Idoxuridine: History, Science, Application, and Future Directions
Historical Development
Idoxuridine’s story began in the late 1950s, just as virology started receiving serious scientific investment in pursuit of treatments for viruses that had long eluded medicine. William H. Prusoff, a pharmacologist, synthesized this compound in 1959. At a time when viral diseases like herpes threatened mostly with no treatments, Prusoff’s group wondered if modifying existing nucleosides would block viral replication. Early lab studies showed striking results: the new compound, now known as idoxuridine, stopped herpes simplex virus in its tracks by mimicking a regular nucleoside but flipping the script once incorporated into growing viral DNA. By 1962, the FDA had approved idoxuridine as the first antiviral drug used in humans. This marked a huge leap forward, setting a precedent for antiviral drug development. The common lesson here is innovation rarely follows a straight path—sometimes, insight grows at the intersection of past failures and a willingness to try something that seems only slightly different from what came before.
Product Overview
Idoxuridine arrived as an antiviral for topical use, especially for herpetic keratitis of the cornea. It did not cure the underlying viral infection but halted symptoms with considerably less damage than older, caustic chemical approaches. Ophthalmologists soon kept it on their shelves, counting on its ability to “trick” replicating viruses without crossing into toxic territory for most patients. It comes in eye drop or ointment form, formulated at low concentrations to balance therapeutic activity and patient comfort. Though many newer drugs have eclipsed idoxuridine in certain uses, hospitals worldwide still stock it because it works where newer options sometimes fail.
Physical & Chemical Properties
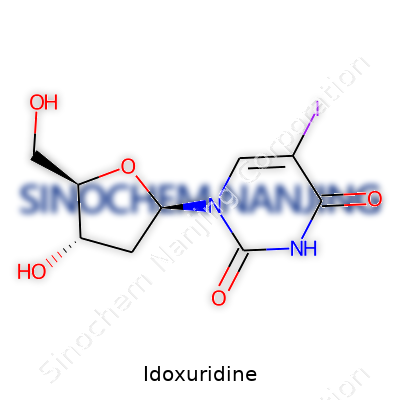
Idoxuridine’s molecular structure looks much like thymidine, one of the four DNA building blocks, but swaps an iodine atom for a methyl group at the 5-position on the uracil ring. The resulting molecule, C9H11IN2O5, holds a molar mass of about 354.1 g/mol. Physically, idoxuridine appears as a white to pale yellow crystalline powder, which dissolves in water and slightly in alcohol. The presence of iodine distinguishes it from its parent nucleoside, influencing its color, solubility profile, and UV absorption characteristics. Its crystalline nature calls for care in preparation, since impure batches or faulty storage can drop its activity or safety.
Technical Specifications & Labeling
Most commercially available forms contain idoxuridine at concentrations between 0.1% and 0.5%, stabilized in sterile aqueous solutions or semi-solid bases for ophthalmic use. Labels require clear identification not just of dosage but of single-use or multi-use container guidelines, since cross-contamination poses serious risk in eye care. Pharmacies must mark expiration dates prominently, as degradation products reduce effectiveness and raise toxicity risks. The product label lists not only the chemical name but regulatory warnings, storage instructions (typically in light-resistant containers at 2–8°C), and the manufacturer’s or compounder’s identification to ensure traceability. This robust set of specifications arose after real-world reports of misuse or storage errors caused harm, highlighting the real need for specific, practical labeling on drug packaging.
Preparation Method
Idoxuridine synthesis tracks a path involving iodination of uridine, usually through a halogen exchange step with iodine monochloride under controlled temperatures. Skilled chemists must maintain reaction conditions strictly to avoid generating unwanted byproducts or wasting expensive reagents. After the iodination, the crude mixture undergoes purification, often via recrystallization, to yield a product suitable for pharmaceutical compounding. The protocol requires filtering out unreacted starting material and side products. This hands-on, batch-focused process means that the quality hinges on the expertise and diligence of the chemist running the show, plus careful quality control at each stage.
Chemical Reactions & Modifications
Idoxuridine itself stems from chemical modification of the native nucleoside structure, bringing an iodo group directly onto the uracil ring. This single-atom substitution blocks the completion of DNA duplexes during viral replication, sticking a wrench in the enzymatic machinery. Further chemical derivatizations of idoxuridine, aimed at tweaking its spectrum or pharmacokinetic properties, have produced compounds like trifluridine—another antiviral with improved bioavailability. The chemistry behind idoxuridine’s effect boils down to a lesson seen often in medicinal chemistry: small changes in molecular structure make outsized impacts on biological activity. Chemists weigh each new idea for analogs with both hope and skepticism, as not every tweak delivers a better medicine.
Synonyms & Product Names
Idoxuridine shows up in chemical catalogs and medical formularies under a variety of names, including 5-iododeoxyuridine, IDU, IDUR, and by trade names like Herplex, Dendrid, Stoxil, and Keravid. Synaptic communication in healthcare depends on this awareness, since the same active ingredient could be listed under any of these labels. This multiplicity can fuel confusion for health professionals and patients alike, especially where drug procurement, substitution, or regulatory oversight cross borders.
Safety & Operational Standards
Workplaces handling idoxuridine, especially bulk powder or during formulation, adhere to safety guidelines established through decades of pharmaceutical use. Inhalation or skin exposure carries risk, so fume hoods, gloves, and goggles serve as much a part of the process as the chemistry itself. For patients, ophthalmic preparations must avoid preservatives that cause irritation or allergic response. Medical staff training, from pharmacists to nurses applying the drops, includes steps to avoid microbial contamination and dosing errors. All this work circles back to one point: high standards in drug prep save eyes, and sometimes lives.
Application Area
Ophthalmology relies on idoxuridine to treat dendritic ulcers of the cornea caused by herpes simplex. It finds use where acyclovir or newer analogs don’t sufficiently halt viral eye infections, or when cost restricts access to the latest therapies. Though regulatory agencies usually bar systemic administration due to toxicity and poor bioavailability, topical application offers strong antiviral activity at the site of disease. Beyond eye care, idoxuridine rarely surfaces, mostly because systemic toxicity outweighs possible benefits in broader viral contexts. Still, the compound’s direct action and solid clinical record keep it on the shelves, even in the era of high-tech antivirals.
Research & Development
Idoxuridine sparked a tidal wave of antiviral drug development that changed how scientists and clinicians approached viral disease. In the decades since its approval, pharmaceutical researchers took its central idea—genetic “decoys” that fool viral enzymes—to shape new generations of drugs. The push for better treatments saw the modification of idoxuridine’s core structure to lower toxicity (such as in trifluridine) or to optimize delivery methods. Scientific papers still cite idoxuridine as a model compound for studying mechanisms of antiviral nucleoside analogs. Researchers track resistance patterns in herpes viruses, aiming to fine tune future therapies not only for efficacy but for sustainability in the clinic. This legacy shows that the original molecule still holds lessons for new generations of chemists and pharmacologists.
Toxicity Research
Toxicological analysis of idoxuridine underscores its double-edged nature. In cell cultures and in vivo models, the molecule triggers cytotoxic effects in healthy as well as infected tissues if applied beyond the recommended window. Systemic absorption—fortunately rare with topical application—leads to bone marrow suppression or gastrointestinal upset, so strict control over route and dose matters. Eye-specific toxicity looms if used beyond two weeks, as corneal epithelium fails to regenerate properly. Regulatory agencies base their approvals on thousands of patient experiences, alongside ongoing post-market surveillance for new adverse effects. This vigilance reflects the medical maxim that even long-used drugs demand respect for their risks as well as their rewards.
Future Prospects
The future for idoxuridine leans toward targeted clinical applications rather than blockbuster status. Research teams look to nanoparticle-based formulations, improved delivery vehicles, and combination therapies aiming to minimize toxicity while boosting the drug’s local effect. Bioengineering advances hint at the possibility of using idoxuridine analogs in gene editing contexts or even in designer therapies for hard-to-treat viral infections. Regulatory and patient demands for safe, cost-effective alternatives ensure it remains relevant in global settings where newer antivirals remain out of reach. Each step forward builds on the foundation set by idoxuridine over half a century ago: one small substitution in a molecule can change the landscape of medicine for good.
What is Idoxuridine used for?
Understanding Why Idoxuridine Matters
Life throws all kinds of curveballs, and eye infections can be one of the most stubborn. Idoxuridine brings help in cases where the infection comes from a virus, particularly the herpes simplex virus. This isn’t the cold sore stuff many people brush off—herpes simplex can settle into the clear front part of the eye, called the cornea, and create some real trouble. I've seen just how much discomfort and damage that can bring. People suddenly lose sharp vision or suffer through pain that screws up their every day. Idoxuridine became important mainly because of its targeted approach against the DNA of these viral invaders.
A Bit of Science in Plain Words
Idoxuridine tricks viruses. Once in the eye, it sneaks into a virus’s genetic code. That virus, now unable to copy itself right, runs into a wall. The herpes simplex virus can’t multiply and make things worse. Compare that to regular antibiotics: they don’t touch this infection at all. This medicine opened new doors in fighting corneal herpes infections back in the day, and the relief was real. Statistics show untreated herpes simplex keratitis, the medical term for this problem, is still a leading cause of blindness from infections worldwide. Giving patients like the ones I’ve met a fighting chance means something.
Idoxuridine: Where It Fits and Where It Falls Short
Doctors only use Idoxuridine as a topical solution, usually as eye drops or ointment. Oral versions never worked out because the drug couldn’t get where it’s supposed to in the body. In my own family, we always kept our hands clean and didn’t share towels for this reason: once a herpetic eye infection spreads, it can do some serious damage fast. Even with this medicine, doctors warn patients to use it exactly as told. Too much, and it starts to harm healthy eye tissue. Not enough, and the virus wins. Patience and careful following of medical advice go hand in hand here. As much as Idoxuridine brought help, it's far from perfect. Today, better drugs with fewer side effects often take over—like trifluridine or acyclovir—but not every case benefits from the new options.
Staying Safe and Thinking Forward
Eye health rarely gets attention until something goes wrong. Most people learn about Idoxuridine after hours spent fighting pain and blurred vision. People who wear contact lenses or take immunosuppressive medications should pay extra attention. Quick care, not ignoring symptoms, and honest conversations with eye doctors make a real difference. Newer antiviral medications keep showing promising results, but cost and availability still shut many out. Education, early action, and regular eye checkups turn minor irritation into a win, not a lifelong struggle.
Supporting Trust by Sharing Facts and Solutions
Doctors still look at every new case on its own terms. No magic pill exists yet, and Idoxuridine’s legacy lives on in the hard-earned lessons it taught. Staying informed helps families spot eye infections and seek out fast care. If someone starts seeing redness, pain, or sensitivity in sunlight, waiting usually makes things worse. Eye protection, clean hands, and skipping the urge to share towels or eye drops go further than most people realize. What matters is that real help continues to reach those who need it most, and that people learn the value of prompt, expert medical advice—whether Idoxuridine sits on the prescription pad or not.
How should Idoxuridine be applied?
An Old Antiviral That Still Matters
Doctors once reached for idoxuridine more often; today, it pops up in eye clinics and specialist practices, mostly to treat viral eye infections like herpes simplex keratitis. This drug changed the game for people with herpetic eye disease decades ago. Science has moved fast, but idoxuridine still finds its way onto prescription pads. Even in a world awash with new antivirals, stories from my own healthcare days remind me how basics matter. One time, treating a persistent corneal ulcer, our team saw quick progress after careful dosing with idoxuridine. Good technique helped avoid setbacks, and the patient kept their sight.
Application Really Means Everything
Idoxuridine comes in both drop and ointment forms. If anybody wants to see results and avoid unnecessary harm, paying attention to the little details counts. Before touching that tiny bottle, patients should wash their hands really well. It sounds simple, but unwashed fingers turn a helpful treatment into a risky routine. Infection control keeps both patient and clinician safer in the process.
For the drops, tilting the head back, pulling down the lower lid to create a small “pocket,” and squeezing one drop right in gives the best shot at getting medicine onto the infected tissue. The bottle tip shouldn’t touch the eye, lashes, or skin. Even a brief touch risks introducing bacteria and makes each future dose a gamble. With ointment, a thin strip inside the lower lid spreads well when the eye blinks a few times. Only use what the doctor recommends—no piling on extra doses—even if the eye feels irritated. More is not always better.
Side Effects: Not Just List Stuff
Some folks dismiss warnings as just legal speak. Red eyes, mild stinging, and blurry sight might follow drug use. Serious problems, like allergic reactions or persistent swelling, demand a quick call to the doctor. Idoxuridine tends to dry out the surface of the eye with overuse; that increases the chances for scarring, making vision worse. I’ve seen a few cases where overzealous self-treatment led to tough conversations and guilt. Health teams must not just hand out a bottle but show patients exactly what to do.
Help Patients Stay Safe
Most pharmacies pack little advice sheets with prescriptions. I always encourage patients: read that paper, yes, but ask real people too. Pharmacists answer questions many web searches never will. Eye specialists know whether idoxuridine’s right for you. Sometimes the drug’s not the best fit. Pregnant women and children need extra caution. A phone call beats regret every time.
Building Trust and Health Habits
There’s no shortcut to learning the method right the first time. Health teams should show every patient the best way to get drops or ointment onto the eye, even with crowded schedules. After all these years, I notice that those who get a quick demonstration in person often need less follow-up and worry less about complications. By taking a little more care with those first few applications, folks can avoid bigger problems down the line. People don’t always remember drug names, but they never forget the help that made tough treatments easier. Proper application, clear communication, and a little patience set up hope for better results—and safer care.
Are there any side effects of Idoxuridine?
What is Idoxuridine?
Idoxuridine has long been used in the treatment of eye infections, mainly to fight off the herpes simplex virus in the cornea. You would usually find it in the form of eye drops or ointment, prescribed by doctors who have to weigh the benefits against the risks in every single case. The pharmaceutical world keeps this medicine handy mainly because viral eye infections show up more than most realize, and untreated, they can lead to pain, sensitivity to light, and even vision loss.
Adverse Reactions: My Own Encounters
People want quick solutions when their eyes start burning and turning red. Personally, after a stint covering health news, those calls for help stick with me. Eyes matter so much in day-to-day life—take away even one day of clear sight, and you remember it. Idoxuridine does its job, but it sometimes brings trouble along.
Redness, irritation, and a burning sensation often show up as part of the package. Many people say the drops sting when they go in. Sometimes, excess tearing follows, and people see mild swelling or even a rash around the eye. Some experience blurred vision for a short time after drops, which makes driving and reading tough. If irritation keeps building or vision gets worse, stopping the drops and calling the eye doctor would be wise.
Rarely, using idoxuridine can trigger hypersensitivity. Swollen eyelids, itchiness around the lashes, or even a scaling rash may develop. I remember my friend, feeling desperate to clear up viral conjunctivitis, ended up with both sore eyes and flaky skin around them for weeks after using this treatment. He later said he wished someone had warned him that what helps one person’s infection can sometimes spark another issue.
Why Monitoring Matters
Medication safety hinges on monitoring and communication. A recent review in the British Journal of Ophthalmology noted that antiviral eye medicines like idoxuridine lead to complications in about one out of every ten users. That’s not rare. For folks with weakened immune systems, those side effects can mean bigger risks: secondary bacterial infections, corneal thinning, or long-term irritation.
The FDA and global health bodies keep idoxuridine on a short leash for this reason. They advise doctors to use it only when the benefits are clear and other options haven’t worked. Many patients wonder whether to try stronger antivirals or newer options with fewer side effects. Physicians now suggest these alternatives if idoxuridine leads to any discomfort lasting more than a few days.
What Helps Reduce Problems
Practical steps matter more than wishful thinking in medicine. Doctors suggest doing a full eye check before writing that prescription. They ask about allergies, other medicines, and previous drug reactions. Using a dropper with clean hands, timing the doses carefully, and avoiding touching the dropper tip to any surface lowers the chance of infection, which sometimes blurs the line between normal side effects and more serious problems.
Reporting side effects as soon as they appear makes all the difference. Pharmacists offer guidance too—some even track reactions and provide feedback to both patient and prescriber, making sure small annoyances don’t balloon into big complications. Clear written instructions help patients avoid overdose or sloppy hygiene, two common triggers for more problems than any virus itself.
Idoxuridine gave doctors hope during a time with few answers for viral eye infections. Its side effects remind us to respect every medication and pay close attention to our own bodies during treatment. Balanced use, honest discussion with healthcare providers, and quick response to new symptoms remain the best ways to keep those risks low.
Can Idoxuridine be used during pregnancy or breastfeeding?
The Real Questions Around Antiviral Eye Drops and Pregnancy
Many expecting mothers worry about safe medication options when dealing with viral eye infections like herpes simplex keratitis. Idoxuridine, one of the oldest antiviral eye drops, often comes up in conversations with ophthalmologists. As someone who has personally been part of these conversations, I’ve seen how tangled the advice becomes, especially when pregnancy and breastfeeding are on the table.
What Makes Idoxuridine Tricky for Pregnant Women?
Idoxuridine works by blocking the DNA replication of herpes simplex viruses, which bring on those painful corneal ulcers and cold sore outbreaks. But drug safety in pregnancy rarely boils down to what works in adults. The reality is that the developing fetus processes chemicals differently and remains far more vulnerable to DNA-targeting compounds.
Animal studies point to birth defects when idoxuridine gets injected in high doses. The problem: researchers haven’t nailed down exactly what a safe dose looks like in humans. The FDA still slots idoxuridine as a pregnancy category C drug—means that researchers saw adverse effects in animal fetuses, but data for pregnant women stays murky.
Antiviral eyedrops like this one don’t enter the bloodstream in large quantities, which certainly reduces risk. Even so, a little medication can add up, especially during the delicate first trimester. For me, this highlights why the instinct to “play it safe” makes sense if other options sit on the table.
Breastfeeding and Eye Drops: A Tough Call
Breastfeeding mothers constantly weigh risk and reward with medications. With idoxuridine, studies haven’t measured how much passes into breast milk—or what happens next. Theoretically, topical eyedrops provide very little systemic exposure for the infant. Still, nobody can promise those amounts stay trivial if a mother uses higher doses or has damaged corneas.
Healthcare teams often choose alternatives like acyclovir ointment or simply advise breastfeeding mothers to minimize application and wipe away excess drops. Some ophthalmologists will only pull out idoxuridine for severe, uncontrolled infections, making sure to reassess at every visit.
The Doctor-Patient Connection Matters
Professional guidance becomes more important than ever here. Obstetricians and ophthalmologists bring experience and access to up-to-date research, and can track the mother’s eye and overall health through the process. Anecdotally, I know mothers who opted to control viral eye infections with compresses and careful hygiene alone, leaning on regular eye doctor follow-ups.
If pressed into using idoxuridine, professionals usually turn to the lowest possible dose, shortest duration, and careful monitoring. Everyone involved—doctor, pharmacist, and patient—needs to communicate openly about risks, infection severity, and possible alternatives.
Safer Routes and Open Discussion
My take is that expectant and nursing mothers benefit from cautious skepticism and open conversations. Antiviral eyedrops should never be chosen out of habit. Milder infections might resolve with warm compresses, lubricating drops, or time, if the doctor agrees. If things escalate, safer antivirals or even oral medication—like acyclovir with a long safety record—may fit better for mother and baby.
It’s worth noting that the drug landscape changes fast. As more data comes out, doctors and patients gain sharper tools for weighing real-world risks. Until then, leaning on shared decision-making and professional follow-up looks like the best way forward.
Do I need a prescription to get Idoxuridine?
The Basics of Idoxuridine
Idoxuridine isn’t something you spot in a neighborhood pharmacy run. This antiviral medicine targets eye infections, especially those caused by the herpes simplex virus. Back in the sixties, it came as a bit of a medical breakthrough for folks suffering from viral eye issues. Eye pain, redness, gritty feeling—all those symptoms spell out trouble, and doctors noticed quick improvement with Idoxuridine drops or ointment.
Why Controlled Access Exists
Getting a prescription for Idoxuridine feels like a barrier to some, but there’s real reason behind the rule. Herpes infections in the eye don’t act like a small cut or scrape—misdiagnosis risks vision loss if mishandled. Doctors often use eye exams and lab tests to get the whole picture before reaching for this drug. Side effects like allergic reactions, stinging, and even damage to your cornea can pop up if used the wrong way. So handing out these drops over the counter would only stir up bigger problems.
Doctor Guidance Matters
There’s more at play than just filling a script. Herpes simplex eye infections tend to reappear, and using the wrong eye medicine can give the virus more fuel to fight back. Steroid eye drops, for example, often make herpes eye problems more severe. If someone guesses wrong and skips straight to Idoxuridine without ruling out other causes, treatment risks outweigh any benefits. With medical oversight, infection gets the right diagnosis and the best shot at a full recovery.
Global Pharmacy Rules
Across different countries, rules about Idoxuridine remain pretty firm. Pharmacies in the US, UK, and Australia don’t hand out this medicine without a doctor’s order. Europe keeps similar standards. In places where rules get loose, problems snowball: resistant viruses, vision problems, and even blindness. The prescription requirement stays practical, rooted in years of experience.
Alternatives and Their Access
Doctors sometimes pick other antiviral drugs like acyclovir, trifluridine, or ganciclovir to tackle the same infections. These also need prescriptions. Pharmacies sell soothing artificial tears to help mild eye irritation, but those won’t clear up a viral infection. Prescription-only policies remain, not out of stubbornness, but because treating the wrong thing brings higher stakes for eye health.
Improving the System
Telemedicine has stepped in during recent years, breaking down some barriers for folks in rural areas or those who can’t easily reach an eye doctor. Video calls and remote consults make it easier to show symptoms, ask questions, and get expert advice—sometimes even leading to a same-day prescription. More work is needed to spread this access, and insurance coverage varies. If someone spots redness, eye pain, and blurry vision, that push to contact a doctor—online or in person—remains the safest move.
Good Health Means Smart Choices
It’s tempting to wish for easy fixes, but eye health sits on a razor’s edge. Idoxuridine counts as one of those drugs where more caution leads to better results. Seeking advice from a professional, sticking to proven treatments, and using new telehealth options carry more weight than risking home remedies or shortcuts. Vision’s worth the trouble.
| Names | |
| Preferred IUPAC name | 1-[(2R,4S,5S)-4-hydroxy-5-(iodomethyl)oxolan-2-yl]-5-methylpyrimidine-2,4(1H,3H)-dione |
| Other names |
5-iododeoxyuridine IDU Iododeoxyuridine Iodouridine |
| Pronunciation | /ˌaɪ.dɒkˈsjʊə.rɪˌdiːn/ |
| Identifiers | |
| CAS Number | 58-42-2 |
| Beilstein Reference | 3565956 |
| ChEBI | CHEBI:5804 |
| ChEMBL | CHEMBL1516 |
| ChemSpider | 1638 |
| DrugBank | DB00249 |
| ECHA InfoCard | ECHA InfoCard: 1000130-777 |
| EC Number | EC 3.2.2.2 |
| Gmelin Reference | Gmelin Reference 83234 |
| KEGG | D00357 |
| MeSH | D007036 |
| PubChem CID | 3678 |
| RTECS number | KV2975000 |
| UNII | 49060FDH99 |
| UN number | UN1851 |
| Properties | |
| Chemical formula | C9H11IN2O5 |
| Molar mass | 354.132 g/mol |
| Appearance | White, crystalline powder |
| Odor | Odorless |
| Density | 1.78 g/cm³ |
| Solubility in water | Slightly soluble in water |
| log P | -0.9 |
| Vapor pressure | 1.91E-7 mmHg at 25°C |
| Acidity (pKa) | 8.06 |
| Basicity (pKb) | 12.08 |
| Magnetic susceptibility (χ) | -80.0e-6 cm³/mol |
| Refractive index (nD) | 1.717 |
| Dipole moment | 3.77 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 528.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -7158 kJ/mol |
| Pharmacology | |
| ATC code | S01AD04 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302 + H312 + H332: Harmful if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-1-2 |
| Flash point | Flash point: 9.4 °C |
| Lethal dose or concentration | LD50 (mouse, intraperitoneal): 105 mg/kg |
| LD50 (median dose) | LD50 (median dose): Mouse intravenous 43 mg/kg |
| NIOSH | WX8750000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.1% |
| Related compounds | |
| Related compounds |
Trifluridine Brivudine Sorivudine Clevudine |

