Looking Beyond the Molecule: Hexafluoropropylene in the Modern World
Tracing the History of Hexafluoropropylene
Hexafluoropropylene, or HFP, stands as one of those stories in industrial chemistry that reminds us how innovation keeps old molecules relevant. This compound first emerged in the heyday of mid-20th-century fluorochemistry, born from curiosity about fluorine’s power to transform the chemical landscape. Early research opened doors to synthetic rubbers and specialty plastics, carving a niche for HFP among architects of modern material science. The drive to produce stable, high-performance polymers made this molecule more than just another laboratory curiosity. By the 1960s, HFP reactions fed directly into the birth of familiar products like fluoropolymers, offering resistance to harsh chemicals and extreme temperatures, traits that fueled the next generation of industrial advancement.
Understanding the Product Itself
HFP doesn’t show up in hardware stores or under kitchen sinks. Its value comes from how it shapes the backbone of high-performance materials. Take Teflon as one example—HFP acts as a key ingredient in making copolymers for nonstick coatings and sealing technologies. Beyond cookware, these materials factor into seals, gaskets, and cable insulation, where reliability isn’t just nice to have—it’s mandatory. Walk through any refinery or chemical plant, and the unseen but vital presence of HFP-derived materials shows up everywhere that hostile environments meet moving parts.
Looking at Physical and Chemical Properties
Chemists see HFP and think “reactive yet stable.” Fluorine atoms crowd around the propylene skeleton, giving the molecule unique sturdiness against heat and breakdown from acids or bases. This chemical backbone doesn’t just shrug off aggressive compounds; it laughs at temperatures that would ruin ordinary plastics. Yet HFP stays reactive enough to stitch into other chemicals, which turns it into the building block behind specialty fluoropolymers. Its volatility means handling demands tight standards, and its vapor pressure calls for storage a step up from casual flammable liquids.
Technical Details and Labeling Concerns
By regulation, HFP gets shipped and managed under serious protocols dedicated to hazardous compressed gases. Labels communicate flammability, the need for airtight containment, and a warning that even a fleeting leak can pose health and environmental risks. That’s not scare talk—good information protects workers and first responders who interact with shipments or maintenance points. The most experienced folks in the chemical business can recount stories of near-misses in handling or transferring HFP, so transparency in labeling and technical sheets isn’t paperwork—it's essential practice.
Preparation and Synthesis: No Simple Task
Commercial routes for producing HFP require careful choreography of temperatures and catalysts. One common method starts with the pyrolysis of polytetrafluoroethylene or similar fluorocarbons under tightly controlled settings. Yields improve with years of plant-level innovation, but the underlying message remains clear: making HFP on scale taxes reactors and operators. Every process tweak has environmental and safety implications. This synthesis highlights how industry builds on careful risk management and continuous investment, even for chemicals that never reach the consumer directly.
Chemical Reactions and What Comes Next
HFP’s utility comes alive in the reactions beyond its synthesis. Copolymerization with tetrafluoroethylene brings to life elastomers used in seals, gaskets, and specialty hoses that serve oil and chemical sectors. Chemists exploit its reactivity to tailor the balance between flexibility and chemical resistance in final products. Modifying HFP’s structure or how it links up with partners brings in properties that regular hydrocarbons simply can’t match. These modifications improve everything from flame retardancy to lifespan under punishing conditions, and push technology forward in industries where downtime gets measured in lost millions.
It Goes by Many Names
Walking through technical journals or regulation documents, you’ll find HFP listed as perfluoropropene or 1,1,2,3,3,3-hexafluoropropylene. Synonyms overlap depending on the context—sometimes reflecting subtle differences in formulation or purity that matter in research or production. If you’re in the business of procurement or process development, knowing the aliases pays off, especially as regulations shift and international standards diverge.
Meeting the Bar: Safety and Operations
Every process using HFP depends on a well-drilled safety regimen. Gaseous toxicity, reactivity, and flammability put workers on alert, so plant procedures aren’t theoretical—people rely on active ventilation, leak detection, and protective equipment daily. Training, not just compliance, shapes the difference between an incident and another safe shift. I’ve stood next to operators checking for leaks with soap solution and hand-held sensors, acting on experience as much as policy. It’s this blend of institutional memory and evolving safety standards that makes hazardous materials handling a constantly moving target.
Why HFP Finds a Home in Industry
HFP doesn’t chase headlines, yet its applications touch almost every field where reliability and resilience matter. In electronics, engineers use HFP-derived polymers for insulation, crucial as microchips shrink and power loads increase. In transportation, aerospace, and automotive companies bank on fluorinated materials for seals and hoses that never see the news unless they fail. Even green technologies bring HFP into the fold, as battery and fuel cell advances depend on stable coatings and membranes. Such reach wouldn’t exist if HFP didn’t bring real-world performance beyond the laboratory bench.
What Researchers Want from HFP
Research teams push HFP in new directions, chasing lower environmental footprints, greater recyclability, and even stronger resistance to degradation. In the lab, HFP offers a test bed for catalysts and mechanisms that could unlock cheaper or greener production routes, or turn difficult waste streams into reusable materials. The scientific literature mixes deep theory—about fluorine’s peculiar chemistry—with brute industrial reality: can the next tweak drop emissions, cut cost, or unlock new performance at scale? Such questions shape grant proposals and technology partnerships across countries and industries.
Digging into Toxicity
The story with any fluorinated compound includes a close look at toxicity, for both short-term worker exposure and longer-term environmental effects. Inhalation presents clear acute risks, driving standards for equipment, training, and emergency response. Chronic impacts track back to how the compound and its breakdown products move through soil and water, sometimes landing HFP and its kin in regulatory crosshairs. Toxicology studies aim to close knowledge gaps, but data sometimes lag behind application as industry races to fill market demand. Those invested in responsible innovation recognize that testing, not assumptions, builds trust with workers and the public.
Where HFP Goes from Here
HFP stands at a crossroads of society’s push for performance materials and its growing unease around persistent chemicals. Calls for greener solvents, circular plastics, and safer industrial processes ask more of every actor in the chain—from frontline workers to policymakers. The future of HFP points toward smarter systems for containment and recovery, more research into alternatives when possible, and relentless commitment to safety and transparency. Some trends, like the development of bio-based or degradable fluorinated materials, rest on how well we understand—and can control—chemistry at the molecular level. Progress never moves in a straight line, but history teaches that both caution and curiosity keep science advancing in the right direction.
What is Hexafluoropropylene used for?
Looking Past the Name
Hexafluoropropylene, or HFP, doesn't sound familiar to most people, but it's probably played a part in your life if you’ve ever handled a non-stick pan, worn a weatherproof jacket, or used a printer. Decades ago, I worked a summer job in a plastics factory. That place smelled sharp and strong, something like bleach and rubber mixed together. I couldn’t wait to get back outside. Only years later did I realize that complex-sounding chemicals like HFP shaped the goods that came off those assembly lines.
Why Manufacturers Count on HFP
People who work with chemicals talk about how HFP acts as a starting point for making other things. Chemists call it a building block. In everyday language, it’s like flour in baking. You use it to produce essential products, not as a final ingredient. The most recognized use connects straight to polytetrafluoroethylene, or PTFE. Most people know this as Teflon. You see Teflon on frying pans, cable insulation, and gaskets. HFP stands behind the surfaces that cook eggs without sticking or keep wires protected from fire and water.
It also shows up in fluorinated elastomers. Car engines and chemical plants rely on these high-tech rubbers. They keep their bounce and hold together even when temperatures dive below zero or engines run hot. Fluorinated elastomers don’t crack, swell, or lose shape like standard rubber. When car makers or engineers need seals or hoses that last, they often turn to materials born from HFP.
Shaping Electronics and Medicine
Technology moves quickly, and a lot of it relies on safety and durability. Printers, phone cables, and circuit boards all need insulation. HFP-based materials handle high voltages and block moisture far better than most plastics. Hospitals need devices that can survive chemical cleaning and intense sterilization. Tools made from HFP derivatives can be cleaned thousands of times without breaking down, lowering the risk of infection.
What Sets HFP Apart
Regular plastics melt, burn, or lose shape under stress. HFP derivatives hold their own. Fire resistance stands out as a key feature. In industries where sparks, heat, or exposure to strong chemicals can cause a disaster, HFP’s backbone is trusted. Think oil refineries, airplanes, or data centers where failure could mean costly shut-downs or even endanger lives. I’ve seen cheap gear fail in harsh environments. You can patch things together for a short time, but materials that simply give up under stress force you right back to the shop floor.
Reducing Risk, Seeking Alternatives
Every useful chemical comes with a downside. HFP doesn’t break down in the environment as quickly as some would like. Once, during a company training, an older engineer told me, “If you want a thing to last forever, you’ll have to deal with it sticking around forever.” Waste from fluoropolymers, including those made with HFP, stays in soil and water for years. Studies raise health concerns, especially around PFAS chemicals sometimes created during processing.
Some researchers push for greener alternatives or ways to recycle fluoropolymers. Factories invest in better filters and responsible disposal methods. Regulators set tighter limits on emissions. These changes cost money and take time, but change is possible. One small plastics company I knew switched to new catalysts to limit byproducts—a step in the right direction.
Taking Responsibility
Hexafluoropropylene shapes tools, technology, and kitchens. Experience shows that every material trades off risk for convenience. Understanding where real danger lies—and how to lower it—lands on everyone involved, from chemists to regulators to consumers. Small changes at the design and production level, carried out by people who see the entire life cycle, offer our best shot at long-term safety and success.
What are the physical and chemical properties of Hexafluoropropylene?
A Closer Look at a Modern Workhorse
Hexafluoropropylene, or HFP, doesn't come up at dinner tables, but it plays a big role behind the scenes. This is a colorless gas, faintly sweet in odor, clear as air, but far from harmless. Pipes, seals, and coatings in car engines and electronics rely on its tough nature. People might find its chemical name daunting, but the usage stays grounded and practical.
Toughness Starts with the Basics
Hexafluoropropylene's formula, C3F6, keeps the structure tight and stable. Under normal conditions, it looks like nothing — an invisible gas hovering at normal temperatures. It boils off at -29°C. This low boiling point demands sealed tanks and thick gloves to keep workers safe during transport and storage. HFP packs a punch with density, nearly three times the heaviness of air. This heaviness means leaks fall to the ground, sliding along floors and into corners, raising risks when mishandled.
Stubborn in the Face of Damage
Companies pick HFP when neither fire nor acid can break it down fast. Its carbon-fluorine bonds laugh at heat and chemical attacks. Most acids, bases, or oxidizers give up. High voltage or intense heat can split those bonds, but typical factory settings won't manage it. No surprise that the chemical industry leans on it to create high-performance plastics like Viton and Teflon, where regular hydrocarbons crumble.
Balance Between Utility and Danger
Every tough chemical brings some threat. HFP won't explode without serious prompting, but it doesn't play nicely with everything. Sparks or open flames can start trouble, and some decomposition produces nasty byproducts: hydrogen fluoride and perfluoroisobutylene, both toxic and dangerous to breathe in. Standard masks usually can’t filter out these fluorides, so safety procedures demand respect. In my work with industrial engineers, training always stressed the need for ventilation and sensors to catch leaks before people walk into trouble.
Riding the Innovation Wave
Environmental questions keep surfacing. HFP doesn't chew up the ozone layer like some older refrigerants, but it holds heat, sticking around in the atmosphere far longer than carbon dioxide. Its global warming potential runs high, making its careful management critical. Companies respond by capturing and recycling HFP where possible, but recycling comes with a price tag, challenging smaller operators pressed by tight margins.
Room for Smarter Practices
Mistakes with HFP don’t just end at the fence line. I've spoken with plant managers who lost sleep after pipe failures vented gas outside. Keeping joints, compressors, and pressure reliefs tight and up-to-date cuts down on risk. Some push for stricter regulations and industry-wide reporting so everyone keeps a closer eye on leaks. Even small efforts like thermal sensors, routine inspections, and retraining old staff make a difference. Sharing real-world fixes, not just lab quirks, helps everyone sleep better at night.
Final Thoughts
People rarely stop to wonder about the backbone chemicals propping up technology and transport. Hexafluoropropylene packs durability, risk, and opportunity into every molecule. Those making, moving, or using it never treat it casually; every ounce of caution pays off in safer workplaces and cleaner skies. Smarter habits still matter, even as technology marches forward.
Is Hexafluoropropylene hazardous or toxic?
Getting Down to What Hexafluoropropylene Really Means for Safety
People who work in chemical plants or around manufacturing lines know hexafluoropropylene by the smell or the label on the tank. Folks outside those circles rarely hear much about it. This chemical shows up mostly in industry, helping create fluoropolymers like Teflon, or used as an intermediate for many specialty products. The safety talk around hexafluoropropylene matters, not just for the handful of specialists who work with it every day. Neighbors around those factories, truck drivers, and even the environment can feel the impact.
What the Research Says About Exposure
Breathing in hexafluoropropylene doesn’t usually happen at the dinner table—it’s gases from leaks or from loading lines in chemical plants, mostly. Years back, regulatory agencies like the U.S. Occupational Safety and Health Administration highlighted that high concentrations can cause lung irritation and breathing problems. Even a short whiff at strong levels burns in the lungs. It can also knock people out if there’s enough of it in a closed space, since it displaces oxygen. That’s not some theoretical problem—workplace accidents over the years prove what happens when ventilation or safety training goes wrong.
Anyone who’s ever had to scrub out a valve and caught a noseful of unknown vapor knows the drill: clear the space, get to fresh air, call for help. The long-term impact isn’t so thoroughly mapped out, but no one signs up to test their luck on the job. Some lab animal studies showed repeat exposure can damage kidneys and lungs over time. Animal studies keep raising questions, and though the jump from rats to humans isn’t perfect, most employers treat it as a flashing red light. That’s common sense.
Hexafluoropropylene in the Environment
Hexafluoropropylene isn’t something you want in your backyard creek or the neighborhood air. The chemical breaks down slowly outdoors, especially if released in bulk from spills or leaks. It can travel far because it’s a gas at room temperature and doesn’t dissolve much in water. The breakdown process makes more persistent compounds, some of which last longer in the soil and water. These fluorinated breakdown products—often called “forever chemicals”—may hang around the environment for generations. Scientists link similar compounds to health concerns ranging from thyroid disruption to cancer. Regulatory pressure keeps inching up, especially in Europe and North America.
Real Solutions—Not Just Paper Promises
Safe handling comes down to more than hazard symbols and data sheets. Workers tell you that emergency drills, real respirator training, and working sensors matter just as much. Everyone from the shift supervisor to the truck loader benefits when equipment gets checked, alarms work right, and everybody understands what to do if things go south. Community groups want more than PR—full reporting and tougher controls near homes and water sources help restore trust. On a bigger scale, government agencies focus on stronger emissions reporting, better spill clean-up, and research into less-toxic alternatives.
At ground level, keeping hexafluoropropylene risks under control doesn’t fall on just one person or company. Regulators, communities, and workers all have stakes in making sure the damage stops before it starts. The smart approach keeps problems contained, lets workers breathe easy, and protects water and air far beyond the fence line.
How should Hexafluoropropylene be stored and handled?
Why Its Safety Story Strikes Home
Hexafluoropropylene, or HFP, shows up in plenty of modern industries. Think fluoropolymer production, specialty plastics, and even lubricants. If you spend any time in chemical plants, or you know someone who does, you quickly realize that HFP doesn’t leave a lot of room for error. Leaks can turn into serious hazards fast, and exposure can harm your skin, eyes, or lungs. I remember walking past a storage area one summer, seeing ice crystals on the valves and piping. It wasn’t the cold outside—it was the liquid inside. One small mistake, and things could have gotten dangerous quickly.
Storage: No Corners to Cut
Experience tells me that with HFP, every container counts. Steel cylinders or high-integrity pressure vessels—nothing less should hold a pressurized, reactive gas like this. Temperature rules come up for a reason. If a plant allows sunlight or heat sources near a tank, pressure builds and risks rise. Engineers I know stick with insulated containers, often tucked away in well-ventilated sheds far from traffic. They’ve seen firsthand what a runaway reaction can do, especially if valves wear out or unexpected jostling happens. Some companies even invest in automatic monitoring—pressure sensors, gas detectors—so there’s a warning before a leak turns urgent.
Handling: Discipline Matters
Personal protective equipment sits front and center. Sturdy gloves, face shields, splash-resistant suits—nothing fancy, just gear that covers skin and eyes. It’s not just busywork. Years ago, a careless moment—untested gloves—meant a coworker spent weeks in recovery. Even a short exposure to HFP vapor can irritate skin and lungs, so trained hands and a cautious pace make all the difference. That also means regular safety drills. Everyone must know what to do if an alarm sounds or if they spot frost building where it shouldn’t be. Nobody wants to scramble under pressure, hunting for a manual.
On-Site Safety Culture Shapes the Outcome
Accidents rarely come from a single mistake. More often, they pile up—rushed inspections, skipped checklist items, missing warning signs. I remember one plant manager making a point to review storage logs every week, checking for patterns in valve checks and gas meter readings. He knew that keeping the pressure in a safe range was not a ‘set it and forget it’ deal. Open communication between operators and supervisors helps too. Hidden leaks or faulty equipment don’t stand a chance if everyone looks out for them and feels accountable.
Solutions That Stick
Sites using HFP don’t need high theory; they need working practices. Regular training stands out most. New hires walk through safety routines until they can recite emergency steps by heart. Maintenance schedules never get skipped, and if a part seems worn, it gets replaced—no exceptions. Alarm systems and remote gas detection save time and, sometimes, lives. Safety data sheets stay close at hand, so nobody wonders what to do if a splash or leak happens. Collaboration with outside safety experts and local emergency crews also adds backup when rare accidents test a site’s preparation.
Handling HFP isn’t just a chemical challenge. It’s about discipline, culture, and practical tools working in sync. Experience teaches that respect for the material lowers risk—and keeps everyone involved out of harm’s way.
What are the safety precautions when working with Hexafluoropropylene?
Understanding the Risks
Hexafluoropropylene, or HFP, brings some tough challenges to people in labs and factories. It’s a gas under standard conditions and gets used for making fluoropolymers and other high-demand specialty materials. HFP won’t show much color or scent, so catching leaks or releases isn’t a simple job for anyone. Exposure means business: breathing even small amounts irritates eyes, nose, throat, and lungs while direct skin contact causes frostbite-like damage. Chronic exposure builds up risks—respiratory problems, headaches, and, at high doses, even worse effects.
The Right Way to Gear Up
Personal experience counts for a lot here. In my years handling chemicals, nothing replaces putting on the right gear every single time. HFP calls for splash goggles, full-face shields, flame-resistant long sleeves, and chemical-resistant gloves—usually made from Viton or PTFE. Forgetting even one piece brings up a tight feeling in your chest, especially knowing HFP penetrates many materials that usually keep other chemicals out. Ordinary nitrile or latex gloves break down fast. Flame-restraint also matters, since HFP burns with toxic byproducts.
Workplace Controls and Monitoring
Air quality should stay front and center. Relying on open windows or fans won’t do the job. Exhaust hoods, high-efficiency scrubbers, and leak-proof storage containers block the invisible risks. Installing real-time gas detectors offers the peace of mind that traditional “sniff tests” never could. I’ve watched teams learn this lesson after expensive false alarms from cheap sensors gave bad readings. Investing in proper detectors with alarms avoids accidents. Regular maintenance and calibration keep them honest.
Handling, Storage, and Transport
Storing HFP right each time matters as much as how someone uses it. Keep it away from heat, open flames, and sharp metal surfaces that might cut or spark. Secure, labeled steel cylinders locked down tightly stop accidental rolling and impact. At one plant, a poorly stored tank rolled into hot equipment—the results cost time, money, and trust. Detailed logs on transfers and storage locations save confusion. Trained staff with clear emergency plans keep the panic down if something goes wrong.
Emergency Plans and Medical Response
Accidents happen even when everyone does their part. HFP leaks can injure fast. Everyone in the area benefits from knowing exactly where emergency showers and eyewash stations live. Well-established communication lines with local medical teams who understand HFP’s effects buy precious minutes if someone needs real help. On-site workers get trained to help with burns, inhalation, or chemical splash. Rescue drills, followed by honest feedback, make the next response better every time.
Learning from the Field
I’ve seen attitudes shift after minor incidents made risks real. No one laughs off training after a co-worker leaves with chemical burns. Sharing lessons from mistakes helps everyone avoid repeating them. Safety culture thrives where leaders own their mistakes, admit gaps, and invite fixes from the team on the floor. Open talk beats short memos about safety rules. Tackling the risk of HFP works best when everyone believes their health counts just as much as the company’s production targets.
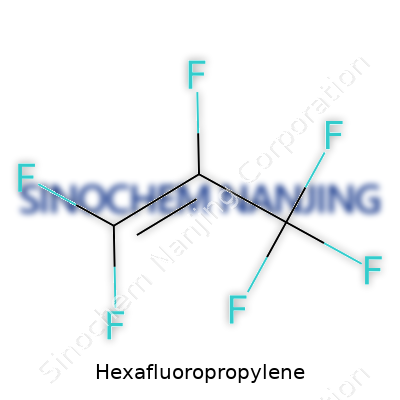
| Names | |
| Preferred IUPAC name | 1,1,2,3,3,3-Hexafluoroprop-1-ene |
| Other names |
Perfluoropropene 1,1,2,3,3,3-Hexafluoropropylene HFP Hexafluor-1-propene |
| Pronunciation | /ˌhɛk.sə.flʊə.rəʊˈprəʊ.pəˌliːn/ |
| Identifiers | |
| CAS Number | 116-15-4 |
| Beilstein Reference | 1201285 |
| ChEBI | CHEBI:35555 |
| ChEMBL | CHEMBL511121 |
| ChemSpider | 12161 |
| DrugBank | DB14096 |
| ECHA InfoCard | 03e859c2-5ebf-4e8c-8ee7-0fe414273d99 |
| EC Number | 204-104-9 |
| Gmelin Reference | Gmelin Reference: **110505** |
| KEGG | C13534 |
| MeSH | D006619 |
| PubChem CID | 7904 |
| RTECS number | UF9100000 |
| UNII | WE229BHZ1H |
| UN number | UN2192 |
| Properties | |
| Chemical formula | C3F6 |
| Molar mass | 150.03 g/mol |
| Appearance | Colorless gas |
| Odor | Gasoline-like |
| Density | 8.238 kg/m³ |
| Solubility in water | insoluble |
| log P | 2.09 |
| Vapor pressure | 2890 kPa (20°C) |
| Acidity (pKa) | pKa ≈ -10 |
| Magnetic susceptibility (χ) | '-9.71 × 10⁻⁶ cm³/mol' |
| Refractive index (nD) | 1.252 |
| Viscosity | 0.23 cP (25 °C) |
| Dipole moment | 0.27 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 348.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1226 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1356 kJ/mol |
| Pharmacology | |
| ATC code | V03AB38 |
| Hazards | |
| GHS labelling | GHS02, GHS04 |
| Pictograms | GHS04,GHS07 |
| Signal word | Danger |
| Precautionary statements | P210, P260, P273, P304+P340, P308+P313, P377, P381, P410+P403 |
| NFPA 704 (fire diamond) | 2-4-3 |
| Flash point | -78 °C |
| Autoignition temperature | 455°C |
| Explosive limits | Explosive limits: 5.5–15% |
| Lethal dose or concentration | LCLo (rat, inhalation): 5000 ppm/4H |
| LD50 (median dose) | LD50 (median dose): Rat oral > 8000 mg/kg |
| NIOSH | SN8710000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Hexafluoropropylene: 0.01 ppm (parts per million) as an 8-hour TWA (Time Weighted Average) |
| REL (Recommended) | 0.1 ppm (0.82 mg/m3) |
| IDLH (Immediate danger) | 45 ppm |
| Related compounds | |
| Related compounds |
Perfluoropropane Tetrafluoroethylene Chlorotrifluoroethylene |

