Hexachlorocyclopentadiene: A Century of Chemistry, Complexity, and Consequence
Historical Development
Looking back at the chemical industry’s evolution over the last hundred years, few compounds inspire such a mix of fascination and wariness as hexachlorocyclopentadiene. Introduced in the 1940s during a wave of innovation for synthetic pesticides, its development rode the tide of demands for agricultural productivity and industrial growth. My own experience studying the chemical literature highlighted how wartime needs and postwar optimism led to widespread search for new compounds, and hexachlorocyclopentadiene quickly turned into a key building block for potent cyclodiene pesticides like aldrin, dieldrin, and endrin. As society came to appreciate the costs of environmental persistence, the legacy of these chemicals grew complicated—pushing scientists and regulators to rethink material progress and responsibility.
Product Overview
What’s striking about hexachlorocyclopentadiene is its dual nature: both as a raw material for manufacturing and as a marker of the collision between innovation and consequence. The chemical shows up as a colorless to pale-yellow liquid, its pungent odor hinting at the chlorinated backbone. It never really passed into public awareness like DDT, but anyone working in industrial chemistry or environmental science recognizes its significance. As regulations on downstream pesticides tightened, attention turned to how and where this chlorinated hydrocarbon enters the world—and often, how it lingers.
Physical & Chemical Properties
Anyone with a background in chemistry gets an instant feel for the stubbornness of this compound just from its structure: six chlorines attached to a five-carbon ring, yielding a dense liquid with a boiling point around 239°C and a relatively high molecular weight. Its stability against water and resistance to breakdown made it attractive for industry, but also created challenges for cleanup. It dissolves in a wide range of organic solvents, remains persistent in soil and sediment, and volatilizes only slowly. For anyone handling it, the volatility and vapor density become more than abstract data—they call for vigilance in any workspace.
Technical Specifications & Labeling
There’s a lot of detail in how hexachlorocyclopentadiene gets classified and labeled. International standards rely on hazard communication: warning labels warn about acute toxicity, environmental risk, and inhalation danger. Pure forms get shipped in steel drums under tight regulation. Some regulatory bodies, such as the US EPA, place it among chemicals of concern for both worker health and broader environmental fate. Safety Data Sheets carry long lists of hazards, but as a chemical worker, I found that real-life safety relies as much on routine habits and a respect for the substance’s ability to cause trouble, as it does on formal paperwork.
Preparation Method
The manufacture of hexachlorocyclopentadiene started as a relatively straightforward process on paper, but controlling it in practice demanded some ingenuity. Industrial synthesis usually begins with cyclopentadiene or its derivatives, reacting with chlorine under specific conditions. The reaction produces a mixture of chlorinated intermediates, which need careful distillation to collect the pure product. In my own research, I saw how subtle shifts in reaction conditions—temperature, chlorine flow, purity of precursors—could push yields up or down, or fill samples with troublesome byproducts. Every improvement in process chemistry aimed to reduce not just waste, but also the risk of explosions or leaks, because the reactants themselves can get out of hand if operators lose focus.
Chemical Reactions & Modifications
On the molecular level, hexachlorocyclopentadiene stands out for its ability to serve as a platform for further synthesis. It’s reactive with a variety of nucleophiles and dienophiles, especially in Diels-Alder reactions that create complex polycyclic structures. For years, organic chemists used it to build new pesticides, flame retardants, and specialty chemicals. The same reactivity makes it a challenge for waste treatment: breakdown products can include both lighter and heavier chlorinated compounds. In my experience, even small mistakes in chemical handling could trigger chain reactions, releasing hazardous chlorinated fumes and requiring emergency containment procedures.
Synonyms & Product Names
Over time, this compound picked up plenty of alternative names, depending on country, intended use, or manufacturer. In textbooks, it often appears as HCCPD or HCCP, just abbreviations to save time. Other synonyms—like hexachloropentadiene, Perchlorocyclopentadiene—reflect the ongoing tangle of nomenclature in chemical commerce. For hazard reporting, standard identifiers such as CAS numbers remove all ambiguity, because there’s little room for error where occupational safety is concerned.
Safety & Operational Standards
For the people who handle hexachlorocyclopentadiene, safety precautions go beyond the normal gloves-and-goggles routine. Engineering controls like sealed reactors, glove-boxes, high-efficiency ventilation, and personal monitors become basic expectations, not extras. Training sessions repeatedly reinforce emergency routines for eye splashes, inhalation, or major leaks—because the acute toxicity and risk of chemical burns are anything but theoretical. I’ve seen facilities where strict storage protocols and real-time leak detection feel less like regulation and more like common sense, learned painfully over decades of industrial incidents.
Application Area
The main applications of hexachlorocyclopentadiene evolved alongside changing public opinion and regulatory action. In the early years, chemical plants gave it pride of place as an intermediate for manufacturing powerful organochlorine pesticides. A push for fire-resistant plastics brought it into the flame retardant industry, especially during decades when flammability standards surpassed toxicity as the focus for product development. Over time, pressure from environmental movements, along with stricter controls on pesticide residues and persistent organic pollutants, narrowed down its legal uses in many regions. The chemical still plays a role in synthesizing specialty chemicals, but large-scale applications dwindled as safer or less persistent alternatives became available. Even so, the legacy of past use lingers in old storage sites and contaminated land, ensuring ongoing attention from hazardous waste managers and chemists alike.
Research & Development
Ongoing research into hexachlorocyclopentadiene involves both new synthesis techniques and efforts to develop remediation approaches for environmental contamination. Scientists work on improving reaction yields, reducing unwanted byproducts, and developing catalysts for selective breakdown. For researchers in my network, the focus shifted over the last twenty years toward how to detect extremely low levels in the environment, how to track its movement through soil and water, and how to engineer microbes or chemical treatments to destroy traces without creating further toxins. Each small win in analytical chemistry or bioremediation gets shared at conferences, with the hope that piecing together technical advances can outpace the spread of contamination.
Toxicity Research
Toxicity studies on hexachlorocyclopentadiene paint a challenging picture. Acute exposures cause burning eyes, respiratory distress, and chemical burns; animal studies show clear evidence of liver and kidney damage, alongside impacts on reproductive systems at higher doses. Over the years, regulators in different countries set exposure limits in air and drinking water, but I’ve seen firsthand how difficult it is to police environmental releases. Unlike some well-known toxins, long-term, low-level exposure data remains incomplete—a gap that worries community activists and toxicologists alike. The chemical’s fate in the body, along with its stubborn persistence in sediments, keeps drawing funding for long-term health studies, especially around contaminated industrial sites.
Future Prospects
Looking ahead, the outlook for hexachlorocyclopentadiene centers on cleanup, safer alternatives, and stricter controls. Environmental chemists continue to refine methods for detecting and eliminating it from waste streams. Green chemistry labs devote resources to wholly new synthesis routes that bypass persistent chlorinated intermediates altogether. To reduce occupational risk, robotics and improved process automation remove workers from direct contact. Policy debates focus on stricter global treaties for persistent organic pollutants; some regions already restrict import and use except in highly controlled settings. No quick solution exists for the contamination already present, but international collaboration and investment in remediation technology offer reason for long-term hope. Each generation of scientists, policymakers, and manufacturers shoulders part of the responsibility—balancing innovation with the duty to avoid repeating old mistakes.
What is Hexachlorocyclopentadiene used for?
The Science Behind It
Hexachlorocyclopentadiene, which often gets called C-56 in industrial circles, isn’t something most people would notice in everyday life. I remember first hearing about it during an internship in a chemical safety office. Back then, I thought it was just another complicated name. Turns out, this chemical shapes a lot of what touches agriculture and health. It serves as a key building block for making pesticides, mostly. Without C-56, many of the bug-fighting products on the market would look different or just not work as well.
Applications that Affect Us All
Most of the volume produced lands in the pesticide business. It helps manufacturers make chemicals like endrin, aldrin, dieldrin, chlordane, and heptachlor. These products target pests that threaten food supply and health, especially in hot, humid regions where insects thrive. Farmers rely on these compounds to keep crops standing and reduce financial loss. I’ve spoken to growers who describe entire seasons riding on how well these chemicals perform.
Apart from its role in agriculture, C-56 finds use in flame retardants and specialty resins. These materials end up in furniture, electronics, and insulation. I caught a news piece about flame retardants making sofas less likely to burn quickly—a clear benefit during house fires. Hexachlorocyclopentadiene acts as one of the chemical bases in these products.
Risks We Face
Chemical progress doesn’t come free. Years spent reading chemical safety bulletins and EPA updates made one thing clear: C-56 is powerful, but also poses serious risks. It can irritate skin, eyes, and lungs. Long-term exposure could hurt the liver or central nervous system. Some of the pesticides built from it collect in the environment and take forever to break down. These compounds have turned up in fish and wildlife, sparking limits and bans in many countries.
During the 1970s and '80s, regulators woke up to the dangers. Communities near manufacturing plants tested higher for contamination. Some areas even faced large-scale cleanups. It’s tough for people living near these sites. From personal conversations with residents in industrial towns, many worry about water safety and possible long-term health issues.
Balancing Need and Safety
The importance of food security and materials that slow the spread of fire can't be dismissed. The issue comes down to using these chemicals responsibly. In my experience, stronger safety standards, better training, and improved containment make a difference. Plants that invest in filtration and real-time air monitoring see fewer leaks and accidents. I’ve visited facilities shifting toward greener alternatives, but it’s not always simple or cheap to swap out chemicals like C-56. Research takes time, and substitutes might not always perform the same way.
Some companies take an extra step and work with local communities, sharing test results and emergency plans. These partnerships help rebuild trust that gets lost after pollution cases. It's no coincidence that the safest facilities often have the lowest turnover—people want to stay where they feel heard and protected. Making process changes, keeping the public informed, and funding clean-up when problems arise all matter more than PR campaigns. We look to experts, not slogans, when it comes to health and the environment.
What are the safety precautions when handling Hexachlorocyclopentadiene?
Why Hexachlorocyclopentadiene Demands Attention
Hexachlorocyclopentadiene, often used in making pesticides and flame retardants, stands out for its reactivity and toxicity. The chemical can cause severe eye and skin irritation, respiratory problems, and organ damage. I have seen workers underestimate how volatile such chemicals can be. They skip on personal protection, thinking a quick task won't cause harm, only to end up with burns or breathing difficulties. The risks go up quickly without solid protection and careful habits.
Personal Protective Equipment Really Matters
Direct contact spells trouble. Skin rashes or chemical burns show up before you know it. I always reach for chemical-resistant gloves—nitrile or neoprene types keep this compound from getting close to the skin. Wearing goggles prevents painful eye exposure and long-term damage. A face shield adds a bit more reassurance for splashy jobs. A lab coat or Tyvek suit stops those nasty droplets from soaking in. Before handling, I make sure my gear is free of holes or wear marks. Breathable masks won’t do here. Full-face respirators with proper filters block toxic vapors and lower inhalation risks.
Engineering Controls Come First
No matter how prepared you feel, relying only on personal gear falls short. Effective chemical handling always starts with using fume hoods or well-designed local exhaust systems. Years ago, a poor exhaust setup filled an entire lab with a choking smell and left staff coughing for days. Workspace airflow makes a difference: high-efficiency ventilation keeps the air clear and cuts down escape of harmful vapors. I’ve learned that skipping maintenance on fans or ducting turns minor leaks into big problems.
Safe Storage Reduces Accidents
Storage demands careful planning. Hexachlorocyclopentadiene reacts aggressively with strong bases or metals, so it belongs in a designated chemical cabinet—never beside acids, solvents, or anything reactive. My old lab labeled all bottles and never kept them above eye-level. Thick, sealed glass keeps leaks in check and prevents accidents caused by thin or cracked containers. Spill trays under containers add another layer of security. Ignoring storage rules sends vapor through a work area and damages more than health.
Spill Cleanup is a Real Team Effort
Spills happen, even with good habits. Having a trusted spill kit nearby saves time and stress. We only use absorbents designed for organochlorines, never sweeping with a broom or rags that might spark or soak through. Evacuating the area and notifying everyone nearby makes a difference every time. My crew learned the importance of clearing the room before suiting up to handle any mess. Sealing waste in strong bags, labeling everything, and sending it for professional disposal keeps others from getting exposed.
Training Empowers Safe Habits
Routine training sticks with me the most. Walking through emergency drills felt repetitive at first, but facing a live incident made everything click. Knowing how to use the gear, recognizing warning signs, and working together helps everyone manage exposure risks. Teams that go over safety sheets every time cut down on errors and stay alert. Reviewing accident reports helps spot habits or shortcuts that can lead to trouble.
Everyday Choices Make a Difference
Making small choices—putting on gloves, checking fume hood flow, double-checking storage—add up to real safety. Overlooking even one step can lead to painful lessons. Health and safety shape how long people stay in this industry. Putting effort into ready equipment, solid training, and reliable storage creates an environment where people get through the workday protected, not exposed.
What are the physical and chemical properties of Hexachlorocyclopentadiene?
Getting to Know its Physical Side
Hexachlorocyclopentadiene doesn’t show up in everyday life unless you work with chemicals or pesticides. You’d recognize it by its strong, pungent smell, a sign to keep your distance. In appearance, it’s a yellow-green liquid, thicker than water. Its boiling point, which creeps above 240°C, keeps it from turning to vapor at room temperatures. It doesn’t mix with water, floating on top if dumped into a pond or river—a worrying trait for anyone concerned about contamination.
I once visited a chemical storage facility. Inspectors wore full protective gear when handling drums of this compound. At room temperature, its vapor can irritate airways, and skin contact brings a burning sensation. Even short handling times demand caution. Companies usually store it in heavy-duty, closed containers, away from sunlight and heat sources. The dense nature of this liquid means spills are not simple to clean up, as it spreads slowly but resists evaporation.
Chemical Behavior: Reactivity and Stability
This compound earns a spot on hazardous lists for good reason. Its structure—a five-carbon ring, decorated with six chlorine atoms—makes it magnetically reactive toward a variety of other chemicals. Manufacturers use it as a building block for things like pesticides and flame retardants because those six chlorines bring power. It reacts readily with strong bases and amines, giving off heat and sometimes forming unpredictable products. Even routine transfer or mixing must be precise and well-monitored.
Researchers have shown it undergoes photodecomposition. Sunlight can split its bonds, breaking it into smaller, often more dangerous pieces like phosgene or hydrochloric acid. That’s why proper storage includes opaque containers. Mixing it with metals like iron or aluminum brings corrosion fast. I remember reading a report where improper storage near old metal piping led to leaks, causing soil and water issues that cost millions to fix.
Risks: Why Care About this Liquid?
People who live near sites using or disposing of this chemical have good reason to ask questions. Low solubility in water doesn’t keep it from causing harm. It builds up in soil and organic materials, sticking around for years. In some studies, animals exposed to even low levels developed liver and kidney damage. There’s evidence it’s genotoxic—able to damage DNA in cells. No one wants to see these effects in the environment or workplace.
Proper handling procedures remain non-negotiable. Regular air monitoring, leak detection, and updated handling manuals are not just bureaucratic requirements. They keep workers healthy and shield communities from risk. Cleanup plans for spills must use absorbents designed for chlorinated compounds, and waste needs high-temperature incineration. Pretending that basic protective equipment will do the trick ignores how quickly this compound can breach safety barriers.
What Can Improve?
Better education ranks high on every solution list. I’ve seen new technicians underestimate this liquid after a short safety briefing—real practice and drills work better. Companies could invest in scrubber systems and sealed process lines, limiting exposure not just for their people, but for anyone nearby. Regulators could step up site inspection rates, keeping pressure on compliance. Sometimes safer alternatives exist, and the push for greener chemistry opens the door for less toxic building blocks.
Some might shrug off the risks until an accident lands in the news, but community voices prove important in driving safer practices. Anyone living or working near chemical plants benefits from demanding transparency and frequent testing results. Sharing what scientists know, in plain language, builds trust and keeps the conversation about risk real and ongoing.
How should Hexachlorocyclopentadiene be stored and disposed of?
Getting Real About Storage
Hexachlorocyclopentadiene is a mouthful to pronounce but harder to manage. This industrial chemical, known for its toxic fumes and chemical reactivity, comes with its baggage. Ignoring the risks—or skipping steps for convenience—ends up hurting people, wildlife, and local communities. From years in research labs and talking with safety officers, tossing this chemical on an ordinary shelf or letting it sit in a rusted drum simply invites trouble.
Safe storage starts with airtight containers made of materials able to handle corrosive content—ideally, glass or high-density polyethylene. Metal drums can rust out, so going for containers that won’t break down under acid-vapor assault makes sense. Avoiding sunlight and keeping the area cool and dry matter even more at places in the South, where this stuff tends to break down faster.
Leaky containers or sloppy labeling mean emergency crews face a guessing game, so clear, chemical-resistant labels and up-to-date inventory hang tags are non-negotiable. Good storage calls for spill-proof secondary containment, whether that's a solid tray or a plastic bin, keeping drips out of floor drains and ground water.
Fire hazards run high. Hexachlorocyclopentadiene reacts with strong bases, metals, or heat. An isolated storage cabinet away from oxidizers, alkalis, or hot equipment gives everyone breathing room. There’s no shortcut; serious injuries stack up in places that cut corners to save a buck or skip maintenance checklists.
Disposal: Less Guesswork, More Protection
Many old-timers remember watching barrels of chemicals get buried or left out back. Now, that carelessness turns up on groundwater and neighborhood medical charts decades later. As regulations strengthened, companies found that safe disposal is a commitment, not a checkbox.
Not all waste handlers can take toxic chemical waste. Only firms licensed and audited under state and federal hazardous waste rules should handle this disposal. My experience with hazardous waste teams showed the best crews work with detailed manifests, dedicated pick-ups, and verified neutralization—or, when needed, high-temperature incineration. Not a single bottle should go into regular trash, down the sink, or into storm drains.
Small spills need special cleanup kits. Full respiratory gear and absorbent pads, plus secure bags, limit toxic exposure. Employees stay healthier, and lawsuits shrink, when emergency response protocols get real-world drills once or twice a year. Those who treat safety drills like an afterthought end up with doctor’s bills and state fines.
Disposal costs money upfront, but accidents or improper disposal cost much more—covering environmental cleanup, community health problems, and legal settlements. In a world where one public report can erase trust built over decades, good stewardship is both ethical and practical.
Solutions for Safer Handling
Facilities working with this chemical do better by training every worker on chemical safety. It’s not enough to depend on a “chemical guy” who holds all the knowledge. Regular training, easily accessible safety data, and new-hire shadowing protect the whole team. Setting up clear, written procedures and response plans turns legal compliance into a culture of respect for health.
It’s worth investing in routine audits—both in-house and from outside experts. Blind spots appear when the same eyes check a room every day. Third-party checks often catch outdated signage, unnoticed container leaks, or blocked exits.
Communities deserve reassurance that businesses near homes, farms, or schools treat chemicals seriously. Direct communication lines, public safety updates, and fast incident reporting help build trust. At the end of the day, safe storage and disposal keep workers, neighborhoods, and the company’s future healthy.
What are the health hazards and first aid measures for Hexachlorocyclopentadiene exposure?
Facing the Hazards
Hexachlorocyclopentadiene, often called “Hex,” has a punch to it that goes beyond its chemical smell. This stuff commonly pops up in places where pesticides and flame retardants are produced. Most folks run into it by accident—either on the job, during warehouse spills, or just by being too close when old drums spring a leak. I once toured a chemical plant where stories of near-misses with Hex made the rounds, and nobody felt shy about the fear it inspires.
Breathing in Hex can leave your lungs burning and your eyes running like you’ve just chopped a bag of onions. Workers have shared stories about hacking coughs, itching skin, and eye pain that didn't quit even after they left the job. Public reports show just how nasty the fumes can get—chest tightness, headaches, even severe breathing problems aren't rare. Some folks end up with chemical burns if the liquid gets on the skin, and if it hits the eyes, it can cause long-term problems. Prolonged or repeated exposure also carries risks for the liver, and animal studies point out real fears about cancer down the road.
Some cases go beyond the immediate pain. Medical journals and OSHA bulletins detail how folks unlucky enough to inhale a strong dose struggled with nausea, vomiting, and symptoms that put them in the hospital overnight. Contact with clothes wet from Hex can lead to blisters and deep rashes. It is not a chemical to shrug off.
Stepping Up to Respond
Hex exposure doesn’t leave much time for hesitation. If it gets on your skin, strip off any clothing that touched it and rinse your skin with running water for at least 15 minutes. A garden hose works if you’re not near a sink or shower. From experience, I can say cold water beats nothing—even if it’s not ideal, flush until help arrives. Avoid scrubbing; friction just makes things worse for an angry chemical.
Eyes need serious attention. Remove contacts fast and break out a clean water source. Hold your eyelid open, letting water run over your eye for at least 15 minutes. Calling for help matters here; don’t try to “tough it out” with a quick rinse.
If the fumes have been inhaled, move into fresh air as fast as possible. Don’t wait for symptoms—just get clear. People who start wheezing or turn pale need prompt medical attention. Loose, fresh air clothing helps while waiting for help and lets the skin breathe. Sometimes medics use oxygen, especially if breathing gets rough.
Swallowing Hex means an immediate call to poison control or emergency services. Don’t make yourself vomit or drink neutralizers unless a professional says so. Every first aid kit at work deserves a card with the number for poison control—far too many jobs skip that basic step.
Preventing the Next Exposure
People handle Hex every day in certain industries, so it makes sense to gear up. No T-shirt or dust mask works here; real protection calls for gloves, goggles, face shields, and well-ventilated workspaces. I’ve seen a plant’s attitude shift after just one incident; eye wash stations went from “maybe next quarter” to front and center by every exit.
Companies get ahead by training workers to spot leaks and spills before the air reeks of chemicals. Regular equipment checks can catch trouble early. Most importantly, listening to workers—because those who sweat through their shifts every day know where corners get cut and risks build up. OSHA and NIOSH guidelines help, too, but everyday vigilance beats any written rule when keeping people safe.
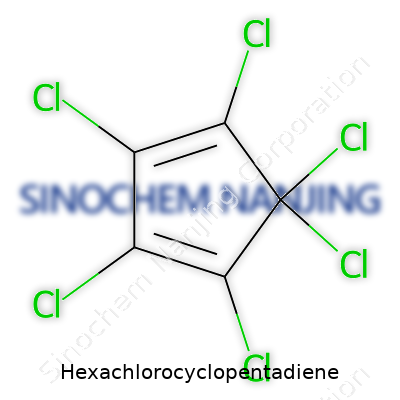
| Names | |
| Preferred IUPAC name | 1,2,3,4,5,5-Hexachloro-1,3-cyclopentadiene |
| Other names |
Perchlorocyclopentadiene Hexachloro-1,3-cyclopentadiene HCCPD C-56 |
| Pronunciation | /ˌhɛk.səˌklɔːr.oʊ.saɪ.kləˈpɛn.tə.daɪ.iːn/ |
| Identifiers | |
| CAS Number | 77-47-4 |
| 3D model (JSmol) | `3D model (JSmol)` string for **Hexachlorocyclopentadiene**: ``` CC1(Cl)C(Cl)=C(Cl)C(Cl)=C1Cl ``` |
| Beilstein Reference | 1361187 |
| ChEBI | CHEBI:38843 |
| ChEMBL | CHEMBL68042 |
| ChemSpider | 6709 |
| DrugBank | DB11385 |
| ECHA InfoCard | ECHA InfoCard: 100.003.203 |
| EC Number | 206-223-3 |
| Gmelin Reference | 83338 |
| KEGG | C11426 |
| MeSH | D006621 |
| PubChem CID | 9583 |
| RTECS number | GV7875000 |
| UNII | OMS547S7C2 |
| UN number | UN2581 |
| CompTox Dashboard (EPA) | DTXSID3020406 |
| Properties | |
| Chemical formula | C5Cl6 |
| Molar mass | 272.77 g/mol |
| Appearance | Light yellow to dark yellow liquid |
| Odor | Pungent |
| Density | 1.65 g/cm³ |
| Solubility in water | 0.077 g/100 mL (20 °C) |
| log P | 3.94 |
| Vapor pressure | 0.00024 mmHg (25°C) |
| Acidity (pKa) | 0.17 |
| Basicity (pKb) | 16.3 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 1.518 |
| Viscosity | 13.1 mPa·s (25 °C) |
| Dipole moment | 2.71 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 340.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -17.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -720.1 kJ/mol |
| Hazards | |
| Main hazards | Toxic by inhalation, skin absorption, and ingestion; causes severe irritation to skin, eyes, and respiratory tract; may cause burns and lung damage; environmental hazard. |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H301, H311, H331, H410 |
| Precautionary statements | P261, P273, P280, P301+P310, P303+P361+P353, P305+P351+P338, P308+P311, P391, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-0-A |
| Flash point | > 224°C |
| Autoignition temperature | 483 °C |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 oral rat 719 mg/kg |
| LD50 (median dose) | 252 mg/kg (rat, oral) |
| NIOSH | GY1225000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Hexachlorocyclopentadiene: 0.01 ppm (0.1 mg/m³) |
| REL (Recommended) | 0.01 ppm (0.1 mg/m3) |
| IDLH (Immediate danger) | 2 ppm |
| Related compounds | |
| Related compounds |
Chlordane Aldrin Dieldrin Endrin Heptachlor Endosulfan Mirex |

