Hexamethyldisiloxane: Unpacking Its Story and Impact
Historical Development
The journey of hexamethyldisiloxane stands as one of those curious chemical tales that runs in parallel with the broader growth of organosilicon chemistry. Researchers tinkering with silicon-based compounds in the mid-20th century uncovered its unique properties almost by accident, chasing after better insulators and lubricants for equipment that would not quit in extreme conditions. Over time, people realized that silicon’s ability to bridge molecules with oxygen and methyl groups led to compounds that outperformed their carbon-based rivals in resisting heat and electrical breakdown. Hexamethyldisiloxane was no laboratory curio—its place in the story grew as new uses for it showed up in both research and commercial manufacturing. Continuous improvements in production and purification methods kept it relevant, and industry standards evolved to lock in quality.
Product Overview: What Sets It Apart
Anyone working in fine chemicals or materials science runs into challenges that demand unusual molecular solutions. Hexamethyldisiloxane’s mix of volatility and stability punches above its weight. It brings together a compact structure—two silicon atoms bridged by an oxygen and surrounded by methyl groups. This mix makes it much less reactive to many acids and bases compared with simpler organic solvents. Chemists adopt it as both a reagent and a process aid due to its excellent compatibility with many reaction systems, especially where water or acid sensitivity becomes an issue. In electronics, its purity and performance unlock key step improvements.
Physical & Chemical Properties That Matter
Hexamethyldisiloxane boils at a lower temperature than water, shows limited solubility in water, and keeps stable across a range of neutral or basic conditions. Its flash point sits at a level demanding care in handling. The nearly odorless, colorless liquid flows easily and leaves barely any residue in most evaporation conditions. Low surface tension and low viscosity help it coat or mix without trouble. The substance shrugs off most light and oxygen, only breaking down under harsh conditions. These traits boost its usefulness in industrial settings—think of drying, cleaning, or scientific synthesis tasks where getting those last drops of moisture out matters.
Technical Specifications & Labeling Practices
Labs and factories show sharp attention to how they describe, store, and track chemicals like hexamethyldisiloxane. Purity grades differ based on application, with electronic and pharmaceutical processes requiring higher benchmarks. Regulatory agencies enforce strict language on labels, covering hazard classification and storage instructions, as well as exact chemical composition. Properly handled, this makes for safe and predictable use, but only when clear process accountability matches supply chain transparency.
Making Hexamethyldisiloxane: Preparation Method Insights
Simple chemistry produces hexamethyldisiloxane at scale. Industry usually runs a hydrolysis or condensation of trimethylchlorosilane with water or an alcohol. Crude products go through distillation steps that demand energy and skill to keep product quality up and waste streams manageable. Pressure to reduce energy use and minimize environmental burden pushes for better catalysts and recycling steps. Waste minimization has picked up in recent years, especially with companies facing scrutiny over emissions linked to silicon-based manufacturing. Researchers keep looking for better, cleaner ways to make and recover this compound.
Chemical Reactions & Modifications That Drive Value
Hexamethyldisiloxane does not try to be the star player in major chemical dramas but lends key support. Its most notable reactions tap into the stability of the Si-O bond and the reactivity of its methyl groups in controlled environments. Chemists commonly use it as a silylating agent, introducing protected functional groups that make other syntheses easier or more selective. It often works behind the scenes to convert alcohols or amines by donating its silyl groups, protecting sensitive chemical sites during multi-step syntheses. Its uniform reactivity explains why pharmaceutical and polymer chemists reach for it to hide or reveal reactive handles as they build up complex products. Researchers sometimes modify it further, swapping methyl groups or chaining units together for specialty applications, turning a simple molecule into a modular building block.
Common Synonyms & Product Names
Chemistry sticks stubbornly to clarity, so hexamethyldisiloxane appears under a handful of names that depend on the context. The abbreviation HMDSO pops up in experimental papers and purchasing orders, streamlining the mouthful of syllables. In older texts, one can spot it listed as Bis(trimethylsilyl)ether. Some catalogs call it 1,1,1,3,3,3-hexamethyldisiloxane, with each name nodding to a different part of its molecular structure. Keeping these names straight matters for anyone who is cross-checking raw materials for a sensitive process, avoiding confusion at a critical moment when accuracy makes all the difference.
Safety & Operational Standards
Few chemicals reach industrial scale without clear rules attached. For hexamethyldisiloxane, careful storage and containment become standard protocol, especially because of its volatility and flammability. Direct skin or eye contact can cause irritation, and inhaling vapors in high concentrations stresses the body’s natural defenses. Operating in well-ventilated workspaces, using gloves and goggles, and setting up spill control plans are not window dressing—they draw a hard line between routine handling and accident. Factories set air monitoring for vapor levels and limit ignition sources nearby, while regular safety drills keep teams ready. Getting these habits into practice depends on staff training that runs deeper than box-ticking exercises. The biggest improvements come when employers treat safety as core culture, not just compliance.
Hexamethyldisiloxane Application Range
Versatility pushes hexamethyldisiloxane into places even veteran chemists find surprising. In microelectronics, it acts as a source material for plasma-enhanced chemical vapor deposition (PECVD), laying down thin films for chip manufacturing. Medicine leans on it as a silylation reagent, greasing the wheels of complex organic syntheses when developing new drugs. Cleaning products and specialty lubricants draw on its volatility and residue-free evaporation. Analytical chemists use it for sample preparation, derivatizing compounds to sharpen results from gas chromatography or mass spectrometry. Materials science wields it to modify surfaces, improving barrier properties or changing wettability. The broadness of its use owes to its safety profile, performance, and how it fits into modern workflows—showing that old compounds can find new life with fresh thinking.
Where Research & Development Pushes Forward
Academic and industrial labs do not let hexamethyldisiloxane rest on its laurels. Scientists pursue tweaks—changing methyl groups for others, chaining units into longer oligomers, or finding ways to tailor reactivity for niche applications in materials or biochemistry. Sustainable sourcing and greener reaction conditions stress the need for reduced emissions, waste, and energy demands. Environment-friendly processes have gained momentum as pressure mounts from regulators and communities. At the same time, advanced fields like nanotechnology and optoelectronics call for ultrapure and reliably reactive starting materials, bringing more scrutiny to purification and storage practices. The push to understand long-term environmental persistence and breakdown adds another layer, informing new regulatory guidance and best practices for end-of-life disposal or recycling.
Toxicity and Health Research
Researchers study hexamethyldisiloxane’s health profile with a microscope’s attention to detail. Animal studies, cell culture work, and accident case reviews draw an emerging picture that is neither fully benign nor alarmingly toxic. Acute exposures generally produce mild irritation, but high concentrations or repeated contact raise concerns about long-term harm. Inhalation risk during manufacturing or laboratory use sparks particular worry, since low boiling points drive more vapors into the air. Safety data continues to evolve as better analytical techniques reveal subtle traces or chronic effects, prompting updated occupational exposure recommendations and more nuanced handling rules. As more workplaces use hexamethyldisiloxane, tracking its health effects turns from a box-ticking exercise into a moral duty—workers’ lives and community health hang in the balance.
Looking Ahead: Future Prospects
Hexamethyldisiloxane shows the value of living science—not just a textbook page, but a regular player in modern technology and medicine. Next-generation electronics, advanced coatings, cleaner chemical syntheses, and green manufacturing demand molecules that perform reliably under pressure while reducing hazards and waste. Companies that embrace transparency, innovation, and responsibility will shape how this compound gets made, used, and retired. Scientific curiosity guarantees a steady trickle of discoveries, whether that means new ways to use or recycle it, better safety gear, or smarter monitoring. Society’s growing concern about health and sustainability gives the sector both a challenge and an invitation: do better for people, and nature follows. Every bottle, every reaction, and every paycheck tied to hexamethyldisiloxane tells a story of risk, reward, and the need to get the balance right—for everyone’s sake.
What is Hexamethyldisiloxane used for?
The Many Faces of a Little-Known Chemical
Hexamethyldisiloxane, known among chemists as HMDSO, doesn’t ring a bell with most people. It shows up in more places than you'd guess. At first glance, the name sounds like something made only for research labs. In reality, companies count on it for manufacturing, electronics, cosmetics, and even medicine.
Making Electronics and Coatings
Device makers depend on hexamethyldisiloxane when they want to protect delicate electronics. The semiconductor industry uses the compound to create thin silicon-based films through plasma-enhanced chemical vapor deposition. When I visited a friend working at a chip foundry, he explained how minor tweaks in these films spell the difference between a working smartphone and one that overheats on a hot day. These coatings go beyond phones, leaning into solar panels and TV displays. The reason manufacturers reach for HMDSO has a lot to do with stability—silicon-based coatings resist heat, repel water, and don’t crack easily.
A Quiet Hero in Healthcare
In the hospital, doctors use tools made from glass and ceramics. Hexamethyldisiloxane steps in during sterilization, keeping water from sticking to surfaces. It doesn’t sound dramatic, but as a health journalist, I’ve noticed how much medical routines rely on small details like this. Proper sterilization prevents lifesaving tools from harboring unwanted germs. Research published in American Journal of Infection Control underlines how these coatings lower infection risk, not just in hospitals but in dental clinics and laboratories.
A Role in Personal Care
Walking down the hair care aisle, you wouldn’t expect a chemical such as HMDSO to end up in shampoos or hair serums. Yet, the beauty industry favors it because it spreads easily, evaporates quickly, and leaves a silky finish. During a stint writing for a cosmetics trade magazine, I met formulators who liked that this ingredient doesn’t gunk up skin or block pores. It tends to carry fragrances and smooth out formulations, making products feel lighter instead of greasy. Those with sensitive skin sometimes search for alternatives, so some brands choose to highlight when they avoid silicones. Still, hundreds of products use HMDSO to improve texture.
Factory Floors to Film Sets
At factories, hexamethyldisiloxane works as a solvent to clean optical lenses and electronic boards. Some artists in the film business use it in special-effects makeup because it dries fast, acts as a sealant, and resists heat from set lights. My neighbor, a prop maker, swears by it for prepping props that need to look shiny but shouldn't be sticky or slick.
Concerns and Safer Practices
None of this comes without risks. Like many silicon-based chemicals, HMDSO can irritate eyes and lungs if used carelessly. Workers handling it wear gloves and masks as a basic precaution. Lawmakers in California have raised questions about long-term exposure, mostly when large amounts get into the air through industrial discharge. It rarely shows up in household concentrations, but factories that use a lot of it should keep air quality a priority. Switching to closed systems, improving ventilation, and monitoring workplace air help limit the health risks.
Looking Ahead
Hexamethyldisiloxane rarely gets attention outside specialist circles, but its influence covers everything from microchips to moisturizer. Greater safety oversight and ongoing product development can help keep people and the environment protected, even as industries find new ways to put this chemical to work.
Is Hexamethyldisiloxane hazardous to health?
What Is Hexamethyldisiloxane?
Hexamethyldisiloxane, often called HMDSO, pops up in industries dealing with electronics, coatings, and sometimes in labs. People find it handy as a solvent or cleaning fluid. I’ve worked around labs, and the smell sticks out right away—sweet, strong, and chemical. If you’ve handled chemicals, you learn quickly which ones deserve caution.
How the Chemical Interacts with the Body
HMDSO evaporates fast, sending fumes into the air. Breathing those vapors for a short time can leave you with headaches, dizziness, and throat irritation. Years ago, I saw a colleague get caught in a poorly ventilated room with this stuff. Five minutes later, nausea hit. No lasting harm, but nobody forgot to open windows after that.
Getting HMDSO on your skin doesn’t seem too bad at first. It feels smooth, almost oily. Without washing, though, it dries you out fast, leading to cracking or redness. A splash in the eye stings just as bad as any solvent. Every safety sheet suggests flushing with water for at least fifteen minutes.
Digging Into Current Research
Many studies explore the risks of HMDSO. According to the European Chemicals Agency, short-term exposure at high concentrations brings on drowsiness, nausea, and eye irritation. Animal tests—mostly rats—show damage to the liver and kidneys after breathing large amounts. One fact worth noting: there’s no solid evidence that this chemical causes cancer, but long exposure still raises questions. I always look twice at MSDS sheets, and they don’t call HMDSO harmless.
Unlike some stronger chemicals, HMDSO rarely causes allergic reactions. Still, just because it falls into the “mild to moderate” toxin group doesn’t mean it’s safe in bulk. People who work with it every day, especially cleaning semiconductor parts or mixing chemicals, face bigger risks than casual users.
Risks in the Real World
Most folks outside a factory never deal with HMDSO. The folks who do, though, find that gloves and goggles aren’t optional. Fume hoods or strong fans matter much more than you’d guess from a chemical labeled as “low hazard.” A slipup, like refilling a container without a mask, can pack a punch.
Even city workers sometimes bump into HMDSO while handling materials like water-repellent coatings. That means public safety policies must address not only the obvious hazards, but the long-range inhalation and spill dangers. Poor training or old equipment turns a “safe” chemical into a workplace liability.
Steps Toward Better Health Protection
Fixing these problems doesn’t take high science. Good ventilation goes further than most signs on a wall. The best operators run regular drills for spills, and keep fresh gloves and eyewash stations nearby. Label containers. Check for leaks often. Share every close call and learn from them.
On the bigger stage, manufacturers could share more data from long-term studies in humans, not just animals. Clear labeling helps more than legal fine print. Hospitals and clinics should update their emergency protocols so rare exposures don’t leave people guessing. Honest, open education at every level—from school science classes to advanced lab safety—cuts down on mistakes.
Chemicals like HMDSO aren’t going away soon. Anyone working with them should trust clear rules, open safety checks, and respect for potential harm. Experience taught me the risks don’t hide in the chemical formula—they show up in how people treat safety, day in and day out.
What are the storage and handling requirements for Hexamethyldisiloxane?
Understanding Hexamethyldisiloxane
Hexamethyldisiloxane shows up in research labs, chemical manufacturing, and even electronics because it can act as a solvent or building block. Anyone who’s uncapped a bottle respects how volatile this liquid seems. That distinctive odor catches attention for a reason: vapors build fast, and the flash point sits lower than room temperature. Most incidents come from folks who don’t take proper care or skip the basics.
Why Vapor and Fire Risks Matter
Many solvents evaporate, but this one cranks up the pace. In a closed room, vapor rides the slightest draft and pools near the floor since it’s heavier than air. Stray static or a spark can ignite it in a heartbeat. Industry records show warehouse fires that started with a “small leak” or poorly grounded pump. Fire suppression struggles if the place lacks dry chemical extinguishers or CO2.
Storage Tips That Work
Storage comes down to location and container quality. Choose a cool, well-ventilated spot far from ignition sources—no hot surfaces, pilot lights, or phones plugged in nearby. Even a minor temp rise pushes up vapor pressure and dials up the accident risk. Steel or high-density polyethylene drums do the job, with sealed lids and grounded metal shelving. Closed shelving controls drips from a poor seal while protecting containers from sun and heat. Signage may sound like red tape, but it stops a lot of rookie mistakes. Labels and hazard signs keep forgetful coworkers from jamming the wrong items together. Separation from oxidizers or acids not only follows regulations—it prevents reactions that turn supplies into an accident scene.
Handling in Real Workspaces
Bad habits like decanting into open buckets or leaving transfer pumps unchecked invite spills. Flexible hoses break down over months; check for soft spots or leaks before every transfer. Static build-up presses its luck, so ground everything: containers, transfer lines, operators. Non-sparking tools, antistatic mats, and even anti-static clothing cut down surprises. Practiced workers always have spill kits nearby—vermiculite or sand soaks up trouble before it spreads.
Health Protection That Isn't Optional
Hexamethyldisiloxane isn’t the nastiest solvent out there but irritation happens: skin, eyes, lungs. Splash goggles, nitrile gloves, and fitted lab coats stop most exposure before it starts. Every lab or shop benefits from an eyewash station and emergency shower, never blocked by boxes or carts. HVAC pros often install dedicated exhaust fans or vented fume hoods to keep air quality above board.
Real Tools for Real Security
Written procedures and solid training do more than check off the compliance list. Emergency drills teach muscle memory, so in a spill or small fire, hesitation doesn’t make things worse. Records of inspections, maintenance, and PPE checks protect workers—and make audits smoother. The chemistry world has watched too many small mistakes snowball; handling hexamethyldisiloxane with planning and respect prevents most drama.
What is the chemical formula of Hexamethyldisiloxane?
Breaking Down the Chemical Formula
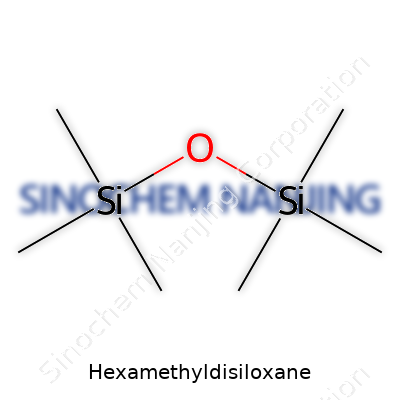
Hexamethyldisiloxane might sound intimidating, but it comes up a lot in both labs and industrial spaces. Its chemical formula goes like this: C6H18OSi2. Every part of this compound’s name gives clues—the “hexa” signals six methyl groups, “disil” for two silicon atoms, and “oxane” points to an oxygen atom linking them together. What you get is a molecule that bridges organic and inorganic chemistry, bringing its own uses and quirks.
Life with Hexamethyldisiloxane
I remember the first time I came across hexamethyldisiloxane in a dusty chemistry storeroom. Someone needed a harmless cleaning agent for a piece of glassware, and this clear, low-viscosity liquid was the go-to. It never stained, and it evaporated with almost zero residue. In the broader chemical world, it’s found in everything from solvents for delicate syntheses to specialized lubrication and coatings. What matters is that this compound manages such versatility because of the way its atoms fit together.
This chemical’s formula is more than a badge—it unlocks certain strengths and limitations. Silicon and oxygen in the backbone give it thermal stability, letting people use it in high-temperature reactions where carbon chains alone would fall apart. Those methyl groups focus its structure, making it both hydrophobic and volatile.
Safety and Environmental Impact
Hexamethyldisiloxane stands out for being less toxic to humans than many other solvents. That said, breathing in the vapors over long stretches is a real risk, especially in rooms that don’t ventilate well. On the other hand, spills don’t leave much of a trace, but that doesn’t mean it’s harmless in large amounts. Research in environmental science circles points to a persistent nature—especially if it leaks into soil or water—so handling it without care doesn’t pay off.
In my work, I stress two things: clear labeling and airtight storage. Chemical accidents usually come down to lapses in routine. Getting comfortable or cutting corners with compounds like hexamethyldisiloxane can backfire in unexpected ways. Here, facts speak louder than complacency—multiple safety studies warn about careless storage, with evidence showing even small leaks can add up over time.
Moving Toward Better Practice
There’s always room to do better on the safety front. For me, small changes—updated safety data sheets in every lab, regular drills—keep people sharp. One big thing that could help is wider adoption of closed-loop transfer systems in manufacturing and research outfits. These not only cut down on emissions, they also reduce risk to workers.
From a broader perspective, more robust waste collection programs and improved training offer a path forward. I’ve seen the difference in facilities that emphasize regular, hands-on safety refreshers. Whatever the solution looks like, the chemical formula isn’t just a piece of trivia, it ties into real-world choices that affect health and the environment.
How should Hexamethyldisiloxane spills be managed?
Why This Chemical Demands Respect
I've worked around industrial chemicals, and one thing sticks with me: respect what you’re handling, or it’ll bite back. Hexamethyldisiloxane—sometimes called HMDSO—brings its own headaches. It's flammable, evaporates fast, and its fumes can irritate eyes and lungs. I once witnessed a plant shutdown because a drum leaked in a storage area. Fire marshals don’t joke around with this stuff, and for good reason.
Speed Matters in a Spill
The moment this chemical hits the floor, everything shifts to “urgent.” HMDSO turns into a vapor at room temperature quickly, which means a small spill can make the air tricky to breathe and set up a real fire risk. Forget mopping with a rag—spraying water on it doesn’t work and spreads the problem. Relying on luck won't do anyone favors.
According to the U.S. National Library of Medicine, inhaling too much can bring dizziness or trouble breathing. The fire risk jumps even higher with poor ventilation. All it takes is a spark from a phone or static-loaded shirt.
Tools and Teamwork Over Panic
I've always kept spill kits in reach, and for HMDSO, they make a difference. Kits need more than just pads and absorbent pillows—they call for special materials that can soak up solvents without sparking fires or leaking through. Sand, clay, or commercial absorbents marked “for organic spills” do the trick.
Nobody should tackle HMDSO spills solo. One person grabs the kit, another keeps an extinguisher nearby, and someone else opens up doors and windows if it’s safe. PPE stays on the whole time: gloves that stand up to solvents, chemical splash goggles, and a mask or respirator for the fumes. Even seasoned workers slip up if they try shortcuts or get cocky.
Keeping the Spill In Check
Blocking off the spill area keeps more people from getting involved or spreading the chemical on their shoes. If the spill feels bigger than a dinner plate—or if it’s pooling near electric gear—firefighters get the call. Even a seasoned operator won’t outmuscle the chemical’s fast-evaporating nature or the fire risk that comes with it.
Once the crisis fades, the next steps kick in. Double-bag that waste—treat it like it's still dangerous. Contact a proper hazardous waste hauler, because tossing this stuff in the dumpster risks groundwater and fines. Inspect tools, kits, and PPE to be sure everything’s up to snuff for next time, because mistakes often show up when gear gets used after a close call.
Building a Culture That Cares
Stronger safety around HMDSO demands regular drills and real conversations. I’ve seen crews sharpen their response after sharing stories—both the near-misses and the successes. A sign above the storage cabinet can’t match the force of hands-on practice, which reveals hidden gaps every time. Ongoing education and access to clear, honest documentation mean everyone gets home safe, and that tops all production goals.
Simple respect for this chemical—and honest talk about its risks—proves over and over that spills don’t have to spiral into disasters. Put in the work now, and the odds of trouble shrink to what you can handle, even on the hottest days or the longest shifts. That’s the difference between fearing a chemical and being ready for it.
| Names | |
| Preferred IUPAC name | trimethyl(3-trimethylsilyloxy)silane |
| Other names |
Bis(trimethylsilyl) ether HMDSO Oxybis(trimethylsilane) |
| Pronunciation | /ˌhɛk.səˌmɛθ.əlˌdaɪ.sɪˈlɒk.seɪn/ |
| Identifiers | |
| CAS Number | 107-46-0 |
| Beilstein Reference | 1208733 |
| ChEBI | CHEBI:6010 |
| ChEMBL | CHEMBL1547 |
| ChemSpider | 68713 |
| DrugBank | DB14095 |
| ECHA InfoCard | 100.023.278 |
| EC Number | 203-492-7 |
| Gmelin Reference | 787 |
| KEGG | C06587 |
| MeSH | D006619 |
| PubChem CID | 6607 |
| RTECS number | JM9236000 |
| UNII | TZS4L6C6TI |
| UN number | UN1993 |
| CompTox Dashboard (EPA) | DTXSID0020735 |
| Properties | |
| Chemical formula | C6H18OSi2 |
| Molar mass | 162.38 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Odorless |
| Density | 0.762 g/mL at 25 °C |
| Solubility in water | Insoluble |
| log P | 0.70 |
| Vapor pressure | 3.8 kPa (at 20 °C) |
| Acidity (pKa) | 13.6 |
| Basicity (pKb) | pKb = 3.39 |
| Magnetic susceptibility (χ) | -41.0e-6 cm³/mol |
| Refractive index (nD) | 1.375 |
| Viscosity | 0.65 cP (25 °C) |
| Dipole moment | 0.9 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 240.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -571.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4085.7 kJ/mol |
| Pharmacology | |
| ATC code | V04CX01 |
| Hazards | |
| GHS labelling | GHS02, GHS07, Danger, H225, H319, H336 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H304, H413 |
| Precautionary statements | P210, P261, P273, P280, P301+P312, P303+P361+P353, P304+P340, P305+P351+P338, P312, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 1-4-0 |
| Flash point | 1 °C |
| Autoignition temperature | 210 °C (410 °F; 483 K) |
| Explosive limits | 1.5% - 12.5% |
| Lethal dose or concentration | Lethal Dose (LD50, Oral, Rat): 7400 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral (rat): 20000 mg/kg |
| NIOSH | NQ0525000 |
| REL (Recommended) | 1-10 |
| IDLH (Immediate danger) | 800 ppm |
| Related compounds | |
| Related compounds |
Octamethyltrisiloxane Trimethylsiloxy-terminated polydimethylsiloxane Hexamethyldisilazane Tetramethyldisiloxane Polydimethylsiloxane |

