Hydroxyethyl Sulfide: A Modern Perspective
A Look Back: The Path to Hydroxyethyl Sulfide
Hydroxyethyl sulfide did not pop up on lab benches overnight. By the late twentieth century, chemists dug into the family of thioethers searching for compounds that featured both functional diversity and a manageable safety profile. The discovery of hydroxyethyl sulfide reflects a period driven by the need for versatile intermediates, especially in sectors where selective modification of molecules provided a real edge. In my years studying organic synthesis trends, I've noticed how research on alcohol and sulfide couplings often starts for cosmetic or agricultural applications, then spills over into completely new directions once industry or government gets involved. The development of hydroxyethyl sulfide stands right at this kind of crossroads, where curiosity, necessity, and commercial aims collide.
Product In Focus
This is not a household name, but hydroxyethyl sulfide often supports the backbone of bigger projects. Chemically, it carries a hydroxyethyl group linked to a sulfur, providing two reactive sites along a small and flexible molecular chain. Its neutral smell and manageable handling let it find its way into chemical syntheses where both the alcohol and the thioether can participate. Think about dye manufacturing, specialty surfactants, or the preparation of custom intermediates for pharmaceutical molecules — hydroxyethyl sulfide frequently plays a hidden but important role in these tasks. Its appeal grows whenever a project demands distinct chemical personalities in a compact molecule.
Physical and Chemical Realities
Spend time with hydroxyethyl sulfide in a lab and its clear, colorless liquid form stands out. Compared to bulkier or strongly odorous thioethers, it carries a moderate smell and stays liquid at standard room conditions. The sulfur atom tends to pull some weight in reactivity, making nucleophilic attacks on the molecule effective under ordinary synthetic conditions. The hydroxy group opens another lane for functionalization and extensions. Hydroxyethyl sulfide dissolves smoothly in common solvents, which comes in handy for process chemists trying to avoid unnecessary mixing problems or phase issues in larger reactors. It sidesteps some of the headaches associated with highly flammable or extremely toxic sulfur species, even while it cannot be described as entirely benign.
Technical Specifics and Labeling
In regulated, well-run labs, labels for hydroxyethyl sulfide highlight its chemical identifier, structural diagram, and hazard warnings. What always catches my attention from experience is the need for clear communication regarding potential skin and eye irritancy. Storage requirements highlight a cool, ventilated space — sulfur compounds do not mix well with strong oxidizers or acids, as even small cross-contamination can create entirely new, sometimes hazardous, substances. Documentation documents both batch purity and the absence of key contaminants, crucial for anyone who relies on reproducibility in multi-step syntheses.
Making It: Preparation Stories
Synthesizing hydroxyethyl sulfide traces directly from mixing ethylene oxide with hydrogen sulfide — a route familiar for industrial scale-ups, thanks to the ready supply of feedstock chemicals. This reaction looks simple on paper, but anyone running it at production scale manages several critical safety risks with each batch. Controlling the pressure, venting, and preventing side-product formation shapes the quality and yield of the finished product. For research-scale labs, using safer alternatives for introducing the hydrogen sulfide unit helps reduce mishap odds. Route tweaking sometimes creates subtle byproducts, leading thorough process chemists to design purification steps against specific impurities.
Getting Creative: Modifications and Reactions
The real utility of hydroxyethyl sulfide comes into focus once it reaches the bench for downstream chemistry. The hydroxy group invites transformations like esterification, etherification, and oxidation, each unlocking branches for further synthesis. The sulfur atom can serve as an anchor for oxidation to sulfoxides or sulfones, or undergo alkylation to make new, more complex organic thioethers. Having both alcohol and sulfide functions on a small molecule allows chemists to build multi-functional products, often conserving both time and resources compared to longer synthetic sequences. Sometimes, modifying the hydroxyethyl sulfide core provides a way to selectively tune physical or biological properties, a valuable trick for those working in drug discovery or material science.
Naming and Synonyms
Over the years, the trade and research literature have referenced hydroxyethyl sulfide under several names: 2-Hydroxyethyl sulfide, thioethanol, or beta-mercaptoethanol. Context matters, as different industries adopt different habits, sometimes leading to confusion for those searching safety data or material inventories. This highlights the ongoing importance of harmonized chemical naming, particularly as regulatory and cross-border chemical trade keeps expanding.
Managing Safety & Setting Standards
Most seasoned chemists recall near-misses involving sulfur compounds. Hydroxyethyl sulfide may not carry the high volatility of mercaptans, but ignoring its skin and eye irritant profile is a quick way to find yourself in the emergency wash station. Good operational standards don’t stop at gloves and goggles: proper venting, accurate labeling, and maintaining separation from strong oxidizers keep everyone out of trouble. Industry uses internationally accepted frameworks for storage, transport, and disposal. Still, I’ve found that real safety culture starts with day-to-day vigilance — safety doesn’t leave room for shortcuts, no matter smaller perceived risks.
Application Space
Hydroxyethyl sulfide performs well as an intermediate for flavor chemicals, fine chemicals, and in certain specialty coating and resin systems. Beyond obvious industrial tasks, research into bioactive molecules sometimes draws on the peculiar balance of hydrophilicity and reactivity in this compound. In the synthesis of pesticides, pharmaceutical precursors, and water-treatment additives, this chemical stands out. It may not headline as the ‘star’ component in finished consumer products, but its ability to bring about selective chemical transformations ensures its continued demand in the background of manufacturing pipelines.
Research and Development: A Steady Push
Academic and industry groups keep finding new roles for hydroxyethyl sulfide, often as part of larger efforts to introduce better-controlled reactivity or to cut down environmental risks from waste streams. Green chemistry approaches look at whether its preparation generates unnecessary byproducts or if the sulfur gets retained in downstream waste, prompting process adjustments. Labs I’ve followed try to engineer functional derivatives or attach the hydroxyethyl sulfide motif onto polymer backbones, chasing new material properties while retaining process reliability. This focus sometimes results in parallel progress for analytical techniques, aimed at identifying minor impurities or degradation products, both for safety and to squeeze extra value from each synthetic run.
Toxicity: Weighing Risks and Rewards
Toxicity becomes a different story for sulfur compounds, especially those with both hydroxy and sulfide groups present. Animal data and cellular studies highlight moderate acute toxicity, manifesting most strongly through inhalation or ingestion. Detailed studies continue to evaluate long-term exposure risks, both in manufacturing and downstream user environments. The compound acts as a skin and eye irritant, but does not present the high toxicity profile of some aryl or chlorinated thioethers. Monitoring programs track both workplace air and effluents to head off accumulated exposure, reflecting both regulatory pressure and industry self-interest. As occupational health standards keep tightening, real-world exposure assessments guide improvements far more than textbook recommendations.
Future Directions and Challenges
No chemical’s story ends with its current set of uses. Applications for hydroxyethyl sulfide grow wherever industries demand new linking motifs, or look to squeeze more performance from a chain of molecular transformations. Its position in bridging sulfur and alcohol chemistry keeps it attractive for the next generation of smart materials and greener synthesis pathways. As process safety and environmental scrutiny both climb, the pressure mounts to overhaul old manufacturing strategies — demanding better process efficiency, lower emissions, and simplified waste management. Policing purity, minimizing trace contaminants, and devising safer synthetic routes all sit on the to-do list for researchers aiming to keep hydroxyethyl sulfide relevant while meeting evolving standards for health and the environment. In my years observing shifts across specialty and intermediates chemistry, I’ve come to see that those who adapt quickly to these pressures gain an edge: innovation now stands inseparably linked to safety and sustainability.
What is Hydroxyethyl Sulfide used for?
Connecting the Lab to the Real World
Walk into any chemical manufacturing plant or lab, and odds are you’ll spot at least one drum labeled hydroxyethyl sulfide. It doesn’t grab headlines, but this clear liquid plays a big role behind the scenes in many industries. Over time, working in chemical distribution, I’ve seen how products like hydroxyethyl sulfide don’t call attention to themselves, yet they keep things running.
The Workhorse in Organic Synthesis
In organic chemistry, building complex molecules starts with simpler pieces. Hydroxyethyl sulfide steps in as a starting material or intermediate, especially when labs need to create molecules that contain both sulfur and oxygen. These two elements show up in pharmaceuticals, fragrances, and specialized plastics. Chemists reach for hydroxyethyl sulfide for its ability to donate a “hydroxyethyl” chunk, helping researchers build targeted molecular structures.
Take drug development as an example. Researchers design small molecules to fit certain proteins in the body. Often, they experiment with different combinations of atoms to tweak how the medicine works or reduce side effects. Hydroxyethyl sulfide offers flexibility here. Its stable structure and accessible functional groups allow labs to attach other atoms or groups, creating a range of test compounds faster than sourcing everything from scratch.
Solvent and Reducing Power
Some people in the chemicals world know hydroxyethyl sulfide as an efficient solvent or as a reducing agent. In dye works, it helps create the right reaction environment, ensuring colors turn out accurate and don’t fade early after washing. Industrial cleaners sometimes turn to this chemical, especially for oil and gas machinery that builds up unwanted residue inside pipes and valves.
The sulfur atom in hydroxyethyl sulfide allows it to react with certain metals. Refineries sometimes use it during the process of removing unwanted metals from petroleum products. Without clean fuels and lubricants, engines and machines lose performance, costing businesses and drivers more in repairs.
Challenges for Safety and the Environment
Sometimes, the behind-the-scenes chemicals like hydroxyethyl sulfide don’t get much scrutiny, but safety concerns should never be ignored. Inhalation or skin exposure over time can lead to health issues. I remember one incident where a spill in a warehouse lingered thanks to delayed detection. Luckily, protective equipment and proper ventilation stopped anything serious from happening. It served as a reminder that workers still remain at risk if companies cut corners or relax training.
The sulfur in hydroxyethyl sulfide raises another issue: what happens after use? Sulfur compounds can release odors and sometimes pose risks for water supplies or wildlife. Disposal must follow strict safety rules, including controlled incineration or trained hazardous waste teams. I’ve noticed some facilities now run more regular audits and encourage anonymous worker feedback to catch problems before they get big.
Looking Forward: Building on What Works
Hydroxyethyl sulfide won’t headline a new battery or pharmaceutical breakthrough, but its steady presence keeps manufacturing and research humming. Keeping the public and workers safe starts by valuing transparency and regular training, not just compliance checklists. In practice, that means open-door policies, reporting close calls, and finding new ways to reduce unnecessary risks in handling and disposal.
Green chemistry keeps growing as a movement. More labs are researching “greener” substitutions for traditional chemicals, seeking decreases in waste and hazards. There’s room here for new derivatives or recycling processes involving hydroxyethyl sulfide that keep the benefits and shed some of the old liabilities. As someone who has spent years hands-on in the business, I believe a mix of open-minded management and innovation from the ground up will give chemicals like hydroxyethyl sulfide a future that fits modern standards—and keeps people, and the world, safer.
What is the chemical formula of Hydroxyethyl Sulfide?
What Makes Up Hydroxyethyl Sulfide?
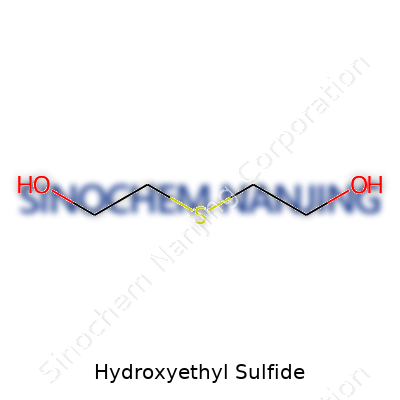
Hydroxyethyl sulfide stands for a molecule built from carbon, hydrogen, oxygen, and sulfur. Its chemical formula, C2H6OS, offers a clue about this structure. Here’s a simple breakdown for those who, like me, often prefer concrete details: there are two carbon atoms, six hydrogens, one oxygen, and a sulfur atom. To those who grew up mixing baking soda and vinegar just to watch the fizz, that mix of elements might sound modest, but the outcomes can be anything but.
Significance in Science and Daily Life
Anyone who studies organic chemistry gets used to spotting patterns in these formulas. The presence of both a hydroxyl group (the “hydroxy” in the name) and a sulfur atom means this compound brings together features from both alcohols and thiols. That blend gives it unique reactivity. Researchers and chemical engineers turn to hydroxyethyl sulfide in labs to create more complex molecules, or sometimes to tweak the properties of a reaction.
Back in college, I spent late nights in the lab with classmates, hunting for ways to improve reaction yields. One friend found that hydroxyethyl sulfide worked as a good intermediate, acting as a stepping stone between more complicated molecules. That hands-on experience left a mark: these little building blocks matter more than people realize outside the lab.
The world depends on chemicals like this in subtle ways. For example, intermediates connect steps in the creation of pharmaceuticals, agrochemicals, and even materials for batteries. Even though most people rarely hear about these compounds, they help make modern life convenient and safe.
Risks, Benefits, and Solutions
Drawing from my time working with chemicals, anything that contains both sulfur and an alcohol group must be handled carefully. Sulfides can release strong odors, and some compounds with sulfur have raised health concerns over the years. Anyone in the lab must follow safety rules, from wearing gloves to handling small quantities under hoods.
There’s a bigger picture here. Chemical manufacturing stands as a major industry, but it can bring contamination and health risks if mishandled. In some cities in the US, for example, chemical accidents have left entire neighborhoods dealing with pollution. Environmental policies have already pushed for stricter guidelines on handling and disposing of such chemicals. The European Union’s REACH regulations suggest that companies collect more safety data before putting new substances on the market, giving a good blueprint for how things could work elsewhere.
Technology brings hope, too. Better sensors and real-time monitoring systems now spot leaks or contamination faster than ever. Digital tracking can follow a batch of a compound from its first reaction to the last drum that leaves the factory.
Transparency matters most. People expect to know what chemicals touch their lives, even if only as an ingredient in something more familiar. Reliable information, research, and a commitment to both innovation and safety will always matter as much as the right formula.
Is Hydroxyethyl Sulfide hazardous or toxic?
Everyday Chemistry Finds Its Way Into the Spotlight
Many folks never hear about hydroxyethyl sulfide unless they dig into chemical databases, work in labs, or pay close attention to what’s inside their household goods. Chemicals with unfamiliar names can sound scary, and searching for answers often brings more confusion than clarity. Let’s break down what’s known about hydroxyethyl sulfide and whether people need to worry.
Digging Into the Science
Hydroxyethyl sulfide (HES) crops up in a handful of chemical processes. Chemists use it more behind the scenes, often as a building block to craft other compounds. It’s clear, colorless, and has a characteristic sulfur odor. Looking at toxicity, true understanding always demands seeing how a substance operates inside the body and how people get exposed in real-world settings.
The Material Safety Data Sheets (MSDS) for hydroxyethyl sulfide indicate it causes irritation if it touches the skin or eyes. Swallowing or breathing in vapors can upset the stomach and lungs. Skin contact sometimes produces redness or swelling, though these symptoms don’t always match what one might see with corrosive or truly poisonous chemicals.
Weighing Toxicity in Context
The fact is, not every chemical that irritates or smells bad means death or lasting damage. Lab tests place hydroxyethyl sulfide at the lower-to-moderate end for acute toxicity. The oral LD50 for rats falls around 2,200 mg/kg. To put that in plain English—this amount of substance per body weight causes death in half of test animals, and that’s a much higher threshold than many household poisons. For reference, acetaminophen (Tylenol) can be far more dangerous in overdose.
No long-term human studies spell out cancer or reproductive risks with HES. It’s rare to see chronic exposure outside of specialty labs. That doesn’t mean it’s perfectly safe. Take a look at the real data: people have worked with similar compounds for decades, and most adverse reactions revolve around poor protective gear or ventilation. No reported disasters or major poisonings exist in the scientific literature.
Workplace Hazards and Good Habits
In my experience working in a chemical research lab, respect for every solvent or reagent pays off. Just because something lands as “moderately toxic” doesn’t mean it belongs down the drain or on bare hands. Workplaces using hydroxyethyl sulfide stick to gloves, goggles, and fume hoods. Spills call for quick cleaning and good ventilation. Most accidents come from shortcuts or a lack of information, not the inherent deadliness of HES.
Putting Health and Safety First
Some countries hold tight rules on importing and using chemicals like hydroxyethyl sulfide. These restrictions don’t always reflect toxicity alone—they often trace back to the compound’s potential as a precursor in chemical synthesis. Storing any chemical safely and training folks on its hazards makes a bigger difference than banning it outright. Public access to honest hazard data fosters smarter decisions, inside and outside the lab.
For everyday people, risk from hydroxyethyl sulfide stays close to zero unless someone works directly with it. Even in the lab, basic protections—those gloves, goggles, and common sense—go farther than fear ever could. Chemical literacy means knowing how to respect powerful tools without letting worry run the show.
How should Hydroxyethyl Sulfide be stored and handled?
Why Safety Steps Matter
Not everybody keeps a bottle of Hydroxyethyl Sulfide under their kitchen sink. This industrial chemical comes with both benefits and risks, and I’ve seen far too many folks underestimate the dangers of chemicals that don’t get much public attention. Back in my early work at a specialty chemical warehouse, stories gathered from seasoned workers helped me appreciate why every protocol exists—because someone, somewhere, cut a corner.
Recognizing Hazards
Hydroxyethyl Sulfide carries a distinct pungency and tends to be flammable. Spills give off vapors that catch fire easily. It doesn’t take much more than a stray spark for things to go sideways. Toxicity is real here, too. Direct exposure, whether by skin or lungs, can irritate and trigger reactions. OSHA’s chemical safety guidelines highlight this compound for a reason. Contaminated hands or casual splashes aren’t just mild inconveniences—they could send you to the ER.
Practical Storage Tips
No fancy lab needed—just solid choices. If you handle this chemical in any fashion, lock up the container in a cool, ventilated spot. Sunlight and heat ramp up the flammability, and closed rooms concentrate vapors. I once had to bust open a back window of a loading room simply because the vent fans broke and nobody had bothered checking. Airflow kept us safe then; it’ll keep you safe now.
Walls and doors mean little against open drums or leaky seals. I recommend chemical-resistant containers with tight-fitting lids. Metal storage can sometimes cause unwanted reactions. Polyethylene drums give peace of mind, and regular inspection for cracks is a must. Labeling goes further than health and safety compliance—it stops mistakes before they happen. I’ve seen two substances mixed in error more than once, and the panic that follows proves labels save lives.
Handling with Confidence
It seems basic, but gloves, goggles, and aprons aren’t old-school overkill. You only need one bad splash in your lifetime to understand their value. Forced ventilation, like a simple fume hood or a good extractor fan, protects lungs from sneaky fumes. One of my mentors always kept a large jug of water close by for rinsing, and insisted anyone handling Hydroxyethyl Sulfide do the same.
Use sealed pumps or dispensers to move the liquid, and work in a defined, uncluttered area. Keeping the bench or floor clear limits trip hazards and spill risks. Absorbent mats or spill kits don’t just sit on a shelf—they prevent minor blunders from turning major.
Training and Response
Experience in chemical environments teaches you to respect the possibility of human error. Regular drills on chemical spills, and knowing exactly whom to call for help, put the odds in your favor. Emergency showers and eyewash stations should be usable, not just present; blocked stations defeat the point.
Never skim over safety data sheets. Keep them in sight. Returning to them before each handling session deepens awareness and keeps new staff from guessing. For storage, stick close to quantities you expect to use—hoarding extra brings extra risks. Government agencies like OSHA and NIOSH set rules built on hard lessons learned by people just like us.
Smart Solutions for Real-World Safety
Hydroxyethyl Sulfide’s best kept secret is vigilance. Good habits matter more than the latest storage gadget. Keep learning, keep inspecting your space, and never rush through handling steps. Sharing stories and knowledge with new team members delivers better results than a box of unused PPE. In my experience, the small investments in time and tools pay back tenfold, with fewer close calls and easier working days.
What are the typical applications of Hydroxyethyl Sulfide in industry?
Industry’s Invisible Workhorse
Hydroxyethyl sulfide doesn’t make headlines. Few people outside chemical plants have ever heard of it. Still, this clear, sharp-smelling liquid keeps some huge wheels turning. I first got a real look at it working in a fine chemicals plant. A barrel tucked into a corner, labeled with little more than its trade name, sometimes holds more influence than a hundred gallons of bleach. In my experience, its role spans a few core industries, and the impact goes way past the factory gates.
Petrochemical Processing: Cleaning Up the Act
The sulfur atom in hydroxyethyl sulfide draws the attention of petroleum engineers. They’re always dealing with how to purify crude oil. Hydroxyethyl sulfide acts as a scavenger, locking up and removing excess hydrogen sulfide gas during refining. This step matters, since hydrogen sulfide is toxic and can corrode equipment. Stopping the gas in its tracks helps refineries meet air quality laws and keeps the air around the plant less dangerous. The international safety community supports this use; reports from both OSHA and the EPA stress the vital need to manage hydrogen sulfide with effective scavengers.
Pharmaceutical Synthesis: Building Blocks for Medicines
Pharmaceutical companies use hydroxyethyl sulfide in complex synthesis steps. Many drugs—especially those for cancer and infection—require sulfur atoms at key positions. Chemists turn to hydroxyethyl sulfide as a way to thread sulfur into complicated molecules. I’ve watched the process take a messy, multi-pot reaction and trim it to a single stage. Drug companies value saves like this because fewer steps mean fewer chances for mistakes and less chemical waste. Medicinal chemists from respected journals like the Journal of Organic Chemistry point to derivatives of this compound in patented syntheses every year.
Agricultural Chemicals: Building Safer Pesticides
Farmers and agri-scientists rely on pest control that doesn’t linger and cause harm outside its target zone. Hydroxyethyl sulfide gives developers an edge when designing ‘degradable’ pesticides. The molecule acts as a sulfur source and modifies active ingredients so they break down into harmless products after use. This reduces risk to groundwater, which matters to anyone who cares about safe drinking water. Field studies published by the American Chemical Society show these derivatives biodegrade much faster compared to older pesticides—a win for both crop yields and public health.
Rubber and Plastics Manufacturing: Tweaking Performance
Rubber and plastic plants often look for ways to tweak the feel, bounce, or durability of their materials. Hydroxyethyl sulfide steps in as a chain transfer agent or curing aid. Its molecular structure helps control polymer length and crosslinking. I’ve seen floor managers use it to stretch the life of industrial hoses or prevent plastics from turning brittle before their time. The right additives translate into products that last longer and require less frequent replacement. Industry white papers highlight these benefits, stressing improvements in material lifespan across automotive and industrial supply chains.
Protecting Workers, the Environment, and the End User
This chemical, like any used on an industrial scale, demands respect. Workers need clear training, from handling and storage to proper disposal. Industry best practices—not just the letter of the law—protect people and the planet. Companies that get sloppy with chemical stewardship face workplace risk, regulatory fines, and loss of public trust. Responsible sourcing and tight safety standards help all of us down the line, whether we’re taking medicine or walking next to a refinery. Our health and safety policies, backed by strong science and ethical oversight, shape how these chemicals benefit society, quietly but crucially, every day.
| Names | |
| Preferred IUPAC name | 2-sulfanylethanol |
| Other names |
2-Mercaptoethanol Ethylene Mercaptan Ethanol, 2-mercapto- B-Mercaptoethanol Mercaptoethanol |
| Pronunciation | /ˌhaɪdrɒksiˈɛθɪl ˈsʌlfaɪd/ |
| Identifiers | |
| CAS Number | 107-03-9 |
| Beilstein Reference | 1321597 |
| ChEBI | CHEBI:16045 |
| ChEMBL | CHEMBL156998 |
| ChemSpider | 74938 |
| DrugBank | DB14004 |
| ECHA InfoCard | 100.003.499 |
| EC Number | 200-579-5 |
| Gmelin Reference | 40965 |
| KEGG | C06133 |
| MeSH | D011850 |
| PubChem CID | 15604 |
| RTECS number | KH8575000 |
| UNII | ZQA8Y388Y3 |
| UN number | UN3334 |
| Properties | |
| Chemical formula | C2H6OS |
| Molar mass | 78.15 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | unpleasant |
| Density | 0.987 g/cm3 |
| Solubility in water | soluble |
| log P | -0.28 |
| Vapor pressure | 0.63 mmHg (at 25 °C) |
| Acidity (pKa) | 15.5 |
| Basicity (pKb) | 7.03 |
| Magnetic susceptibility (χ) | -57.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.488 |
| Viscosity | 1.93 mPa·s (20 °C) |
| Dipole moment | 2.01 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 191.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -164.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | −523.2 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D02AX |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. H315: Causes skin irritation. H319: Causes serious eye irritation. H335: May cause respiratory irritation. |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P280, P303+P361+P353, P305+P351+P338, P370+P378 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 104°C |
| Autoignition temperature | 215°C |
| Explosive limits | Explosive limits: 2.6%–17.0% |
| Lethal dose or concentration | LD50 Oral Rat 736 mg/kg |
| LD50 (median dose) | LD50: Rat oral 960 mg/kg |
| NIOSH | SN3850000 |
| PEL (Permissible) | PEL: 2 ppm (6 mg/m3) as TWA |
| REL (Recommended) | 0.05 mg/L |
| IDLH (Immediate danger) | 200 ppm |
| Related compounds | |
| Related compounds |
Ethanethiol Diethyl sulfide Ethylene glycol 2-Mercaptoethanol Ethylene oxide |

