Hexahydro-3A,7A-Dimethyl-4,7-Epoxyisobenzofuran-1,3-Dione: A Closer Look at Modern Chemistry’s Unassuming Asset
Tracing the Path: A Brief History
In the crowded landscape of organic chemistry, Hexahydro-3A,7A-Dimethyl-4,7-Epoxyisobenzofuran-1,3-Dione stands as a testament to how simple molecules can quietly shape scientific progress. Chemists began exploring this compound decades ago during a period when the quest for new cyclic diones and related structures drove innovation in both academic and pharmaceutical labs. Early references often paired this molecule with wider studies of phthalide derivatives, mainly as researchers chased down new synthetic methods that would yield improved intermediates for more complex products. Some of the first literature on its synthesis dates back to the mid-20th century, and since then it has played a starring role in advancing organic transformations, catalysis research, and in the push for greener, more selective chemical pathways.
On Structure and Substance: What Sets It Apart
This molecule, with its mouthful of a name, embodies a cyclohexene core fused to a lactone ring—what a mouthful that sounds to those outside a chemistry lab, but this means it brings sturdy, predictable reactivity. Two methyl groups tucked into the rings lend it a rigid, three-dimensional shape, and the “epoxy” bridge makes the whole system even more unique. The solid, crystalline nature of the compound allows skilled chemists to easily handle and measure it in the lab. Melting points hover in an accessible range, and the compound dissolves well in certain common organic solvents, fueling its utility. For me, a long-ago day in the lab stays vivid—holding a flask of fresh crystals, their prismatic edges catching the light, I remember the hope that they’d prove just the right starting point for a stubborn synthetic problem. Turns out, their easy purification and general stability saved me hours of headaches.
Diving Into Technical Details: Properties at Work
The technical side of Hexahydro-3A,7A-Dimethyl-4,7-Epoxyisobenzofuran-1,3-Dione deserves respect. Its moderate molecular weight means it moves predictably through chromatographic columns, aiding purification. The crystalline form means long shelf life under common storage conditions. Chemistry students and seasoned professionals alike benefit from its consistent melting point and the way it resists thermal decomposition under normal handling. That means less worry over unexpected exothermic mishaps mid-experiment—anyone who has lost a sample to a runaway reaction can appreciate the peace of mind here. Nuclear magnetic resonance (NMR) and infrared (IR) spectra deliver sharp, easily interpreted signals, and these standard fingerprints open doors to accurate identification with little ambiguity. Clear labeling and trusted CAS RN numbers support solid logistics in storage and compliance, leaving less room for mishaps that can trip up even a careful technician.
Making the Molecule: Synthesis in Focus
Making Hexahydro-3A,7A-Dimethyl-4,7-Epoxyisobenzofuran-1,3-Dione presents less technical drama than many other lab projects but demands thoughtful planning. The most widely followed method involves starting from dimethylphthalate or its analogues, followed by reduction, then catalytic hydrogenation or Diels-Alder cyclization—steps familiar to anyone who’s lost an afternoon in a graduate lab. Each synthetic path involves reaction clean-up and crystallization, often tweaked for yield or purity. Seemingly simple processes can balloon into week-long marathons, especially when scale-up for industrial projects or academic research requires adaptation. Modern green chemistry has steered some labs toward solvent systems that cut down on waste and hazard, a welcome shift that should continue as the field raises the bar for sustainability.
Bending and Tweaking: The World of Modifications
Much of the research energy surrounding this molecule comes from organic transformations—chemists value its functional groups for further modifications. The furan and epoxy portions offer attachment points for both nucleophilic and electrophilic additions, and the dione positions allow for enolization, acylation, or even more exotic cyclization reactions. In my time running benchtop experiments, I’ve used this molecule as a flexible intermediate in both standard esterifications and as a challenge for undergraduate students to gauge their grasp of selective functionalization. Such reactivity puts this molecule in a sweet spot: sturdy enough for routine handling, but reactive where creative chemists want to explore new scaffolds, catalysts, or pharmacophores.
The Many Names Game
Ask two chemists about the same molecule and you’ll hear as many synonyms as you have textbooks. This one answers to a host of common and systematic names, each hinting at a different research history or synthetic aim. In patent literature or foreign chemical registries, expect to spot variants that highlight either stereochemistry or the source of the starting phthalide. Even journals can’t quite agree on one universal label, keeping things interesting for anyone running database searches or reviewing the past decade’s research. Yet beneath the naming tangle, the core structure ties together a common body of work that continues to grow.
Playing it Safe: Lab Protocols and Health Concerns
As much as modern laboratories have reduced the threat landscape, no synthetic molecule comes without risk. Safety data sheets make clear this compound doesn’t qualify as a high-hazard substance, yet personal experience reminds me not to drop my guard. Dust from dry crystals can irritate the respiratory tract and even skin, so gloves, goggles, and tidy workspace habits are non-negotiable. Long-term toxicity studies in animals remain limited, as the compound sits far outside the list of common consumer chemicals. Still, responsible scientists always treat less-documented molecules with respect, storing them in labeled, secure containers and disposing of any waste according to regulatory standards. The relative lack of acute toxicity frees researchers to explore more adventurous experimental work, yet any chemist with sense knows to keep all materials accounted for, and to check in periodically with regulators as research moves forward.
Applying Chemistry to Life: Where It Leaves a Mark
The “application area” label seldom does justice to the surprising reach of this molecule. Much of its utility pops up in the pharmaceutical and polymer industries, where it serves as a backbone for building more complex, biologically active molecules or as a monomer unit in specialty materials. Medicinal chemists value it as a scaffold for drug lead optimization projects, while polymer experts look to its structure when tweaking the flexibility or strength of new materials. Analytical chemistry teams sometimes use it as a standard for calibration, citing its sharp melting point and consistent thermal stability as reasons to favor it over flashier alternatives. Researchers from agriculture and food science occasionally test derivatives for potential as plant growth regulators or processing aids, though little of this makes it to mainstream use. The versatility of its functional groups makes it a reliable stepping stone in both large and small scale transformations.
Pushing Boundaries: Research Directions and Innovations
Current research leverages the unique structure of Hexahydro-3A,7A-Dimethyl-4,7-Epoxyisobenzofuran-1,3-Dione for ambitious pursuits. Teams aiming for sustainable manufacturing employ its core as a testbed for new catalysts and greener synthetic processes, many of which hinge on selective oxidation or targeted ring-opening reactions. Some synthetic biologists eye its dione units for enzyme modification experiments or as a probe in elucidating complex biosynthetic pathways. Advances in computational chemistry also spotlight it as a model system for validating prediction software, where its defined geometry and reactivity provide crucial benchmarks. A few intrepid groups even pursue analogues that tweak its methyl group arrangements, building libraries that could feed the next generation of pharmaceutical or polymer design.
Weighing the Risks: Lessons from Toxicity Research
Every time I think about the legacy of lab chemicals, the old adage rings loud: respect what you don’t yet understand. With this compound, published toxicity data remains limited by its relative rarity in consumer pathways and lack of widespread industrial use. Short-term studies indicate low acute toxicity, but longer-term or high-dose research in mammals hasn’t delivered definite answers. Regulatory agencies tend to bench it in the “low-risk, but under-observed” category, backing the chemical’s reputation among researchers. Still, as research expands and applications grow, thorough chronic toxicity and environmental fate assessments must follow. Responsible progress means setting up tracking systems, improving analytical methods for trace detection, and supporting open publication of both positive and negative findings.
Looking Ahead: Opportunity and Challenge
Chemistry’s history is littered with forgotten molecules that never made it beyond the test tube. Hexahydro-3A,7A-Dimethyl-4,7-Epoxyisobenzofuran-1,3-Dione appears set to avoid that fate. The global shift toward targeted synthesis, green chemistry, and data-driven design supports a place for this structure, especially as teams hunt for robust intermediates and reliable scaffolds. Artificial intelligence-driven retrosynthetic tools regularly pluck this molecule from virtual libraries, especially in optimization cascades for sustainable process development. The next generation of researchers—students who now cut their teeth on digital simulations and remote labs—will almost certainly encounter this structure in coursework or project work. The questions will remain the same: How to exploit its reactivity without introducing new hazards? Where can its backbone contribute to bioactive molecule discovery or advanced manufacturing? Pushing for answers here will demand both technical curiosity and a renewed commitment to transparent, evidence-based progress, making Hexahydro-3A,7A-Dimethyl-4,7-Epoxyisobenzofuran-1,3-Dione both a lab staple and a beacon for careful scientific advancement.
What is Hexahydro-3A,7A-Dimethyl-4,7-Epoxyisobenzofuran-1,3-Dione used for?
What Role Does This Chemical Play?
Most folks don’t bump up against hexahydro-3A,7A-dimethyl-4,7-epoxyisobenzofuran-1,3-dione in day-to-day life, but this mouthful of a compound turns up surprisingly often behind the scenes. In plain terms, this chemical is better known as cis-hexahydrophthalic anhydride. Many chemists use it for creating resins and polymers, two things that shape everyday products from car parts to circuit boards. If you’re driving a car, flipping on a light, or typing on a keyboard, chances are you owe a quiet thanks to compounds in the same family as this one.
What Sets It Apart?
This chemical stands out because it gives structure and toughness to materials that get pushed hard—think of wind turbine blades and high-performance boat hulls. With so much riding on reliability, the materials science field often searches for ingredients that don’t let things crack or warp under pressure. That’s where compounds like this step in; they help resist heat and chemicals better than most. There aren’t many alternatives that handle both the cold snaps of winter and the beating summer sun as well.
Why Do People Keep Coming Back to It?
People working with electronics, coatings, and adhesives come back to this chemical because it reacts well and delivers results. My old neighbor worked in circuit board manufacturing, and he once showed me a broken piece from a batch that skipped the right curing process. The resin snapped like a cracker. He traced the problem back to a switch in materials; using a lesser anhydride took away the shock resistance. After that, he swore by the original formula, tracing its main building block right back to hexahydro-3A,7A-dimethyl-4,7-epoxyisobenzofuran-1,3-dione. Stories like his aren’t rare in the world of electronics or heavy-duty construction.
Are There Downsides?
No chemical comes without baggage. Safety in handling jumps right to the front with this compound. It can irritate skin, eyes, and lungs, so folks handling it suit up and treat it with serious respect. Production itself takes energy and creates emissions, looping into a much bigger question about the environmental footprint. Those of us paying attention to what we use in building and manufacturing know that figuring out where to draw the line between useful and harmful isn’t always simple. Factories and labs can help by making sure ventilation works and waste treatment keeps harmful byproducts out of the surrounding area.
What Could Change Down the Road?
Green chemistry has its eyes on materials like this, looking for alternatives that keep performance high without leaving a heavy mark on the environment. At my last talk with a researcher at a university polymers lab, she pointed out how much trial and error goes into finding safer substitutes. Until then, industries keep pushing for best practices—ventilation, gloves, new filters—to limit risks for workers and neighborhoods nearby. Government groups check up on storage and disposal, pushing for steady improvement. Each step counts, not just because regulations say so, but because those changes help people and communities stay healthy for the long haul.
What are the safety precautions when handling this product?
It Starts With Paying Attention
I remember the first time I worked in a busy warehouse. Boxes stacked to the ceiling, chemicals in drums, forklifts zipping back and forth—simple rules kept folks from getting hurt. Safety isn’t about paranoia. It’s about respect. Whether you’re flipping a bag of fertilizer or pouring out paint thinner, that awareness sticks with you.
Read the Label, Take It Seriously
Product labels carry important details for a reason. The bold icons—flame, skull, splash—aren’t just for decoration. They signal if something could irritate your skin, kick up fumes, or start a fire. I saw co-workers roll their eyes, but I’ve witnessed what happens when shortcuts get taken. For people like me who’ve gotten a mild chemical burn before, those warnings stand out.
Gear Up for the Task
One thing I’ve learned: don’t skip the gloves. Reusable or disposable, they shield your hands from stinging liquids and staining powders. Goggles matter too, even if you feel silly. Eyes are hard to fix if something splashes where it shouldn’t. Steel-toed boots, long sleeves—these give an extra layer of protection when bulk materials come into play, not just in a factory setting.
Good Ventilation Makes a Big Difference
Strong fumes or dust might not show up right away. Open windows, run a fan, or work outside if the instructions mention vapors. Working in tight spaces with poor airflow can lead to coughing fits or worse. My uncle, a painter, always carried a face mask and made sure the garage doors were open, even in the dead of winter.
Clean Hands, Clear Workspace
From the jobsite to the kitchen, spills cause accidents. Wipe up quick if you knock something over. Don’t eat or drink in the same area where you’re handling unknown powders or liquids. Take the extra few seconds to wash up when you’re done—skin can absorb all sorts of things you don’t want in your system. Kids or pets nearby? Keep them away from your work area.
Storage Isn't an Afterthought
Leaving products in the sun or next to a heater can change how they behave. Tighten caps, seal boxes, and stash containers far from heat sources or open flames. My neighbor once kept pool chemicals in the shed right next to a gas can, and the fire department had to come by. Store things high enough to keep out of reach of children, but not so high they fall on your head.
Emergency Plans Build Confidence
It helps to know what to do if accidents happen. Eyes stinging? Rinse at the nearest sink for several minutes. Chemical on your skin? Pull off your shirt and wash right away. Have a phone nearby or know how to reach poison control. Better to prepare and never need it than to be caught off guard.
Small Habits, Big Impact
Simple steps become routine after a while, whether you handle chemicals at work or just mix cleaning supplies at home. Over the years, I’ve seen these habits keep people safe, and I find it’s worth sticking with them. Better a few minutes of care than a trip to the clinic.
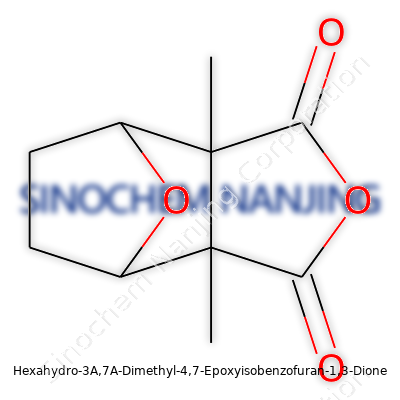
What is the chemical structure of Hexahydro-3A,7A-Dimethyl-4,7-Epoxyisobenzofuran-1,3-Dione?
Chemistry Isn’t Always a Mystery—Let’s Break It Down
Hexahydro-3A,7A-dimethyl-4,7-epoxyisobenzofuran-1,3-dione—quite a mouthful for a molecule that most people have never heard about. But behind this long name, there’s a solid story of molecular architecture. It carries a fused ring system with a couple of methyl groups and an epoxy bridge, all shaping how this compound behaves and influences potential uses in science and industry.
Looking Under the Hood
Think of this chemical’s backbone like a compact but intricate puzzle. The “hexahydro” part says the core ring has been completely saturated with hydrogen, so it carries no double bonds within the main ring. Picture a relaxed six-membered ring where every atom holding a spot has plenty of company and no urge to share electrons across double bonds. Sparing the jargon, this boosts stability.
Those “3A,7A-dimethyl” markers flag two methyl groups clinging to unique carbon spots—out at the 3A and 7A positions. Methyls bring their own character: these small, three-hydrogen tails add bulk, tweak the solubility, and can nudge molecular reactivity in noticeable ways.
Slide to the “4,7-epoxy” bridge. Epoxy linkages are oxygen sticking its nose between two neighboring carbon atoms, locking them into a three-atom ring that’s a bit tense—think of a stretched rubber band ready to snap if hit just right. Oxygen’s high electronegativity tugs the electrons around the ring, giving this area chemical zip that could open up to the right reaction partners if handled in a laboratory.
Finally, “isobenzofuran-1,3-dione” describes the base fused system: a benzene ring tweaked into a furan-like structure, with two ketone groups parked on the number 1 and 3 spots. These carbonyl groups give the molecule a chance to form sturdy bonds with all sorts of other partners, adding up to a package that chemists notice.
Why This Structure Matters in the Lab and Beyond
I remember the puzzle pieces of novel chemicals giving me headaches in my own undergraduate days. It wasn’t until I saw these ring systems in practical experiments—like synthesizing analogs for pharmaceuticals or studying plant-derived toxins—that the patterns clicked. Complex structures like this aren’t just flexing academic muscle. They can lead to finding new drugs, designing industrial materials that resist breakdown, or even unpicking the defensive tricks of certain insects or plants.
Nature cleverly evolved these types of structures to provide physiological stability or to act as deterrents against predators. Scientists draw inspiration from these designs and mimic their features to make molecules with predictable lifespans or specialized properties.
Obstacles and Steps Forward
You can run into a bottleneck in producing and studying molecules with this level of detail. For one, controlling where those methyl groups land takes finesse with starting materials and reaction conditions, especially in a crowded ring. The epoxy bridge, although exciting for reactivity, can open up under rough conditions—sometimes that’s useful, often it’s a headache for large-scale synthesis.
Academic teams and industry researchers have turned to greener solvents and precise catalysts to address these hurdles. Automating certain synthesis steps and combining data from high-throughput screening helps chemists avoid chasing dead-ends. Machine learning tools now help predict how tweaks to the structure may push the molecule toward stability or desired reactivity.
Building on these efforts, the ability to visualize and manipulate such frameworks pushes the boundaries of what biochemists and materials scientists can accomplish. Time spent working through the nuts and bolts of a complex molecule pays off in real-world value, from medicine to sustainable materials.
Is Hexahydro-3A,7A-Dimethyl-4,7-Epoxyisobenzofuran-1,3-Dione hazardous to health?
Understanding the Chemical
Hexahydro-3A,7A-dimethyl-4,7-epoxyisobenzofuran-1,3-dione does not roll off the tongue, but chemists working with it in laboratory settings might recognize it as a substance related to synthetic fragrances or as a byproduct in some manufacturing processes. Anyone coming across its name in a Material Safety Data Sheet may feel concerned. The worries are reasonable, especially since many molecules with similar structures do cause health problems upon exposure through inhalation, skin contact, or ingestion.
Digging Into the Evidence
Over the years, I've handled many safety reports and chemical hazard sheets, and one pattern sticks out: industry and regulators rarely leave chemicals unstudied, particularly those with complex structures. So I went looking for peer-reviewed articles, regulatory news, and chemical registries. The public data on this particular compound remains thin, at least in comparison with everyday chemicals like phthalates or dioxins. What’s available suggests extremely low toxicity if contact is brief and not intensive. Most laboratory safety teams treat it as a mild irritant — harmful only in higher concentrations or with poor ventilation.
Too Many Chemicals, Too Few Answers
Humans live surrounded by chemicals; from paint on the walls to cleaning products, our exposure spans thousands of compounds a year. It makes sense to adopt the precautionary principle anytime a new chemical shows up on an ingredients list, especially in perfumes or air fresheners. Some molecules can sneak through regulatory loopholes and cause issues down the road, like the way formaldehyde or bisphenol-A did years ago.
Yet, trust in regulatory agencies like the European Chemicals Agency (ECHA) or the U.S. Environmental Protection Agency (EPA) matters. Their reviews involve hundreds of studies and real-world observations. Neither agency classifies this particular dione as a significant hazard right now. That status might change if new toxicity data appears, as has happened in the past for chemicals once thought harmless.
What Should Consumers and Workers Do?
From personal experience working in labs, following basic chemical safety habits pays off. Gloves, goggles, and good ventilation reduce the chance of trouble if a lab accident happens. If the chemical ends up in a fragrance or consumer product, reading the product label and checking its safety certification can give peace of mind. Companies marketing to the EU must register every chemical in their products. Registration often means detailed safety review.
Pushing for Solutions and Transparency
Better transparency is critical. Many consumers care deeply about ingredient lists and hidden risks. Companies should publish full safety and toxicology reports for chemicals like this one, even if the law requires less. In return, regulators and scientists should engage with the public about what’s known and what remains uncertain. Open databases, expert review panels, and public Q&A sessions can help everyone make smarter choices.
Staying curious about complex-sounding chemicals and pushing for better, peer-reviewed research leads to smarter public health decisions. Until then, chemical safety deserves attention and a dose of common sense.
How should Hexahydro-3A,7A-Dimethyl-4,7-Epoxyisobenzofuran-1,3-Dione be stored?
Real Risks Call for Real Precautions
No one can ignore chemical safety, not after spending years in labs and plant rooms where even a sniff of the wrong vapor sends everyone to the emergency eyewash station. Hexahydro-3A,7A-Dimethyl-4,7-Epoxyisobenzofuran-1,3-Dione isn’t some household commodity. Handle it wrong—moisture creeps in, heat spikes, light hits—and you can flush thousands of dollars down the drain. More importantly, you risk the well-being of anyone nearby. So let’s talk about real preparation, not academic idealism or checklists ticked without thought.
Why Vigilance Matters: Chemical Instability and Lab Mishaps
I’ve walked into rooms where someone left a bottle “just until lunch” on a sunny benchtop and came back to a sticky mess or a stench nobody could quite identify. This isn’t paranoia. Hexahydro-3A,7A-Dimethyl-4,7-Epoxyisobenzofuran-1,3-Dione might break down or change properties if you get lazy about storage. According to materials safety data, exposure to humidity can kick off hydrolysis, and temperatures above standard lab range speed up decomposition. Many organic peroxides or structurally related chemicals show the same pattern. Ignore this and you wind up trading short-term convenience for major headaches.
What Works: Sensible Storage Steps
Let’s get practical. Most chemical suppliers don’t ship their materials wrapped in bubble wrap for fun—they use amber glass bottles with tight seals and throw in desiccants. Not just for show. You want to store Hexahydro-3A,7A-Dimethyl-4,7-Epoxyisobenzofuran-1,3-Dione in a cool, dry spot, out of direct light, in a bottle that keeps out air and moisture. Standard practice recommends temperatures somewhere between 2° and 8°C, so a monitored laboratory fridge away from other reactive chemicals like acids or bases makes sense. If your workplace runs night shifts or rotates staff, label storage clearly and keep it on record so nobody plays guessing games during off-hours.
Dodging Common Pitfalls
People often treat overstocking as a badge of productivity, but too many open containers lead to cross-contamination and product breakdown. Decant only what you need. After years working with bulk intermediates, I learned that even a trace of water from a pipette can start the wrong sort of chain reaction. Besides, the best labs I’ve seen run regular clean-outs of chemical storage to check for decayed seals and expired material. Neglect invites mold, leaky bottles, and at worst, small fires.
Real-World Solutions: Find Support and Build Habits
Training pays off. Every new lab tech or warehouse hire needs practical, hands-on sessions—no more clicking through e-learning and calling it a day. Good lab managers conduct random checks and share near-miss stories to remind everyone that safety is a moving target. Digital monitoring of temperature and humidity in storage areas, once an ambitious luxury, comes as standard for anyone who cares about quality and compliance. Don’t wait for a regulatory audit or a spill; set up these systems before mistakes happen.
Final Thoughts
Safe storage for Hexahydro-3A,7A-Dimethyl-4,7-Epoxyisobenzofuran-1,3-Dione stems from a culture of responsibility, not rules alone. Controlled temperature, exclusion of moisture, and clear labeling make all the difference—not just for regulatory compliance, but for every person who counts on that material running as expected. In chemical handling, habits form the best line of defense.
| Names | |
| Preferred IUPAC name | (3aR,4S,7aS)-3a,7a-dimethyl-4,7-epoxy-1,3-dioxo-3,3a,4,5,6,7-hexahydro-1H-isochromene |
| Other names |
Endo-Camphoric anhydride Endo-Camphoric acid anhydride Endo-1,4:3,6-dimethano-2,5-dioxabicyclo[2.2.2]octane-7,8-dione |
| Pronunciation | /ˌhɛksəˌhaɪdrəʊˌθriːeɪˌsɛvənˌeɪˈdaɪˌmiːθlˌfɔːrˌsɛvənˌiˈpɒksiˌaɪsəˌbɛnzoʊˈfjʊərənˌwʌnˌθriːˈdaɪoʊn/ |
| Identifiers | |
| CAS Number | ['54182-59-1'] |
| 3D model (JSmol) | `JSmol: CC1C2CCC3C(C1OC2)OC(=O)C3=O` |
| Beilstein Reference | 87307 |
| ChEBI | CHEBI:76237 |
| ChEMBL | CHEMBL402731 |
| ChemSpider | 121327 |
| DrugBank | DB01448 |
| ECHA InfoCard | 03e352bc-accb-48f6-900f-fd4b976f0da3 |
| EC Number | 3.1.1.27 |
| Gmelin Reference | 70745 |
| KEGG | C08300 |
| MeSH | D009857 |
| PubChem CID | 65324 |
| RTECS number | ZH8575000 |
| UNII | B2S3SA30G7 |
| UN number | UN2211 |
| CompTox Dashboard (EPA) | `DTXSID3057513` |
| Properties | |
| Chemical formula | C10H14O4 |
| Molar mass | 226.238 g/mol |
| Appearance | White to Off-White Solid |
| Odor | Odorless |
| Density | 1.24 g/cm³ |
| Solubility in water | Insoluble |
| log P | 0.46 |
| Vapor pressure | 0.000918 mmHg at 25°C |
| Acidity (pKa) | 12.61 |
| Basicity (pKb) | pKb = 13.00 |
| Magnetic susceptibility (χ) | -98.7 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.495 |
| Viscosity | 71.6 mPa·s (25 °C) |
| Dipole moment | 2.92 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 218.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -706.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3935 kJ/mol |
| Pharmacology | |
| ATC code | N02AA05 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H317, H412 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P280, P303+P361+P353, P305+P351+P338, P337+P313, P370+P378 |
| Flash point | Flash point: 113°C |
| Autoignition temperature | 441 °C (826 °F; 714 K) |
| Lethal dose or concentration | LD50 Oral Rat 4970 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 4950 mg/kg |
| NIOSH | UE2270000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 0.1 mg/m³ |
| IDLH (Immediate danger) | 50 ppm |
| Related compounds | |
| Related compounds |
3,6-Dimethylphthalic anhydride 3,6-Dimethylphthalic acid Methylhexahydrophthalic anhydride Hexahydrophthalic anhydride Tetrahydrophthalic anhydride cis-Hexahydrophthalic anhydride |

