Hypophosphorous Acid: A Commentary on Its Role, Development, and Future
Historical Development
Long before chemists could tune reactions with precision, hypophosphorous acid emerged in the scientific record. Early researchers learned to handle phosphorus out of both curiosity and necessity. Exploring the byproducts of white phosphorus led to the discovery of this simple yet reactive acid. Over time, industrial methods moved beyond rudimentary glassware, and the process of isolating hypophosphorous acid became more controlled. The acid found a spot among chemical reagents as scientists realized it played a reliable role in reducing reactions. The nineteenth and twentieth centuries pushed this compound into mainstream chemistry labs, not because it was rare, but because it could do things other acids couldn’t — especially stripping oxygen from materials in a predictable way. Even as new compounds appeared, hypophosphorous acid held its place, favored by those aiming for targeted reductions without unnecessary side products.
Product Overview
Folks working with hypophosphorous acid see it mainly as a colorless liquid, delivered in bottles sporting clear hazard labels. Industries and labs rely on this acid not for flash or novelty, but for utility and reliability. It plays a key role in electroless nickel plating, controlling metal coatings on surfaces ranging from microelectronics to automotive components. Chemists often grab it as a reducing agent for manufacturing applications and organic synthesis steps. It does the job in pharmaceuticals where gentle reduction keeps fragile molecules intact. In practical terms, hypophosphorous acid is one of those workhorse chemicals — not fancy, but always showing up where reliability matters.
Physical & Chemical Properties
The liquid form of hypophosphorous acid carries a faint, pungent odor. Its chemical structure, H3PO2, packs two hydrogens bonded directly to phosphorus, which affects its reducing power. It resists easy classification, behaving neither like a strong inorganic acid nor like simple organic acids. Instead, it offers a balance that suits both industrial and research labs. Chemists searching for selectivity gravitate toward R-group retention when hypophosphorous acid steps into a reaction. There’s a certain trust in physical reliability — it stays stable under routine conditions but demands respect because of its potential to spark fire in contact with oxidizers.
Technical Specifications & Labeling
Tracking purity matters, especially in electronic or pharmaceutical fields. Most bottles arrive labeled with concentration and clear hazard pictograms. Regulatory guidelines require mention of its corrosiveness and toxicity, so no one mistakes it for benign household acids. Purity ranges, consistently above 50%, crucially determine outcome in sensitive syntheses. Many suppliers provide certificates of analysis to document meticulous control. These steps don’t just keep customers informed; they serve as reminders that handling hypophosphorous acid isn’t casual work. Label information ranges from UN numbers to directions for dilution and storage, rooted in a history of incidents where careless handling proved costly.
Preparation Method
Most commercial producers rely on the reaction of white phosphorus with an alkali hydroxide, followed by acidification. This method builds on classical chemistry but scales with rigorous checks against unwanted side reactions. Laboratory chemists often introduce sodium hypophosphite to a solution, add acid, and capture the hypophosphorous acid that forms. Safe handling sits at the core of each step in the process. Controlling reaction rate and keeping impurities low takes both experience and care. Even small adjustments in temperature or reagents translate into meaningful differences in the finished product’s purity and stability.
Chemical Reactions & Modifications
The true utility of hypophosphorous acid shows up when it steps into redox chemistry. It strips halides from aromatic rings, forges new C-H bonds, and, most notably, reduces transition metal oxides to free metals. One underappreciated reaction removes diazo groups under mild conditions — a technique quietly adopted by countless synthetic chemists. Its salt, sodium hypophosphite, underpins electroless plating: the nickel deposits cleanly onto surfaces without electricity, thanks to the reducing power unique to hypophosphorous acid. Modifications, including forming esters and salts, broaden the functional range for industry, often smoothing the path for custom applications in coatings and manufacturing.
Synonyms & Product Names
The chemical world carries hypophosphorous acid under several flags: phosphinic acid, HPA, and even ‘hydrogen phosphonate’ among certain researchers. The IUPAC name, phosphinic acid, turns up in technical literature, but few outside academic chemistry use it. Supply shipments usually call it hypophosphorous acid or mention the concentration next to the name. The variety of labels testifies to the acid’s movement through both technical and trade circles.
Safety & Operational Standards
Incidents from the past underscore the need for vigilant handling. Hypophosphorous acid poses risks the moment it leaves the bottle. It irritates skin, eyes, and lungs on contact. Vapor inhalation can trigger serious health effects in unprotected users. It reacts vigorously with oxidizers, presenting fire and explosion risks that demand strict segregation from incompatible materials. Modern operational standards ask for full personal protective equipment, ventilation, and airtight containers. Institutions addressing long-term occupational health follow guidelines set by agencies like OSHA and the European Chemicals Agency. Regular training and emergency procedures aren’t bureaucratic hoops — they are non-negotiables in facilities handling significant volumes. Facilities that cut corners end up in news headlines, reminded that chemistry always punishes complacency.
Application Area
The list of uses for hypophosphorous acid branches into distinct industries. Metal finishing plants depend on it as a reducing agent in electroless plating, coating everything from circuit boards to car parts with a fine layer of metal. Organic chemists turn to it for selective reductions, especially during steps where alternatives might break down delicate molecules. In water treatment, its derivatives manage corrosion and scavenge oxygen, extending the life of pipes and reactors. Drug manufacturers use it to safeguard sensitive intermediates on the journey toward active ingredients. Even library science circles have debated using hypophosphorous acid to halt paper degradation, though toxicity issues often limit direct application. Its reach is not about volume but about delivering solutions that few other acids offer.
Research & Development
Research teams keep revisiting hypophosphorous acid, searching for new ways to harness its potential. Scientists investigate better synthetic pathways, aiming to balance yield, safety, and waste. Catalysis work continues to probe what else can be tailored with mild, selective reductions. Environmental chemists look for ways to capture and break down phosphorus waste, part of the larger push toward greener industry practices. Material scientists have begun to fiddle with modified hypophosphites in coatings that resist wear and tear, hinting at future uses beyond classic plating. Pharmaceutical research sometimes leans on new derivatives to protect fragile molecules or tune their properties, suggesting that hypophosphorous acid’s contribution to drug development could grow if toxicity concerns can be mitigated.
Toxicity Research
Toxicology labs spend a lot of attention on hypophosphorous acid because phosphorus compounds often show unpredictable effects in biological systems. Low-level exposure can cause headaches or nausea, while more significant contact threatens the kidneys and liver. Chronic exposure, especially in the workplace, piles on risks like skin sensitization and respiratory issues. Animal studies unearthed problems with metabolic disruption, though translating these findings into human risk depends on dose and exposure time. Safety reviews highlight the acid’s acute toxicity but also note that stringent protocols have prevented large-scale incidents in modern facilities. Regulators wrestle with the balance between allowing essential applications and keeping workers and communities out of harm’s way, leading to periodic reassessments of occupational limits and disposal rules.
Future Prospects
Looking forward, hypophosphorous acid faces both challenges and promise. Industries seek to cut costs, reduce hazardous waste, and improve safety for workers, all while pushing for higher performance. Alternatives in plating and reduction haven’t fully displaced hypophosphorous acid, in large part because of its unique role in selective modifications. Future research into novel catalysts and replacements will determine whether its industrial share grows or contracts. If scientists can tune its reactivity, reduce its toxicity, or recover it more efficiently from waste streams, hypophosphorous acid could keep its relevance even as stricter environmental standards appear. For now, this unassuming acid carries a legacy born of chemistry’s early years — a reminder that the old tools often have more to offer, especially as researchers, regulators, and manufacturers converge on solutions that work for people and the planet.
What is Hypophosphorous Acid used for?
A Chemical That Punches Above Its Weight
Hypophosphorous acid, often seen in labs simply as HPA, takes up a spot on the toolbelt for chemists and manufacturers alike. This compound slips into conversation mostly in circles where reducing power is needed, and that job shows up in more places than most folks realize. You see it working behind the curtain where a lighter touch is needed, not plowing through with brute force like some other chemicals.
Making Metals Shine and Last
Anyone who’s worked with metal finishing knows the pain of watching a freshly made part corrode or fade. HPA steps in during electroless nickel plating, which adds a tough, corrosion-resistant layer to steel and other metals. Here, the acid provides electrons that help metal atoms settle where they belong on a surface. Factories use this to toughen car parts, improve electronics connections, and even protect kitchen gadgets. Reports from industrial case studies highlight that electroless nickel coatings made with HPA can extend product life and keep things working clean and smooth for years. That kind of payoff matters to both businesses and anyone buying those products.
Bringing Pharmaceuticals to Life
Walk into any pharmacy, and you’ll spot the end results of a massive web that begins in the chemistry lab. HPA finds a role here, too, acting as a gentle reducer when chemists craft active ingredients for medication. Many modern antibiotics, painkillers, and specialty pills start out as base chemicals that must be precisely altered. One wrong step, and you’ve got an unusable substance. HPA helps turn the right kind of molecules into forms that fight infection or control blood pressure, offering more control than older or riskier chemicals. Regulatory agencies pay close attention to what goes in the cauldron, so anything that makes processes safer and more predictable stands out for good reason.
Protecting Wood, Wire, and Beyond
HPA also ends up on the frontline of fire protection. In the building trades, manufacturers rely on specially treated wood that’s less likely to burn or spread flame. Adding HPA to fire retardants helps keep homes and offices safer, giving people more time to escape fires or reducing damage when accidents happen. The National Fire Protection Association outlined several cases where materials treated with phosphate-based chemicals, including those made from HPA, slowed the spread of flames during laboratory tests. HPA also keeps another low profile in electrical manufacturing. It helps strip insulation from copper wire, an essential task before wires get bundled into electronics or vehicles.
Risks and Room for Improvement
No discussion of HPA skips past safety concerns. Like many potent chemicals, it demands careful handling. Toxicity and environmental hazards can’t be swept under the rug, especially as misuse has sometimes linked it to illicit activities. Labeling and training become critical on shop floors, in research labs, and in shipping warehouses. Smart controls and regular oversight protect workers and keep chemicals out of the wrong hands. There’s also a push among industry leaders to keep refining processes so that less residue ends up in wastewater, and more gets recycled or reused.
What Lies Ahead
Using chemicals carries responsibilities, and HPA demonstrates just how thin the line is between making lives better and creating risks. Stronger regulation, new recycling methods, and tighter workplace policies point the way forward. At the same time, the everyday products people rely on—from cell phones to safe buildings—get a little better, all thanks to compounds like HPA that do their work quietly, molecule by molecule.
Is Hypophosphorous Acid hazardous or toxic?
The Real Risks Behind Hypophosphorous Acid
Everyday life doesn’t bring science lab chemicals across most people’s kitchen tables. Some industries do handle a few that pop up more than others. Hypophosphorous acid stands out in some factories and labs because people talk about it in the same breath as “hazardous” or “toxic.” It’s true: this chemical shows up in places that make electronics, pharmaceuticals, and even some types of resins. But unless someone spent time on the inside of those operations, the mentions rarely travel much beyond chemistry class or a passing regulatory headline.
This acid looks clear, almost innocent, and sometimes gives off a bit of a sour smell. In science, appearances mislead. On contact, hypophosphorous acid stings skin, scorches eyes, and harshly irritates respiratory passages. Workers on shop floors know this after only one mistake—just a little mist or spill can leave a nasty burn or a cough that lingers. Facts say that even a small exposure, over time, adds up to chronic breathing issues if not given respect. Official safety data lays it out clearly for a reason: irritation, tissue damage, and risk of fire come with this one.
Why Toxicity Isn’t Just Hype
Plenty of chemicals wear the label “dangerous.” When regulators, like the U.S. Environmental Protection Agency (EPA) or the European Chemicals Agency (ECHA), assess risk, toxicity takes a front-row seat. Hypophosphorous acid carries the potential to react strongly and catch fire on contact with oxidizers. There’s no hint in its appearance, but even fumes hang around longer than people expect, bringing the hazard home through air rather than touch.
Long-term studies highlight damage to the nose, throat, and lungs. There’s reason to listen to warnings. A few decades back, some illegal drug producers started using hypophosphorous acid as a reagent. This led agencies to track its movements closely. It didn’t earn that attention by acting harmless.
Looking Out for Health and Safety
In manufacturing spaces where people encounter hypophosphorous acid, safety rules become daily routine. It isn’t just paper policy. Gloves with the right resistance, goggles that actually fit, and air systems that keep vapors out of everyone’s lungs show up in every well-run shop. I’ve seen firsthand how even a small lapse, like leaving a glove with a pinhole, can send someone to the first aid room with blistered skin. Once, a poorly ventilated workspace sent a whole shift coughing out the door, forcing everything to shut down till the exhaust fans worked again.
Official agencies advise keeping this acid far from heat, sparks, or incompatible chemicals. Written reminders matter, but in real life, storage cabinets marked with bold signage and regular inspections save more accidents than memos ever could. Labeling, spill kits, and fire extinguishers stand within reach—not because of paranoia but because of experience.
Modern Solutions and Safer Practices
As industry keeps evolving, green chemistry continues to push for safer substitutes and cleaner processes, but some applications still lean on this acid’s unique properties. Digitization now helps keep accurate records of usage and storage, minimizing human error through automation. Training—real, hands-on and interactive—beats bored scripts and drills. Workers who feel respected have shown time and again they pay more attention to these risks. No one likes cutting corners when the reminder sits fresh in a team member’s memory.
Chemicals like hypophosphorous acid call for layered defenses and honest conversations. Transparency, thoughtful design of plant layouts, and a steady stream of up-to-date information keep real people from becoming statistics. Risks don’t disappear with paperwork, but vigilance, simple safety steps, and a solid respect for the facts make a world of difference in every shift where it matters.
What are the storage requirements for Hypophosphorous Acid?
Why Hypophosphorous Acid Demands Respect in Storage
Anyone who’s had to store chemicals like hypophosphorous acid knows the job isn’t just about clearing some shelf space in a storeroom. This acid, with its corrosive and reactive nature, asks for real attention. Handling it directly, I've seen gloves degrade quicker than expected if they aren’t the right material. Improper practices can turn a tidy workspace into a hazard zone overnight. People often underestimate a clear, slightly oily liquid. But hypophosphorous acid has a knack for showing who’s boss when rules are bent.
Core Storage Considerations
Keeping hypophosphorous acid stable starts with the right container. Standard practice uses tightly sealed containers made of materials resistant to strong acids—think high-density polyethylene. Metal containers? Big mistake. They corrode fast, sometimes with enough heat to cause burns or worse. I’ve witnessed rusting drum lids after contact with acid fumes, leading to leaks no one wants to clean up. Clear labeling matters, too; mix-ups with more benign substances have triggered costly evacuation drills in some labs.
Mind the temperature and the moisture. Hypophosphorous acid breaks down slowly, but heat speeds up the process. Warm storage rooms aren’t suitable here. Anything above room temperature signals trouble, as excess heat can increase the rate of dangerous hydrogen phosphide gas formation. Humidity can also increase corrosion and trigger unsafe reactions with metal shelving. An overlooked puddle under a bottle, if left unchecked, grew into a crusty mess and stained the bench for weeks at a site I visited.
Ventilation and Isolation—More Than Just Buzzwords
One of the keys to safe storage is proper ventilation. Hypophosphorous acid gives off fumes that pose a risk not just to the person working, but to anyone in proximity for extended periods. Setting up a storage cabinet with dedicated venting is a detail that can't be skipped. This isn’t about ticking a box on a safety checklist; it keeps the workspace safe for real people.
Separation from incompatible chemicals shapes every storage layout. Combining hypophosphorous acid with oxidizers or heavy metal salts can start fires or explosions. I’ve seen near disasters from simple mistakes, like a delivery person stacking acids together. Storing away from open flames, heat sources, and direct sunlight helps maintain stability—best done in a cool, dry area well-marked for corrosives.
Real Solutions: Building a Safer Routine
High standards in labeling, inventory control, and employee training make a big difference. Digital inventory systems let the team spot stockpiles nearing the end of their shelf life, reducing the risk of breaks or leaks from deteriorating containers. Emergency spill kits, including absorbent materials and neutralizers, cut down on panic in a spill event. Clear instructions posted near storage areas improve confidence, which goes a long way in high-stress moments.
Personal protective equipment—gloves, goggles, and acid-resistant aprons—should sit within arm’s reach. Proper gear turns a potentially risky job into a manageable one. Supervisors who walk the talk by enforcing rules inspire safer habits and fewer shortcuts.
All of this comes down to respect—for the material, for the people working around it, and for the space itself. Chemicals like hypophosphorous acid don’t offer second chances. Smart storage is a daily commitment, backed by experience, facts, and a shared goal of keeping everyone safe.
What is the chemical formula and purity of Hypophosphorous Acid?
A Look at Hypophosphorous Acid
Science classrooms and factories both use some chemicals that most folks have never heard about. Hypophosphorous acid goes by the formula H3PO2. It often shows up in a colorless liquid form and gets recognized for its strong reducing properties. I first ran into it during a college chemistry lab, where we needed a reliable reducing agent. People in electroplating or certain organic syntheses often need hypophosphorous acid for its dependable performance.
The Role of Purity in The Real World
Purity means a lot in lab work or any industrial process, especially when talking about acids. Chemical reactions often depend on it—impurities can throw off results, mess with product yields, or even spark dangerous side effects. In everyday industry, a substance like hypophosphorous acid usually comes rated around 50% purity in water solution. This isn’t just marketing. Water dilutes the acid to a standard concentration that suppliers can promise. For specific pharmaceutical or high-tech processes, companies might offer purities above 99%, but outside of these niches, the 50% benchmark meets most practical needs.
Where It Matters: Safety, Regulations, and Trust
Purity isn’t just about getting clean test results. Take plating, for example—the final color, adhesion, and corrosion resistance of metal finishes rely on the absence of contaminants. I once saw a metal batch scrapped because off-the-shelf acid, sourced without checking its certificate of analysis, contained trace iron and copper. Mistakes like that don't just waste time; they eat into budgets and can lead to regulatory headaches. Regulatory bodies, including the U.S. Environmental Protection Agency (EPA) and the European Chemicals Agency, place strict guidelines on the chemical purity grades acceptable in various industries to minimize harmful byproducts and ensure public safety. Trust between supplier and end-user depends on consistent documentation, verified through certificates of analysis, not just the word of a sales brochure.
Purity: How Is It Checked?
Labs use tests such as titration, high performance liquid chromatography (HPLC), or ion chromatography to figure out how pure this acid is. Each method lets you catch specific contaminants—things like iron, sulfates, and nitrates. I remember labs where a single test result showing excessive metals forced entire production lines to pause. In more regulated fields, manufacturers will release batch-specific purity reports. This transparency helps people make informed decisions and protects public safety, especially in pharmaceuticals and electronics, where even a little contamination can mean failed batches or compromised components.
Steps Toward Improved Safety and Reliability
Factories and labs can avoid a lot of headaches by demanding clear, recent certificates for any shipment of hypophosphorous acid. Chemists and technicians should store acids properly, away from incompatible chemicals such as oxidizers. Regular training, safety reviews, and strict access controls reduce the chance of accidents. Engaging with responsible suppliers—those with a track record of transparency—usually proves cheaper and safer over time than chasing the lowest up-front price. For companies on tight budgets, buying smaller certified lots may lower risk compared to bulk purchases from unknown sources. Over time, good habits protect both workers and final products, all starting with something as basic as checking the label and certificate before that first drop leaves the bottle.
How should Hypophosphorous Acid be handled and disposed of safely?
Why Safe Practices Matter for Hypophosphorous Acid
Plenty of labs and industries rely on chemicals as sharp and reactive as hypophosphorous acid. Its key role in reducing reactions and its popularity among niche industrial applications make it useful but far from harmless. Exposure risks include burns, breathing trouble, and in serious cases, poisoning. I’ve observed colleagues in both research settings and small-scale production jobs treat this acid with real caution, and it’s not just paranoia—the stories of painful skin injuries or sudden clouds of noxious gas stick for a lifetime.
The chemical can catch fire in contact with air or other chemicals—believe me, it’s happened—and it releases phosphorus oxides, which are never things you want in your lungs. Personal experience shows a quick spill can catch everyone off guard. In those moments, only strict protocols keep small mishaps from becoming emergencies.
Direct Approaches to Safe Handling
Every bottle or drum I’ve handled arrived with clear hazard markings, often a skull or a corrosion pictogram that drives the point home. Use thick nitrile gloves, goggles with side protection, lab coats, and chemical-resistant aprons. Gloves matter: too many folks cut corners there and live to regret it when their hands sting for days.
Work in spaces with solid ventilation. Fume hoods aren’t optional toys—they suck up fumes fast enough to stop them from leaking into the room. One lesson I picked up early: never pour or mix hypophosphorous acid without a sturdy, labeled secondary container. Glass helps for short-term handling, but never trust it for storage unless specifically rated—stress or age can catch up fast.
Spills demand speedy action. Keep enough baking soda or a proper acid neutralizer close by, and treat even minor splashes seriously. It doesn’t pay to let tiny leaks go unnoticed, especially around warm machinery or open flames, since this acid can ignite surprisingly easily in the right conditions.
Disposal: Doing It Right
Disposing of hypophosphorous acid boils down to diligence. Dumping it down the drain pollutes waterways and puts treatment plant workers at risk. I’ve seen company teams coordinate disposal runs, using sealed drums with triple labeling, for the real outflows. Each drum heads to an approved hazardous waste facility—never just the regular dumpster or hospital bin.
The acid doesn’t do anyone favors when mixed into municipal waste streams; phosphorus runoff feeds algae, kills fish, and damages entire ecosystems downstream. Neutralization stands out as an important first step. Mix the acid with excess sodium hydroxide or carbonate under close supervision, always tracking the pH. The reaction gives off heat and gas, so run it in small batches. Once the mixture reaches neutral, it transforms into a safer phosphate salt, which still heads to hazardous waste—but without the explosive volatility.
Better Training, Better Results
Most mishaps I’ve witnessed came down to missing training or cutting corners, never equipment failure alone. Everyone from students to senior engineers needs regular refreshers on chemical and personal safety. Posters above chemical benches help, but nothing replaces a hands-on run-through of what rises and what falls apart in a spill or a fire.
Public awareness also plays a role. Community hazardous waste disposal programs accept materials from home labs or small workshops. Sharing tips in safety meetings and offering real examples from the field keeps the lessons memorable.
Moving Toward Safer Choices
There’s no substitute for safe, direct handling and strict disposal routes with hypophosphorous acid. Industries, schools, and home experimenters alike have shaped safer routines through trial and error. Every label read, glove replaced, and bottle properly stored adds up—not just to a cleaner work zone, but to lasting health for workers and the wider environment. The difference speaks for itself, even years down the line.
| Names | |
| Preferred IUPAC name | Phosphinic acid |
| Other names |
Phosphinic acid Dihydroxyphosphine Hypophosphite acid Acide hypophosphoreux Acido ipofosforoso |
| Pronunciation | /ˌhaɪ.poʊ.fɒsˈfɔːr.əs ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 6303-21-5 |
| Beilstein Reference | 14611 |
| ChEBI | CHEBI:24646 |
| ChEMBL | CHEMBL1201643 |
| ChemSpider | 79104 |
| DrugBank | DB14157 |
| ECHA InfoCard | ECHA InfoCard: 100.030.191 |
| EC Number | 231-913-4 |
| Gmelin Reference | 516 |
| KEGG | C02650 |
| MeSH | D006928 |
| PubChem CID | 1077 |
| RTECS number | WM4900000 |
| UNII | WX7H99QKD3 |
| UN number | UN1803 |
| Properties | |
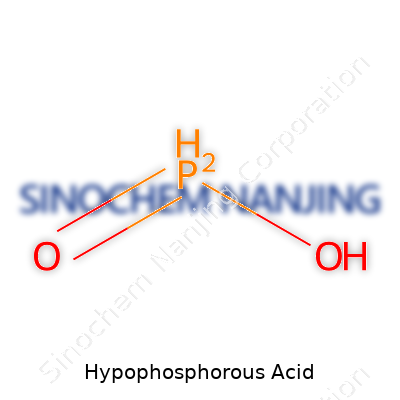
| Chemical formula | H3PO2 |
| Molar mass | 65.99 g/mol |
| Appearance | Colorless or slightly yellow fuming liquid |
| Odor | Odorless |
| Density | 1.144 g/cm³ |
| Solubility in water | miscible |
| log P | -0.77 |
| Vapor pressure | 0.03 mmHg (20 °C) |
| Acidity (pKa) | 1.2 |
| Basicity (pKb) | 1.2 |
| Magnetic susceptibility (χ) | Magnetic susceptibility (χ): -35.8·10⁻⁶ |
| Refractive index (nD) | 1.509 |
| Viscosity | 30 mPa·s (25 °C) |
| Dipole moment | 1.13 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 90.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −534.6 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -538.9 kJ/mol |
| Pharmacology | |
| ATC code | V03AB06 |
| Hazards | |
| GHS labelling | GHS02, GHS05 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | Hazard statements: Causes severe skin burns and eye damage. Harmful if swallowed. |
| Precautionary statements | P210, P280, P301+P330+P331, P305+P351+P338, P370+P378, P501 |
| NFPA 704 (fire diamond) | 3-0-2-W |
| Autoignition temperature | 240 °C (464 °F; 513 K) |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 oral rat 252 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 3,180 mg/kg |
| NIOSH | WAQ24630SY |
| PEL (Permissible) | PEL: 3 mg/m³ |
| REL (Recommended) | 500 mg/L |
| IDLH (Immediate danger) | IDLH: 5 ppm |
| Related compounds | |
| Related compounds |
Hypophosphite Phosphorous acid Phosphoric acid Phosphine |

