Looking Into Hydroxyethyliminodiacetic Acid: Importance, Progress, and Practical Impact
Historical Development
Chemistry moves in leaps and bounds, sometimes because a small change in a molecule leads to bigger changes in industry. Hydroxyethyliminodiacetic acid (HEIDA) popped up on the scientific radar as long ago as the mid-twentieth century, when industries started pushing for gentler, greener alternatives to traditional chelating agents. Back then, folks worked with harsh chemicals like EDTA that stuck around in the environment. Scientists spent years in labs tweaking amine and acetic acid derivatives, working through long nights to chase molecules less stubborn but just as powerful. Exploring the family tree of aminopolycarboxylic acids, researchers zeroed in on HEIDA, betting on it for smoother metal ion control. Publications began appearing by the 1960s, detailing an easier time stripping troublesome ions out of water or chemical processes. This historical push for sustainability and control shines through every time someone brings up HEIDA in a lab or factory.
Product Overview
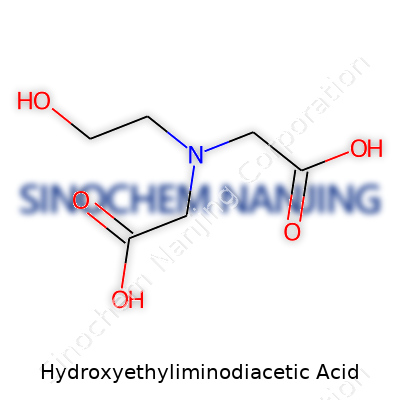
Diving into what makes HEIDA useful, you quickly find a compound tailor-made for chelation. Built from an ethylene backbone with two acetate arms and an extra hydroxyethyl group, it stands out for doing the same heavy metal clean-up as better-known EDTA, but with a gentler approach. The product usually lands on shelves as a white to off-white solid, sometimes in a granular powder. Its appeal stems from an ability to bind troublemakers like calcium, magnesium, copper, and iron. Once in hand, operators use HEIDA to prevent scale, boost detergent formulas, and soften water. There's a growing buzz with manufacturers eyeing its milder toxicity and lower persistence in the natural world. In factories and plants, decision-makers like having a chelator that backs up green chemistry commitments. For anyone who has handled barrels of the old classics, HEIDA means better air and water at the end of the shift.
Physical & Chemical Properties
HEIDA doesn’t make a secret of itself, from its white solid look to its straightforward chemical behavior. The compound carries a molecular formula of C6H11NO5, usually weighing in at around 177 grams per mole. With a melting range above 200°C (often decomposing instead of melting cleanly), it is robust under most storage conditions. While not especially soluble in plain water, it quickly forms water-soluble salts alongside sodium or potassium cations. Its acid groups (pKa values sit between 2 and 10) give scientists a wide pH range for applications. As a chelator, HEIDA stands up against mineral acids but keeps its stability with most common organic chemicals. At work, it takes on the heavy jobs without breaking down or forming hazardous byproducts.
Technical Specifications & Labeling
Factories churning out batches of HEIDA need a clear set of rules to keep industries on track and regulators satisfied. Most technical documents push for purity levels of 99% or higher, with strict constraints on moisture content, traces of metals, and organic impurities. Labels on sacks or drums list batch numbers, net weight, chemical composition, and storage instructions. Regulations demand hazard statements for skin and eye irritation risks, plus guidance on safe disposal. Good labeling keeps everyone from warehouse handlers to chemists in the loop, making slip-ups in dosing or mixing less likely.
Preparation Method
In labs and plants, making HEIDA relies on smart chemistry rather than brute force. The recipe usually starts with glycine and formaldehyde in a controlled reaction, then blends in monoethanolamine or monochloroacetic acid. Temperatures and pH swing back and forth as the mixture gets stirred for hours or even days, leading to a rich soup of intermediates. Purification follows, typically through recrystallization or precipitation, until the product reaches the purity levels set by regulators or buyers. Lab veterans know that tweaking temperatures and feed rates can mean the difference between a clean, efficient batch and an expensive do-over.
Chemical Reactions & Modifications
HEIDA steps into reaction vessels ready for action, not content to just float along. Its backbone hosts multiple carboxyl and amine groups, creating hotspots for further chemical creativity. In research or custom synthesis, chemists attach extra functional groups at those sites to tune solubility, binding power, or reactivity. Many applications call for the sodium or potassium salt—transforming the acid into a more user-friendly, water-dissolving powder. Other times, folks graft HEIDA onto polymers to create slow-release agents or filter materials for water treatment. The flexibility in modification gives manufacturers a way to hit more specific targets, like harder-to-chelate metals or custom applications in electronics.
Synonyms & Product Names
This compound doesn’t always show up on paperwork as hydroxyethyliminodiacetic acid. Trade names are easy to spot, but technical folk just as often refer to it as HEIDA, HIDA, or N-(2-Hydroxyethyl)iminodiacetic acid. Some suppliers market it as a chelation enhancer or heavy-metal cleaner, but the real chemistry folk stick to the IUPAC name. For researchers, knowing these aliases avoids confusion, especially with supply chain complexity across regions and regulatory environments.
Safety & Operational Standards
Every chemical requires respect, and HEIDA holds its own safety playbook. Folks working with bags of the substance follow best practices: gloves, goggles, and dust masks keep minor irritations or allergies at bay. Well-ventilated rooms matter, even though the compound lacks a strong smell or dangerous fumes under normal lab conditions. Plants adopt spill clean-up procedures, eye-wash stations, and fast-acting containment plans. Long-term storage works fine in sealed containers away from high humidity and strong oxidizers. Local and international standards often echo similar guidance, with special attention to worker health, packaging, and shipping.
Application Area
HEIDA’s reach grows with environmental demands and changes in water management rules. Major utilities use it for water softening, stripping out hard water ions before they clog pipes. Industrial plants lean on it as a scale inhibitor and anti-precipitant, keeping machines running longer and cutting sulfuric acid use. The detergent sector turns to HEIDA for laundry additives that help soap work better even with poor-quality water, reducing soap scum and residue. In pharmaceuticals, its role in stabilizing formulations brings consistency to therapeutic products. Metal finishing and cleaning operations value it for rust removal, surface prep, and extending the life of expensive equipment.
Research & Development
University labs and R&D departments keep finding new roles for HEIDA, targeting both greener chemistry and better process control. Teams investigate ways to blend HEIDA into recyclable polymers or water filtration membranes for trace-metal cleanup. Projects chase alternatives to legacy chelators in agriculture, aiming for healthier crops and less environmental run-off. Ongoing research looks at the nuances of binding kinetics, molecular modeling, and compatibility with other additives. Integrating artificial intelligence and automated reaction monitoring speeds up breakthroughs, which means faster product cycles and safer chemical tools in the field.
Toxicity Research
Data from animal studies show HEIDA carries lower acute toxicity compared with traditional workhorses like EDTA, both for humans and aquatic life. Environmental scientists put in hours tracking degradation pathways—finding that microbial action tends to break HEIDA down faster after disposal. Reports from occupational health teams point out minor irritant risks to skin and eyes, while long-term exposure appears to carry less bioaccumulation risk. Regulatory bodies keep updating exposure limits as new research comes out, but so far, HEIDA fits into the trend for safer, less persistent chelating agents.
Future Prospects
Next-generation chemistry often leans on the building blocks that work today, modified or adapted for new rules. With HEIDA, there’s a steady march toward even lower toxicity and higher biodegradability, especially in places where drinking water and aquifers face pollution from heavy metals. Markets look strong in Asia and Europe as industries pivot from older chelators. Green technology pushes more research into dual-function materials, such as catalysts that both bind metals and self-clean. If environmental setbacks push for tighter restrictions on persistent organic pollutants, demand for HEIDA could climb higher. Lab teams already test its limits in pilot projects for desalination, mining, and advanced manufacturing. Staff working in the field feel the change—cleaner work spaces, less harm to local environments, and broader support from regulators all mark a shift driven by this unassuming molecule.
What is Hydroxyethyliminodiacetic Acid used for?
What Hydroxyethyliminodiacetic Acid Actually Does
Ask any chemist about the tools for binding metals, and this chemical, usually called HEIDA, will show up in the conversation. I’ve seen it most often in labs that handle water treatment projects or those testing soil for heavy metals. HEIDA works as a chelating agent. In plain language: it grabs onto metal ions, stopping them from running amok. This quality gives it a surprisingly broad set of uses, some of which might not be immediately obvious.
How It Helps With Water and the Environment
Now and then, municipal water plants hit trouble keeping metals like iron and copper out of tap water. High levels aren’t just a plumbing nuisance; they can affect people’s health. Compounds like HEIDA help take these metals out of water before the water reaches anyone’s faucet. Compared to some classic chelators, HEIDA can grab hold of certain metals even when the water pH creeps a bit higher or lower than expected. I’ve seen HEIDA work in this setting to help utilities avoid pipes getting clogged or corroded. Less pipe damage means fewer costly fixes for cities and a safer supply for homes.
Applications in Laboratories and Medicine
Any setting where metal ions mess with an experiment becomes a test field for chelating agents. Researchers often rely on HEIDA in samples where controlling every variable matters, because stray metals can skew results. Some diagnostic kits use chemicals like HEIDA to trap unwanted metal ions that would otherwise mess up accuracy. This accuracy directly influences patient diagnoses and treatment plans, so the stakes run high.
Relevance to Agriculture and Industry
Agricultural soil doesn’t tell its own story without some help. To detect toxic metals, labs add a chelator like HEIDA to soil samples. The agent grabs onto metals, making them easier to measure. Knowing what’s stuck in the dirt informs decisions about fertilizer, crop choice, or environmental cleanup. In industry, HEIDA sees a place in textile processing and paper manufacturing, where traces of metals in water can mean weaker fabric or discolored paper. Textile engineers tell me that having the right chelator keeps product quality up and wastage lower, a win both for profits and sustainability.
Environmental and Human Safety
Every new chemical in town should face scrutiny before routine use. The good news is that HEIDA doesn’t rank high on toxicity charts. People handling it in labs or factories still take precautions. Gloves go on, spills get contained right away, and nobody lets it touch their eyes or mouth. Used properly, the benefits for pollution control and water safety outweigh most hazards.
How We Could Improve Use and Oversight
As with any chemical, the big picture needs more information sharing. Industries using HEIDA should track how much gets into wastewater, not just to meet regulation, but to keep the surrounding environment healthy in the long run. Partnerships between research institutions and manufacturers can help create cleaner processes. It’s easy to see a safer route forward: better monitoring, stronger worker education, and investment in greener alternatives for any tasks where old habits still cause unnecessary pollution. Community buy-in matters. When neighbors, industries, and scientists talk openly about what goes into water and soil, everyone wins—whether it’s a lab tech or a family drinking water at dinner.
What are the chemical properties of Hydroxyethyliminodiacetic Acid?
A Look at the Molecule
Hydroxyethyliminodiacetic acid, known in some labs simply as HEIDA, catches a chemist’s eye for its full spectrum of functionalities. There’s a hydroxyethyl arm, two acetic acid groups, and a central amine. This structure means the molecule rocks both acidic and basic sites. In practice, the carboxylic acid groups give strong acidity, typically showing pKa values around 2 and 9. The hydroxyethyl on the nitrogen atom brings extra water-love (hydrophilicity) and a bit of extra bite for ion exchange. You get a molecule that mixes easily in water, forms clear solutions, and hangs onto metal ions with serious grip.
Why Chelation Changes the Game
This acid acts like an octopus for copper, nickel, iron, and other metals. Chelation happens when more than one part of the organic molecule locks onto a single metal ion. In this case, two arms from the acetic acids and the nitrogen team up to grab atoms, forming stable, water-soluble complexes. This knack for chelating comes from the geometry — the amine and carboxylate groups fall in just the right spots. It means HEIDA binds transition metals even at relatively low concentrations. At a wastewater site, for example, dumping in HEIDA keeps heavy metals from falling out as sludge. They stay in check, dissolved, and ready for later removal.
Resistance, Stability, and Limits
HEIDA doesn’t just dissolve and disappear. It stands up to light, swirling oxygen, and most neutral to mildly acidic conditions, so the stuff doesn’t break down sitting on your shelf or in your beaker. Heat it too much or go into extremely alkaline territory, and the functional groups can start to hydrolyze. That’s less of a concern in daily industrial use. What really matters is that up to pH 12, HEIDA keeps structure and holds onto metals. But you can’t toss it with strong oxidizers. Bleach, for example, will chew through its hydroxy and nitrogen sites and make a mess of breakdown products.
Why This Matters Outside the Lab
The chemical story of HEIDA brings real-world impact, especially for industries chasing cleaner water. In my years following municipal water treatment trends, relying on simple precipitation can leave trace metals in the final effluent. Chelating agents like HEIDA pick up the slack and reduce the leftover metals that regulations target. Farmers mixing micronutrient fertilizers also get help, as HEIDA-bound metals don’t lock up in soil but stay available for crops. There’s a growing call to balance effectiveness and environmental persistence. HEIDA’s biodegradability lags behind common amino acids, sparking research into greener alternatives and more complete treatment after use.
Possible Steps Forward
Chemical engineers, regulators, and environmentalists all have a stake here. Reusing chelating agents, refining destruction methods, and designing analogs with faster breakdown give pathways forward. Advanced oxidation — using UV or ozone — shows promise for breaking down spent HEIDA complexes and cutting environmental load. Innovations like these demand solid testing, honest risk evaluation, and open communication with the public. Reliable water, cleaner crops, and responsible waste management depend on getting the chemistry right — not simply knowing a molecule’s formula but understanding what it really does out in the wild.
Is Hydroxyethyliminodiacetic Acid safe to handle?
What I’ve Learned About Handling This Chemical
Working in a laboratory environment, you get to know the dos and don’ts of different chemicals. Hydroxyethyliminodiacetic acid isn’t a household name, but plenty of folks in water treatment, chemical processing, and research labs handle it. You crack the container open and you’re not facing the nastiest stuff on the shelf, but you still can’t treat it like sugar or table salt. My experience has taught me that familiarity with a chemical’s risks shapes how people approach safety—sometimes for the better, sometimes not.
Health Risks Deserve Respect
Contact with hydroxyethyliminodiacetic acid usually brings up eye or skin irritation. You splash a bit onto your hands or breathe in a dust cloud during weighing and you might not even notice right away, but hours later those red patches or a sore throat make for a lousy evening. According to safety data sheets, short-term effects rarely turn serious, though the discomfort can’t be ignored. Some studies point out that repeated skin contact causes dermatitis, and handling the powder without gloves has left plenty of colleagues I know with chapped hands.
Inhaling particles poses a greater problem. Standard research suggests consistent exposure, without masks or ventilation, can turn a nuisance into a respiratory issue. Chemical safety agencies rank this acid lower on toxicity than many lab regulars, but ignoring basic rules increases risk. Nobody wants to discover the hard way that lung irritation makes breathing tough or ruins a project with lost workdays.
Learning from Laboratories and Factories
Protective gear and good habits stick with people for a reason. In every place I worked, the labs that had eye wash stations stocked and fresh nitrile gloves always on hand dodged most troubles. Emergency training sometimes gets ignored, but seeing what happens when it pays off is enough to convince me—people remember the coworker who rushed to safety after a spill and avoided a chemical burn.
The acid itself won’t catch fire, but it doesn’t belong near strong oxidizers or metals. A surprising number of accidents come from mixing leftovers, so paying attention to what goes down the drain really matters. Outside the lab, factories move drums of the stuff. Spills on the loading dock bring different challenges, and chemicals like this can eventually find their way into groundwater without sharp monitoring.
Finding Sensible Solutions
Training stays at the top of my list. Walk into any site handling hydroxyethyliminodiacetic acid: the safest teams keep up with training and know the symptoms of exposure before something escalates. Material safety data sheets cover the basics, but regular drills make the difference between theory and calm, effective action during a spill or leak.
Personal protective equipment talks tend to sound repetitive. Still, cheap gloves and old goggles wear thin quickly. Investing in solid gear pays off, especially for frequent handling. Companies setting up fume hoods and ventilation do more than follow the rules—they keep their staff healthy and avoid downtime. Clear labeling lowers the odds of anyone mixing up containers or skipping a step. Finally, simple awareness matters—no chemical, no matter its reputation, gets handled casually.
Years ago, I saw a technician ignore protocol, figuring the acid was “mild.” Within minutes, he learned otherwise. He recovered, but the lesson spread fast. Safety means respecting even the “less dangerous” chemicals, not by fearing them, but by meeting every step with knowledge, good gear, and sharp attention.
What are the typical applications of Hydroxyethyliminodiacetic Acid?
A Look at Metal Cleanup
Rust stains can be stubborn, and industrial water systems have to stay clean to run smoothly. Hydroxyethyliminodiacetic acid comes in handy because it grabs onto metal ions. This acid, with its specific structure, forms tight bonds with iron, copper, calcium, and more. That’s extremely useful in water treatment plants, where removing heavy metals keeps machines healthy and helps protect the environment. This approach isn’t theoretical—I’ve seen factories avoid large repair bills just by switching to better chelating agents in their pipes. It’s easy to overlook what’s floating in water, but removing stray metal ions pays off by keeping pumps running and workers safe.
Supporting Medical Diagnostics
Labs don’t often get the limelight, but accuracy in blood tests or imaging can shape someone’s future. Hydroxyethyliminodiacetic acid appears in certain diagnostic kits, often not as the star ingredient, but as a helper. The acid’s ability to corral metals helps stabilize solutions used in blood analysis, letting labs measure trace elements or run liver function scans without interference. I’ve spoken with technologists who say these chelators help them get reliable results, even from samples that have sat too long or traveled a long way, so patients aren’t left guessing.
A Boost in Agriculture
Farmers want healthy crops, but not every field gives plants the nutrition they need. Some soils bind up micronutrients, locking away iron, zinc, or manganese. Plant leaves turn yellow, growth slows, and yields drop. Enter hydroxyethyliminodiacetic acid, mixed in crop sprays or soil supplements. It helps micronutrients stay dissolved, so plants soak up what they need. This isn’t just a chemical fix. I grew up around farms that struggled with iron chlorosis in soybeans—after switching to chelated micronutrients, losses dropped and the fields looked greener. Tools like this don’t replace good soil management but give a real edge when dealing with tricky ground.
Industrial Cleaning and Maintenance
No one likes spending weekends scrubbing limescale off bathroom tiles, so imagine dealing with hard-water deposits on industrial equipment. Hydroxyethyliminodiacetic acid finds a job in tough cleaning formulations. It breaks down scale and dissolves mineral crusts in cooling towers, boilers, and heat exchangers. Facilities teams rely on it to keep energy bills down and cut downtime. It’s not just about making things look shiny; scale buildup can overheat systems and wreck performance.
Reducing Environmental Impact
Some traditional chelating agents hang around for years and don’t break down well after use. Hydroxyethyliminodiacetic acid is more biodegradable, so it’s a better pick for manufacturers who want to meet tighter water discharge regulations. Communities benefit—the rivers and lakes don’t get clogged with persistent chemicals, and wildlife stands a better chance.
A Few Thoughts About the Future
Chemical tools that pull double duty—powerful yet more eco-friendly—are right in step with what industries and farmers want today. Research keeps looking at ways to improve both performance and safety. I see more collaboration among chemists and engineers, not just for better profits but for healthier workplaces and cleaner surroundings. Investing in alternatives that break down and don’t poison our water seems less like an option and more like a responsibility. Hydroxyethyliminodiacetic acid helps bridge that gap.
How should Hydroxyethyliminodiacetic Acid be stored?
Why Storage Matters
Labs carry risk, even without considering rare compounds. Hydroxyethyliminodiacetic acid isn’t a kitchen cabinet chemical, but it shouldn’t be in the back of a cluttered supply closet either. Researchers and technicians handle it for metal chelation, lab processes, and sometimes as a building block for more complex chemistry. Neglecting storage details is risky, both for personal safety and for the experiments that rely on this material staying pure and stable.
Know Your Enemy: Chemical Properties
Every handler benefits from learning more than just the name on the label. This acid stands out for drawing ions out of metal solutions, but its chemical personality includes some stubborn quirks. Hydroxyethyliminodiacetic acid holds up well in closed drums, away from light and moisture. Some acids might break down or corrode quickly, but this compound likes stable, dry, and cool spaces. Extreme heat or wild swings in humidity can trigger slow breakdown, leaving a less effective product behind and possibly creating troubleshooting headaches.
Practical Steps for Safe Storage
Forget about tossing this bag into a box under a sink. Storage spaces should stay cool, typically under 25 degrees Celsius, but not subject to freezing. Storing it on shelves out of direct sunlight goes a long way. Humid basements invite clumping or slow hydrolysis, so airtight containers are not a bonus—they’re essential.
Labeling matters. It’s easy to assume nobody would mistake this for a food additive or benign buffer powder, but mistakes pile up where busy people rush through inventory. Clear, bold labels prevent mix-ups—especially where multiple chelating agents live side by side. Write the date you received or opened the drum. Older chemical stocks may not work as intended, and expired inventory shouldn’t slip into experiments where results depend on clean, uncontaminated ingredients.
Mind the Neighbors
Hydroxyethyliminodiacetic acid shows little tendency for dramatic reactions, but nothing stays safe if incompatible substances spill together. Keep acids and bases separate. No strong oxidizers nearby. I once visited a small lab with chemical drums resting against each other in an old wine cellar. A leak from a bottle of bleach nearby led to a sticky mess and unexpected chemical odors. Stacking storage or crowding containers can lead to undetected drips and dangerous combinations.
Good Habits Protect Everyone
Safety isn’t just about rules; it’s a habit formed over time. Stores like gloves and goggles near the acid, and easy access to spill kits, mean safer routines. Routine checks of the storage area should happen monthly. One veteran tech kept a simple sheet taped to the wall and signed it after visual checks—an easy reminder and protection against forgetfulness.
There’s no substitute for experience, but even experts trip up if spaces get messy or protocols turn lazy. Talking through safety routines as a team, inviting feedback, and updating the group when something changes is one of the best ways to prevent trouble. Seek out up-to-date guidance from chemical suppliers and follow workplace protocols to make smart decisions.
Keeping Science on Track
Hydroxyethyliminodiacetic acid, well cared for, performs as promised. Otherwise, it’s wasted money—or worse, a safety issue waiting quietly for its moment. Stay alert, stay organized, and save yourself from trouble before it starts.
| Names | |
| Preferred IUPAC name | 2-[(2-Hydroxyethyl)iminodiacetic acid] |
| Other names |
HIDA N-(2-Hydroxyethyl)iminodiacetic acid 2-Hydroxyethyliminodiacetic acid HHEIDA Iminodiacetic acid, N-(2-hydroxyethyl)- |
| Pronunciation | /haɪˌdrɒksiˌɛθɪlɪˌmiːnoʊdaɪəˈsiːtɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 3587-60-8 |
| Beilstein Reference | 1744651 |
| ChEBI | CHEBI:24670 |
| ChEMBL | CHEMBL3181857 |
| ChemSpider | 23274 |
| DrugBank | DB02307 |
| ECHA InfoCard | 06c0860d-1e49-4439-a2a1-6e3031404672 |
| EC Number | ["3.8.1.5"] |
| Gmelin Reference | 9589 |
| KEGG | C14248 |
| MeSH | D006881 |
| PubChem CID | 17658 |
| RTECS number | KK5075000 |
| UNII | 8T9J2J2U27 |
| UN number | UN3077 |
| Properties | |
| Chemical formula | C6H11NO5 |
| Molar mass | 207.17 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.332 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -3.05 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 8.05 |
| Basicity (pKb) | 7.71 |
| Magnetic susceptibility (χ) | -7.93·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.529 |
| Viscosity | Viscosity: 30 mPa·s (25°C) |
| Dipole moment | 5.51 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 205.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1132.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1836 kJ/mol |
| Pharmacology | |
| ATC code | A16AA13 |
| Hazards | |
| Main hazards | May cause respiratory irritation. Causes skin irritation. Causes serious eye irritation. |
| GHS labelling | GHS07, GHS05 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | Hazard statements: Causes serious eye irritation. |
| Precautionary statements | Precautionary statements: P261, P264, P280, P301+P312, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | > 230 °C |
| Lethal dose or concentration | LD50 (oral, rat): > 5000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat Oral 4000 mg/kg |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.5% |
| Related compounds | |
| Related compounds |
Iminodiacetic acid Nitrilotriacetic acid Ethylenediaminetetraacetic acid (EDTA) Diethylenetriaminepentaacetic acid (DTPA) Methyliminodiacetic acid N-(2-Hydroxyethyl)ethylenediaminetriacetic acid |

