Looking Closer at Hydroxylamine Sulfate: Chemistry’s Quiet Workhorse
Historical Roots and Growing Role in Industry
Hydroxylamine sulfate first poked its head above the ground in the late 19th century, back in an era when chemists were still sorting out what went into building the molecules that power industry and medicine today. The compound quickly found a spot in the chemist’s toolbox, and not by accident. The world needed reducing agents, and this one got the job on account of its chemistry and affordability. In my years following industrial trends, it’s been striking how some of these old inventions keep returning with new purposes. Hydroxylamine sulfate isn’t a household name, but it’s nudged its way into countless manufacturing and research settings as time has rolled forward, transforming with every shift in what the world demands from its chemicals.
What Sets Hydroxylamine Sulfate Apart
On the surface, it looks as ordinary as you’d expect from a salt: white, crystalline, and dry. Its chemical formula—NH2OH·H2SO4—keeps it firmly in the stable, non-volatile camp, making storage and transport easier than more unpredictable reagents. That stability has been key for labs and factories alike. Chemically speaking, it packs a punch. As a reliable source of hydroxylamine, it doesn’t give off the same strong fumes or require the same careful handling that the pure compound would. The slow, thorough way in which it reacts has made it a steady favorite in processes where gentle reduction and precise modifications are needed. In practical terms, that means fewer headaches and smaller risks for the team responsible for handling it.
Specifications and What Labels Mean in the Real World
Labels on bins and bottles may sound dry, but they’re the first line of defense in making sure the compound gets used correctly. Industry specs for hydroxylamine sulfate spell out things like chemical purity, residual moisture, and pH in water solution because small variations can change how it works in a reaction or what results you see at the end. Whether for pharmaceuticals, textiles, or semiconductor cleaning, these specs blunt the chances of surprises, and the best suppliers back them up with real data. Years of work with industrial partners have shown the difference that good labeling makes—not just in safety, but in keeping entire production lines running smoothly.
From Synthesis to Shelf: How It’s Made
The most common way to make hydroxylamine sulfate goes back to straightforward redox chemistry. Typically, a solution of hydroxylamine is produced by reducing nitrogen compounds, like nitric oxide, with a reducing agent such as hydrogen or electrolytic methods. The practical beauty comes when chemists capture and stabilize the result by combining it with sulfuric acid, forming the sulfate salt. The process often calls for temperature control, careful monitoring of byproducts, and constant attention to safety, since unchecked reactions can quickly run into trouble. In commercial manufacture, the steps have been honed for decades to squeeze out as much product as possible, while containing side reactions. Seeing these processes up close drives home the balance of chemistry, economics, and safety that modern plants juggle every day.
Core Reactions and Fresh Paths in the Lab
Hydroxylamine sulfate’s chemical versatility helps explain its long life in labs and factories. It can turn carbonyl groups into oximes, a key step for making pharmaceuticals, pesticides, and specialty polymers. The reduction of nitro compounds, transformation of aldehydes, and the preparation of caprolactam for nylon all lean on this chemical. Every semester, chemistry students come to grips with its reactivity during core organic experiments. More recently, research teams have started tweaking the molecule itself, or blending it into new reaction systems, to invent gentler ways to achieve classic transformations, or to open up new routes that shun harsh conditions or expensive catalysts. The appetite for cleaner, more sustainable chemistry has pulled hydroxylamine sulfate back into the spotlight as researchers hunt for alternatives to fossil-fuel-based reagents.
The Web of Names: Synonyms in Science and Industry
If you keep an eye on technical documents, it doesn’t take long to notice that the same compound travels under a wardrobe of titles—hydroxylammonium sulfate, NH2OH·H2SO4, and various trade labels that come and go with suppliers. This jumble can trip up even seasoned engineers or researchers, especially when moving information between countries and across regulatory borders. In my conversations with teams navigating international procurement, the message is always clear: standardization saves money, time, and mistakes, but it remains a slow process.
Doing it Right: Safety, Handling, and Operational Truths
Safety in handling hydroxylamine sulfate comes down to respect for its chemistry. It doesn’t have the volatility or flammability of some industrial chemicals, but it brings its own risks—especially in contact with strong bases, oxidizers, or if it gets destabilized by heat. Cases of runaway reactions and accidents in the past have spurred both new handling protocols and stricter training. The lessons are always written in the regulatory guidance, but I’ve found real operational safety on the plant floor comes from tight routines, experienced teams, and a culture where speaking up about odd smells, unexpected residues, or even worn labels is encouraged. In settings where I’ve seen safety go right, managers listen to operators and updates to protocols happen often, not just in response to disasters.
Where the Compound Earns Its Keep
From industrial chemistry to everyday consumer goods, the reach of hydroxylamine sulfate can surprise those who have never handled it directly. One major area is caprolactam synthesis—the step between raw chemicals and the nylon fibers in clothing and carpets. Manufacturers lean on its reliability in semiconductor cleaning, removing oxidized surfaces without trashing delicate circuitry. In textiles, paper, pharmaceuticals, and even agricultural fields, hydroxylamine sulfate has a habit of turning up anywhere a gentle but effective reducing agent makes work cleaner or more efficient. My own introduction came through the agricultural channel—watching its use in seed treatment and pest management, where the tolerance for residues and byproducts remains razor-thin due to consumer safety standards.
Research: Digging for Greener and Smarter Uses
The last decade has seen a second wind for research on classic compounds like hydroxylamine sulfate. Scientists tackle head-on the challenge of reducing hazardous waste, cutting water and energy use, and designing better pathways to the chemicals that modern life demands. In academic and industrial labs, teams test new catalysts, rework reaction media, and search for bio-based replacements for fossil-sourced nitrogen. Research journals have documented how tweaks in preparation or dosing give more control over yield and purity, shrinking the environmental footprint. Seeing fresh ideas blossom from old chemistry brings a sense of optimism: tradition and innovation don’t have to sit on opposite benches in the lab.
Toxicity: What Decades of Testing Have Taught
Hydroxylamine sulfate’s toxicity comes into play if it’s swallowed, inhaled as dust, or if skin gets long exposure, which spells out the need for strong protective routines in workplaces. Animal studies have shed light on its acute and chronic effects, spurring regulatory limits in air and water. Field examples make the human side sharp—overexposure can lead to headaches, skin redness, and worse, making site monitoring and medical check-ins a necessity, not a box to tick. Checking equipment, using good air flow, running regular training: these steps end up saving real people from injury. In pockets of the globe where weaker safety enforcement still rules, a push from international partners and industry groups may do more to protect health than decades of paperwork.
Future Prospects: Where the Compound Goes from Here
Hydroxylamine sulfate seems set for a third or even fourth act, as environmental regulations, global supply chains, and green chemistry practices reshape industrial priorities. Companies want reagents that satisfy both performance and sustainability demands, often under tighter cost controls and heavier scrutiny from the public and investors. Energy storage, water treatment, and specialty manufacturing keep drawing interest. Every breakthrough in lowering process waste, cutting emissions, or opening new synthetic routes feeds the case for keeping the compound in the industrial inventory. From my own vantage point, the chemical’s ability to adapt—not just in how it's made, but in how it's used—will likely decide its shelf life as a workhorse, or whether rising alternatives finally put it out to pasture.
What is Hydroxylamine Sulfate used for?
A Common Player in Chemistry Labs
Hydroxylamine sulfate makes frequent appearances across all sorts of industries, especially for folks who work in chemical manufacturing. I remember from my own days in an undergraduate lab that this compound didn’t just sit on shelves; it played an active role in day-to-day experiments. Its main job, more often than not, involves reducing agents. In basic chemistry, that means it helps transform other substances, driving reactions forward in a controlled way. That reliability makes it an important tool for chemists tackling everything from research trials to regular production processes.
Photography: Film Development’s Unsung Helper
Hydroxylamine sulfate once held its own in darkrooms all around the world. Black-and-white film developers leaned on its performance to bring images out of negatives. The chemical helped reduce silver halides in exposed film so that pictures could fully emerge. Even as digital images replaced film, the compound sticks around in specialty labs and educational settings. Schools often use it to teach the basics of photographic development, blending a little science with a hint of history.
Textile and Dye Industries
Factories that work with dyes keep stocks of hydroxylamine sulfate close at hand. Dyeing textiles isn’t just about color; it’s about controlling reactions to avoid waste and ensure colors remain true. The chemical interrupts unwanted side reactions, keeping dyes stable and colors vivid. This has always struck me as one of those behind-the-scenes supports: people rarely notice or talk about it, but take it away and the results go sideways. The textile industry keeps using it because it works well time after time.
Pharmaceutical Processes
The pharmaceutical field asks for precision at every step, especially during the early stages of drug design and synthesis. Here, hydroxylamine sulfate often helps in making key building blocks for medications. Drug makers count on its abilities because it inserts itself neatly into certain chemical reactions, supporting the production of life-saving compounds. Efficiency and reliability carry real weight. Any mistake in synthesis risks delays for patients, but this compound helps keep things on track.
Purification and Metal Treatment
Some folks use hydroxylamine sulfate to purify precious metals, including platinum and palladium. Even in small amounts, impurities can ruin a batch of metal or compromise electronics that depend on these rare elements. Gold refiners and electronics recyclers frequently rely on it to improve the quality of recovered material. Cleaning up metals at the molecular level demands speed and accuracy, two things this compound can deliver when handled carefully by skilled professionals.
Potential Risks and Smart Solutions
No chemical comes without risks, and hydroxylamine sulfate deserves the same respect. Mishandling can lead to hazardous releases or health incidents, sometimes with serious consequences. Anyone using it should wear gloves, goggles, and work with good ventilation. Industry guidelines exist for a reason—neglecting them can endanger whole teams. Training helps, but ongoing safety checks make a real difference. Lab managers and educators can run regular refreshers, making sure everyone from newcomers to veterans remembers the risks. Investing in safer chemical alternatives also makes sense for certain applications, cutting down on exposure and keeping operations running smoothly.
Looking Forward
As industries shift with new technology, the uses of hydroxylamine sulfate might change. Right now, its steady performance guarantees a lasting place in labs and factories. By keeping safety at the front of every process and exploring greener swaps when possible, everyone benefits in the long run. My experience tells me that the best outcomes happen when people combine old knowledge with new ideas, never getting too comfortable with “how things have always been done.”
Is Hydroxylamine Sulfate hazardous or toxic?
The Real Risks in Everyday Use
Hydroxylamine sulfate lands in a chemical category that always gets my attention: useful but not to be taken lightly. It helps make pharmaceuticals, photographic developers, and even some pesticides. That’s not something you keep under the kitchen sink. I’ve worked around chemical storerooms before, and you can spot the warning labels from across the shelf—hydroxylamine sulfate ones tell you right away, “Don’t fool around with this.”
Health Hazards are Real
The stuff can irritate the skin, eyes, and respiratory tract. Not just a mild itch, but burning and redness or even blistering. I’ve seen coworkers not pay attention, pull off a glove too soon, and a tiny splash means a whole afternoon in the safety shower. Inhaling powder is a real risk, since fine particles go straight to your airways; a few minutes around a poorly sealed bag, and folks are coughing. Lab safety sheets – the kind you see pinned to walls in university basements – don’t exaggerate here.
Hydroxylamine sulfate acts as a reducing agent, so it tweaks red blood cells and can set off something called methemoglobinemia, where oxygen can’t bind properly in blood. Symptoms might start with a headache or dizziness but paying attention early is the only way to avoid a trip to urgent care. One time an accidental small spill during an undergraduate research project sent someone to get a blood test, just to be safe.
Fire and Explosion Potential
Even stored right, this chemical has a bit of a reputation. It breaks down and releases gas if it gets too hot — that’s why you never store it near strong oxidizers. One spark or open flame, and you’re dealing with more than just a mess. Rarely, if it dries out or mixes with certain other chemicals, it can set off a reaction hot enough to cause real damage. That’s the sort of thing you learn the hard way if someone ignores training.
Environmental Impact and Disposal
Anything going down the drain or tossed without a plan means trouble for water treatment plants and eventually for streams and lakes. Fish and aquatic plants don’t handle sudden chemical exposure well. Many places now enforce special disposal barrels and label systems to keep this compound out of normal garbage trucks. It’s not the kind of chemical you can simply dilute and forget about, at least not unless you want surprise regulatory fines and angry calls from environmental agencies.
Balancing Utility and Safety
I understand both sides—researchers and workers see its value; safety folks lose sleep over possible accidents. Wearing gloves, goggles, and using proper ventilation makes a real difference. So does sticking to small quantities unless there’s a strong reason. Most labs and factories keep detailed records of every bit brought in or used, and for good reason—it makes it harder to forget just how seriously to take it.
Strict rules about handling, storage, and disposal all come from past lessons. Training sessions support real safety and prevent health emergencies before they ever become a story. Even if the chemical sits behind a locked cabinet for months, its potential hazard stays the same.
Practical Takeaway
Hydroxylamine sulfate has its place in science and industry, but none of us can afford to treat it like just another routine material. Experience with safe handling and up-to-date knowledge saves lives, reduces health risks, and protects the waters and soil outside our labs. Respect for the hazards isn’t just policy—it’s personal.
How should Hydroxylamine Sulfate be stored?
Some chemicals come with a reputation, and hydroxylamine sulfate fits that bill. As someone who once had to dig through countless safety sheets to keep a small lab running, I paid extra attention to this compound. Hydroxylamine sulfate doesn’t leave much room for error: it reacts to temperature, moisture, and even its neighbors on a storage shelf. Those who deal with this chemical know that careless handling can mean property damage or worse.
What Makes Hydroxylamine Sulfate So Fussy?
This compound picks up water from the air. Open containers greet humid days by clumping up or, worse yet, starting uncontrolled reactions. Stories pop up in the chemical industry about containers swelling or even rupturing when left unchecked. At the same time, this material won’t tolerate severe heat: at high temperatures, it breaks down, sending out toxic gases. Nobody wants to breathe nitrogen oxides and sulfur oxides in a poorly ventilated space. All this doesn’t just read like a warning label—it’s routine reality for folks on the ground.
Setting Up the Right Storage Conditions
Before adding any container to a storeroom, think about air and temperature. Dryness trumps all. Desiccators or low-humidity spaces make life easier, reducing headaches down the line. Store it below room temperature when possible, but definitely keep away from any place that sees spikes above about 30°C (86°F). At home or in small labs, dedicated chemical fridges without food or drink offer more than just peace of mind—they keep accidents at bay. Locks on those fridges don’t just protect kids; they limit the risk of untrained hands grabbing the wrong jar.
The Role of Containers and Labeling
Glass or high-grade plastic containers work, as long as they close tightly. Seals matter. Non-reactive liners defend against leaks or unexpected reactions with air. Each container deserves a sharp, easy-to-read label: product name, concentration, and clear hazard warnings. In my own lab, faded labels led to a close call with a bottle that had sat gathering dust for longer than anyone could track. We adopted a schedule, and monthly checks became as important as the storage rules themselves.
Stay Far From Incompatible Substances
Hydroxylamine sulfate can’t stand strong oxidizers, alkalis, or pretty much anything flammable. Chemical storage experts recommend dedicated shelves—never mix it with peroxides or metal powders. Emergency services respond faster and safer if chemicals follow this separation, because fewer surprises in an emergency mean fewer injuries.
Building a Culture of Care in Storage
Training staff and students becomes less of a checkbox and more of a shared habit. People make mistakes if they don’t know why policies exist. Clear rules about PPE—gloves, goggles, lab coats—and spill kits placed nearby set the tone right away. My own mentor always drilled us: “Respect the rules, not just for yourself, but for everyone you work with.” Big accidents start small. Simple habits like closing lids every time or checking humidity gauge readings save lives and money.
Solutions for Small and Large-Scale Stores
Big outfits might install ventilation hoods and monitor air quality; smaller places can make do with silica gel packs and regular inspection. For transport, UN-approved containers limit leaks and breakage, and keeping detailed logs lets everyone track who accessed which chemical and when. Technology helps: some labs use RFID tags for tracking. In practice, though, nothing beats hands-on walks through a storeroom and a well-trained eye spotting a swelling cap before it pops.
What is the molecular formula of Hydroxylamine Sulfate?
Breaking Down the Basics
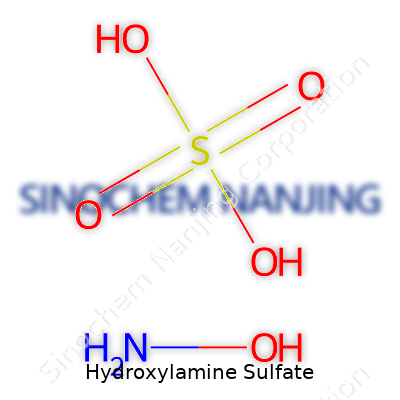
Hydroxylamine sulfate carries the molecular formula (NH2OH)2·H2SO4. People often call it a handy chemical for both laboratory and industrial settings. This formula shows two molecules of hydroxylamine for every sulfuric acid unit. The compound looks simple at a glance, yet holds a strong place in science.
Why the Formula Matters
Understanding what makes up this chemical isn’t about memorizing letters and numbers. It’s about safety, practical reality, and problem-solving. Hydroxylamine sulfate gets used everywhere from pharmaceuticals to water treatment—but it can also become risky if mishandled. By knowing exactly what’s inside, chemists and technicians make decisions that protect people, equipment, and the wider world. Recognizing the formula means recognizing exactly what kinds of reactions might happen, what precautions should be taken, and how to respond if something goes wrong. Reports of chemical exposure or runaway reactions often list hydroxl- and amine-related chemicals, so it helps to stay informed.
Practical Applications and Caution
Having worked with chemical inventories, I’ve watched how proper labeling and formula knowledge make or break a workday. With hydroxylamine sulfate, workers can create antioxidants, photographic developers, and rubber stabilizers. In water treatment, it helps clear out chloramines. Without an understanding of its formula, someone might underestimate how it reacts—leading to unsafe environments or contaminated products. Every decision in a lab or plant, from ordering raw materials to mixing solutions, depends on clear chemical identification.
Regulatory bodies, including the Occupational Safety and Health Administration (OSHA) and the Environmental Protection Agency (EPA), highlight the need to match product labels with molecular data. Even a small mismatch can mean the difference between routine work and an emergency call. Real-world incidents have proven that relying on structure, not just name or appearance, saves lives and money.
Challenges Faced in the Field
Hydroxylamine can decompose unexpectedly or interact with metals and other chemicals—something that textbook formulas put in concrete terms. From my own lab experience, I’ve seen confusion at the storeroom level when similar-looking chemicals are shelved together. Hydroxylamine’s sulfate form appears as white crystals, yet its makeup gives it stronger reactivity than many expect. Trained staff spot the signs of decomposition, but everyone benefits from clarity on the original molecular formula during first contact.
Accuracy in chemical identification supports everything from product quality to emergency response planning. Factoring in the molecular structure keeps teams one step ahead during audits and inspections. Data from chemical safety databases and case studies illustrate how knowledge gaps cause preventable accidents. Being precise isn’t a burden—it’s a daily practice that starts with knowing what you’re working with.
Pathways to Better Handling
People should look beyond labels to understand chemical formulas in every workplace. Everyone using or storing hydroxylamine sulfate gets a stronger grip on safety and handling by learning its full structure. Training sessions, easy-access Material Safety Data Sheets, and cross-checks in digital inventories help grow confidence and reduce risk. Hydroxylamine sulfate serves industries well—but only when its molecular formula anchors every decision.
How do you safely handle and dispose of Hydroxylamine Sulfate?
Working with Hydroxylamine Sulfate Without Trouble
Anyone who’s spent time in a lab knows the sting of chemicals that don’t give second chances. Hydroxylamine sulfate grabs attention thanks to its powdery appearance and sneaky danger. I learned early on to respect anything that can cause skin burns with a quick splash. Gloves carry more weight than ego in these settings. Goggles and a sturdy lab coat follow close behind. Lax habits or shortcuts rarely end well.
Spills draw a crowd for good reason. The compound gives off vapors strong enough to irritate lungs. Even a minor whiff goes straight for the nose and throat. Simple mistakes start with an open bottle or a messy weighing job. Anyone tasked with moving this chemical around can’t afford to act casual. I store it in a cool, dry spot, away from heat and sunlight, far from anything flammable or reactive. A closed container prevents moisture from making the powder clump and react.
Facts About Hydroxylamine Sulfate Hazards
Reports tie this chemical to several lab explosions over the years. It reacts fiercely with metals, oxidizers, and bases. Always check labels and containers twice, especially if many chemicals stand side by side on the same shelf. A good rule—no eating or drinking in the workspace—cuts down on accidental poisoning. Simple rules save lives.
No one should touch hydroxylamine sulfate bare-handed, and regular training makes a huge difference. Emergency showers and eye washes stay ready in facilities that take risks seriously. Most places that use this chemical have strict procedures and insist on written logs for every transfer, use, or storage move.
How to Get Rid of Hydroxylamine Sulfate Safely
Years back, someone tossed a small bottle into the sink—no thought, just muscle memory. The drain pipe corroded from the inside, turning a quick cleanup into a plumbing nightmare. Hydroxylamine sulfate must never go down regular sinks, into municipal garbage, or out with other office trash.
Sealed, labeled containers stand as the only way to prepare chemical waste for collection. Local rules require contacting a certified hazardous waste handler, and I always check their credentials and records. Most companies insist on storing spent chemicals in secondary containment. This step prevents accidental spills from spreading if the main jar cracks or leaks.
Smart Solutions going forward
A culture of double-checks and peer reviews cuts down on near-misses. I keep updated safety data sheets handy and run quick refresher drills once a season. Collaboration between users and the facility’s safety staff helps spot overlooked risks. Shared experience matters more than fancy degrees here.
Digital tracking solves the mystery of “where did that bottle go?” Barcode scanning and inventory software let us trace each step. Disposal logs and real-time reporting bring problems into the open faster. Encouraging open talk about mistakes sets the stage for steady improvement.
Safe handling and responsible disposal of hydroxylamine sulfate comes down to habits, not secrets. Trust grows with every safety briefing, honest question, and practical demonstration. I have seen labs with zero accidents for years—proof that steady, no-nonsense routines outweigh luck.
| Names | |
| Preferred IUPAC name | hydroxylammonium sulfate |
| Other names |
Hydroxylammonium sulfate Oxammonium sulfate Sulfuric acid, hydroxylammonium salt Hydroxylamine, sulfate (2:1) |
| Pronunciation | /haɪˌdrɒksɪlˈæmiːn ˈsʌl.feɪt/ |
| Identifiers | |
| CAS Number | 10039-54-0 |
| Beilstein Reference | 1691223 |
| ChEBI | CHEBI:61408 |
| ChEMBL | CHEMBL50461 |
| ChemSpider | 22441 |
| DrugBank | DB11345 |
| ECHA InfoCard | 100.043.392 |
| EC Number | EC 232-265-7 |
| Gmelin Reference | 82280 |
| KEGG | C06389 |
| MeSH | D017744 |
| PubChem CID | 6025 |
| RTECS number | MW4850000 |
| UNII | B1202Q2F8P |
| UN number | UN2865 |
| Properties | |
| Chemical formula | (NH2OH)2·H2SO4 |
| Molar mass | 164.14 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 1.88 g/cm3 |
| Solubility in water | Very soluble in water |
| log P | -4.0 |
| Vapor pressure | Negligible |
| Acidity (pKa) | pKa = 5.9 |
| Basicity (pKb) | 8.06 (for hydroxylamine) |
| Magnetic susceptibility (χ) | -6.1e-6 cm³/mol |
| Dipole moment | 1.47 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 214.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -563.7 kJ/mol |
| Pharmacology | |
| ATC code | V03AB38 |
| Hazards | |
| Main hazards | Oxidizer, harmful if swallowed, causes burns, may cause allergic skin reaction, toxic to aquatic life |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | Harmful if swallowed. Causes serious eye irritation. May cause respiratory irritation. |
| Precautionary statements | P264, P270, P273, P280, P301+P312, P305+P351+P338, P330, P337+P313, P501 |
| NFPA 704 (fire diamond) | 2-1-1 |
| Flash point | > 157 °C (315 °F) |
| Autoignition temperature | > 307 °C (585 °F; 580 K) |
| Lethal dose or concentration | LD50 Oral - rat - 844 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 842 mg/kg |
| NIOSH | MW3675000 |
| PEL (Permissible) | PEL: 1 mg/m3 |
| REL (Recommended) | 1 - Recommended |
| IDLH (Immediate danger) | IDLH: 80 mg/m³ |
| Related compounds | |
| Related compounds |
Hydroxylamine Hydroxylamine hydrochloride Hydroxylamine nitrate Ammonium sulfate |

