Hydroxylamine Nitrate: An In-Depth Look
Historical Development
Few chemicals reveal so much about the intersection of science, industry, and safety as hydroxylamine nitrate. Early research started in the first half of the twentieth century, with scientists exploring its potential in both military and civilian realms. During the 1940s and 1950s, demand for energetic materials nudged researchers into fine-tuning the synthesis and stabilization of this compound. It didn’t take long for it to show up at nuclear and chemical research facilities, where its high reactivity challenged chemists and engineers alike. As the Cold War era fueled advancements in explosives and propellants, hydroxylamine nitrate became a key player. Over the decades, improved safety protocols and deeper understanding of nitrate chemistry have allowed its use to expand cautiously, but recklessness in handling this substance still ends badly. From early mishaps to today’s carefully monitored usage, the learning curve hasn’t been gentle, and history shows there’s no room for shortcuts.
Product Overview
Many folks who don’t work in labs haven’t heard of hydroxylamine nitrate. Those who have, treat it with respect. This compound, usually appearing as a colorless, crystalline solid in its pure form, acts as a strong oxidizer and reducing agent—an unusual combination. Guidance for transportation and storage comes packed with warnings, and regulatory agencies watch it like a hawk. Commercial sources often provide it as an aqueous solution, which slightly lessens the risk of catastrophic decomposition, but doesn’t remove it entirely. Quality depends on purity, water content, and the absence of metal ions that might catalyze breakdown.
Physical & Chemical Properties
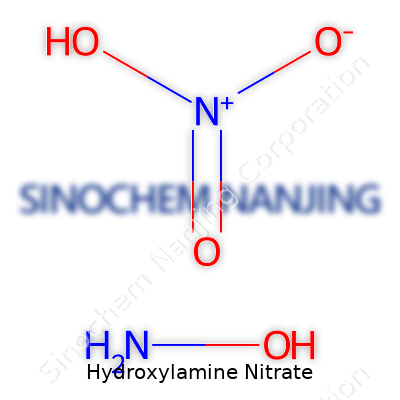
Hydroxylamine nitrate, chemical formula NH2OH·HNO3, stands as a salt yielded from the reaction between hydroxylamine and nitric acid. This compound dissolves easily in water and methanol, yet keeping it dry and warm spells trouble. It breaks down violently above 70°C or when allowed to dry out, ejecting nitrous oxide, nitrogen, and heat. As an oxidizer, it stirs up fast redox reactions and fits right into chemical syntheses needing an energetic boost. Its density ranges from 1.2 to 1.3 g/cm³, and the molar mass clocks in at 96.06 g/mol. A sharp, acidic odor gives a subtle warning, but not enough for someone to risk inhaling this stuff. Electrostatic sensitivity, impact risks, and rapid self-decomposition raise its hazard class beyond what most industrial chemicals carry.
Technical Specifications & Labeling
Regulators call for detailed labeling and hazard identification on hydroxylamine nitrate. Specifications include concentration in solution, precise pH value, and limits on metallic contaminants that may speed up decomposition. The United Nations classifies it as a dangerous good, and shipping documents will list UN number 3379 along with flammable and oxidizer identifiers, marked “toxic by inhalation.” Material safety data sheets do not mince words about potential for runaway reactions or violent detonation. Good suppliers run extra analysis to provide assurances about composition, solubility, and long-term stability, because a slip on these numbers has tragic consequences. In practice, users document every transfer, every storage vessel, and every exposure condition, since this product’s track record demands accountability.
Preparation Method
Most commercial-grade hydroxylamine nitrate comes from reacting hydroxylamine sulfate or hydrochloride with barium nitrate or sodium nitrate. Chemists mix the reactants in cold water, precipitating out by-products like barium sulfate and then filtering everything under a blanket of inert nitrogen to guard against accidental ignition. The process scales up under rigorous control, because the reaction rates accelerate at higher concentrations or temperatures, sometimes producing heat that catalyzes further breakdown or explosion. The final clear solution is tested several times before it ever leaves the lab, and heavy reliance on cooling, stirring, and anti-static protocols speaks volumes about the tightrope chemists walk to make it safely. Attempts to shortcut final purifying steps, or slip in recycled nitrate, resulted in more than one infamous lab incident.
Chemical Reactions & Modifications
Chemists admire hydroxylamine nitrate’s reactivity on both oxidative and reductive sides. They use it with nitrosating agents, hydrazines, and other energetic compounds to generate new materials for rocket propellants and explosives. Laboratory workers often mix it with other salts to alter thermal stability, seeking to reduce its unpredictable breakdown. Some efforts go into stabilizing solutions with antioxidants or sequestering agents, but not every attempt pays off. In nuclear fuel reprocessing, it takes part in redox reactions that separate uranium and plutonium, a practice dating back to the dawn of atomic technology. Its nitrosylation reactions show up in pharmaceutical syntheses, where managing hazards matters just as much as the chemistry. Recent papers describe attempts to modify the molecule’s structure, but any change that lowers sensitivity or increases shelf-life faces uphill battles with fundamental chemistry.
Synonyms & Product Names
Hydroxylamine nitrate hides beneath several names in technical documents and shipping manifests: it appears as “HAN,” “hydroxylammonium nitrate,” and “nitric acid, compound with hydroxylamine (1:1).” Some procurement lists use “NH2OH·HNO3.” In military and space programs, it turns up under code names or proprietary blends, especially as formulations for monopropellants or mixed oxidizers. Old safety manuals from decades past may mention “HAN solution” in the context of accelerator or propellant development. For a material with less pop culture notoriety, it moves in influential scientific circles.
Safety & Operational Standards
Anyone handling hydroxylamine nitrate learns quickly: even small mistakes carry massive implications. Direct contact causes burns, blisters, and chronic dermatitis, while inhalation irritates lungs and may trigger severe allergic responses. Storage calls for cool, well-ventilated, explosion-rated facilities, plus clear segregation from organic solvents and metals. Stringent regulations outlaw bulk or domestic storage outside licensed operations, and users keep full records by law. Specialized containers, sometimes lined with polymers, limit contact with the structural materials that can destabilize the compound. Staff receive thorough hazardous materials training, and emergency response drills prepare teams for worst-case scenarios. Lessons from past disasters, such as the 1999 explosion at Concept Sciences in Pennsylvania, led to tighter local and federal oversight, highlighting the need for vigilance at every stage.
Application Area
Despite the dangers, hydroxylamine nitrate continues to find uses in the chemical, aerospace, and nuclear sectors. Major application sits in “green” monopropellants for satellite thrusters, where HAN solutions power advanced engines without the extreme toxicity of hydrazine. In chemical synthesis, it acts as a selective reducing agent for precious metal catalysis and a precursor in specialty explosives. Nuclear reprocessing uses it to control oxidation state transitions during actinide separation. Scientists working in pharmaceutical research employ the compound’s nitrosating abilities for complex synthetic steps, but always under layers of containment. In practice, only hardened, well-equipped labs with robust training can safely exploit its reactivity, which means HAN’s promise often bumps against regulatory hurdles and operational risk. Still, where performance outweighs risk, demand endures.
Research & Development
Research into hydroxylamine nitrate pushes two fronts: improved chemical handling and new energetic applications. Materials scientists experiment with solution stabilizers, chelating agents, and novel co-solvents to lower the risk without sacrificing performance. Engineers design safer storage systems, with better sensors for heat or pressure spikes inside containers. The advent of electric spacecraft has breathed new life into HAN research, as engineers seek to replace toxic hydrazines with more sustainable, less regulated monopropellants. Universities pursue greener manufacturing techniques, reducing dependence on rare earths for catalyst preparation. Papers continue to surface on modifying the nitrate’s thermal properties, tuning the release of gases in controlled explosions or propulsive reactions.
Toxicity Research
A single exposure accident involving hydroxylamine nitrate teaches lessons on the spot. Studies in rodents and cell culture show that ingestion or inhalation damages tissue rapidly, causes swelling of internal organs, and disrupts oxygen transport in blood. Chronic exposure, rare but documented in poorly ventilated facilities, ends in neurological symptoms and lasting respiratory trouble. Early research underestimated risks, as early labs lacked sufficient protections. Modern toxicology confirms the need for airtight respiratory protection and regular air monitoring. The compound’s notorious instability means accidents release a stew of oxides and acids, which calls for specialized decontamination procedures. Epidemiological reviews after large-scale incidents have reinforced strict exposure limits in industrial hygiene practices.
Future Prospects
The trajectory for hydroxylamine nitrate looks tied to broader shifts in industry and technology. Commercial satellites and deep-space missions demand propellants with better energy density and safer handling, boosting interest in HAN blends. Stricter rules on hazardous materials push industry towards better safety engineering, both in containment and in operator training. Academic labs, working with public and private partners, hunt for modifications that lower risk without killing performance, but progress moves in small steps, not leaps. Environmental concerns over the by-products of nitrate chemistry hint at continuing changes in waste treatment and remediation standards. Even as new competitors in energetic materials research show up, there’s no simple replacement in sight, and the long history of this compound serves as both warning and inspiration for chemists hoping to do things better the next time around.
What is Hydroxylamine Nitrate used for?
A Quiet Cornerstone in Chemistry
Hydroxylamine nitrate doesn’t get headlines, but its impact stretches across a handful of industries. I remember my first internship at a chemical plant; the engineers always paid close attention to this compound when prepping material for further reactions. In labs and factories, folks rely on its unique blend of stability and reactivity. Its official description marks it as a white, water-soluble solid—nothing flashy at a glance, but its punch comes from its chemical structure.
Explosives: Risk, Control, and Reality
The biggest claim to fame for hydroxylamine nitrate comes from its use in making explosives. That may sound dramatic, but it’s not movie-style bomb making. This compound helps with the controlled production of energetic materials. Military and mining applications see it as a building block, helping transform other chemicals into finished products that can produce energy on demand. My uncle, who worked at a mining facility, told stories about the strict safety checks in place—for good reason, since even small mishandling could cause accidents.
The underlying risk highlights why professionals receive special training to handle this substance. The balance of safety and danger is a daily reality. Records from the United States Occupational Safety and Health Administration show that strict ventilation, temperature control, and protective gear remain non-negotiable. Incidents involving hydroxylamine nitrate, though rare, have pushed companies to overhaul their safety policies from the ground up. I’ve seen coworkers go through extra drills just for shipments containing this material.
Rocket Propulsion: Powering Dreams and Satellites
Satellites you check every day for weather updates owe a debt to propellants based on hydroxylamine nitrate. Scientists at NASA and private companies use it as an ingredient in monopropellant mixes, valued for its high energy and clean exhaust. Labs have demonstrated that these propellants offer higher performance and fewer emissions than traditional hydrazine-based fuels. Engineers want both reliability and reduced environmental impact, something this compound delivers. In my own research days, the buzz was always around finding ways to tame its instability for greater use in orbital launches.
Industrial and Laboratory Uses
Beyond explosives and space gadgets, hydroxylamine nitrate finds work in research labs and manufacturing. It acts as a reducing agent. Chemists put it to use in synthesis, purifying metal ores, and even some pharmaceutical steps. I still remember my awkward student days, carefully measuring the compound while a professor watched like a hawk—not out of distrust, but out of honest concern for the volatility. Mistakes in weighing or mixing could spell disaster, and everyone in the room knew it.
Challenges and Safer Alternatives
This compound comes with a tough reputation. Accidents over the past several decades, like the 1999 plant explosion in North Carolina, ended in tragedy. Regulators and manufacturers pivoted fast, investing in better training and storage protocols. Chemical engineers today work overtime testing new stabilizers and containment strategies. Some companies now turn to alternative chemicals with lower risk profiles, but trade-offs with effectiveness and cost persist. Even with alternatives on the table, industries keep a close eye on ongoing research for safer blends and stronger protocols.
A Call for Better Safety Culture
There’s no escaping the hazards. Proper education, modern equipment, and transparency with workers keep everyone safe. My own experience taught me that knowledge and a healthy dose of caution matter more than bravado. The wider public might never hear the name hydroxylamine nitrate, but its legacy—good or bad—sticks around in every process it touches.
Is Hydroxylamine Nitrate hazardous or explosive?
Handling Chemicals Carries Real Risk
Working with chemicals, especially ones with names most folks haven’t heard of, brings a certain amount of caution. Hydroxylamine nitrate fits that bill. In my time around labs and plant floors, few substances get both chemists and safety managers to perk up as much as this one. Plenty of data backs up their concern.
The History and Uses
Hydroxylamine nitrate isn’t something you find on grannies’ pantry shelves. It comes up mainly in specialized industries like rocket propellant research, explosives, and some types of chemical synthesis. During the Cold War, scientists in the US and Soviet Union both experimented with it to push boundaries of fuel performance. Its properties made it attractive in certain formulas, even if its stability ranked somewhere between “questionable” and “risky.”
The Hazards Hit Close to Home
This stuff can fool even trained eyes. The nitrate part brings oxygen to the table, basically feeding flames in the right (or wrong) situation. The hydroxylamine piece? That adds a dash of unpredictability. Together, they make a chemical that behaves differently from the ingredients alone. Several times over past decades, facilities suffered fires or blasts after a lapse in protocol, sometimes fatal. OSHA and the Chemical Safety Board dug into those incidents, warning industry and academia alike.
In warmer conditions, or if mixed improperly (too concentrated, contamination, left sitting too long), hydroxylamine nitrate can decompose rapidly. That means energy escapes as heat, pressure climbs, and suddenly a workspace turns into a bomb. Even something like friction or a spark can set off a chain reaction. No one in chemical safety wants a repeat of what happened at Napp Technologies in 1995, where a similar chemical led to an explosion and multiple tragic deaths.
Beyond the Explosion Fear
People often focus on the drama of explosions, but danger doesn’t stop there. Hydroxylamine nitrate gives off toxic fumes if it burns—stuff like nitrogen oxides or ammonia. These gases harm lungs, eyes, and can linger in closed environments. Splash it on the skin, and you risk chemical burns. Breathe in dust? It irritates the respiratory tract and leaves some workers with coughing fits they remember for days. So exposure isn’t just a matter of catastrophic events. Chronic mishandling can slowly chip away at health, too.
Tried and Tested Precautions
Experience teaches old chemists to double-check every label and work slowly with reactive compounds. Protective gear isn’t negotiable—gloves, goggles, and face shields stand as standard. Mixing or transferring needs to happen in small amounts, away from ignition sources or metal, with grounded equipment. Emergency showers and eyewash stations stand close by. Training doesn’t just cover what to do; it stresses why those rules matter, drawing on real-world cases.
Facilities with this chemical use continuous monitoring to catch temperature or pressure rises early. They keep detailed logs and have evacuation plans tailored to the actual risks. Regulatory oversight—like EPA’s Risk Management Plan rules—ensure companies don’t cut corners. Disposal also commands attention, calling for neutralization and specialist handling. Regulators issue clear rules, but industry must bring the discipline.
Always Respect the Power
Whatever the chemistry textbook claims, nature doesn’t cut anyone slack. Hydroxylamine nitrate earns its reputation from both lab research and hard lessons on the job. Whether you’re an old hand or just starting your career, understanding what goes wrong keeps everyone safer. A chemical only seems boring until it’s mishandled—and then it’s too late.
What are the storage requirements for Hydroxylamine Nitrate?
Risks Around the Corner
Hydroxylamine nitrate brings a special set of challenges. It’s not one of those chemicals found on every industrial shelf, and some shops treat it with more caution than they do for stronger acids. It reacts easily and can start decomposing with just a little heat or the wrong kind of touch. Add a bit of metal, stray sparks, or loose copper to the equation, and its risks increase. Thinking about what happened in past chemical mishaps — including plant accidents traced back to poor storage — shows how easy it is to underestimate its hazard under ordinary conditions.
Temperature Isn’t Just a Number
Hydroxylamine nitrate likes to break down above room temperature. Around 30°C (86°F), the pace picks up. By 50°C (122°F), a real disaster is possible. I've worked in facilities where summer meant careful checks on warehouse AC units, making sure they actually cool the chemical rooms instead of just humming along. One night, a sensor failed, and somebody rushed to fix a stuck relay. Without it, I’m sure we’d’ve been reading about ourselves in the morning paper. Anyone managing this compound must think hard about temperature controls, dependable monitoring, and plain, cold redundancy.
Container Choices Make or Break Safety
Some people think any sturdy drum works for everything. Not so with hydroxylamine nitrate. Steel sets off decomposition. Even copper traces can act as triggers. In my own shifts, plastic tanks have prevented trouble more than once. Polyethylene, for instance, resists interaction. Glass does the job too, but plastic stands up to the knocks in daily handling. Manufacturers ship it with care for this reason, and good operators never transfer it into “just any” recycled drum. One forgotten screw cap, replaced with a slightly corroded metal one, often leads to slow trouble: pressure builds up over weeks, and then, one Monday morning, the warehouse manager finds a bulging lid or worse.
Rule of Dilution and Cleanliness
Hydroxylamine nitrate works best stored as a solution. Water slows down breakdown, and the risks decrease at concentrations below 40%. Still, leaving crystals behind or letting evaporation go unchecked in storage leaves everyone exposed. From my own rounds, a false “empty” barrel hiding damp residue still carries risks. We trained staff not just to follow the sheet on “proper disposal” but also to do physical checks; corners cut here have turned cleanup jobs into news stories at several plants in the last decade.
Vent, Label, and Think Ahead
Every container needs a working vent. Pressure sneaks up as the substance decomposes, and vessels rupture more quietly than people expect. Labels must tell the real story. I’ve seen reused containers with old stickers that confused the incoming crew. The risk isn’t just paperwork — it’s someone bringing the wrong tool, wearing no respiratory protection, or stacking barrels where they shouldn’t. Safety drills feel tedious until a real-world test proves their worth. But anyone skipping the basics is rolling dice with health and with the law.
Simple Steps Prevent Tragedy
Routine checks. Regular staff training. Rigid separation of incompatible chemicals. Anyone charged with holding onto hydroxylamine nitrate owes this level of care to their community — not because regulators demand it, but because lessons from history demand it. By keeping processes strict, storing only in the right materials, and respecting what this compound is capable of, accidents become less likely. These aren’t just lab rules; they’re rules written in the memory of every plant that learned the hard way.
How should Hydroxylamine Nitrate be handled safely?
Understanding the Material
Hydroxylamine nitrate doesn't get much attention outside of labs or industrial settings, but its hazards are well known to anyone with hands-on chemistry experience. This compound holds a unique spot; with strong oxidizing properties and sensitivity to heat, shock, and contamination, it’s not something you pop on a shelf and forget about. Just remembering the infamous explosion at Knapsacker Hülsenfabrik in 1991, where hydroxylamine nitrate played a role, serves as a gut-check for how these supplies demand respect and attention.
Risk Factors and Realities
In real work environments, I’ve seen that small oversights become big problems. Hydroxylamine nitrate reacts with a range of organic and inorganic materials. Even tiny bits of grease, metal filings, or stray dust can trigger violent behavior. While gloves and goggles are a given, those alone don’t address the critical issues. Static discharge can spark an incident, so simple steps—grounding containers, making sure tools are clean and non-sparking—do a lot of heavy lifting.
Environmental Controls Matter
Temperature and humidity controls aren't just boxes to check. Hydroxylamine nitrate decomposes if it overheats or dries out, which brings risks nobody wants to face. A well-ventilated area with climate control stops vapor buildup and reduces decomposition speed. Maintaining concentration below critical limits also helps manage risk. From my view, regular monitoring with reliable sensors and good old-fashioned checks beat fancy automation. One missed alert can turn into an emergency in no time.
Education: The First Line of Defense
Training counts for everything. You want people running these operations to know the material, not just by the numbers from a textbook, but from hands-on drills and clear communication. I always preferred straightforward training programs, where workers could ask questions and walk through scenarios—for example, what to do if a container tips over, or a fire starts nearby. Teams need to feel comfortable stopping production if something seems off, and that comes from real trust and practice, not laminated checklists posted on the wall.
Emergency Planning and Response
You can't predict every hazard, but you can prepare for common trouble. Spill kits should be specific—using only compatible absorbents, carefully marked for each chemical. Fire extinguishers have to match the risk; regular dry chemical extinguishers don’t help much for energetic nitrates. Plans must cover evacuation routes, emergency communication, and how to help first responders get in safely. In real experience, clear labeling and humility in asking for backup make a big difference.
Storage and Container Choices
For storage, I always looked for dedicated areas with controlled access, no sunlight, and no heat sources. Sturdy packaging—often high-density polyethylene or similar materials—keeps reactions in check. Segregation from acids, fuel, paper, and flammable solvents keeps risk down. Staff should check containers for bulging, leaks, or rust during every shift, and report anything unusual, not keep quiet to avoid hassle.
Staying Accountable
Regulation drives much of this—OSHA, EPA, and local fire codes all have strong recommendations, and for good reason. In my experience, the best-managed sites treat those rules as starting points and always do more. Communication between management and staff goes a lot further than blind trust in paperwork. Transparency around reported incidents and follow-up steps help teams feel responsible for each other’s safety.
Solutions: Respect, Simplicity, and Ongoing Vigilance
Handling hydroxylamine nitrate without accidents boils down to constant vigilance, not overcomplicating safety systems, and keeping communication open. Regular retraining, honest reporting, and a clear policy for addressing mistakes build a safety culture everyone buys into. It isn’t about fear—it’s about respect for substances that can change lives in a single moment.
What is the chemical formula of Hydroxylamine Nitrate?
The Value of Clear Chemical Knowledge
Standing in a school laboratory for the first time, I remember staring at a shelf cluttered with bottles. Each one held a different substance, and each label seemed like a secret code. That’s where a chemical formula comes in. It strips away confusion, slicing straight to the heart of what a compound truly is. Hydroxylamine nitrate has a formula that packs power and risk: NH2OH·HNO3. This combination sits at the intersection of chemistry’s usefulness and its dangers.
The Pieces That Make Up Hydroxylamine Nitrate
Pulling apart the formula NH2OH·HNO3, you find two building blocks. One is hydroxylamine (NH2OH), a reducing agent with a knack for giving up electrons, and the other is nitric acid (HNO3), an aggressive acid and an oxidizing force. Bring them together, and you get an energetic salt used for propellants and explosives. Picking up a chemistry textbook, I saw plenty of reminders about the risks attached. Hydroxylamine nitrate opens a door to both practical uses and real hazards.
Safety Lessons From Hard Accidents
In 2001, a chemical plant in the U.S. South faced a disaster. Hydroxylamine nitrate played a starring role, showing how a simple formula can pack an unpredictable punch. Heat, pressure, or physical shock can send this compound past its breaking point. Even small misjudgments, like warming it up too much or storing it the wrong way, can turn a busy workplace into a scene of chaos. Chemical safety reports underline this: hydroxylamine nitrate needs strict guidelines, skilled hands, and the right environment every time.
Uses on the Frontier—and the Tightrope Walk
Energy industries and defense sectors put this material on center stage. Because NH2OH·HNO3 breaks down fast, it finds work as rocket fuel or in specialty explosives. Balancing performance with risk sounds like a tall order, but modern engineering meets that challenge every day. Synthetic chemists with years behind them lean on lab coats, goggles, and even robot arms to keep accidents at bay. The internet offers a flood of data sheets and government-backed safety standards that hold everyone accountable.
The Reality of Responsible Handling
Saying “handle with care” takes on weight when explosive hazards are real. A chemical formula only scratches the surface. Behind those letters and numbers, there's a long chain of training and preparation. A person who works with hydroxylamine nitrate learns to trust their team, double-check procedures, and plan for the worst. Regular drills, clear packaging, and constant environmental monitoring all become part of daily life. Trusted groups like the Occupational Safety and Health Administration post updates and guides to update practices with fresh research.
Moving Toward a Safer Future
Manufacturers and regulators keep refining storage and handling practices. New safety features—pressure relief valves, automated monitoring, better chemical stabilizers—are showing up across the industry. Researchers look for substitutes that bring the energy of NH2OH·HNO3 without the same risk. Open communication between chemists, regulators, and emergency responders keeps the next generation of workers safer and better informed. In the world of chemical safety, every clear formula tells its own story of potential and responsibility.
| Names | |
| Preferred IUPAC name | hydroxylammonium nitrate |
| Other names |
HAN Hydroxylammonium nitrate Nitric acid, hydroxyammonium salt |
| Pronunciation | /haɪˌdrɒk.sɪlˈæm.iːn ˈneɪ.treɪt/ |
| Identifiers | |
| CAS Number | 13465-08-2 |
| Beilstein Reference | 1460118 |
| ChEBI | CHEBI:75189 |
| ChEMBL | CHEMBL1233476 |
| ChemSpider | 16314 |
| DrugBank | DB11445 |
| ECHA InfoCard | 100.029.139 |
| EC Number | 236-813-4 |
| Gmelin Reference | 133152 |
| KEGG | C00508 |
| MeSH | D006929 |
| PubChem CID | 159190 |
| RTECS number | NL1050000 |
| UNII | W16X8T7T3E |
| UN number | UN1515 |
| Properties | |
| Chemical formula | NH3NO3 |
| Molar mass | 64.021 g/mol |
| Appearance | White crystalline solid |
| Odor | odorless |
| Density | 1.84 g/cm³ |
| Solubility in water | Very soluble |
| log P | -4.50 |
| Vapor pressure | 0.04 mmHg (25 °C) |
| Acidity (pKa) | 4.71 |
| Basicity (pKb) | 8.02 |
| Magnetic susceptibility (χ) | −7.6×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.385 |
| Viscosity | 3.46 cP (20°C) |
| Dipole moment | 5.95 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 143.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -146.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -311 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V03AB35 |
| Hazards | |
| Main hazards | Oxidizer, explosive, toxic, corrosive, harmful if inhaled or swallowed. |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS01,GHS03,GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H272, H302, H314, H317, H332, H335, H341, H373, H410 |
| Precautionary statements | P210, P220, P221, P260, P280, P305+P351+P338, P308+P313, P370+P378, P501 |
| NFPA 704 (fire diamond) | 3-1-1-OX |
| Flash point | 85 °C |
| Autoignition temperature | 220 °C (428 °F) |
| Explosive limits | Lower: 2.4% Upper: 99% |
| Lethal dose or concentration | LD50 oral rat 72 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 71 mg/kg |
| NIOSH | PC9625000 |
| PEL (Permissible) | PEL: Not established |
| IDLH (Immediate danger) | 300 mg/m3 |
| Related compounds | |
| Related compounds |
Ammonium nitrate Hydroxylamine Hydroxylammonium sulfate Nitric acid |

