis stretch for pages, especially for semiconductor-grade stocks, listing sodium, iron, copper, and even sub-ppm boron contamination. Storage instructions always get a bold font: cool, dry, tightly sealed jars—no exceptions. Those who skip careful labeling risk regulatory headaches, especially in North America and Europe where trade and transport laws for nitrates require strong compliance.
Preparation Method
Lab syntheses usually start by dissolving pure indium metal or, less often, indium oxide in nitric acid under gentle heat. The metal’s chunk by chunk reaction with the acid pumps out hydrogen gas. Patience goes a long way here—rush it and you lose product to side reactions or get incomplete conversion. After full reaction, evaporation under reduced pressure or in open dishes leads to crystallization. Sometimes, extra ethanol helps coax the product out of solution. In bulk production, process control means constant monitoring of temperature, ventilation, and acid concentration to catch any runaway reactions. Getting rid of excess acid before storage matters—leftover fumes create storage hazards, and any misstep ends up hurting both yield and purity.
Chemical Reactions & Modifications
Indium nitrate carries a knack for dropping indium oxide during careful heating, giving users a handy route to pure indium oxide thin films. Add an alkali and you get indium hydroxide, which then dries back into the oxide. Its reactions with ammonium salts, rare earth elements, and other transition metals open access to mixed-metal oxides and catalysts, key items for anyone tinkering with ceramic superconductors or transparent electrodes. In organic settings, indium nitrate catalyzes some hydrogenation and oxidation reactions, and chemists rarely run indium-based organic syntheses without at least glancing at its nitrate cousin. The material doesn’t play well with strong reducing agents or concentrated alkali, as both can unleash ammonia or explosive nitrogen oxides fast enough to ruin a day’s work.
Synonyms & Product Names
Most catalogs simply list it as “Indium(III) Nitrate” or “Indium Nitrate Hydrate.” Anyone chasing older research might find “nitric acid, indium (3+) salt,” “trinitrate d’indium,” or just “In(NO3)3.” Still, the CAS number 13465-09-3 cuts through confusion when ordering or looking up data. Watch out for mistakes—some listings jumble it with other indium salts, but the difference in hydration or counterion can sometimes make trouble for inexperienced buyers.
Safety & Operational Standards
Most labs treat indium nitrate with tough respect. Its dust stings the eyes and skin, even at low exposure, and nobody enjoys breathing in stray dust or vapors produced during heating. Standard PPE means gloves, goggles, and—if weighing powders—masks or full-face shields. Local exhaust or fume hoods turn on for even tiny prep batches. Storage under lock and key, away from organic matter or reducing agents, cuts down risk of fire or accidental reactions. Bigger outfits write safety protocols with oversight from both chemical hygiene officers and health advisors, since nitrate compounds get flagged for regulatory attention. In spills, workers rush to dilute with lots of water before mopping up, because indium nitrate’s solutions corrode pretty much anything with a hint of metal. Most training sessions tell the same story: treat it with the same care as other exotic metal nitrates, always following up with routine air and surface monitoring in busy lab spaces.
Application Area
Indium nitrate played a mostly hidden part in electronics until the push for flat-panel displays drew serious investment. Now, it’s best known as a precursor for indium tin oxide—a transparent conductor in touchscreens, LCD panels, and solar cells. Artists, jewelers, and glass manufacturers use indium nitrate for colored glass and specialty coatings, where it beats out some lead compounds for environmental safety. In high-tech industries, thin film deposition from indium nitrate solutions leads to smooth, well-bonded coatings. Catalysis experts study its actions in hydrogenation reactions, often looking for activity in organic synthesis or pollution reduction. Medical researchers have tinkered with indium nitrate, trying to label antibodies or deliver radioisotopes, yet safety and availability tend to keep most projects on the cautious side for now.
Research & Development
Today, university and industrial labs chase better methods for making indium oxide films or nanoparticles, betting that cheaper processing or new chemical blends might trim the cost of next-generation displays and efficient photovoltaics. Nanotechnology teams spend hours finding tricks to control indium nitrate’s hydrolysis and particle size, improving transparency and conductivity at the same time. In the world of green chemistry, some groups test alternative solvents or milder acids to lower the waste made by classic indium nitrate manufacturing, hoping to cut out polluting nitrogen oxides. Most researchers agree that while indium is rare, process recycling and re-use can stretch supplies much further, yet industry standards do not always catch up to best academic practices.
Toxicity Research
Indium compounds do not get the same notoriety as lead, cadmium, or mercury, but several studies show long-term exposure may affect lung, kidney, and liver health. Technical staff and researchers remain wary about direct skin or inhalation exposures, especially since occupational standards in the United States and Europe have started to tighten in response to new data. Animal studies point to possible chronic toxicity after repeated dosing, though clear links in humans stay under debate. Waste disposal remains a hot topic—companies face fines or site audits if they dump nitrate solutions down ordinary drains, so most collect wastes for off-site treatment or metal recovery. In practice, the small amounts handled limit environmental releases, but an eye toward cumulative risk keeps indium nitrate on the chemical safety watch-list.
Future Prospects
Indium nitrate’s future rides on both the continued push for better electronics and the drive to make renewable energy more affordable. As demand for touchscreen and solar cell materials grows, researchers keep searching for ways to stretch the indium supply through recycling, better deposition methods, or even partial substitution with less expensive metals. Some hope lies in using indium nitrate to produce oxide films at lower temperatures, which could cut down manufacturing costs in energy-hungry industries. Next steps probably mean tighter controls on purity, more transparent supply chains, and stricter environmental practices—none of which come easy or cheap. Yet, for now, indium nitrate remains a keystone for both serious research and emerging technology, with every advance in processing likely to ripple across multiple industries.
What is Indium Nitrate used for?
The Role of Indium Nitrate in Technology and Science
Indium nitrate might sound unfamiliar to most people, but its applications touch parts of everyday life and advanced research. My experience working in academic labs has put me face to face with some of the lesser-known chemicals that keep progress moving. Indium nitrate’s story isn’t as headline-grabbing as lithium or silicon, but its importance keeps growing.
Indium nitrate stands out in the world of specialty chemicals because of its connection with the electronics sector. The demand for smarter, more efficient tech keeps going up, and materials like indium nitrate quietly shape that journey. One major use appears in the production of indium tin oxide (ITO), which forms transparent electrodes for touchscreens, flat-panel displays, and solar panels. The chemical acts as a useful precursor. Its ability to dissolve easily in water and break down at relatively low temperatures makes processing a lot less complicated for manufacturers.
Indium Nitrate in Research and Development
Working in a research environment, I noticed that chemicals like indium nitrate open doors for creating new materials and testing out cleaner methods for producing products. Academic institutions use indium nitrate to synthesize high-quality indium oxide nanoparticles. These particles can serve a variety of purposes, from thin-film transistors to experimental sensors. The production process tends to be straightforward and gives researchers a way to tune particle sizes or structures by tweaking simple factors like temperature or solution composition.
Solar energy researchers take a closer look at indium nitrate for its role in developing more efficient energy conversion devices. Critical breakthroughs in photovoltaic cells rely on substances that can absorb or transmit light in exactly the right ways. Indium nitrate helps build the kind of transparent electrodes required for these technologies. The push for renewable energy and better touchscreen devices hinges on the basic work of preparing and manipulating these compounds.
Health, Safety, and Environmental Concerns
Lab experience also drives home the fact that using chemicals like indium nitrate comes with responsibilities. The compound can irritate skin and eyes, and inhaling the dust isn’t safe. Safe storage and handling need attention to avoid accidental exposure. This isn’t just about following a rulebook; it's about protecting people working directly with these materials, whether in an industrial setting or a university lab.
On the environmental side, waste disposal demands careful attention because indium belongs to the group of rare metals. Recovering and recycling indium from electronic scraps becomes more urgent as demand carries on rising. Indium nitrate eventually finds its way into the recycling stream through old screens or panels. More support for closed-loop recycling networks could cut down on mining and help protect resource reserves for future generations.
Moving Forward
The world might not talk much about indium nitrate, but its fingerprints show up in electronics, renewable energy research, and nanotechnology. Industry, education, and government can each play a part in improving recycling, handling, and innovation so that this valuable resource keeps contributing to new breakthroughs.
What is the chemical formula of Indium Nitrate?
Getting to Know Indium Nitrate
Indium nitrate comes up pretty often in labs and industrial spaces, thanks to its interesting properties. The formula sits clear: In(NO3)3. This combination puts one indium atom together with three nitrate groups. That number three actually means a lot because it hints at the chemical structure and how people end up using the compound. Anyone who’s handled chemical equations recognizes the difference that small numeric details can make—getting that wrong leads straight to ruined experiments or plant downtime.
Why Indium Gets Matched With Nitrate
Most folks remember indium as a soft, silvery metal, kind of tucked between cadmium and tin on the periodic table. A century ago, nobody gave much thought to indium compounds, but as electronics got smaller and made with more precision, indium salts like this one picked up speed. Indium nitrate isn’t flashy by itself, but it breaks down into other indium compounds used in touchscreens, thin-film coatings, or specialty solders. Factories lean on it, glassmakers turn to it, and research labs rely on it for pure indium ion sources in solution.
Why the Chemical Formula Matters
Most people see a formula like In(NO3)3 as just another set of symbols. In the world of production and lab work, each symbol spells out very real decisions about storage, safety, and even costs. One wrong digit during ordering or inventory can mean a shelf full of unusable material. Indium nitrate loves moisture—left sitting out, it draws in water and changes weight and quality. That property often gets overlooked until everything seems to stick together or dissolve at the wrong time.
My own introduction to indium nitrate didn’t start in a text book; I got to know it by cleaning up a work bench where someone left a jar open on a humid afternoon. The powder had clumped up, sticking to everything, and the label smudged. After a few hours, the lesson sticks: handling starts with knowing exactly what’s inside the jar, right down to those little numbers in the formula.
Handling Hazards and Finding Safer Solutions
Indium compounds don’t usually get listed in the top charts of hazardous materials. Still, anyone with experience can tell you that nitrates in general can get feisty—under heat or when mixed with the wrong chemicals, nitrate groups break down and release gases. This means nobody can treat indium nitrate like common table salt.
From a health perspective, careful attention helps avoid long-term exposure risks. The dust can irritate the nose and lungs, and spill protocols vary depending on whether you’re working as a hobbyist or running a large operation. A good lab keeps proper ventilation and decent training so people aren’t surprised by unexpected reactions. Over time, companies that handle indium nitrate at scale build up recycling and waste systems so leftover material doesn’t make its way into water or soil.
Fresh Approaches for Practical Problems
There’s always room for safer storage—airtight, moisture-proof containers work much better than whatever’s left in the corner of a busy lab. In larger operations, environmental controls and regular checks make a clear difference. On the chemistry education side, clear labeling and small, practical demonstrations have more impact than just repeating “In(NO3)3” for the sake of memorization.
Once people see not just the formula, but the real uses and risks, indium nitrate becomes more than just a curiosity or a hazard. It’s a real part of making things work, one small batch at a time.
Is Indium Nitrate hazardous or toxic?
Understanding What’s at Stake with Indium Nitrate
Indium nitrate isn’t a household item, but it shows up in labs, electronics factories, and some specialty manufacturing circles. Its role in making semiconductors and liquid crystal displays puts it on the radar for plenty of people working in tech and research. Most folks outside those fields haven’t thought about it, but anyone near industrial chemicals owes it to themselves to stay informed about what’s being handled in the workplace.
The Hazards No Worker Should Ignore
From a personal perspective, I’ve seen more than a few colleagues brush off the importance of safety sheets and hazard warnings, especially with obscure chemicals like indium nitrate. Indium nitrate can irritate the skin, eyes, and airways. If inhaled, it doesn’t take long for coughing and a burning sensation in the throat to show up. In stronger doses, it has even caused lung damage in animal studies. Extended contact might affect the liver or kidneys. Its danger isn’t always immediate—over time, little mistakes add up. Neglecting masks or gloves costs people in the long run.
No one should ignore nitrates as a group. High exposures can affect how blood carries oxygen or mess with organs. There’s enough overlap in how different nitrates harm the body to make respect for every member of that chemical family a smart choice. Indium itself isn’t as widely studied as lead or cadmium, but early evidence suggests it deserves the same level of caution. Japan’s electronics industry dealt with lung diseases linked to indium dust—workers with direct exposure faced a higher risk of lung disease and scarring. Even if indium nitrate isn’t the exact cause, past incidents should serve as wake-up calls.
Simple, Reliable Ways to Stay Safe
Experience has taught me that basics matter most. Good labs and responsible employers build safeguards into daily routines, like proper ventilation, chemical-resistant gloves, and eye protection. Clean workspaces and easy access to safety showers or eyewash stations can mean the difference between a minor scare and a trip to the ER. Sometimes, the best protection is just knowing how to respond if a spill or splash happens. Rapid cleanup and personal decontamination reduce both fear and harm.
Labeling and storage habits also make or break chemical safety. Indium nitrate doesn’t spread itself around, but a tipped-over bottle on a crowded shelf has caused dozens of close calls. Workers should demand clear signage and well-kept inventory lists to avoid surprises. MSDS sheets shouldn’t gather dust in a back room; they ought to hang near workstations. In my own job, I’ve found that reviewing these documents as a team every quarter puts everyone on the same page—and even keeps the newbies asking the right questions.
Building a Healthier Work Culture
To move forward, companies should invest in more training, and add regular check-ins on chemical handling. Leadership needs to walk the floors, ask about unsafe practices, and listen to workers’ concerns. Regulators could offer more up-to-date, easy-to-understand resources on chemical exposures. Many smaller shops fall through the cracks, lacking both in awareness and resources for protective gear. But no worker should trade health for efficiency.
Staying cautious with indium nitrate isn’t about overreacting. It’s about expecting that people deserve best practices, not just good enough efforts. Folks in leadership and on the floor both gain from a culture of respect, communication, and ongoing attention to what goes in—and out—of the lab.
How should Indium Nitrate be stored?
The Everyday Risks Hiding in a White Powder
Plenty of folks overlook safety issues until a headline or an accident wakes them up. Indium Nitrate, looking almost harmless as a white powder or crystal, is a substance that can cause burns, respiratory problems, and environmental headaches if stored like just another chemical. People who work with it—lab techs, warehouse crew, research grad-students—deserve setups proven to keep them out of the ER and off those accident charts.
How I’ve Learned To Store Chemicals Without Regret
Years of working around chemical stocks in university labs and at a specialty materials distributor have hammered in some basics. Don’t stack things right next to incompatible substances. Don’t let humidity or sunlight mess with your storage areas. Don’t skip the label-reading. In my experience, shortcuts or boredom lead straight to damaged packaging and poorly-contained spills.
Indium Nitrate’s trouble spot? Water. This compound pulls moisture from the air, eventually breaking down into nitric acid and indium hydroxide. If a jar leaks or gets damp from loose humidity, you haven’t just wasted expensive material—you might end up scrubbing acid from steel shelving or patching up corroded floors.
Real Storage Advice—Not Just Checklist Filler
Forget the generic safety binder rules for a moment. Let’s get practical. A dry, cool space with tight temperature control works best. Pack Indium Nitrate in thick, tightly-lidded polypropylene or glass bottles. No metal caps—corrosion is a silent wallet-drainer. Shelve the bottles somewhere off the ground, not next to acids, bases, organics, or any oxidizing stuff.
Humidity sneaks into buildings from places you wouldn’t expect—cracked sealing around doors, poor HVAC units, clogged vents. Toss desiccant packs into storage cabinets as backup. An extra humidity monitor fixed near your chemical stock can save you the price of a new floor or an insurance claim.
Label everything right down to batch date and quantity. Don’t rely on memory or handwritten marks that fade or flake off. If you ever walked into a storeroom looking for ‘that white bottle’ only to realize the last guy switched it with something else, you know what a costly mistake it is.
People and the Environment Deserve Precaution
Exposure ain’t rare. Inhaled dust irritates lungs, especially for folks with asthma or allergies. Skin burns, nosebleeds, headaches—these are the common complaints if spills aren’t managed right away. Protective gloves, good ventilation, and sealed goggles make spills optional, not regular.
Down the drain, a little can go a long way—Indium isn’t just another harmless metal. It’s tough for water treatment plants to filter out, letting it slip into rivers or groundwater. Disposing of the stuff as ‘labs have always done’ is a problem. Follow local hazardous waste codes. Lock the material in a leakproof waste drum, and schedule pick-up with a vendor who traces their disposal, not just dumps it.
Small Steps That Pay Off Later
If a supervisor or safety officer shrugs at specific storage for Indium Nitrate, point out the cost of lost production, cleanup fees, and health claims. It’s not a mountain of work. Plugging leaks, double-checking containers, teaching good practices, and updating safety labels are simple investments.
Indium Nitrate won’t explode your warehouse, but stored carelessly, it has a way of causing harm that sneaks up on you. Keep things dry, labeled, and sealed, and you’ll barely think about it—exactly the way it should be. Experience counts, and the old basics work best.
What are the common methods of handling and disposing of Indium Nitrate?
Staying Safe in the Lab or Plant
Indium nitrate turns up in research, electronics, even solar panels. Most folks in industry cross paths with this stuff as a white, water-soluble powder or in liquid form. If you’ve worked with indium compounds, you know they don’t smell or look dangerous, but that isn’t much comfort if you’ve seen the safety data sheets.
There’s no sugarcoating it—breathing in the dust means risking lung irritation. Getting it on your skin can bring out redness or burns. In shops where I spent time, everyone wore nitrile gloves and splash-proof goggles from start to finish. Long cotton lab coats helped, too. Some bigger operations lock down process steps inside fume hoods, cutting airborne risks and wiping down workbenches at the end of every shift.
One truth learned over the years: You don’t sweep up spills with a broom. Wet methods—damp towels, mops—stop dust from kicking up. Keeping a spill kit right by the workbench or reactor pays off, especially on busy days. Any waste—rags, gloves, towels—goes straight into labeled, closed containers. There’s just no shortcut, not if you care about your lungs or the folks working after you.
The Disposal Question Nobody Wants to Dodge
Once a batch gets made or an experiment wraps up, leftover indium nitrate becomes hazardous waste. You can’t just toss it in regular trash or pour it down the sink. Type in “indium toxicity” and you’ll pull up animal studies showing organ damage, so no one wants this stuff finding its way into rivers.
EPA rules treat spent indium nitrate as hazardous. That fits with every state-level regulation I’ve seen across companies. The common path is simple on paper: Gather the waste, tag it, and ship it off to a licensed chemical waste contractor. These professionals either neutralize it by chemical treatment or lock it into a solid matrix meant for hazardous waste landfills. The right storage for this waste means using corrosion-resistant drums or bottles. Flimsy packaging risks leaks on trucks—that kind of costly mess makes news and leaves a black mark on companies.
Why Regulations and Training Matter
Some shops get lazy, skip the training, or leave out the paperwork. That’s where small mistakes haunt a business. Local inspections focus on waste logs, safety plans, and routine training drills. Some years ago, one facility I visited paid a five-figure fine for letting waste bottles pile up without documentation. If every worker knows the hazards, and supervisors push for quarterly refreshers, mistakes stay few and far between.
One thing stands out: No matter how careful people get, someone eventually drops a container or mislabels a waste bottle. Best practice says review every step at least yearly and stay on top of any legal updates. That protects your team and keeps the operation humming.
Better Methods for the Future
Tech is moving, and green chemistry research looks at safer alternatives to indium nitrate. Until those become cost-effective, every lab or plant manager needs to treat indium nitrate with the seriousness it deserves. Closed-loop processes, recycling options, or safer analogues could make a real difference. For now, staying honest about what leaves your building—and how—remains the only fair way to keep communities and workers protected.
| Names | |
| Preferred IUPAC name | indium(3+) trinitrate |
| Other names |
Indium(III) nitrate Indium trinitrate Nitric acid indium(3+) salt |
| Pronunciation | /ˈɪn.di.əm ˈnaɪ.treɪt/ |
| Identifiers | |
| CAS Number | 13465-09-3 |
| Beilstein Reference | 3589841 |
| ChEBI | CHEBI:52060 |
| ChEMBL | CHEMBL4298878 |
| ChemSpider | 23865662 |
| DrugBank | DB11272 |
| ECHA InfoCard | Indium nitrate (ECHA InfoCard): 100.033.811 |
| EC Number | Indium Nitrate" EC Number is "236-397-7 |
| Gmelin Reference | 84902 |
| KEGG | C18627 |
| MeSH | D011857 |
| PubChem CID | 25013 |
| RTECS number | NL1050000 |
| UNII | 3078J3T52S |
| UN number | UN3288 |
| Properties | |
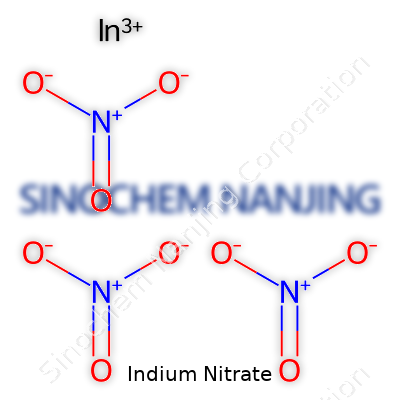
| Chemical formula | In(NO3)3 |
| Molar mass | 300.83 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 4.376 g/cm³ |
| Solubility in water | Soluble |
| log P | -4.73 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 6.64 |
| Magnetic susceptibility (χ) | -56×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.540 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 231.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -274.0 kJ/mol |
| Pharmacology | |
| ATC code | V09GX02 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes severe skin burns and eye damage, may cause respiratory irritation. |
| GHS labelling | GHS07, GHS05, GHS09 |
| Pictograms | GHS05, GHS07 |
| Signal word | Danger |
| Hazard statements | H272, H302, H315, H319, H335 |
| Precautionary statements | P234, P261, P264, P270, P271, P273, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P332+P313, P337+P313, P362, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-0-2-OX |
| Lethal dose or concentration | LD50 Oral Rat 365 mg/kg |
| LD50 (median dose) | LD50, Oral, Rat: 1130 mg/kg |
| NIOSH | RS2065000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Indium Nitrate: 0.1 mg/m3 (as Indium, OSHA PEL, TWA) |
| REL (Recommended) | 2 mg/m³ |
| Related compounds | |
| Related compounds |
Indium(III) chloride Indium(III) oxide Indium(III) sulfate Indium(III) acetylacetonate |

