Hexafluoroacetone: A Clear-Eyed Look at an Unmistakable Chemical
Tracing the Road: How Hexafluoroacetone Came Into the Picture
Curiosity about hexafluoroacetone (HFA, also known by names like perfluoroacetone and 1,1,1,3,3,3-hexafluoro-2-propanone) goes back further than most would guess. Early research into organofluorine compounds set the stage for HFA’s first appearance in the laboratory. Back in the days when chemical exploration was pushing deeper into synthesis and new industrial materials, HFA stood out for its strong reactivity and unique structure. Companies and academic groups both latched onto hexafluoroacetone because fluorinated chemicals seemed to hold the key for growth in plastics, coatings, and advanced electronics. Before folks had strict environmental regulations, the handling of HFA veered recklessly close to cavalier. Over decades, with more respect for risk and tighter control, research and commercial production of HFA have settled into routines that fit today’s safety standards.
Unpacking the Substance: What Hexafluoroacetone Brings to the Table
Nobody mixes up HFA with anything else once they’ve worked around it. The liquid evaporates quickly, carries a sharp odor, and hits with a volatility most chemists learn to treat with care. Six fluorines anchor themselves to a central carbon backbone, shaping a molecule resistant to many ordinary reactions but surprisingly eager to pair up under the right conditions. The chemical formula, (CF3)2CO, spells out a story of high electronegativity and low reactivity toward water unless catalysts are around. HFA turns up colorless but leaves no doubt about its presence in a lab through aggressive fumes that demand proper ventilation. Most flammable substances of this volatility trigger alarm bells, but HFA dodges ignition thanks to the stabilizing effect of so many fluorines. These same characteristics also allow HFA to function as an effective building block in fluorinated organic chemistry, especially where a tough, unreactive unit is needed.
Setting the Rules: Handling, Labeling, and Technical Details
Walking through a storage facility that deals with HFA, you realize very quickly that precision guides every move. Containers built with corrosion-resistant materials, clear hazard labels, and well-documented safety instructions are not there for show. HFA demands a meticulous approach because it forms toxic gases if spilled and reacts violently with strong bases and nucleophiles. Technicians test product quality using gas chromatography and spectroscopic checks, ensuring the purity matches strict industry benchmarks. Accurate labeling, adherence to transportation rules, and emergency plans all go hand in hand. Storage temperatures and humidity controls help keep unwanted hydrolysis at bay. These precautionary steps developed over years of hard lessons and reflect a field that values both innovation and worker safety.
Making HFA: Synthesis Methods That Shaped an Industry
Those familiar with fluorine chemistry know that most routes to HFA start with common fluorinated substances like hexachloroacetone or perfluoropropylene. Electrochemical fluorination methods once dominated, and for good reason—the process reliably exchanges chlorine atoms for fluorine and delivers impressive yields. Another approach used the reaction of perfluoropropylene oxide with hydrogen fluoride under pressure. Many modifications in synthesis protocols stemmed from cost concerns, environmental restrictions, and the steady push for cleaner processes. Exhaust systems, waste capture, and onsite neutralization units show up at most facilities bulk-producing HFA. Improvements in process scaling and closed-loop recovery systems now keep both costs and emissions manageable. Watching chemical engineers tinker with reactor conditions, catalyst choices, and temperature swings drives home the point that this compound’s preparation continues to evolve.
Reactions, Adaptations, and Chasing New Possibilities
HFA stands out as both a reactive center and a stabilizing anchor, depending on its application. Chemists exploit its capacity as a ketone to undergo condensation reactions and addition of nucleophiles. Anyone looking to craft other fluorinated products values HFA for its ability to donate or share its tough CF3 groups. For example, its reactivity with amines, alcohols, or even water (with proper control) creates new classes of intermediates that form the backbone of high-performance polymers. Researchers tinkering with side chains or semifluorinated surfactants often start with HFA or its derivatives. Even the pharmaceutical world—where stability and resistance to metabolic breakdown attract serious interest—has turned to HFA-based building blocks for exploring novel drug candidates. Changing the substitution on HFA, or attaching it to larger ring systems, opens the door to materials with fresh properties.
What’s in a Name? Synonyms and Global Usage
Most chemists recognize HFA under various names, which reflects regional naming conventions and supply chain quirks. Besides hexafluoroacetone, you’ll find listings for perfluoroacetone, 1,1,1,3,3,3-hexafluoro-2-propanone, and sometimes even under legacy trade designations. Checking the CAS number remains the best way to avoid confusion when ordering or referencing HFA in multi-national projects. Fluorochemical manufacturers and research suppliers all use subtly different branding, but the underlying compound doesn’t change. A seasoned chemist or formulary manager keeps lists of these synonyms close at hand to cut through catalog clutter.
Investing in Safe Operations: Lessons from the Lab and Factory Floor
Safety training around HFA never gets perfunctory. This compound brings risks that demand respect—acute toxicity at relatively low inhalation levels and corrosivity to the eyes and mucous membranes. Environmental controls, leak detection, and personal protective equipment become non-negotiable. No excuses for skipping gloves, goggles, and proper respirators. Facilities with experience in organofluorine chemistry pursue continuous risk assessment programs, not just regulatory checkboxes. Opening a valve without a fresh review of standard operating procedures can have lasting consequences. Emergency drills and training on antidote use (such as treating exposure with calcium gluconate) stand as standard practice in every forward-thinking operation that deals with HFA. Stories circulate about accidents from earlier eras—enough to convince most old hands that vigilance backs every innovation.
HFA at Work: Main Applications and Why They Matter
Hexafluoroacetone’s popularity stems from its versatility across multiple industries. Polymer manufacturers rely on it for producing polyimides that stand up to searing heat and corrosive chemicals. These polymers find homes in spacesuits, electrical insulation, and coatings for critical engine components. Other uses spring from its capacity to launch new surfactants and wetting agents found in everything from industrial cleaning to advanced microfabrication. The chemical’s unusual blend of stability and reactivity helps electronics makers lay down thin, functional films using vapor deposition techniques. HFA has a less visible but critical role in chemical syntheses—building molecules too sensitive for most organic solvents or seeking the high-electron density that only fluorine brings. The fact that HFA-based materials show high durability and resist breakdown under hostile conditions explains their appeal to aerospace and defense sectors. A push for new pharmaceuticals and specialty polymers keeps research labs reaching for HFA derivatives, always stretching for better performance.
Steering Research and Development: Where Are We Headed?
Research into HFA rarely stands still. Innovators keep pressing to reduce waste, green up processes, and discover derivatives that push new boundaries. Growing public concern over persistent chemicals drives teams to find less hazardous production chains or engineer safe degradation pathways for HFA-derived products. Some labs focus on capturing the amazing stability of HFA, aiming for medical devices and diagnostic reagents with long shelf lives and resistance to contamination. Analytical chemists keep refining detection techniques to monitor environmental impact, while engineers chase upcycling strategies for used and spent HFA-containing materials. Progress seems to depend less on a lone breakthrough and more on the cumulative lessons and persistence of many hands around the world trying to coax better, safer, and more responsible chemistry from this already well-studied molecule.
Understanding the Risks: Toxicity and Occupational Health
No one ignores toxicity risks when working with HFA. Studies in both animals and cell cultures paint a clear picture—this compound, inhaled or absorbed through the skin, disrupts normal metabolic pathways and hits hard at the lungs, liver, and nervous system. Short-term exposure can trigger respiratory distress, irritation, and neurological symptoms that merit immediate intervention. Chronic exposure, even at low levels, brings concerns for long-term organ damage and heightened cancer risks, based on animal models and occupational case studies. Workplace monitoring remains essential, and every responsible employer invests in air sampling, medical surveillance, and enforcing exposure limits. Moving toward less toxic derivatives and improving containment systems gives some hope for the future, but the facts remain: HFA is not a chemical for casual handling.
Looking Ahead: The Road for Hexafluoroacetone
The future shape of HFA production and application will probably depend on a few undeniable forces. Stricter environmental rules, pressure to cut down on persistent chemicals, and end-of-life recycling for fluorinated materials all shape industry strategies. Emerging research looks to design HFA alternatives with reduced toxicity and easier degradation. Breakthroughs in catalysis and reactor design could keep driving down environmental footprints for HFA synthesis. Partnerships between industry, academic groups, and regulators go a long way in balancing innovation with stewardship and transparency. My experience has taught me that progress in chemistry always comes layered with compromises—pushing toward safer, more sustainable use will keep HFA in the conversation well into the next era of advanced materials and chemical engineering.
What is Hexafluoroacetone used for?
More Than Just a Tough Name
Most people won’t find hexafluoroacetone on a shelf at the local store. The name sounds like something straight out of a lab manual, and that’s accurate—it’s a chemical not meant for everyday hands, but it impacts daily life more than most folks realize. Chemists rely on this compound in specialized settings, particularly in places where things have to work reliably and safely under stress.
Why Do We Use Hexafluoroacetone?
Companies producing advanced materials look to hexafluoroacetone as a building block. That’s because its core structure helps make products that face extremes—harsh environments, high heat, strong chemicals. Most notably, this chemical goes into making fluorinated polymers, especially some high-performing plastics like poly(hexafluoroacetone) and polymers such as PEEK with tough, chemical-resistant coatings. If you see wire insulation in electronics that can handle serious abuse, or you notice seals and gaskets in engines that stay tough after years of work, hexafluoroacetone has helped shape those materials.
Semiconductor makers also prize this compound. Technology companies need ultra-pure, reliable materials, and even a slight contamination can throw off the accuracy of delicate processes. Hexafluoroacetone helps in producing specialty etchants and surface treatment agents, allowing chip makers to keep up with demand for smaller, faster processors.
Real-World Examples and My Own Take
I got my first look at advanced fluorinated plastics in an industrial plant specializing in high-end cabling. The engineers showed me samples of wiring insulation that stayed flexible and strong long after exposure to oils, coolants, and even open flame. One of them explained that using hexafluoroacetone derivatives meant less downtime and more confidence in safety gear. In environments where a wiring failure could cost thousands—or worse, put lives on the line—every advantage counts.
Medical device manufacturers also rely on this chemical. Surgical instruments and implantable devices have to resist both corrosive body fluids and multiple rounds of sterilization. The non-stick, “slippery” coatings, often based on fluorinated chemicals made from hexafluoroacetone, help medical tools go through critical use cycles without letting dangerous bacteria hide out on their surfaces. I’ve had family members undergo surgery where every detail matters, down to the coatings on the surgical scissors. Knowing the science behind these tools gives me a deeper respect for the people and processes involved.
Risks and Responsible Use
There’s a flip side to big chemistry. Hexafluoroacetone comes with serious health risks. It’s toxic, volatile, and strict safety controls must stay in place wherever it’s handled. That means proper fume hoods, specialized training, and rigorous air monitoring matter as much as what the chemical is used for. Manufacturing facilities juggle efficiency with regulation, putting a premium on continuous oversight. Safe disposal and spill response plans become standard practice, not just best practices. The goal is to protect people on-site and in the surrounding community.
Green chemistry holds some potential for safer alternatives but, to date, few replacements deliver the same mix of heat and chemical resistance at scale. Researchers keep digging for solutions that offer these benefits without the health trade-offs. It’s one of those situations where society enjoys the results—clean electronics, safer cars, effective healthcare—while a handful of experts bear the responsibility for every step of the supply chain.
What’s Next?
Hexafluoroacetone offers a glimpse into the backbone of advanced technology and manufacturing. Its story highlights the ongoing balance between innovation and public health—a balance that demands expertise, honesty, and attention to detail at every stage.
What are the safety precautions when handling Hexafluoroacetone?
What Makes Hexafluoroacetone So Hazardous?
Hexafluoroacetone is no everyday chemical. Harsh on lungs and eyes, and pretty unforgiving if it leaks or spills, this compound brings real risk to people who handle it in labs and factories. Even after years of experience around chemicals, I pay closer attention when I see that name. Inhalation can trigger coughing, breathlessness, and chest pain. Skin contact stings and can burn. Few chemicals inspire the room to go silent like this one.
Personal Protective Equipment Saves Lives
Full-face respirators with cartridges for acid gases, splash-resistant goggles, and heavy gloves—these items turn routine lab habits into daily rituals. I remember a colleague skipping gloves for a quick connection, only to end up in the emergency shower seconds later. Chemical-resistant aprons offer coverage, but only if worn over the whole front. Closed shoes that cover ankles provide another layer of insurance that I never neglect.
Well-Ventilated Spaces Matter
Proper airflow is non-negotiable. Fume hoods, not just a cracked window, keep hexafluoroacetone vapors moving away from lungs and skin. A well-maintained ventilation system makes daily work safer without much thought. Airflow sensors alert staff if something goes wrong; without them, exposure can go unnoticed until someone feels ill. I had a scare once in a room where the fan slipped off, and within minutes people started to cough—no one let that happen twice.
Storage Requires a Careful Approach
Locked cabinets insulated from heat sources, far from food or common break areas, offer reliable safety. Compatible containers—usually fluoropolymer or stainless steel—stop leaks and reactions with the container itself. Labels warn people quickly if they see a bottle where it shouldn’t be. Inventory checks catch issues before a missing bottle turns into a dangerous guessing game.
Small Actions, Lasting Results
Accidents shrink with simple habits. Gathering a spill kit before work, keeping an eye on container seals, and reviewing MSDS sheets help stave off careless mistakes. The day someone asks, “What do we do if we spill some?” is not the moment to realize the steps aren't clear. Posting safety data sheets where people work with hazardous materials benefits newcomers and veterans alike.
Training Builds Confidence
My old supervisor drilled every emergency scenario until it felt second nature—alarms going off, chemicals hissing, and the clock always ticking. Regular drills make evacuation routes, first aid for chemical burns, and containment plans sink in for everyone. People freeze less often during real trouble if they’ve practiced.
Simple Steps Add Up
Hexafluoroacetone rewards respect but punishes shortcuts. Wearing the right protection, keeping containers secure, and never working alone mean most exposures wind up as stories, not emergencies. After years in the lab, my appreciation for these simple precautions keeps growing—no job or experiment is worth a trip to the hospital. Safety, in my experience, starts with everyday habits and a willingness to speak up any time corners get cut.
What is the chemical formula of Hexafluoroacetone?
What Is Hexafluoroacetone?
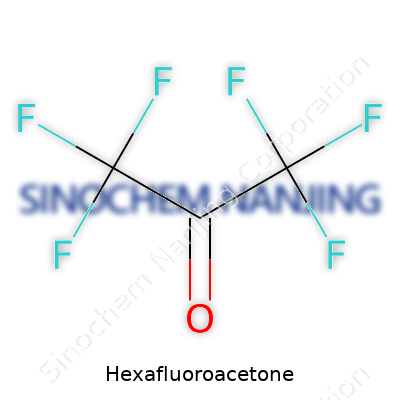
Hexafluoroacetone isn’t something you run across in everyday life. This compound, which scientists know as CF3COCF3, shows up in the world of chemistry labs and specialty manufacturing. Its chemical formula points to a structure where two trifluoromethyl groups (CF3) flank a central carbonyl group (CO). The presence of six fluorine atoms in this molecule gives it some unique properties and demands a careful approach to safety and handling.
Importance in Industry and Research
Hexafluoroacetone plays a behind-the-scenes role in making fluorinated chemicals, special polymers, and pharmaceuticals. Researchers and chemists value it for the strong electron-withdrawing influence that the fluorine atoms provide. That makes it a helpful intermediate for certain reactions, especially when other approaches to fluorine-rich molecules stall or don’t yield high enough purity. In my own stint at a research lab, we reached for chemicals like this when synthesizing stable, heat-resistant materials for tough environments.
CF3COCF3 doesn’t stay on the shelf for long because its high reactivity can help produce other advanced building blocks, including compounds used in non-stick coatings and robust insulating materials. In practice, this molecule can jump-start processes in ways that older, less specialized chemicals can’t. Chemists find that using hexafluoroacetone cuts out extra steps and harsh reaction conditions. The demand for efficient, resource-conscious processes keeps driving its use, especially as sustainability gains focus in chemical manufacturing.
Health and Safety Considerations
Hexafluoroacetone delivers results in the right hands, but ignoring safety would be a big mistake. Exposure—even a small amount—can cause irritation to lungs, eyes, and skin. I learned quickly to respect the fume hood and personal protective equipment after hearing stories of accidental exposure. Inhalation brings symptoms that no worker wants to endure, ranging from persistent coughing to serious damage in sensitive tissue.
Anyone handling CF3COCF3 must follow strict protocols: leakproof containers, adequate ventilation, and prompt cleanup for spills. Training for emergency situations helps, but prevention stands as the best solution in busy lab conditions. I’ve seen researchers refuse to cut corners because the risks of exposure outweigh the few minutes they might save.
Smart Solutions for the Future
As demand for hexafluoroacetone rises, conversations focus on engineering controls and personal safety culture. Substituting less hazardous fluorinating agents still challenges the industry, but green chemistry principles are gaining ground. Engineers collaborate with toxicologists to refine standards and invest in equipment that reduces manual handling—the less direct contact, the lower the risks.
Some companies run closed systems that basically never open until the waste is neutralized. Automated monitors for leaks help keep workers safe and catch problems before they escalate. In university labs, safety audits and regular emergency drills keep people sharp, while new digital tracking for chemical use and disposal pushes accountability.
Demand for high-performance materials won’t slow down, but continued investment in research holds potential for safer substitutes or more efficient processes that reduce exposure to hexafluoroacetone. Until then, respect for this molecule and its risks sets the foundation for every successful project.
How should Hexafluoroacetone be stored?
Direct Encounters With Dangerous Chemicals Change Perspectives
I have walked through industrial storerooms stacked with hazardous materials. Over the years, I’ve seen more caution signs and chemical barrels than I care to count. Hexafluoroacetone stands out on the list of substances that demand special respect. Extreme volatility, high toxicity, strong reactivity—this stuff doesn’t just burn skin or eat away metal, it can damage lungs and trigger fires with little provocation.
Hexafluoroacetone—No Room for Error
Industry veterans know Hexafluoroacetone by its sharp odor and its knack for escaping containment. This compound boils at a low temperature, has a vapor heavier than air, and reacts fiercely with water. The lesson is simple: forget a minor detail and you might trigger an evacuation or a hospital visit. One incident with a leaking valve forced a facility shutdown in my early days. The cleanup team wore full-face respirators, handled only by workers properly trained, and nobody got near the vapors without thorough training.
The Importance of Temperature and Ventilation
Storing Hexafluoroacetone calls for proper temperature control. Cool, dry environments slow down unwanted reactions. I’ve seen what happens when storage rooms get too warm—the pressure builds inside containers, sometimes venting hazardous fumes into the air. Good facilities keep the room temperature comfortably below ambient—no risk of containers swelling from heat.
Hexafluoroacetone needs containment that won’t corrode or degrade. Stainless steel with the right internal coating stays stable for years. Some operators try plastic or glass, looking to save on costs, but that path never ends well. Experienced handlers always choose metal tanks built with chemical-resistant linings.
Moisture—The Hidden Enemy
Moisture in the air or leaks in storage seals spells trouble. Even a small trace of water can set off a violent reaction. Dozens of stories float around the community about lab workers caught off-guard by unexpected splashes and invisible condensation. The answer: store Hexafluoroacetone with desiccants and airtight lids, checking each container for signs of corrosion or compromised seals.
Air Handling and Fume Extraction
Nobody enjoys walking into a stuffy chemical storeroom. Proper air exchange isn’t just about comfort, it’s about safety. Hexafluoroacetone vapors can hang around at floor level, making it critical to circulate air from the base as well as overhead. Effective venting systems, regular air monitoring, and working carbon scrubbers keep everyone safer. I’ve seen labs install panic buttons to trigger emergency ventilation and rapid evacuation routes if sensors detect rising vapor levels.
Security and Emergency Response—The Final Barrier
Only trained personnel get anywhere near Hexafluoroacetone. Sites lock it away with restricted access, signs warn, and spill kits sit right nearby. In one memorable incident, an outdated emergency shower system left a worker exposed for seconds too long—now everyone I know double-checks that decontamination stations work. Fast response saves lives and reduces damage.
Moving Forward With Awareness
Every mistake with Hexafluoroacetone leaves a lesson. Better training, stricter rules, and smarter facility design lower risk. Industry bodies offer up-to-date guidance on best practices, and the most diligent always keep learning—never assuming this powerful compound behaves like anything else in their inventory.
What are the potential health effects of exposure to Hexafluoroacetone?
Understanding What’s at Stake
Hexafluoroacetone pops up in chemical manufacturing, semiconductor processing, and even in labs shaping polymers and pharmaceuticals. This stuff doesn’t carry a friendly warning on the bottle, and the health effects get brushed aside unless you’ve worked somewhere fumes drift through the halls. People hear the name and tune out—who wants to learn about another harsh chemical unless it starts making news? Yet hexafluoroacetone poses some real risks, especially to workers in those industries.
Short-Term Exposure: Breathing and Burning
If hexafluoroacetone escapes into the air, even small amounts can irritate eyes and skin. At higher levels, it attacks the respiratory system. Folks exposed to it have reported coughing, a burning sensation in the throat, and watery eyes. I remember visiting a polymer plant years ago—the safety officer showed off an emergency eyewash station, saying you only need it once to never forget. It didn’t sound theoretical. Getting this stuff on bare hands might cause redness or blisters within minutes. Direct inhalation brings headaches and tightness in the chest. Left unchecked, exposure adds up fast in a packed space, especially if someone doesn’t catch a leak right away.
Chronic Effects: What Long-Term Exposure Does
Workers on the same production line week after week know it’s not always about one bad day; small exposures can set you up for stronger problems. Repeated contact increases the risk of chronic bronchitis and lung damage. Inhaling low amounts, day after day, can scar lung tissue—sometimes for the long haul. The Environmental Protection Agency spells out the dangers of repeated inhalation, listing lasting breathing trouble and heightened sensitivity to respiratory infections. There’s also evidence in the research that hexafluoroacetone can screw with your nervous system and kidneys when exposures drag on or spike above usual limits.
Preventing Exposure Is Not Optional
In shops and chemical plants, exposure to serious hazards shouldn’t get shrugged off as “just how it is.” Proper ventilation and airtight handling systems go a long way. I’ve watched experienced operators always check their seal and double up on gloves. Effective personal protective equipment—think face shields, respirators, not just goggles—keeps workers out of harm’s way, even during maintenance. Safety culture counts. Quick spill response and proper training help stop accidents becoming full-blown emergencies. Regulations don’t get around the fact that mistakes and shortcuts cost health in real ways. The American Conference of Governmental Industrial Hygienists sets strict occupational exposure limits for a reason. I’ve only seen safety culture pay off if people follow rules not out of fear of the boss, but respect for each other’s well-being.
Beyond the Factory Floor
Handling waste and emissions demands oversight. Accidents can affect whole communities if storage or disposal falls short. Strong environmental monitoring and reliable reporting systems make a difference. Practical solutions—scrubbers, better leak detection, frequent air quality checks—cut down on the risks. Inside and outside the plant, better transparency lets people know exactly what’s in their air. Trust comes from honesty and giving folks a stake in their own health. Hexafluoroacetone shouldn’t make headlines through preventable accidents, but through responsible innovation and care for workers who keep critical industries running.
| Names | |
| Preferred IUPAC name | 1,1,1,3,3,3-Hexafluoropropan-2-one |
| Pronunciation | /ˌhɛk.səˌflʊə.roʊ.əˈsiː.təˌnoʊ/ |
| Identifiers | |
| CAS Number | 684-16-2 |
| 3D model (JSmol) | `JSmol\_model\_string = "C(C(=O)C(F)(F)F)(F)(F)F"` |
| Beilstein Reference | 1361116 |
| ChEBI | CHEBI:43014 |
| ChEMBL | CHEMBL135787 |
| ChemSpider | 52007 |
| DrugBank | DB14096 |
| ECHA InfoCard | 20b0fb07-fb2d-4ed3-a292-cd3e406fc6e6 |
| EC Number | 206-199-1 |
| Gmelin Reference | Gmelin 1160 |
| KEGG | C06355 |
| MeSH | D006600 |
| PubChem CID | 7908 |
| RTECS number | KI2325000 |
| UNII | 74ON5U5L8A |
| UN number | UN3220 |
| Properties | |
| Chemical formula | C3F6O |
| Molar mass | 166.04 g/mol |
| Appearance | Colorless gas or colorless liquid with a pungent odor |
| Odor | Pungent |
| Density | 1.589 g/cm³ |
| Solubility in water | Very soluble |
| log P | 0.4 |
| Vapor pressure | 32.06 psia ( 1760 mmHg ) at 20°C |
| Acidity (pKa) | 5.4 |
| Basicity (pKb) | 4.2 |
| Magnetic susceptibility (χ) | -38.6 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.252 |
| Viscosity | 0.425 cP (25 °C) |
| Dipole moment | 2.29 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 348.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1210 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1755 kJ mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS02,GHS06,GHS05 |
| Signal word | Danger |
| Hazard statements | H301, H311, H331, H314 |
| Precautionary statements | P210, P260, P261, P264, P271, P285, P301+P310, P304+P340, P320, P311, P330, P363, P370+P376, P403, P405, P501 |
| NFPA 704 (fire diamond) | 3-4-2 |
| Flash point | -17 °C (1.2 °F; 256 K) (closed cup) |
| Autoignition temperature | 460°C |
| Explosive limits | Explosive limits: 5.2–15.2% |
| Lethal dose or concentration | LD50 inhalation rat 202 ppm/4H |
| LD50 (median dose) | LD50 (median dose) of Hexafluoroacetone: 250 mg/kg (rat, oral) |
| NIOSH | NIOSH: **KN5250000** |
| PEL (Permissible) | PEL = "0.005 ppm (0.024 mg/m3) as TWA |
| REL (Recommended) | 1 ppm |
| IDLH (Immediate danger) | 300 ppm |
| Related compounds | |
| Related compounds |
Hexafluoroisopropanol Perfluoroacetone Hexafluoropropylene |

