Histamine Dihydrochloride: Practical Insights and In-Depth Commentary
Historical Development
Histamine and its salts, including histamine dihydrochloride, have roots running back over a century. Scientists like Sir Henry Dale and Charles Barger uncovered the mysterious actions of histamine on animal tissues in the early 1900s. Throughout the 20th century, researchers learned to isolate, modify, and eventually synthesize histamine and its salts in the lab. Pharmaceutical labs saw promise in this compound, especially after evidence showed it could trigger and modulate several key physiological reactions. In the 1990s, the search for immune system modulators and new cancer therapies brought histamine dihydrochloride to clinical trials, especially in combination with cytokines. Trusted by clinicians in certain contexts but approached with caution, this compound’s story shows how scientific progress evolves by building on small discoveries, learning from clinical setbacks, and betting on more precise biochemical targets. From being a simple mediator of itching and dilation of blood vessels, histamine entered the world of complex immunology and oncology. Careful documentation and rigorous standards have shaped its modern production and clinical usage.
Product Overview
Histamine dihydrochloride draws attention in both clinical medicine and laboratory settings. The pharmaceutical form appears as a white to off-white crystalline powder, packaged in tightly sealed vials or containers to maintain purity. Researchers and clinicians use the salt to stimulate immune responses, particularly in leukemia and related clinical trials. Pharmacy teams stock it with detailed labeling and handling instructions. For chemical suppliers, purity grades matter—anything below 98% gets set aside, as minor contaminants could jeopardize experiments or patient safety. This product rarely sits on a retail shelf; professionals acquire it through specialized distributors who follow strict regulations, and traceability remains non-negotiable.
Physical & Chemical Properties
The compound holds the formula C5H11N3·2HCl. Its molecular weight, 184.08 g/mol, comes up repeatedly in supply catalogs. Easily dissolving in water but less so in ethanol, the powder carries a faint odor reminiscent of ammonia. It melts between 256 and 260ºC, decomposing as it nears those temperatures. Its shelf life hinges on low humidity and protection from light, so manufacturers supply it in amber containers or foil pouches. Under electron microscopy and powder diffraction, histamine dihydrochloride reveals a simple, stable crystalline lattice. pH levels play an important role in its stability; users tend to mix only what they need before administration due to its tendency to break down outside optimal conditions.
Technical Specifications & Labeling
Technicians and pharmacists depend on precision. Commercial packs of histamine dihydrochloride come with batch-specific certificates of analysis. Labels display concentration, lot number, expiration date, recommended storage, and synthesis route. All vials and containers feature hazard pictograms required by the Globally Harmonized System (GHS) along with guidelines for immediate spill response and first-aid measures. Expected levels of heavy metals, residual solvents, and other impurities appear clearly on the documentation, based on pharmacopeial standards like USP or EP monographs. The Material Safety Data Sheet (MSDS) includes instructions for fire, chemical exposure, and waste disposal, and suppliers train staff to interpret and act on each instruction as they receive shipments.
Preparation Method
Bulk synthesis of histamine dihydrochloride usually starts with imidazole and chloroacetaldehyde. Chemists orchestrate condensation and reduction steps, milking the process for a pure intermediate before adding hydrochloric acid to yield the stable dihydrochloride salt. Each batch goes through a series of filtrations and recrystallizations. Large-scale manufacturers optimize these steps for consistent particle size, ensuring both uniform dissolution and manageable filtration during final purification. Laboratories performing smaller runs work under fume hoods, using high-quality reagents to minimize contamination. Routine testing follows every stage, not just for purity, but for by-products that could trigger adverse reactions in patients or experimental animals.
Chemical Reactions & Modifications
Chemists working with histamine dihydrochloride view it as both a target and a starting point. Under mild conditions, the primary amine group reacts with acids, anhydrides, or isocyanates to form a range of derivatives, often serving as research tools in immunology or pharmacology. N-alkylation or acylation generates new analogues that land on the bench for evaluation in neurotransmission studies, allergy research, or even veterinary applications. The compound also undergoes oxidation or reduction to explore subtle shifts in bioactivity. More advanced researchers exploit the imidazole ring for metal complexation, designing catalytic systems or diagnostic agents by attaching labels and chelators onto the scaffold. Each modification aims to answer practical questions—Can new analogues block certain receptors? Might a tweak in structure reduce unwanted inflammation during cancer therapy?
Synonyms & Product Names
Anyone reading clinical or chemical literature has likely seen histamine dihydrochloride under different aliases. Beyond its IUPAC designation, the compound shows up as Histaminum Dihydrochloridum, 2-(1H-Imidazol-4-yl)ethanamine dihydrochloride, and even the shorter 'histamine hydrochloride'. Product catalogues sometimes use trade names; medical supplies reference Levact, Ceplene, or other branded forms standardized for injection. In regulatory databases, every synonym receives a meticulous cross-reference. For medical teams, these naming conventions avoid confusion, especially with similar-looking but pharmacologically distinct agents. The industry’s reliance on strict nomenclature keeps dosing errors and dangerous substitutions at bay.
Safety & Operational Standards
Working with histamine dihydrochloride mandates clear safety protocols. Only trained personnel handle and measure the powder, wearing gloves, eye protection, and in some cases, respirators filtered for fine dust. Accidental skin exposure triggers immediate washing, as the compound can cause severe redness, itching, and inflammation. Medical professionals carefully dispose of all sharps, vials, or contaminated materials in compliance with hazardous waste rules. OSHA standards and similar international guidelines dictate ventilation, spill control, and emergency procedures. Pharmacists administering the compound to patients provide clear risk disclosures, warning of potential hypotension, tachycardia, and bronchospasm. Each institution crafts its protocols rooted in national and international regulations, learning from documented incidents to patch vulnerabilities.
Application Area
Hospitals, research labs, and biopharmaceutical companies turn to histamine dihydrochloride for its ability to modulate the immune system. In cancer immunotherapy, the compound teams up with interleukin-2 (IL-2) to fend off relapse in acute myeloid leukemia, helping reactivate immune effector cells. Although major health authorities restrict its use to carefully screened patient groups, immunologists continue to explore its roles in boosting natural killer cell function. Some European clinics rely on the drug in specific protocols, backed by trial data showing improved survival for certain patient populations. Laboratories test it for use in allergy research, signal transduction studies, and as a reference standard in histamine release assays. Its clear-cut physiological effects make it a mainstay in undergraduate and graduate education, giving students tangible examples of vasodilation, bronchoconstriction, and immune stimulation.
Research & Development
Pharmaceutical companies and academic researchers pick up histamine dihydrochloride as a linchpin for trials probing immune response. For oncologists, the ongoing challenge is to maximize antitumor activity without tipping the scales toward dangerous cytokine storms or excessive tissue inflammation. Researchers also track its role as a pharmacological probe in mast cell and basophil biology. Fresh research focuses on developing analogues with the same immunomodulatory punch but a lower risk of side effects. Teams experiment with new routes of administration—transdermal, sublingual, or sustained-release devices—hoping to improve patient experience and drug levels. Collaboration spans continents, and global regulatory agencies share clinical trial data to guide future approvals and therapeutic guidance.
Toxicity Research
Animal studies and human trials reveal a mixed bag for toxicity. Acute exposure causes redness, headache, flushing, and at higher doses, dramatic drops in blood pressure. Researchers catalog and predict these responses using animal models, but human variability always surprises. Chronic toxicity studies remain rare, since patients rarely receive extended courses. Carcinogenicity and mutagenicity tests in rodents provide some reassurance, but medical professionals remain wary, especially when considering use outside of controlled clinical settings. Toxicology teams recommend close monitoring for systemic effects, and most hospitals require resuscitation equipment in treatment rooms. Regulators demand that pharma companies report all observed adverse events, tracking even subtle patterns in laboratory animals or volunteer cohorts.
Future Prospects
Clinical use sits at something of a crossroads. Personalized medicine, digital monitoring during treatment, and better side effect management could shape future therapies featuring histamine dihydrochloride or its derivatives. As immunology uncovers new pathways, the compound’s precise mode of action faces further scrutiny, which may inspire the next generation of bioactive molecules for oncology, allergy, or even autoimmunity. Industry rumor points to new delivery systems on the horizon—microspheres, encapsulation, or implantable devices that deliver tightly controlled doses. Researchers expect more collaboration between pharma companies and regulatory bodies to smooth out trial design, patient selection, and follow-up. Some see potential in veterinary medicine and companion animal oncology. As understanding grows and biotechnology evolves, histamine dihydrochloride’s story has new chapters waiting to be written, shaped by the needs of doctors, patients, and researchers looking for answers where old therapies fall short.
What is Histamine Dihydrochloride used for?
A Closer Look at the Uses
From my experience, few people outside the medical field know much about histamine dihydrochloride. Most think of antihistamines and allergies, but there’s something more remarkable going on, especially in hospitals and cancer clinics. Doctors use this compound as part of cancer therapy, specifically for certain types of leukemia. Acute myeloid leukemia, to be precise, causes chaos in the bone marrow. Patients who battle hard through chemotherapy face another major hurdle—the disease coming back.
To improve patient outcomes, clinicians look for new approaches. Research pushed forward by large-scale studies has shown that histamine dihydrochloride, given with interleukin-2 (IL-2), can keep cancer from returning. This combination works as a maintenance treatment, meaning it helps prolong remission after initial therapy. Medical teams in Europe adopted it following studies in the 2000s, where results hinted at higher leukemia-free survival rates. Without options like this, too many patients relapse.
How It Works
Immune defenses sometimes struggle to keep cancer at bay, especially right after chemotherapy batters the body. Natural killer cells, which act as the body’s own security detail, need a boost. Histamine dihydrochloride steps in here. It blocks enzymes that weaken the immune system's attack. Most immune experts say it protects the killer cells from suppression, keeping them active in hunting down stray leukemia cells. Adding interleukin-2 further ramps up the immune response, so the combination has a solid biological foundation. Studies—peer-reviewed and published in respected journals—show fewer relapses in patients treated with the combo.
Why It’s Important
Cancer affects families on every street. Even one new chance at more years in remission means something. A loved one of mine survived a leukemia diagnosis, and knowing there are focused treatments like this offered some hope. Not all treatments work for everyone, but giving doctors an extra tool matters. For patients whose leukemia snuck back after intense chemo, the prospect of a maintenance therapy changes the outlook. Hospitals in many European countries now use this regimen, giving solid evidence to back up the manufacturer’s claims.
Unlike broad chemotherapies, histamine dihydrochloride targets immune mechanisms. That translates to fewer side effects for many patients. Listening to patients in support groups, the idea of treatment that allows them to stay at work, take care of their kids, or just live a life with less time in hospital matters as much as remission statistics. The body’s own defenses, with a little chemical guidance, can do remarkable things.
Current Limitations and Looking Ahead
Not all countries have approved or adopted histamine dihydrochloride for leukemia maintenance. Getting this type of treatment into more clinics comes down to regulatory hurdles, cost, and continued science. Governments and hospitals everywhere need to look at real-world results and push for coverage when there’s solid evidence behind it. Scientists haven’t stopped searching for even better results through new combinations and dosing schedules. For families, hearing that more maintenance therapies might appear brings new hope.
With cancer, the fight never stops. Histamine dihydrochloride might not be a household name, but it gives new options to those who need every chance they can get.
How is Histamine Dihydrochloride administered?
Administration Steps and Everyday Impact
Histamine dihydrochloride isn’t a household name, but in medical oncology, many know it as part of combination therapy for acute myeloid leukemia. Doctors deliver this compound through subcutaneous injection, sliding a fine needle just under the patient’s skin. In my experience helping family members through cancer care, I’ve seen how a process that looks straightforward can feel daunting—especially when needles and medication meet anxious minds and bodies.
Unlike pills or mouth sprays, subcutaneous delivery asks for trust in preparedness. Healthcare staff select a sterile syringe with the precise dose. Skin, often around the abdomen or thigh, gets swabbed with alcohol, then pinched gently. The needle pierces, the medication goes in, and then there’s careful withdrawal—a process that lasts only seconds but matters a lot. After each injection, the healthcare team monitors for local reactions: swelling, redness, or rare allergic responses. Decades of safety data support this approach, but vigilance never drops.
Why the Injection Format?
Many wonder why doctors don’t offer an easier route, like swallowing a tablet. It comes down to reliability and science. Histamine dihydrochloride doesn’t play nicely with stomach acid or digestion, so the shot avoids those hurdles. It gets into the body fast, right where the immune system can get the message. Many cancer treatments share this delivery method for similar reasons—some medications break down too quickly otherwise.
Medication schedules usually build a rhythm; histamine dihydrochloride often follows a twice-daily pattern for three weeks during treatment cycles. These patterns can test patience and pain tolerance. The site of the shot changes to avoid irritation, and nurses coach patients or caregivers through a routine that turns foreign tasks into familiar ones. My neighbor, fighting leukemia, learned to give her own shots at home, gaining confidence with practice. That control over a scary part of life matters.
Reducing Barriers in Real-Life Care
Many patients face hurdles such as fear of injections, travel to clinics, or financial stressors around frequent appointments. Programs that empower patients with proper training for self-injection offer tangible relief. Digital resources with video demonstrations, step-by-step checklists, and helplines give real peace of mind. In rural areas or during times like the pandemic, this kind of support stands out even more.
Pharmaceutical providers, hospitals, and advocacy groups can also smooth the path by streamlining prescription access and explaining costs clearly. Transparent insurance support and robust patient education matter just as much as the right syringe. From public health reports, we know missed doses reduce success, so lowering every barrier counts.
What’s Next?
Ongoing research looks at improved formulations or even new routes, though for now, subcutaneous injection remains the gold standard for this therapy. Real people drive this research—survivors, doctors, nurses, caregivers—pushing for new ways to balance rigorous treatment and daily living.
Histamine dihydrochloride may not be a familiar name outside cancer circles, but how we deliver it reflects the real-life mix of medical progress and human connection. Each injection is a moment where science meets resilience. That’s the part that always sticks with me.
What are the possible side effects of Histamine Dihydrochloride?
Understanding What Really Happens
Most people won’t find themselves thinking about histamine dihydrochloride unless a doctor brings it up. This compound shows up in some cancer treatments, especially for certain types of leukemia. Whenever a new medication enters the picture, people start asking about side effects. That question matters just as much here. So, what can happen to your body if you take this drug?
Why Side Effects Matter in Real Life
People might hear “side effects” and only picture a mild rash or an upset stomach. For me, knowing about side effects turned a medical mystery into something manageable. I once watched a close friend get blindsided by a new prescription—only because no one in the clinic walked through the possible short-term and long-term changes in a practical way. It left us scrambling for answers in the middle of a tough moment. Being honest about risks isn’t just a courtesy, it builds trust between health care providers and patients.
The Body's Response: Flushing, Headache, and More
Many folks taking histamine dihydrochloride notice skin flushing or get a headache. That’s not surprising, since histamine plays a central role in blood flow and immune signals. It prompts blood vessels to open up, which can leave your cheeks feeling warm. With this drug, that feeling ramps up. It isn’t always just a nuisance—headaches sometimes disrupt the day and flushing makes people feel self-conscious.
Some report chills, fever, or fatigue. Doctors observe these regularly. Whenever your immune system gets a nudge, even a small one, your system can feel worn down. In the context of cancer treatment, every bit of strength counts. No one running on empty wants another drain on energy.
Throat Tightness and Breathing Worries
A few people feel tightness in the chest or throat, or develop trouble catching their breath. For someone who already has allergies or asthma, those symptoms provoke more than anxiety—they can put you at real risk. These aren’t things to just watch and wait. Anytime a medication threatens your ability to breathe, ignoring it only raises the stakes. Quick communication with your doctor is key.
Digestive System Issues
Upset stomach, diarrhea, and nausea tie into histamine’s effects throughout the body. Since histamine receptors show up everywhere, the gut can react almost as quickly as the skin. Nobody likes to think about spending more time in the bathroom, but it’s a real-life issue for someone already stressed by illness.
Rare But Serious Complications
Low blood pressure gives some patients a scare, especially if dizziness kicks in. Heart palpitations aren’t rare. For anyone with heart disease in the family (like in mine), doctors take these symptoms more seriously with regular checks and more conversations at every appointment.
Open Conversation and Better Outcomes
There’s no shortage of drugs with long lists of side effects, but what matters is honesty and strategy. Laying out real experiences—headaches, chills, chest tightness—helps patients and families brace for what could come. Doctors can help chart a course, offer practical support, and explain which symptoms call for urgency. Smart care comes from clear talk and real partnership between patients and medical teams.
What Steps Make a Difference?
Doctors can’t predict every problem, yet they can bring science and experience to the table. Keeping lines open for reporting new symptoms goes a long way. Patients play a role, too, logging what they notice and sharing details, even small ones. As treatment advances, better monitoring and up-to-date research create more hope for predicting and limiting the toughest side effects of drugs like histamine dihydrochloride.
Is Histamine Dihydrochloride safe during pregnancy or breastfeeding?
Digging Into the Facts
Pharmacies offer a lot of options for medical treatment, but history shows not every drug fits every situation. Histamine dihydrochloride, used in some cancer treatments, has drawn some attention on the question of safety during pregnancy or breastfeeding. Many hear “histamine” and think of allergy pills. This is something different—an immune-modulating agent sometimes paired with interleukins in treating acute myeloid leukemia. It goes beyond the scope of your basic antihistamine, making this a serious topic.
Expectant mothers and people caring for newborns deserve drug information they can trust. Deciding whether to continue treatment or start a new medicine can weigh heavily on families who’ve been blindsided by a leukemia diagnosis. Information from real-world studies helps bring clarity here, but research results don’t always provide clear lines.
Current Findings
No major clinical trials have focused on histamine dihydrochloride use in pregnant people. Preclinical animal studies, according to published drug labels, sometimes showed harmful effects on developing embryos at high doses. Regulatory agencies like the FDA and EMA report insufficient human data to pull firm conclusions. They usually err on the side of caution, and healthcare professionals tend to avoid prescribing it during pregnancy unless no safer alternatives exist and the absolute need outweighs risk.
With breastfeeding, the story isn’t much brighter. Because there’s no concrete data about whether this drug passes into breast milk, it sits in a gray zone. Given the nature of most anti-cancer drugs, experts worry about the potential for even trace amounts reaching a newborn’s system. The American Society of Clinical Oncology and similar groups routinely recommend pausing breastfeeding during active cancer therapy, unless there’s strong evidence suggesting zero risk.
Why It Matters
Sifting through these issues makes you realize how uneven the research patchwork remains—especially for rare drugs or treatments that mostly serve adults with aggressive diseases. Safety studies in pregnant or lactating individuals often lack funding and fall far behind research into adult-only populations. No parent or patient should have to make blind choices or trade informed care for medical advances.
Asking oncologists and pharmacists about the latest, even if the answer is “we just don’t know,” pushes everyone toward safer, more cautious medicine. Avoiding unnecessary medicines during pregnancy has protected generations from harm. Older drugs once seemed routine until birth defects or infant complications started showing up years later.
Where to Go from Here
Transparent, shared decision-making between healthcare professionals, parents, and patients charts a safer path. Anyone facing this dilemma needs room to ask tough questions at the pharmacy, the oncologist’s office, or with the care team. Each case comes with unique factors: the urgency of cancer treatment, timing in pregnancy, available alternatives, and family choices about breastfeeding.
Research dollars should prioritize studying drugs in populations that need them most. In 2024, technology allows more real-time reporting on safety signals than ever before. Electronic records and patient registries can close gaps researchers missed years ago. If you’re in this situation, keep records, report unusual symptoms, and let your care team know about any exposure. It may save not just your family, but countless others.
Does Histamine Dihydrochloride interact with other medications?
Living With Treatment Decisions
I remember volunteering in an oncology clinic some years back. People didn’t just worry about cancer; they worried about every pill, every symptom, every trade-off. Questions like, “Can I take my regular medicine with this new one?” didn’t show up in textbooks—these came up in hushed voices or when someone caught me coming down the hallway. They stick with me because behind each question sits fear and hope tangled together. So if you’re staring at a bottle of histamine dihydrochloride and wondering what else you should know, you’re not alone.
What the Evidence Tells Us
Histamine dihydrochloride has found a place in treating certain types of leukemia, especially as part of maintenance therapy after remission. Doctors prescribe it in combination with interleukin-2 for acute myeloid leukemia (AML). But, mixing drugs is never simple. Histamine acts on the immune system, and so do a fair few medications—think steroids, anti-inflammatory drugs, and even allergy medications. Scientific reports warn against just assuming your established medication list won’t matter. Data points to possible amplification or weakening of effects. If steroids or immunosuppressants stack up, they can reduce how well histamine dihydrochloride works.
Chemotherapy regimens get re-evaluated for every new patient because everyone’s metabolism runs differently, and not everyone tells the whole story about their other pills. Antihistamines, which a surprising number of people keep in their home medicine cabinet, fight against what histamine dihydrochloride is trying to accomplish. These could sabotage treatment, without ever looking suspicious on paper.
Sorting Out the Details with Real People in Mind
One big lesson from direct patient care: the most dangerous drug interactions are often unintentional. Folks forget to list vitamins, supplements, or over-the-counter painkillers. Histamine dihydrochloride doesn’t live in a vacuum. Acetaminophen, NSAIDs, and some antibiotics change the way the body handles both cancer treatment and their day-to-day health. A recent review in Drugs showed drug-drug interactions often lead to unexpected side effects, missed doses, or serious complications. The worry grows when drugs affect the immune system, because the consequences aren’t always obvious until much later.
Doctors have software that flags drug interactions, but no algorithm catches the nuance of real lives. I’ve sat with nurses sifting through a patient’s list, asking follow-ups, double-checking before anyone gets a new prescription. People with leukemia, for example, aren’t only seeing their oncologist—they meet with their dermatologist for rashes, their pulmonologist for respiratory infections, their primary doctor for regular checkups. Every new prescriber means a fresh chance for an accidental mix-up. After all, medical records don’t always keep up in the real world.
Solutions Begin with Conversation
I can’t think of a stronger argument for taking your medication list—an actual, written list—everywhere. Hand it to every doctor, nurse, or pharmacist who asks. In my time at the clinic, the simplest, most effective intervention was a pharmacist reviewing all meds before a new drug started. That catch prevented several near-misses. Pharmacists track down rare interactions and explain risks in plain English.
Don’t feel awkward bringing up everything you take, even if it “doesn’t count” in your mind. Tea, herbal drops, sleep aids from the grocery store—they all count when you’re dealing with something as sensitive as immune system therapy. If you have any doubts, ask your provider directly: “Does anything I take get in the way of this treatment?” Push for details. It’s your life and your peace of mind at stake.
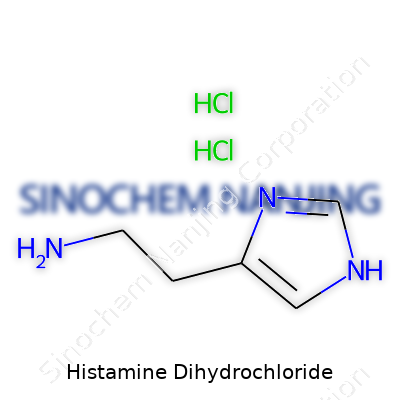
| Names | |
| Preferred IUPAC name | 2-(1H-imidazol-4-yl)ethanamine dihydrochloride |
| Other names |
Histamine dihydrochloride Histamine hydrochloride EINECS 200-465-7 Histaminum dihydrochloridum 2-(1H-Imidazol-4-yl)ethylamine dihydrochloride |
| Pronunciation | /ˈhɪstəmiːn daɪhaɪdrəˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | 56-92-8 |
| Beilstein Reference | 358691 |
| ChEBI | CHEBI:5977 |
| ChEMBL | CHEMBL1200902 |
| ChemSpider | 4384083 |
| DrugBank | DB06825 |
| ECHA InfoCard | 100.015.258 |
| EC Number | 1.4.3.22 |
| Gmelin Reference | 82145 |
| KEGG | C01533 |
| MeSH | D006638 |
| PubChem CID | 218295 |
| RTECS number | MW7850000 |
| UNII | E42ON78A2K |
| UN number | UN3467 |
| Properties | |
| Chemical formula | C5H11Cl2N3 |
| Molar mass | 184.07 g/mol |
| Appearance | White to almost white crystalline powder |
| Odor | Odor: odorless |
| Density | 0.82 g/cm3 |
| Solubility in water | Very soluble in water |
| log P | -2.2 |
| Vapor pressure | 0.0 hPa (20 °C) |
| Acidity (pKa) | 9.8 (pKa of the imidazole ring) |
| Basicity (pKb) | 10.04 |
| Magnetic susceptibility (χ) | -6.2×10⁻⁶ cm³/mol |
| Dipole moment | 0.89 D |
| Pharmacology | |
| ATC code | R06AX05 |
| Hazards | |
| Main hazards | May cause allergic reactions; causes eye, skin, and respiratory irritation; harmful if swallowed |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P264, P270, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 1-3-0 |
| Flash point | 160 °C |
| Lethal dose or concentration | LD50 oral rat 90 mg/kg |
| LD50 (median dose) | LD50 = 214 mg/kg (Rat, oral) |
| NIOSH | WFH42870 |
| PEL (Permissible) | PEL (Permissible exposure limit) for Histamine Dihydrochloride: Not established |
| REL (Recommended) | 500 mg |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Histamine Histamine phosphate Cimetidine Ranitidine Famotidine Diphenhydramine |

