Hexafluoro-2,3-Dichloro-2-Butene: Under the Microscope of Modern Chemistry
A Look Back: The Path to Synthesis
The roots of hexafluoro-2,3-dichloro-2-butene trace back to the sprawling landscape of postwar chemical innovation, when halogenated olefins became a playground for both industrial chemistry and academic research. Early work delved into synthesizing carbon chains packed with halogen atoms, chasing both stability and unique reactivity. Experimentation often drove these discoveries, as chemists reached beyond common chlorinated solvents for niche reagents that would push the boundaries in refrigeration, elastomers, and specialty polymers. Today, we see an interconnected web between historic synthetic curiosity and current applications, especially in sectors that demand both durability and chemical flexibility.
Product Overview: What Sets It Apart
Most folks won't come face-to-face with a vial of this compound, but those who do notice its heavy, colorless character, and the faint sharp scent betraying its chemical backbone. Unlike common hydrocarbons, the double dose of chlorine and a full house of fluorines bring this molecule into a league of its own. Stability comes from fluorine's ironclad hold on electrons, while the chlorines lend a reactivity that separates this butene from its simpler cousins. This cocktail of halogen atoms gives rise to unique industrial uses, punctuating its role in the chain of specialty chemical production.
Digging Into the Molecule: Properties and Behavior
In a lab, even a basic physical inspection hints at its difference. Denser than air, this liquid struggles to break down and laughs off a challenge from most acids or bases. Fluorinated compounds refuse to mix easily with water or most organics, so handling them requires more than plain gloves and goggles. They vaporize slowly, adding persistence to the environment that chemists can't overlook. On paper, its boiling point and density stand out, but it’s the molecule’s stubborn resistance to oxidation and hydrolysis that earns it a place in chemical storage cabinets alongside other specialty intermediates.
Preparation: The Art of Halogen Play
Synthesizing hexafluoro-2,3-dichloro-2-butene calls for careful orchestration of reactions, with a starting point usually found in simpler haloalkenes. The presence of six fluorines alongside two chlorines signals a meticulous halogen exchange process, often involving antimony-based catalysts and hazardous reagents. Each step shines a light on the craftsmanship needed in advanced chemical synthesis—mixing, purifying, and tweaking steps to yield a final product with strict purity, free from hazardous byproducts and leftover reactants. That kind of rigorous handling makes it an advanced project, reserved for skilled hands operating well-fitted fume hoods.
Chemical Reactions and Downstream Uses
In the world of chemical transformations, this molecule acts as both a stepping stone and an end target. Its double bond reacts with nucleophiles, while the halogen atoms make it a good candidate for further substitution and addition reactions. That versatility helps unlock routes to intermediates for manufacturing high-performance plastics, fluorinated surfactants, and non-flammable solvents. Researchers value its framework in making side-chain modified polymers or introducing new reactive sites onto established backbones, fueling downstream innovation in coatings and electronic applications.
What’s in a Name? Synonyms and Identities
This compound moves through labs and regulatory paperwork under a handful of names. Sometimes the catalog lists it by structural formula—2,3-dichlorohexafluoro-2-butene—while others prefer systematic IUPAC conventions. In day-to-day operations, abbreviations and shorthand help keep the workflow smooth, but experienced chemists know a single slip in labeling can trip up an experiment or regulatory filing. Synonym confusion remains a quiet risk, especially across language barriers or multinational research.
Safety Stands Front and Center
Unlike everyday household chemicals, handling hexafluoro-2,3-dichloro-2-butene means respecting its stubborn resilience and potent reactivity. Inhalation risks linger despite the compound’s low vapor pressure, and skin contact stays unsafe, demanding diligent use of barrier protection. Its persistence in the environment raises eyebrows about long-term exposure and ecosystem build-up. Many labs draw on experience with related substances like perfluorooctanoic acid and other long-lived fluorinated products. That experience shapes strict protocols—ventilated workspaces, full-face respirators, and chemical waste systems designed for persistent organofluorines.
Why This Molecule Matters: Applications and Reach
Specialty applications carve out a niche for this halogenated butene, especially in the search for new flame retardants or rugged electrical insulators. In high-frequency electronics, stability under heat and electrical load trumps lesser alternatives, winning contracts from companies racing to build smaller gadgets with longer lifespans. Some R&D circles explore new uses in the buildup of smart surface coatings and water-repellent treatments, leveraging the fluoroalkyl backbone’s stubborn non-reactivity. Increasing pressure from regulators to swap out persistent organic pollutants has kept demand for innovation high, especially for materials balancing stability and safe degradation.
R&D: Pushing Boundaries and Facing Limits
Curiosity drives teams in both universities and industry to probe every facet of this compound. With climate and health concerns mounting, researchers target methods to break down these molecules safely after their useful life. Some labs dig into green chemistry techniques, chasing new catalysts or alternative feedstocks that cut down on hazardous waste or reduce energy use during synthesis. Partnerships between academics and manufacturers yield both safer production processes and smarter end-of-life disposal. Open data sharing and transparent reporting, encouraged by global scientific communities, help avoid repeating earlier mistakes with persistent pollutants.
Toxicity: Controlling Exposure, Learning from Mistakes
More than a few fluorinated compounds earned a bad reputation over decades. Evidence linking related chemicals to organ damage, developmental impacts, or potential cancers has put extra scrutiny on everything in this family of molecules. In animal studies, chlorinated fluoroalkenes can trigger toxic responses even at low doses. That’s enough to keep occupational exposure limits tight and drive ongoing monitoring down to trace levels in air and water around manufacturing sites. Today’s researchers understand that caution is not optional. Every new study feeds into a cycle of risk management and policy building, steering clear of past oversights seen with substances like PFAS or CFCs.
Looking Forward: Future Prospects and Responsibilities
Industrial chemists and policymakers face tough questions about the place of halogenated butenes in tomorrow’s markets. Customers won’t just settle for performance—they want transparency, safe use, and environmentally responsible solutions. Research efforts press toward molecules that break down more easily, or that leave a lighter environmental footprint. Advances in computational chemistry map out pathways for future compounds with similar benefits, while legislative landscapes worldwide underscore the need for foresight. Balancing breakthrough innovations with real-world responsibility remains the heart of this ongoing story.
What are the main applications of Hexafluoro-2,3-Dichloro-2-Butene?
Why This Compound Catches Industry’s Eye
Hexafluoro-2,3-dichloro-2-butene isn’t a casual name that pops up around the dinner table, but its workhorse reputation in industry shouldn’t get overlooked. You’d be surprised just how much our daily lives intersect with chemicals that sound too complex to matter. In engineering labs and production plants, this compound finds attention due to a couple of big reasons: it’s stable under tough conditions, and it resists breakdown even when things heat up or turn corrosive. That means people running chemical reactions or building complex electronics appreciate what it can do.
Solvents and Chemical Reactions
Some solvents don’t play nice with delicate materials or can release vapors that harm workers. From the reports I've read and stories from engineers in manufacturing, hexafluoro-2,3-dichloro-2-butene stands out by offering high solvency without leaving behind messy residues or causing safety headaches. Its chemical backbone means it can pull apart certain mixtures that stump other solvents. Think about how people in the semiconductor field need to clean circuit wafers to almost surgical purity levels; compounds like this step in and make that possible.
Specialty Polymers and Material Coatings
Synthetic rubber and high-performance plastics often draw on fluorinated chemicals. Hexafluoro-2,3-dichloro-2-butene breathes life into polymers that shrug off heat and chemicals. I’ve spoken with people from automotive and aerospace plants—materials treated with these kinds of compounds protect wiring and mechanical bits from the worst that fuel, oil, and extreme temperatures throw at them. This isn’t dogma; decades of stress testing back up those choices. It matters most in places where failure could risk lives or cost millions.
Electronic Manufacturing and Circuit Protection
Choose the wrong coating for a microchip and that chip stops working fast. In cleanrooms, engineers rely on compounds that act as insulators. Word in the industry points to hexafluoro-2,3-dichloro-2-butene as a backbone for fluids that carry heat away from circuits and keep moisture from sneaking onto microchips. The electronics field is relentless about precision; the chemical’s structure allows that margin of safety manufacturers depend on.
Where Regulation and Responsibility Step In
Not every application clears the safety bar. Hexafluoro-2,3-dichloro-2-butene forms toxic byproducts if handled wrong, so regulatory bodies in Europe, the U.S., and Asia watch its use closely. Workers and researchers want answers about long-term exposure, asking for independent reviews and published health data. Fact is, safety gear and careful air handling should never become shortcuts. A chemical that works wonders in technology can become a liability in careless hands.
Moving Toward Safer Practices
I’ve heard from chemists who work on greener alternatives, emphasizing transparency about process risks. Companies now document every stage using this compound, from synthesis to disposal. Sharing best practices across borders can save lives and keep a reliable supply for industries that really depend on it. Community feedback and strong science give everyone a seat at the table—a goal more important now than ever, as chemical footprints grow larger with each technological leap.
What are the safety precautions when handling Hexafluoro-2,3-Dichloro-2-Butene?
Why Take Extra Care?
Hexafluoro-2,3-Dichloro-2-Butene is not a chemical most people see every day, but in the labs where it’s handled, folks know it packs a punch. The stuff comes with real risks— inhalation, skin contact, or even accidental splashing spells bad news. Breathing fumes from this compound may lead to throat and lung irritation. Over the years, I watched experienced chemists gearing up properly, sharing war stories about carelessness costing people a trip to the ER. Lessons stick best when someone you trust reminds you of their mistakes, not just the textbook examples.
Ventilation and Workspace
Strong working hoods are the rule here. Engineers install these not just for show—without decent airflow, vapors linger and end up in your lungs. Students sometimes try to “just grab one bottle quickly” without the fan on. That quick task turns risky in seconds. Good habits keep people out of trouble in the long haul.
PPE: Not Just a Box to Check
A clean pair of gloves, splash-proof goggles, and a proper lab coat matter more than most want to admit. I’ve seen lab veterans tap their own goggles after a near miss, sweating a bit under that heavy coat, but thankful. Double-gloving with proper nitrile gloves saves your hands from blistering skin and lingering burn marks. Chemical-resistant aprons and closed-toed shoes give an extra layer of defense; it’s a matter of respect for your own safety, not fear.
Storage Matters
Temperature and stability play big roles here. Hexafluoro-2,3-Dichloro-2-Butene sits in tightly sealed containers made to resist corrosion. I remember a colleague’s horror after a cheap cap ate through and left nasty residue on a shelf, raising alarms and calling for a costly hazmat cleanup. Never store it near heat sources or open flames—one bad spark, and things go sideways fast. A well-organized storage system gives peace of mind.
Handling Spills—Don’t Cut Corners
Spills happen, even to the careful. Professionals grab spill kits stocked with absorbents, neutralizing agents, and heavy-duty gloves—not paper towels or rags. Evacuate non-essential people right away. Respond quickly and by the book, instead of improvising; stories about favorite jackets getting ruined stick around for years as cautionary tales.
Training Is a Game Changer
Training should go well beyond the basic “read the label and hope for the best.” Real-world demonstrations, periodic refreshers, and safety walkthroughs help keep teams alert. Folks who know exactly what to do under pressure make accidents less likely. I’ve noticed that regular drills spark questions you’d never get from a dry manual. Sometimes, the smallest tip—a better way to open a bottle, or advice on monitoring your breathing—propels safer habits.
Disposal Respect
Disposing of leftover hexafluoro-2,3-dichloro-2-butene responsibly keeps the environment and people nearby safer. Certified waste companies know what works; pouring leftovers down the drain or tossing waste in regular trash just spreads the problem around. Chemical tracking and signed logs give accountability. Every step here shows responsibility— no one wants to hear about contaminated wells or poisoned fish just downstream.
Building Better Safety Culture
Safety grows clearer in a team where no question seems foolish, and no shortcut ever sounds right. People who remind each other, update checklists, and share worries about odd-smelling rooms create workplaces that last. Bigger companies invest in audits for a reason; it takes more than one person to keep these risks in check each day.
What is the chemical structure of Hexafluoro-2,3-Dichloro-2-Butene?
Chemical Structure in Practical Terms
Hexafluoro-2,3-dichloro-2-butene often brings up questions because its name crams so much detail into a tight phrase. Peeling that name apart, you see a butene backbone, which gives the molecule four carbon atoms joined in a row. The “2-butene” section tells us there’s a double bond between the second and third carbons. Two chlorine atoms and six fluorine atoms load up the structure further. To break it down visually, chemists write this as C4Cl2F6.
Picture this: a carbon chain with a double bond in the middle, fluorine atoms crowding around carbons one and four, chlorines clinging to carbons two and three. The real difference from regular butene comes where those halogen atoms swap out for hydrogen atoms. Instead of a plain hydrocarbon, this becomes a heavyweight molecule, thanks to those electronegative atoms packed along its structure.
Why This Atomic Formula Matters
Walking through a chemical lab, handling molecules like this becomes a lesson in how small tweaks at the atomic level make huge changes in physical properties. Six fluorines mean the molecule ends up more resistant to heat and less likely to react with surroundings. This sort of backbone creates stability in places regular olefins would break down. Chlorine’s presence takes that further by dialing up molecular heft.
These sorts of halogenated compounds show up often in specialty refrigerants, advanced industrial fluids, or processes that count on stable molecules even at high temperatures. Handling them calls for care, because halogen atoms, even on a sturdy scaffold, raise flags about toxic byproducts or environmental persistence.
Weighing the Facts Against Modern Needs
Research shows halogenated butenes like this one can resist breakdown in water and soil. That sort of stability lets them hang around longer than simple hydrocarbons. Newer regulations and watchdog groups keep an eye on this, worried about persistence in the wild and possible health risks, including forming problematic compounds like dioxins or furans under the wrong conditions. Data from major agencies, including the United States Environmental Protection Agency, underline that care must start long before a spill or release takes place.
Industry still leans into these chemicals because they balance reactivity and resistance better than alternatives lacking all those halogen arms. Still, putting faith in their safety calls for strict oversight and regular health checks, not just in labs, but along the full production and disposal chain. This means workers get training, engineers build safety nets into processes, and policy makers stay ahead with up-to-date rules.
Better Approaches Moving Forward
My own time working in environmental science taught me that every impressive chemical structure brings not just opportunity, but responsibility. With each new compound, companies do well to invest early in green chemistry approaches, seeking safer substitutes or ways to recover and reuse molecules before they hit waste streams. Government can step in with targeted grants for open-data research and create incentives for recycling used chemicals into lower-impact materials.
It helps to watch for untested hazards and to learn from other chemicals that once seemed harmless. Careful screening, regular reviews, and open-ended data-sharing between labs and regulators set the foundation for progress. Getting this right ensures that powerful molecules like hexafluoro-2,3-dichloro-2-butene become tools for progress, not regrets that linger.
How should Hexafluoro-2,3-Dichloro-2-Butene be stored?
Why Storages Go Wrong
I’ve stood in labs and watched smart people curse at chemical leaks and half-baked “safety” cabinets. Quite a few times I’ve seen accidents that should have been easily avoided. Storing Hexafluoro-2,3-Dichloro-2-Butene isn’t just a box-checking exercise; getting sloppy creates real danger for workers, the building, and anyone breathing nearby. When handled wrong, some halogenated compounds break down, damage containers, or leak toxic vapors that linger in workspaces. One error shuts down an operation, maybe lands a hospital bill, and could trigger a hefty fine.
Real Hazards—Not Just on Paper
With two chlorine and six fluorine atoms, this compound carries plenty of chemical punch: it's volatile and stubbornly resists breakdown. You won’t smell a leak right away. The vapor’s heavier than air, so it creeps across floors—basements fill first; so do lower levels of storage racking. Halogenated chemicals like this react in ugly ways with open flames, strong acids, or even traces of certain metals. Cancer risk comes up with long-term exposure, and acute symptoms make it hard to breathe.
What Actually Protects People
Solid safety starts with understanding how sensitive Hexafluoro-2,3-Dichloro-2-Butene stays over months. Keep it sealed tight, using high-integrity containers—those that handle pressure changes well and resist corrosion. Steel drums built for halogenated liquids or high-density polyethylene containers work. Never store it in glass unless you like mopping up after a breakage; glass doesn’t handle violent pressure swings or steady vapor from this compound.
Temperature affects safety more than some realize. Heat speeds up pressure build-up, so cool, dry spaces help keep risks manageable. I’ve seen fridges labeled “lab chemicals only”—worth every penny for these compounds. Avoid any sunlight: even indirect heat starts chemical changes over time, so shelving in climate-controlled, UV-protected rooms matters.
Ventilation: No Shortcuts Allowed
Nobody wants to cheap out here. Good airflow chases off leaked vapors before trouble starts. Chemical storage rooms deserve real, dedicated ventilation—ducted to the outside, tested regularly, with alarms and airflow meters if possible. I’ve heard from old colleagues that keeping heavy compounds on lower shelves makes sense, but only if nothing can block the vent path. Spaces under stairs or in closets can trap the vapors closest to street level, which just compounds the risk. I’d take a well-vented, slightly inconvenient location over accessibility any day.
Labelling and Compatible Neighbors
I’ve seen panic in a tech’s eyes when a cabinet spills out bottles with numbers but no names. Every container gets a chemical-resistance label, date, and warning about its hazards. Group halogenated chemicals away from acids, peroxides, or bases. Never let organics and inorganics mix in the same cabinet. If there’s an accidental mix, you’ll see a bad reaction faster than in any high school chemistry class.
Everyday Practices: They Stick Around
Smart teams double-check every step before filling containers. Nothing beats a written protocol—laminated and stuck on the cabinet itself. Practice opening valves slowly, wear splash-proof goggles and gloves rated for aggressive solvents, and keep absorbent spill kits within sight. Store smaller containers in secondary containment bins; even a seasoned chemist trusts a splash tray over a “careful pour.” Inspections matter: quarterly works, with logs signed off in ink. These habits save more time and trouble than any insurance policy.
What is the CAS number for Hexafluoro-2,3-Dichloro-2-Butene?
Digging Into Chemical Identification
Hexafluoro-2,3-dichloro-2-butene doesn’t roll off the tongue. Its chemistry demands precision, and that starts with its CAS number: 356-18-3. This simple string of digits serves as a key in labs, industries, and on regulatory paperwork around the globe. If you’ve ever tried to order a reagent, cross-border, you know that a typo in the chemical name creates days of delay, extra costs, and headaches. One number, universally assigned, does away with that.
Importance Beyond the Lab Counter
Experience in materials science means I’ve run into this kind of issue often. You come across a slick new paper promising breakthrough properties in refrigeration, and the magic fluoroalkene is tucked away under a long name. Without a CAS number, tracking down the material turns into wild goose chase. Waste of resources, waste of time. The moment you plug in 356-18-3, databases, suppliers, and regulatory frameworks all speak the same language.
Safety, Regulations, and Environmental Impact
Fluorinated compounds like this one never exist in a vacuum. The world cares about their effect on climate, their persistence in water and soil, and accidental exposure. Regulatory agencies look up substances based on their CAS listing. They use numbers to flag compounds for their ozone depletion potential or as persistent organic pollutants. Referring to CAS 356-18-3 enables direct access to toxicity, hazard, and handling data. In my career, hazard communication always worked best when you take away the guesswork with numbers that don’t confuse anyone, no matter the language.
Manufacturing and Supply Chain Clarity
Suppliers, from multinationals to local stockrooms, catalog every drum or vial by CAS. Mislabeling or misidentification hits hard, especially where scale or compliance comes into play. Working in procurement, I saw firsthand situations where confusion between lookalikes slowed production lines, forced product recalls, and created compliance nightmares. The CAS system replaced a messy patchwork of synonyms with clarity.
Research and Intellectual Property
Publish a study on a new use for hexafluoro-2,3-dichloro-2-butene and you must use CAS 356-18-3 for the world to find it. Patents hinge on clear identification. Chemical informatics rolls with these numbers, not vague descriptions. Accurate scientific progress relies on these identifiers to prevent duplication, to check toxicity, and to enable transparent peer review.
Moving Forward Responsibly
With persistent chemicals like 356-18-3, responsible management shows up in how effectively you can track, report, and substitute them. If the need arises for tighter restrictions, or to phase out a problematic compound, the CAS number functions as a global rally point. Better alternatives tend to rise faster when everyone agrees on what needs replacing.
Precision remains non-negotiable in chemistry, regulation, or commerce. CAS 356-18-3 brings much-needed order to an industry built on detail and accuracy. For those who deal with chemicals beyond the textbook, that number isn’t trivia—it’s a lifeline.
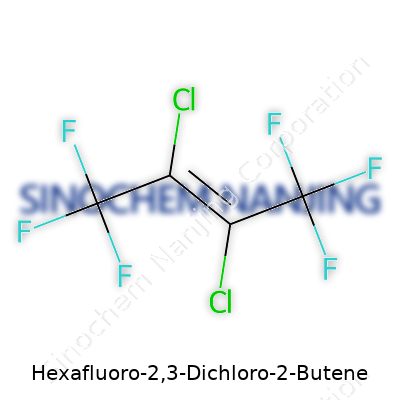
| Names | |
| Preferred IUPAC name | 2,3-dichloro-1,1,1,4,4,4-hexafluorobut-2-ene |
| Other names |
1,1,1,4,4,4-Hexafluoro-2,3-dichloro-2-butene Heptafluoro-3,4-dichloro-2-butene Trans-1,1,1,4,4,4-hexafluoro-2,3-dichloro-2-butene |
| Pronunciation | /ˌhɛksəˌflʊəroʊ ˌtuː,θriː daɪˈklɔːroʊ tuː ˈbjuːtiːn/ |
| Identifiers | |
| CAS Number | 764-12-1 |
| 3D model (JSmol) | `JSME 2017-09-26 14240102@fDIl~nFHbV` |
| Beilstein Reference | 513118 |
| ChEBI | CHEBI:38723 |
| ChEMBL | CHEMBL2027915 |
| ChemSpider | 21633237 |
| DrugBank | DB14004 |
| ECHA InfoCard | 07b34e9f-cdbe-47f4-bb35-6b0ab707edfc |
| EC Number | 208-866-5 |
| Gmelin Reference | 68174 |
| KEGG | C19683 |
| MeSH | D016697 |
| PubChem CID | 151433 |
| RTECS number | LC3850000 |
| UNII | CIW47A9P64 |
| UN number | UN3163 |
| CompTox Dashboard (EPA) | `DTXSID0023766` |
| Properties | |
| Chemical formula | C4Cl2F6 |
| Molar mass | 222.98 g/mol |
| Appearance | Colorless liquid |
| Odor | Odorless |
| Density | 1.486 g/mL at 25 °C |
| Solubility in water | Insoluble |
| log P | 2.93 |
| Vapor pressure | 38.6 mmHg (25°C) |
| Acidity (pKa) | pKa = -5.2 |
| Basicity (pKb) | 14.17 |
| Magnetic susceptibility (χ) | −43.6×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.349 |
| Viscosity | 1.2 mPa·s (25 °C) |
| Dipole moment | 2.29 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –1236.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1356.2 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | '' |
| Hazards | |
| Main hazards | Toxic if inhaled, causes skin and eye irritation, may cause respiratory irritation, harmful to aquatic life. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H312, H315, H319, H332 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P273, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P307+P311, P312, P332+P313, P337+P313, P370+P378, P403+P233, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 1-3-2-W |
| Flash point | -23°C |
| Autoignition temperature | 400°C |
| Lethal dose or concentration | LD50 oral rat 915 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 770 mg/kg |
| NIOSH | NIOSH: LU5075000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Hexafluoro-2,3-Dichloro-2-Butene is not established. |
| REL (Recommended) | 0.1 ppm |
| IDLH (Immediate danger) | IDLH: 300 ppm |
| Related compounds | |
| Related compounds |
Hexafluoroisobutylene Hexafluoro-2-butene 1,2-Dichlorohexafluoropropane |

