Hexafluoroethane: A Closer Look at Its Past, Present, and Where It’s Headed
Charting the Historical Development
Anyone diving into the story of hexafluoroethane notices its rise tracks with the explosion in the electronics and refrigerants industries during the 20th century. Chemists first synthesized this gas in laboratories interested in what fluorinated compounds could do. Decades later, people leaned on hexafluoroethane’s properties to solve practical problems in semiconductor production and medical imaging. My own introduction to hexafluoroethane happened in the early 2000s, as industry literature buzzed about the demands modern chip manufacturers faced. Clearly, its usefulness didn’t just arise by accident. Demands for more stable, inert, and effective gases shaped research and production, pushing hexafluoroethane from chemical curiosity to industrial staple. Tracing its applications, you find collaboration across companies and universities, each advancing the reliability and use cases for this persistent, nonflammable compound.
Product Overview in Industry Context
Hexafluoroethane’s role breaks out wherever high reliability and chemical stability take precedence. From etching silicon wafers to acting as a refrigerant under tough conditions, this gas has made itself essential. Its broad use in plasma etching reflects how industry grows more demanding as integrated circuits shrink. Where impurities might destroy a batch of microchips, companies choose hexafluoroethane for its robust purity standards. The world’s growing reliance on data centers and smart devices owes something to this quiet, colorless companion swirling through those fabrication plants. Not only does it make processes cleaner; it sidesteps a lot of the corrosion and reactivity issues that plagued earlier alternatives.
Physical and Chemical Properties in Focus
Hexafluoroethane isn’t just another colorless gas. Chemically stable even under strong electric fields, it shrugs off many aggressive chemical conditions that break down similar compounds. Its boiling point clocks in much lower than water’s, making storage and transport a challenge unless handled with the right pressure controls. Its chemical formula—C2F6—reflects six fluorine atoms tightly bound to two carbons, which accounts for its impressive thermal and chemical resilience. This kind of stability proves critical, especially in electronics manufacturing. The choices manufacturers make often come down to finding compounds like this, where high fidelity and low contamination allow precision that some call “mission critical.” Its low solubility in water and high density compared to air also leave a mark on how teams handle and store hexafluoroethane in the workplace, squeezing for both efficiency and safety.
Technical Specifications & Labeling from a Real-World Lens
In the workplace, you won’t see hexafluoroethane pass through unmarked cylinders or loosely tracked vessels. Regulations require labels tracing purity, batch origin, and chemical composition. From my experience in laboratory settings, these details can’t be skipped—the sharp eyes of safety officers and engineers have seen accidents when shortcuts tempt hurried hands. Standard specification sheets often demand at least 99% purity, and this consistency helps keep processes like semiconductor etching or refrigerant recovery running without interruption. Having worked alongside technicians who rely on clear labeling, I’ve watched fortunes saved by well-trained staff catching a mislabeled cylinder before it reached the clean room.
Preparation Methods: From Theory to Practice
Manufacturers typically synthesize hexafluoroethane using processes that pass fluorine gas over carbon-containing feedstocks, like chloroform or dichloroethylene, in the presence of catalysts. These controlled reactions squeeze out impurities, using either high temperature or specific catalysts to guide the outcome. Laboratories and factories must weigh the trade-offs of yield, byproduct management, and cost, choosing between direct fluorination and other routes, such as electrochemical fluorination. Facilities producing this gas often juggle the risk of releasing harmful byproducts, relying on continual monitoring and updated equipment to strike the balance between productivity and responsibility.
Chemical Reactions & Modifications: Navigating the Options
While hexafluoroethane’s main advantage sits in its reluctance to react, clever chemists have nudged it into transformations under extreme conditions—think plasma environments or catalytic settings. Breakdowns in arc discharges, for example, have been studied to reclaim valuable fluorine atoms. Some processes in research settings aim to split the molecule for specialized fluorination, while other teams experiment with recycling reactors that minimize emissions. The gas’s stability forces a creative approach, revealing new ways tech teams reuse or reconstitute fluorine-rich gases, keeping expenses and waste in check.
Synonyms and Product Names: Decoding the Language
Walk into a lab or browse a supplier catalog, and hexafluoroethane might show up as perfluoroethane, R-116, or simply C2F6. I’ve seen some old-timers in the field call it by a trademark or refrigerant designation, but most teams today stick with R-116 or perfluoroethane for clarity. While brand names exist for specific grades, knowledgeable technicians and buyers keep to these terms to ensure clear communication—nobody wants confusion over which fluorinated gas sits in that cylinder.
Meeting Safety & Operational Standards
Nobody working with hexafluoroethane can ignore the safety side. Although non-toxic and non-flammable, its ability to displace oxygen brings real risk of asphyxiation in confined spaces. I’ve worked in facilities where leak detectors, ventilation fans, and alarm systems run around the clock just to catch a whiff of any escaping gas. International regulations—like those observed by the semiconductor industry—require strict operational standards, frequent pressure checks, cylinder tracking, and enforced ventilation plans. Everyone on the shop floor picks up regular training, knowing that even a small mistake could expose a team to a silent hazard. Gloves, goggles, and face shields come standard, especially where pressurized systems and transfer lines cross paths.
The Breadth of Application Areas
Semiconductors and microelectronics represent the beating heart of hexafluoroethane usage today, where it etches features onto silicon wafers with microscopic accuracy. Beyond these clean rooms, the medical world taps into this gas for use in imaging (like ultrasound contrast agents), and researchers chasing novel refrigerants explore its lower-pressure operating points. During the last decade, climate researchers also began to track its growing presence as a greenhouse gas, pushing industries to work toward responsible use, capture, and destruction systems. This creeping focus on long-lived gases like C2F6 opened new areas in environmental engineering—carbon abatement teams and emissions specialists count every molecule to keep companies in step with tightening regulations.
What’s New in Research & Development?
Ongoing research efforts target both the continued use and the environmental challenge that hexafluoroethane presents. Scientists in integrated circuit design constantly test ways to make etching more selective, faster, and less wasteful. I’ve read recent papers describing combinations of perfluorinated gases, pulsed plasmas, or new byproduct capture systems, all promising lower environmental impacts while maintaining high output. Environmental scientists are now designing reactors to destroy C2F6 at end-of-life, instead of letting it escape to the atmosphere as a persistent greenhouse gas. Teams are also modeling how reuse and recycling infrastructure for rare gases could cut down both emissions and costs.
Looking Deeper into Toxicity Research
On the toxicity side, hexafluoroethane carries a reputation more for suffocation than for classic poisoning. Since it doesn’t get absorbed into tissues or cause chemical burns, the risks show up asphyxiation and frostbite in high exposures. Decades of exposure studies, both in workplace investigations and animal models, flag the importance of proper ventilation and leak detection. I recall sharing a lab with a specialist who always harped on oxygen monitors, never letting up on reminding new staff how odorless and invisible these risks could be. These routines may seem tedious, but enough incidents have shown what happens when operational discipline slips, especially in lower-ceilinged facilities or maintenance zones.
Future Prospects: Steering Toward Sustainability
The world can no longer treat fluorinated gases as disposable resources. Regulators, industrial players, and researchers now put energy into closing the loop on C2F6, capturing emissions and substituting less persistent alternatives when possible. Looking forward, I expect to see real innovation as companies and labs adjust their practices, maybe even working toward wider adoption of safer gases or greener recycling technologies. Progress here doesn’t just help the bottom line or regulatory compliance—it builds trust with communities and reassures a world concerned about the climate footprint of advanced tech. People working with hexafluoroethane, from manufacturers to disposal specialists, carry the responsibility to stay vigilant, blending technical curiosity with a clear eye on sustainability.
What is Hexafluoroethane used for?
Silicon Chips and Precision Cleaning
Hexafluoroethane sounds like something out of a lab, but it’s closely tied to everyday objects—particularly the computers and phones people use. Semiconductor factories use this gas during the making of chips because it etches silicon wafers with incredible accuracy. Etching helps create pathways and patterns embedded on microchips, which power everything from personal laptops to electric cars. Hexafluoroethane helps clear out minute debris or unwanted deposits where there’s little room for error. Based on manufacturing trends reported by the Semiconductor Industry Association, the demand for gases like this one grows each year, tracking with the rise in connected devices.
Plasma and Cleaning in the Electronics Industry
People who’ve walked into a cleanroom know dust and residue pose serious threats to electronics. Hexafluoroethane often becomes part of a plasma, used to clean not only silicon wafers but also the chambers and equipment that interact with these sensitive materials. Plasma cleaning using this gas removes contaminants at the molecular level, giving engineers the clean slate they need for next steps. Producers value consistency, so they reach for gases with stable behavior, and hexafluoroethane tends to deliver.
Medical Device Manufacturing
Surgical tools, implantable devices, and diagnostic pieces all require high standards. This gas offers a non-corrosive, non-flammable option in settings that can’t risk chemical residue. It manages to sterilize surfaces and eliminate potential for infection or malfunction. Regulatory agencies like the U.S. Food and Drug Administration set heavy rules about what materials touch medical devices; hexafluoroethane passes muster thanks to its purity and performance in controlled environments.
Refrigeration and Fire Suppression
On the practical side, hexafluoroethane also works its way into refrigeration technology. Certain commercial and industrial cooling systems use this gas as a refrigerant or as a component in refrigerant blends. Its low reactivity makes it manageable in closed systems that keep perishable items cold. Fire suppression is another field that exploits the gas’s properties. Unlike old halon-based fire extinguishers that attacked the ozone layer, hexafluoroethane doesn’t respond in a way that tears apart oxygen molecules, so it offers a safer approach in protected spaces like server farms.
Environmental and Safety Concerns
Despite its utility, I see a real need for caution. Reports from the Intergovernmental Panel on Climate Change show that hexafluoroethane holds a global warming potential thousands of times higher than carbon dioxide. So leaks, poor containment, or careless disposal create lasting damage. Any company using or disposing of this gas should take real steps to recover and recycle it, not just vent it into the air. Regulatory frameworks like the Kigali Amendment push the industry to use alternatives or tighter containment. Some manufacturers now invest in collection and destruction technology, or search for gases that offer similar performance with a lower climate price.
Room for Improvement
People who rely on technology rarely see the links — a mobile phone call or fresh fruit in the supermarket connects all the way back to choices made about industrial gases. While hexafluoroethane does a remarkable job in high-tech production and medical manufacturing, the hidden downside arrives in its climate impact. Strict leak detection systems, responsible end-of-life management, and the hunt for greener alternatives belong on the table for everyone invested in tech, medicine, or the planet’s future.
What are the safety precautions when handling Hexafluoroethane?
Understanding the Risks
Hexafluoroethane doesn’t show up on most folks’ radar, unless your job brings you to semiconductor factories, electronics labs, or refrigeration plants. This gas has no taste or smell, so it doesn’t warn you when it leaks. Breathing in too much can starve your blood of oxygen, and in higher concentrations, can knock you out or even stop your heart. A lot of people underestimate the risks because it seems so benign: clear, odorless, and cold. But that's exactly the reason small mistakes turn serious fast.
Real-World Precautions
Personal experience tells me that you need to respect this stuff in the same way you respect electricity: hands off unless you know exactly what you're doing. Protection starts with good ventilation. I’ve worked in labs where a fume hood hums all day–no one trusts the building’s regular air flow. It’s not paranoia. Closed rooms gather leaks, and because hexafluoroethane doesn’t smell, you could breathe it before realizing.
Next comes protective gear. Gloves, safety goggles, and face shields aren’t just for looks. Once, I watched a careless move send a cold blast of compressed gas across a bench. In seconds, frost formed on metal clamps and glassware. If that hits skin or eyes, you’re looking at frostbite. People often forget how fast these gases expand and freeze things.
Storing the cylinders the right way matters. Keep them upright, chained or strapped so they don’t tip over. One time, someone set a cylinder on a wheeled cart, with no strap, in a busy workshop. Fifteen minutes later, the cylinder rolled halfway across the floor, hitting a table. If the valve had snapped, that room could’ve turned into a missile launch site. I’ve seen it happen with other gases, and the memory keeps me cautious.
Reading the Signs and Responding Fast
If you handle hexafluoroethane often, get a portable gas detector. Modern detectors give early warning before the air hits dangerous levels. It’s tempting to skip this gear, especially for small jobs or quick setups, but early warning systems have saved lives. I’ve seen someone press an emergency stop button in time because an alarm started beeping out of the blue.
Know the symptoms of overexposure: dizziness, headaches, feeling weak or lightheaded. Train yourself and your team to spot these signs in each other. The moment someone looks pale or wobbly, pull them into fresh air and call medical help. Every minute counts. Even if you feel fine, don’t trust your senses alone. Hexafluoroethane doesn’t play fair by letting you smell trouble coming.
Training and Teamwork
Everyone who works with specialty gases has stories of close calls. Real safety comes from regular training, keeping calm under pressure, and looking out for friends and coworkers. No one likes endless drills, but running through emergency routines means people know what to do before panic sets in. It’s not about following rules for the sake of it–it’s about making sure everyone leaves work in one piece, every day.
So while hexafluoroethane sounds like just another chemical, don’t treat it like an afterthought. Respect the risk, gear up, stay informed, and watch each other’s backs. That’s how you come home safe.
What is the chemical formula and molecular weight of Hexafluoroethane?
Learning the Details: What’s Behind the Name?
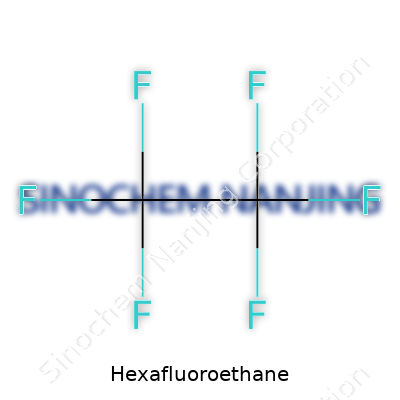
Hexafluoroethane doesn’t sound like something most folks talk about over coffee. Yet this compound keeps showing up in places you wouldn’t expect. Hexafluoroethane has the chemical formula C2F6. Its molecular weight registers at 138.01 g/mol. These numbers sum up a lot about where this substance fits into science and industry, but out in the world, they also tell a story about why we keep coming back to it.
Hexafluoroethane in Action
Plasma etching comes to mind. That’s the kind of job where precision matters. Semiconductor manufacturers rely on the predictable nature of C2F6, carving patterns only a few atoms wide. They use this gas to create the microchips that end up powering everything from phones to electric vehicles. Without compounds that behave exactly as expected, manufacturing would become a gamble, and chips wouldn’t function for long.
Working in a chemistry lab, I spent more hours than I’d like to admit studying what makes such substances tick. C2F6 played a role in more experiments than just chip-making. Its stability, low reactivity, and almost complete lack of odor meant researchers found ways to use it as a calibration gas and in refrigerants. I learned firsthand how much accuracy counts. Get the wrong gas or a mixture with unknown impurities, and entire experiments get tossed.
Looking at the Risks: Environmental Concerns
With all its uses, C2F6 also carries a heavy burden. It’s an extremely potent greenhouse gas. According to the Intergovernmental Panel on Climate Change, a single molecule of hexafluoroethane can trap thousands of times more heat than the same amount of carbon dioxide. It sticks around in the atmosphere for ten thousand years or more. Researchers keep a close eye on emissions for good reason. These gases don’t just disappear after being vented off a factory roof. I remember seeing the concern ripple in meetings about compliance with the Kyoto Protocol and new reporting requirements for fluorinated gas releases.
Approaching Solutions
Solving this environmental challenge takes more than just switching chemistry sets. Real progress shows up in recycling efforts at factories and adopting alternative etching techniques that use less harmful gases when possible. I’ve witnessed how staying ahead of regulations doesn’t always come cheap, but the push for better containment and recovery has fostered new technologies and jobs. With pressure from both governments and customers, businesses have become more accountable for their emissions, treating every kilogram saved as a win for the world.
Why C2F6 Matters
The formula C2F6 might look simple, but the conversations it sparks are anything but. Whether it’s the role in next-gen technology or the challenge of protecting our atmosphere, hexafluoroethane reminds us that chemistry never operates in a vacuum. Each molecule packs a punch far beyond its raw numbers, shaping decisions that reach into labs, factories, and up into the sky.
How should Hexafluoroethane be stored and transported?
Facing the Real Risks
Anyone working in industries like electronics manufacturing knows hexafluoroethane packs a punch. While often thought of as just another colorless gas on the production line, it comes with a set of challenges. Left unchecked, pressure or heat can turn this efficient etching agent into a genuine hazard. People who have spent any time in a facility that relies on this gas understand the nervous respect it commands.
Why Secure Storage Matters
Accidents remind us that cutting corners with compressed gases carries steep costs. Hexafluoroethane takes up space at room temperature, but it’s usually stored in thick-wall cylinders made from high-grade steel or aluminum. Ordinary storage or plastics just won't cut it—they crack, leak, or just fail. Keep these cylinders upright. I’ve seen what happens when one falls over—connections rattle loose, fittings shear, or worse: gas escapes and puts workers in immediate danger.
Isolation stands as the first line of defense. Put hexafluoroethane cylinders in a well-ventilated spot, not just any storage closet or shed. Fans and open airflow lessen the chance of gas building up if a small leak starts. Don’t store cylinders in direct sunlight, next to heating vents, or anywhere heat could build up. My first job in a warehouse involved monitoring temperature logs by hand, which made the threat of a heat spike stick in my mind. An overheated cylinder can burst, so keep it cool and shaded whenever possible.
Security around compressed gas cylinders isn't overkill. It’s common sense. Put up clear signs—no smoking, toxic gas, use gloves, eye protection. If your team works shifts, make sure everyone knows the safety layout, not just the night supervisor. Emergency shut-off valves and gas detectors go a long way, but only if everyone knows where they are and how to use them. I've witnessed drills where confusion reigned supreme; training drills really do matter.
Transportation Done Right
Moving hexafluoroethane between sites or across the country brings its own headaches. Secure every cylinder with real straps, not just a rope tossed over the top. Cylinders rolling loose in a truck can smash valves, cause leaks, or worse. The Department of Transportation calls for labeling and documentation for a reason, including those big-green diamond hazard placards plastered on the truck's side. Drivers must know exactly what they’re hauling and the potential consequences.
During transportation, a vented cargo area is key. Never cram cylinders into sealed compartments, trunks, or car backseats. Unexpected leaks in closed spaces can knock out drivers in minutes. If you think it’s safer to travel in cooler weather, you aren’t wrong, but never count on the weather staying friendly for the whole trip.
Check for leaks or worn fittings at every stop. A buddy of mine used a soapy water spray on all connections and spotted a hissing pinhole just before loading. Fast thinking avoided a major problem. Every stop along the chain requires eyes and hands-on checks—not just paperwork.
The Role of Experience and Science
No workplace wants to wind up as the subject of an accident report. The basics: cool, upright storage, routine checks, real training, and trusted hardware form the foundation. Science shows inhaling high levels of hexafluoroethane suffocates without warning. The Environmental Protection Agency and OSHA both set strict control limits: maintain air concentrations below 1000 ppm for worker safety.
Engineers keep searching for even sturdier valves, better leak detectors, and automated shut-off systems. Some facilities now employ real-time remote sensors connected to alarms—a huge jump over my early days walking the floor with a gas sniffer. Even the most high-tech system, though, only works when people use it correctly. Hands-on training and common sense can’t be replaced.
The Path Forward
Every worker deserves to go home healthy. Clear procedures, tough storage solutions, steady oversight, and good old diligence keep things running safely. Mistakes around gases like hexafluoroethane are rarely small, so give the process the respect it demands. If something looks off, call it out—no one regrets choosing caution.
What are the potential health and environmental hazards of Hexafluoroethane?
Why Hexafluoroethane Catches Attention
Hexafluoroethane often comes up in discussions about greenhouse gases and industrial chemicals. This synthetic gas lands in etching work in the electronics sector and also appears when making certain refrigerants. Working in a high-tech factory, seeing canisters stamped with unfamiliar names, you start to wonder what might drift unseen through the air. Most folks don’t wake up thinking about hexafluoroethane, but its footprint stretches further than the signatures in a lab notebook.
Breathing Concerns in the Workplace
Occupational safety really matters. Engineers and plant workers exposed to leaking processes or storage tanks risk inhaling this substance. Hexafluoroethane, if breathed in too much, can displace oxygen and provoke headaches, dizziness, or worst-case suffocation. A colleague once told me about a time the alarm triggered in the fab at 2 a.m.; nobody waited around to see how painful chemical gases could get before filing out. Short-term effects usually fade after fresh air, yet repeated exposure means compounding risk.
Official organizations like OSHA and NIOSH keep exposure limits for a reason. Testing for leaks, improving ventilation, and regular health checks save trouble down the line. Safety training should always cover more than the bare minimum. People at the frontlines deserve real-life info about what escapes from those glossy metal pipes.
Hexafluoroethane and the Environment
Hexafluoroethane sticks around in the atmosphere. Its chemical structure—two carbon atoms, six fluorine atoms—doesn’t break down quickly. Each molecule traps heat better than carbon dioxide by a huge margin. Just one ton pumped into the sky locks in heat as strongly as over 12,000 tons of CO2 over a hundred-year stretch. The consequences connect right back to climate talk. Rapidly rising temperatures, shouting headlines warning about wildfires, droughts, lost crops—much of that comes down to these emissions.
UN climate treaties and national rules treat this gas as a serious greenhouse threat. Tracking and reporting have grown better but not perfect. Big chipmakers and refrigeration suppliers now tweak old processes or hunt for less harmful chemicals. Still, total bans don’t arrive overnight. Scrapping out old gear brings its own challenges; leftovers still seep out if someone skips the right disposal steps.
Pushing for Solutions
Cutting down hexafluoroethane releases starts with equipment. Good maintenance means fewer leaks. Plant operators swap in sensors and keep logs because small unnoticed leaks add up. Firms that invest in real-time monitoring or increase staff safety training give the industry a reputation for responsibility. In meetings, I’ve pushed to see if alternative gases could replace fluorinated options—with some experiments falling flat, others opening up real hope.
Regulations can push companies to act faster, but building a culture of accountability matters most. Directors and engineers must admit where the dangers lie instead of looking the other way until someone gets hurt. Providing recycling stations and developing destruction technologies for used gases—like plasma treatment—show science gives tools to reduce risks. Customers and downstream partners can push by asking questions, digging beyond the nice brochures, demanding more environmental data.
Hexafluoroethane can’t hide behind a wall of jargon. Its risks—toward health and the planet—are plain. The biggest step comes from recognizing problems early and not letting them grow into bigger ones.
| Names | |
| Preferred IUPAC name | 2,2,2,2',2',2'-hexafluoroethane |
| Other names |
Ethane, hexafluoro- Perfluoroethane Freon 116 R 116 C2F6 |
| Pronunciation | /ˌhɛks.ə.flʊə.rəʊˈiː.θeɪn/ |
| Identifiers | |
| CAS Number | 76-16-4 |
| 3D model (JSmol) | `3D model (JSmol)` string for **Hexafluoroethane**: ``` C(C(F)(F)F)(F)(F)F ``` |
| Beilstein Reference | 1901267 |
| ChEBI | CHEBI:38837 |
| ChEMBL | CHEMBL17564 |
| ChemSpider | 7736 |
| DrugBank | DB02386 |
| ECHA InfoCard | 100.001.249 |
| EC Number | 204-646-6 |
| Gmelin Reference | 139 |
| KEGG | C13768 |
| MeSH | D006593 |
| PubChem CID | 6393 |
| RTECS number | KI5775000 |
| UNII | K1PB7U692H |
| UN number | UN2193 |
| Properties | |
| Chemical formula | C2F6 |
| Molar mass | 138.01 g/mol |
| Appearance | Colorless, odorless gas |
| Odor | Odorless |
| Density | 8.17 kg/m³ |
| Solubility in water | 0.026 g/L |
| log P | 0.78 |
| Vapor pressure | 5840 mmHg (20 °C) |
| Acidity (pKa) | ~-25 |
| Magnetic susceptibility (χ) | −30.8×10⁻⁶ |
| Refractive index (nD) | 1.233 |
| Viscosity | 0.0144 cP at 25°C |
| Dipole moment | 0.0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 337.0 J·K⁻¹·mol⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1220 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1541 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V03AB12 |
| Hazards | |
| GHS labelling | GHS02, GHS04 |
| Pictograms | GHS04,GHS05 |
| Signal word | Danger |
| Hazard statements | H220, H280 |
| Precautionary statements | P210, P260, P271, P304+P340, P312, P403 |
| Autoignition temperature | 550 °C |
| Explosive limits | Explosive limits: 5.1–14% |
| Lethal dose or concentration | LCLo (rat) 75000 ppm/2h |
| LD50 (median dose) | > 1700000 ppm (rat, inhalation, 4 hours) |
| NIOSH | NIOSH: KT6475000 |
| PEL (Permissible) | 1000 ppm |
| REL (Recommended) | REL: 250 ppm |
| IDLH (Immediate danger) | 8000 ppm |
| Related compounds | |
| Related compounds |
Tetrafluoromethane Octafluoropropane |

