Hexafluoroacetone Hydrate: More Than a Chemical Curiosity
Looking Back: The Road to Discovery
Chemistry turned a new page the moment fluorine chemicals came into play. Hexafluoroacetone hydrate didn’t jump into lab shelves overnight. The world started taking fluorinated ketones seriously around the mid-20th century, once folks realized these compounds didn’t just resist attack—they could help invent things that had simply never existed before. Academic groups exploring the reactivity of perfluorinated compounds kept bumping into hexafluoroacetone hydrate, noticing that it behaved differently from almost anything else they studied. Its story isn’t just about test tubes and flasks, though—it's also driven by needs that appeared in refrigerants, polymers, and high-performance materials. Practically every innovation with this molecule traced back to people experimenting and stubbornly trying to get more out of simple chemical building blocks.
Understanding the Stuff: Properties and Character
Anyone who's spent time with hexafluoroacetone hydrate will talk about its sharp, pungent odor. Its physical form usually comes off as a clear, colorless liquid, but what catches attention is the way it absorbs water easily, forming a hydrate almost instantly on exposure to moist air. This aggressive hydration marks a line between straight hexafluoroacetone and its hydrate—taste of moisture and the molecule latches onto water like glue. Chemically, the presence of three fluorines on each side of the carbonyl group sets up a volatile, highly reactive environment. Once inhaled, it can irritate the respiratory system, reflecting its potent action. The boiling point sits low enough that spills quickly turn to vapor, not something you want lingering without protection. Compare it to more familiar acetone, and you see just how much fluorine changes the rules.
Finding the Right Label: Names and Marketplace Confusion
Working in the lab means juggling synonyms like perfluoroacetone hydrate, or sometimes 1,1,1,3,3,3-hexafluoro-2-propanone hydrate. If you talk to people in industrial chemistry or material science circles, they might mention fluoroketone hydrate, especially if the conversation circles around applications. The confusing mix of names hints at this chemical’s spread across industries, and the tendency of research teams and suppliers to pick naming conventions that fit their own literature or commercial priorities. It’s easy to see why even professional chemists wind up double-checking labels before opening a fresh bottle.
On the Workbench: Making Hexafluoroacetone Hydrate
Producing hexafluoroacetone hydrate means grappling with tricky feeds of anhydrous hexafluoroacetone and precise water control. Industrial routes often rely on the oxidation of hexafluoropropene, a highly fluorinated olefin. This reaction uses catalysts—like palladium on carbon or silver nitrate—to help turn the propene into the ketone. Add controlled water at the right stage, and you get the hydrate. The yield, as with all fine chemicals, depends on care, skill, and a relentless focus on keeping things dry until hydration is really wanted. Any slip in moisture control means product loss or, worse, unmanageable hazards. In small-scale labs, researchers busy themselves with distillation setups under inert atmospheres, always wary of the substance clouding up with excess water vapor.
The Reactivity Question: More Than a Simple Ketone
People drawn to hexafluoroacetone hydrate often chase its high electron-withdrawing power. Each of the six fluorines pulls charge from the central ketone, setting up one of the strongest carbonyls around. This property gets exploited in a wide family of nucleophilic addition reactions, condensation steps, and even polymerization processes. It’s a favorite option for introducing fluorine tags onto other molecules, making it handy for pharmaceutical research where tracking or stability matters. In organic syntheses, the hydrate sometimes gets used as an oxygen donor or hydration agent, bringing new life to reactions that simply stall in the presence of ordinary acetone or standard hydrated ketones. Researchers have teased out more exotic chemistry too—modifying natural products by swapping out labile hydrogens for fluorine through the hydrate’s influence, crafting analogs with wholly new biological properties.
Keeping Work Safe: Hazards and Handling
Anyone experienced with hexafluoroacetone hydrate doesn’t need an incident to know respect comes first. This is a respiratory, eye, and skin irritant with a low threshold for causing harm. Even glimpsing the safety data sheets, folks see its acute toxicity: inhalation can trigger coughing, tissue burns, and sometimes delayed pulmonary effects. There’s a good reason for the serious gloves, face protection, and fume hoods you see in every lab working with this stuff. Training and vigilance matter more here than almost anywhere else. Industrial plants rely on scrubbers and sealed transfer lines, while researchers just trying to measure a few grams focus on minimizing any exposure at all. The standards for operational safety build on known toxicity data—no amount of experience excuses skipping full PPE, secondary containment, or fail-safes.
From Research to Real-World: Applications and Impact
Chemists don’t just marvel at the reactivity of hexafluoroacetone hydrate—they push it hard in the lab and industry. Its biggest claim to fame lands in polymer chemistry, especially the synthesis of polyimides and fluorinated copolymers with remarkable heat and chemical resistance. The aerospace sector pulls it into specialty coatings and matrix materials, desperate for anything that stretches the envelope of durability. Pharmaceuticals find a use where rugged fluorinated backbones slow breakdown in metabolic processes. Even in electronics, traces of this hydrate show up in the pursuit of stable, high-performance dielectric materials and etchants. Some researchers look for new ways to patch environmental weaknesses—trying to engineer the molecule into next-generation refrigerants or solvents that cut down on ozone depletion or climate impact, compared to older fluorocarbons.
The Toxicity Angle: Balancing Potential and Risk
Despite its high-tech charm, hexafluoroacetone hydrate forces a grim conversation about health. Studies trace acute exposures leading to tissue corrosion and, at sufficient concentrations, irreversible lung damage or systemic toxicity. Chronic effects remain less understood; animal tests hint at possible long-term organ impact, but there’s room for debate about dose, duration, and species sensitivity. Regulatory attention means constant evaluation for workplace exposure limits and environmental discharge tolerances. The tricky part comes when balancing cutting-edge application against possible public health risks—historically, underestimating fluorinated chemical hazards produced cases of severe environmental persistence and bioaccumulation. Some of the best minds in toxicology now train their focus on this class of chemicals, with active debates about better detoxification routes, improved PPE, and safe disposal.
New Ideas: Pushing the Chemical Frontier
Every year, new publications emerge hinting at previously unknown reactivity or application for hexafluoroacetone hydrate. Synthetic chemists chase elusive reaction pathways, always trying to build more complex, useful, or esoteric molecules. There's a growing push for green chemistry approaches: designing safer variants or using milder conditions to reduce risk without giving up the power of those six fluorines. Environmental researchers track breakdown, seeking ways to neutralize waste streams that hold onto the hydrate or its transformation products. Engineering teams look for new containment tech and more efficient water-scavenging methods to keep storage stable outside tightly controlled cleanrooms. Industry groups form coalitions to share best practices for handling and eventual disposal, pushing everyone toward less hazardous ways to get the benefits without the historical baggage of previous fluorinated chemicals.
Looking Down the Road: Where It Heads Next
A familiar tension sits between bold innovation and responsible stewardship for hexafluoroacetone hydrate. Material scientists, regulatory bodies, and frontline chemists all try to squeeze more performance from every fluorinated molecule without losing sight of long-term impact. The tool remains valuable—its presence in new semiconductors, high-performance fibers, and precision etchants ensures it won’t disappear from the conversation soon. Some researchers go after bio-based or partially-fluorinated alternatives, hoping to recover similar advantages at a lower ecological price. Industry has begun to confront legacy pollution with active remediation studies and more circular-thinking around process waste. The story, ultimately, swings between optimism for what new chemistry can solve and the humility learned from tough lessons about toxicity, persistence, and the unpredictability of large-scale adoption. People in the field know the work is never done: real progress comes by asking hard questions, sharing knowledge, and always remembering that innovation has to pair with accountability.
What is Hexafluoroacetone Hydrate used for?
The Quiet Force Behind Innovative Materials
Hexafluoroacetone hydrate enters the scene quietly, usually tucked away in specialized chemistry labs. Folks outside the chemical industry rarely hear about it. Yet, a lot of the products we use trace back to compounds made using this substance. It comes as a clear liquid and packs a punch with its reactivity due to the six fluorine atoms locked into its core structure.
Transforming Manufacturing
Many companies lean on hexafluoroacetone hydrate as an intermediate in making specialty polymers. These aren't the plastics covering soda bottles, but advanced materials built to stand up to heat, friction, and chemical exposure. I remember interviewing a polymer chemist who summed it up: "This molecule gives us the stability we need for high-performance coatings and membranes." Industries like electronics and aerospace bet on this kind of durability.
A prime example: polyimide films, used for insulating wires in jets and electric cars, need molecular strength and insulation. Hexafluoroacetone hydrate helps with the chemical backbone of these films. Its influence reaches into things like circuit boards, not just for insulation but for allowing ever-thinner components without burning out.
Pharmaceutical and Agricultural Impact
Beyond plastics, this compound steps into pharmaceuticals. Medicinal chemists often need building blocks that offer chemical resistance and unique reactivity. Hexafluoroacetone hydrate brings both to the table, introducing fluorine atoms that change how drugs interact with the body. Drug designers chase these interactions, hoping for better targeting or longer-lasting effects.
In agriculture, this chemical acts as a stepping stone to certain pesticides and herbicides. Fluorinated molecules often persist longer in fields and resist breakdown by sunlight or rain, so controlling their use becomes important. Regulators and chemists keep close tabs on these processes, weighing up the benefits and risks in terms of health and environmental outcomes.
Risks and Responsible Handling
Exposure to hexafluoroacetone hydrate can harm human health. Its vapor irritates eyes and lungs, and spills spell trouble for groundwater and soil. Every chemist working with this stuff wears heavy-duty gloves, goggles, and works in specialized hoods built to suck away harmful fumes.
Concern isn’t just about direct contact. Accidental releases—during transport or disposal—run up against local safety laws. Tracking and labeling rules stay strict for a reason. Some suppliers include instant on-site neutralization kits with shipments. That makes it easier to squash leaks before they spread. Inspection teams also check waste handling carefully, knowing that leaking barrels lead to regulatory fines and community outrage.
Shaping Smart Solutions
Chemists and manufacturers need to strike a balance between utility and safety. R&D teams search for alternatives, but few other chemicals deliver the same performance, especially for heat- and chemical-resistant materials. So, the focus shifts to handling—using closed systems, double-sealed containers, and better employee training. This approach protects both workers and the public.
Public transparency matters. Over my career, I’ve seen community pushback lessen when companies hold open-door sessions, showing people the safety steps and listening to their fears. If rules keep evolving and reporting stays honest, this compound’s value can thrive without putting health at risk.
What are the storage requirements for Hexafluoroacetone Hydrate?
Understanding the Risks
Every seasoned chemist learns fast that hexafluoroacetone hydrate is one substance you handle with real respect. This chemical, often found in research facilities and specialized manufacturing, brings a unique blend of volatility and reactivity. Its watery form—the hydrate—might trick newcomers into dropping their guard, but that would be a serious mistake. Stories float around about accidental releases causing respiratory irritation and serious corrosion, so careful storage isn’t just a checkbox—it's about keeping people and equipment safe.
Why Humidity and Temperature Matter
In my early days at the university lab, there was a storeroom where the air always felt slightly damp. We once stored a bottle of hexafluoroacetone hydrate there. Days later, the outer cap showed the first signs of etching. Water vapor will coax this substance into breaking down, releasing acidic fumes. From incidents like this, it becomes obvious: damp, warm storage introduces risk. The ideal storage environment stays dry and cool. Many sources point to temperatures below 8°C. Regular refrigeration offers one layer of safety, but dedicated chemical refrigerators stand out. Domestic refrigerators sometimes fail, but chemical-grade units add extra layers of containment and control.
Container Choices and Compatibility
After that early incident, we switched from simple screw-cap bottles to tightly sealed glass containers designed specifically for aggressive chemicals. No one trusts plastic with a substance like this—solvents often chew up plastics, and a slow leak leads to invisible vapor hazards. Containers with secure Teflon-lined seals prevent escaping fumes, and clearly labeled bottles (with hazard warnings) keep everyone alert. No fussy, generic containers here; only robust, purpose-designed vessels deserve a spot on the shelf.
Preventing Unseen Hazards
A bottle sitting quietly on a shelf causes no worry—right up until it leaks or reacts. To reduce surprise, placing compatible absorbents around the storage area goes far. Granulated carbon or inorganic absorbents, tucked into spill trays, help manage unplanned drips. Some labs set up small trays lined with chemicals that neutralize acids. It’s a small investment for peace of mind.
Ventilation and Isolation
Experience quickly teaches that shared storage invites problems. Hexafluoroacetone hydrate earns its own corner, away from acids, bases, and especially water-reactive metals. Dedicated ventilation keeps vapor buildup in check. If a lab lacks a chemical fume hood adjacent to storage, local exhaust ventilation reduces the risk of worker exposure—no one wants to find out what inhaling those fumes feels like. Regular checks keep the environment controlled and address small leaks before they matter.
Education and Emergency Planning
No amount of rules prevents mistakes if people don't understand the risks. Ongoing education—training refreshers, safety meetings, open discussions about recent accidents—keeps safety issues at the forefront. Spill response kits, eye wash stations, and emergency ventilation systems need regular testing. This builds trust, not just in procedures and devices, but in each other. Mistakes shrink when people share experiences and stay alert.
Building a Culture of Safety
Decades of stories—some scary, some minor—show that storing hexafluoroacetone hydrate well means keeping a sharp eye on basics: dryness, cool temperatures, compatible containers, and informed staff. The smallest details make the difference. That’s what separates a smooth-running lab from the ones people talk about for the wrong reasons.
Is Hexafluoroacetone Hydrate hazardous to health?
Not Just Any Industrial Chemical
You walk past a plant or research lab, see the word "hexafluoroacetone hydrate" on a label, and wonder—so what’s the deal with it? Plenty of chemicals earn a bad reputation, some of it deserved, some exaggerated. This one? It deserves a hard look. It’s used in the making of polymers, certain pharmaceuticals, flame retardants, and other specialized products. But the story behind those lab coats and plastic safety goggles tells plenty about risk and responsibility.
Breathing and Skin Contact: No Small Matter
Anyone who’s spent time around chemical facilities knows the feeling: itchy eyes, a scratchy throat, or that sour scent wafting from a spill. With hexafluoroacetone hydrate, those red flags matter. The compound releases fumes that irritate airways, bringing on coughing, shortness of breath, and, if you keep breathing it, real damage to the lungs. Getting the stuff on your skin isn’t much better. Rashes, burns, and sometimes blisters come up fast—a clear sign this isn’t a substance you’d want with bare hands.
Numbers Don’t Lie: Exposure Limits and Risks
Regulatory bodies set limits for a reason. The National Institute for Occupational Safety and Health (NIOSH) sets a recommended exposure limit for hexafluoroacetone at 0.1 parts per million over eight hours. For context, that’s a tiny number; just a hint of gas means someone's at risk. Inhaling more than that can land workers in the hospital with lung inflammation or worse. Chronic exposure raises the stakes, as some studies link the compound to liver and kidney damage after weeks or months on the job.
Just How Bad? What Does the Research Say?
Toxicologists point to the molecule’s structure—highly fluorinated substances often hang around the body and the environment longer than anyone wants. Research from several universities suggests long-term, low-level exposure could mess with organs beyond the lungs. The compound breaks down in the body to form substances just as nasty. Nothing about this reads like scare tactics; scientists have documented these routes in animals and flagged the same red lights for people.
Personal Story: Seeing the Effects Up Close
I worked a summer at a specialty plastics plant out of college, running clean-up after maintenance. We handled barrels that had warning stickers featuring hexafluoroacetone hydrate. Even suited up with gloves and masks, a spill during a valve change pushed two coworkers into the medical bay with breathing trouble and skin reactions. No one loved the paperwork, but after that, no one questioned why it took time to suit up and log every hazard. If you see folks sweating over safety data sheets, that’s not paranoia, just learned behavior from accidents they lived through.
What Makes a Difference? Common-Sense Safety
Factories and labs that use this chemical usually go heavy on the safeguards: local exhaust fans, full-face respirators, and strict training. Hazard communication—simple, ongoing, and clear—keeps everyone sharp. Managers who invest in good ventilation and skin barriers save money on lost labor and keep health complaints lower. Beyond safety gear, real improvement comes from culture: open channels for workers to flag leaks and near-misses, and bosses who take it seriously. In my experience, that’s still not the rule everywhere, but convincing stories beat thick binders of procedure every time.
Room for Improvement
Not every shop follows best practices all the time, and not all workers feel free to press pause and double-check the plan. Regular audits help, but nothing replaces training that feels real—hands-on sessions, stories from frontline workers, time to review what happens if things go wrong. Companies who put people before production numbers gain trust and avoid the slips that can lead to ER trips.
Bottom Line
Hexafluoroacetone hydrate poses real threats for anyone handling or working around it. Ignoring the danger invites harm that’s predictable and severe. With the right tools, training, and openness, teams can protect workers and keep a necessary but nasty chemical from turning a tough day at work into a life-altering event.
What is the chemical formula of Hexafluoroacetone Hydrate?
Breaking Down the Chemistry
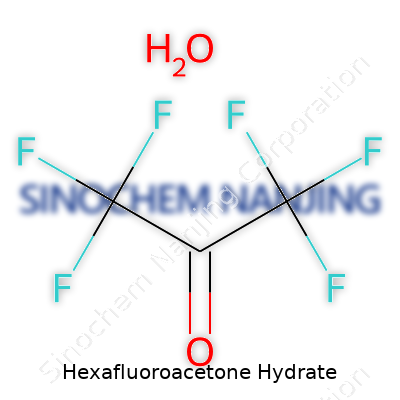
Hexafluoroacetone hydrate may not sound like something you keep under the kitchen sink, but dig into a chemical manufacturing facility or a research lab and you find it can be pretty important. Chemically, the hydrate means you pair water with hexafluoroacetone. That changes its behavior and reactivity. The chemical formula for hexafluoroacetone itself reads as (CF3)2CO. Add water, you get hexafluoroacetone hydrate: C3HF6O2. The water molecule links by attaching across the double bond of the acetone portion. It sounds technical, but it’s just an extra water hanging on and changing how the molecule acts in the real world. Hydration shifts its toxic profile and manages to make storage simpler and safer. That isn’t a small thing if you have handled strong reagents in the lab world.
Why Structure Matters in Real Situations
Hexafluoroacetone hydrate pops up mostly in specialty applications—think pharmaceuticals, polymer production, or advanced organic synthesis. The presence of hydration means you have a reliable way to transport and use a pretty good fluorinating or acylating agent without sacrificing safety. Dry hexafluoroacetone, as a gas or liquid, gets aggressive. Reliable hydration lets people move, store, and use this chemical with a lot less risk of leaks or toxicity. That practical shift from a risky, volatile reagent to something you can weigh on a scale with a gloved hand is a big deal for researchers and industry alike.
Toxicity: The Double-Edged Sword
Fluorinated chemicals require serious respect. I remember working through safety briefings that flat-out banned some fluorine reagents unless you could prove you needed them. Hexafluoroacetone fits that pattern. By itself, it can irritate the eyes, skin, and lungs. Hydrate it and you end up with something that’s less hazardous (though by no means safe for careless handling). Laboratories often opt for the hydrate form because the extra water stabilizes it and reduces vapor pressure—no clouds of toxic gas floating around. So, you have a material with industrial utility, but you need procedures, proper ventilation, and training to handle it right.
Tackling Safety and Environmental Challenges
Hexafluoroacetone hydrate offers something chemists like: control. The hydrate form isn’t exempt from environmental impact. Most fluorinated compounds resist breaking down and can accumulate. That persistent, bioaccumulative nature means accidental spills or improper disposal can cause headaches later, both for the environment and regulatory compliance. Safe handling starts with rigorous training and equipped storage—clearly labeled bottles, secondary containment, spill kits at the ready. Efforts to control run-off and emissions make a difference. In practice, labs and plants use closed systems, treat waste streams, and follow up-to-date regulatory guidance from agencies like the EPA or local equivalents.
A Broader Look: Innovation and Responsibility
Demand for specialty reagents like hexafluoroacetone hydrate won’t go away soon. These compounds move innovation forward in areas like new materials, electronics, and advanced medicines. Those advances walk hand-in-hand with growing calls for stewardship—using safer alternatives wherever possible, investing in greener chemistry, and designing smarter processes that reduce exposure across the board. Fixing legacy problems from persistent chemicals takes more than one breakthrough, it means constant vigilance and adaptation. Research, solid safety culture, and respect for these potent molecules steer the field in the right direction.
How should Hexafluoroacetone Hydrate be handled safely?
Understanding Real Hazards
Hexafluoroacetone hydrate isn’t something people use around the home. It shows up in labs and chemical plants with good reason—it packs a punch. This colorless liquid gives off a sharp odor. Skin, eyes, and lungs react badly to even a small exposure. The issue stems from its real tendency to get into the air and trigger health problems fast. Breathing in the vapors brings on coughing, shortness of breath, and long-term trouble for anyone taking safety lightly. A friend once forgot to double-check his respirator seal working with a similar chemical. He headed home sick that same day. Mistakes like that aren’t uncommon, so taking safety seriously matters.
Personal Protection Each Step of the Way
No one starts work with hexafluoroacetone hydrate before putting on the right gear. Nitrile or fluoropolymer gloves, safety goggles, and full-face shields become standard apparel. Lab coats or chemical suits keep the stuff from soaking through to skin. People handling this chemical count on working in a fume hood. Air flow carries fumes away before workers ever breathe them. I’ve seen coworkers forget to check for holes in gloves and pay the price with red, irritated skin. Double-checking gear stays second nature once you’ve watched someone scramble to rinse burns under an emergency shower.
Storage Demands Respect
Too often, chemical storage gets sloppy. Hexafluoroacetone hydrate needs stainless steel or glass containers with tight seals. Common plastics don’t hold up; this chemical eats through them. Each container sits in a secure, cool, dry spot away from acids and bases. Fire protection stands ready nearby because leaks or spills spell big trouble. Spills don’t offer second chances. I saw a near-miss when someone left a cap loose. A strong smell tipped us off before fumes built up. Only training and tools prevented a real emergency that day.
What to Do if Trouble Hits
Every lab using this chemical has plans drilled in for spills or exposure. Absorbent neutralizers, shut-off valves, ventilation fans—everyone knows what to grab, which buttons to hit, and where the showers stay. Speed means everything. Rinsing skin or eyes for at least a quarter-hour counts for more than any paperwork after. Calling for help comes next, and nobody should work alone in these settings. Once, I saw quick thinking make the difference between a scare and a hospitalization.
Long-Term Solutions and Training
One day of safety training doesn’t cut it. Review sessions and hands-on drills keep sharp edges razor-ready. Regular checks for leaks or rust around containers help spot minor problems before they get out of hand. Audits by outside experts catch gaps people nearby may miss. I’ve seen labs switch to smaller batch handling or look into alternatives with better safety records. Sometimes, working with less dangerous materials proves smarter, especially for teaching settings or smaller operations.
Responsibility Goes Beyond the Rules
Rules matter but don’t tell the whole story. People on the floor pick up unofficial skills—like spotting faulty seals or knowing the smell that means danger. Sharing those tips helps new team members avoid old mistakes. In chemical safety, looking out for one another beats any checklist. That’s a lesson I keep seeing reinforced in every lab and plant that handles tough stuff like hexafluoroacetone hydrate. In the end, real safety comes down to respect, teamwork, and never cutting corners.
| Names | |
| Preferred IUPAC name | 2,2,2-Trifluoro-1-(trifluoromethyl)ethane-1,1-diol |
| Pronunciation | /ˌhɛk.sə.flʊə.rəʊ.əˈsiː.təʊn ˈhaɪ.dreɪt/ |
| Identifiers | |
| CAS Number | 6840-30-4 |
| Beilstein Reference | 3563853 |
| ChEBI | CHEBI:38981 |
| ChEMBL | CHEMBL504660 |
| ChemSpider | 89315 |
| DrugBank | DB04124 |
| ECHA InfoCard | 03fa419f-b479-4379-ade2-4dbfc819d002 |
| EC Number | 200-912-2 |
| Gmelin Reference | 59708 |
| KEGG | C18356 |
| MeSH | D006603 |
| PubChem CID | 67674 |
| RTECS number | AB1925000 |
| UNII | 0Y3OQ71K8I |
| UN number | UN3341 |
| CompTox Dashboard (EPA) | `DTXSID7031723` |
| Properties | |
| Chemical formula | C3H2F6O2 |
| Molar mass | 166.08 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | pungent |
| Density | 1.6 g/mL at 25 °C |
| Solubility in water | Miscible |
| log P | -0.27 |
| Vapor pressure | 23.2 mmHg (25°C) |
| Acidity (pKa) | 0.4 |
| Basicity (pKb) | 3.8 |
| Magnetic susceptibility (χ) | -40.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.276 |
| Viscosity | 3.24 cP (20°C) |
| Dipole moment | 2.48 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 191 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1205.6 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1994 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H314: Causes severe skin burns and eye damage. |
| Precautionary statements | P210, P233, P260, P264, P271, P280, P301+P330+P331, P304+P340, P305+P351+P338, P312, P314, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3 0 2 W |
| Flash point | 52 °C |
| Autoignition temperature | 140°C |
| Explosive limits | Explosive limits: 4.4-16% (as Hexafluoroacetone) |
| Lethal dose or concentration | LD50 Oral Rat 1210 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 920 mg/kg |
| NIOSH | RS6130000 |
| PEL (Permissible) | PEL: 0.005 ppm (as Hexafluoroacetone) |
| REL (Recommended) | REL: 0.01 ppm |
| IDLH (Immediate danger) | 50 ppm |
| Related compounds | |
| Related compounds |
Hexafluoroacetone Trifluoroacetone Trifluoroacetic acid Hexafluoroisopropanol |

