Hexachlorobenzene: Legacy, Lessons, and the Path Forward
The Checkered History of Hexachlorobenzene
Hexachlorobenzene, once a staple in chemical manufacturing, traces its roots back to the early twentieth century. Our industrial ambitions drove its creation, and for decades, global agriculture leaned on chemicals like this to control fungus in crops like wheat. I remember flipping through old science textbooks and seeing pesticide charts filled with unpronounceable names, Hexachlorobenzene always near the top. Governments promoted it as a technological boon, convinced that its stability and effectiveness could only help boost yields. This optimism masked the cost. Only after communities began noticing health issues and persistent environmental residues did the world start asking uncomfortable questions.
Understanding the Chemical at Its Core
At its heart, Hexachlorobenzene comes together from six chlorine atoms clinging to a benzene ring, creating a snow-white crystal. It doesn’t burn in your hand or dissolve easily in water. Reliability drew folks to it—predictable melting points, a knack for withstanding heat, resistance to breakdown in harsh environments. These same features underline much of the trouble. Unlike more fleeting substances, Hexachlorobenzene lingers. It binds to soil, clings to fat in living things, rides air and water far from the original site. What appears durable and unreactive on a chart looks more concerning when you start tracing where residues show up.
Technical Standards and Labeling Challenges
Manufacturers crafted guidelines over the years so users could handle Hexachlorobenzene safely. Labels mention purity percentages, melting points, and stability details. But on the ground, the complexity goes deeper. Regulations exist, but enforcement breaks down in many parts of the world. Some countries banned it for agricultural use by the late 1970s, others lagged behind. I’ve talked with environmental researchers who combed through old warehouses still storing banned chemicals, labeling faded, accountability lost in time. People need clear, honest communication about dangers and requirements, not bureaucratic jargon or bare minimum compliance.
Making and Modifying Hexachlorobenzene
Hexachlorobenzene doesn’t grow on trees—it’s a product of industrial synthesis, most often spun out as a byproduct in the manufacture of other chlorinated compounds. Plants mix benzene and chlorine gas, using catalysts, and after several refining steps, Hexachlorobenzene ends up in storage drums, often together with related compounds. Niche uses appeared in dye and rubber manufacturing too. Chemists long explored tweaking the molecule—swapping chlorines, breaking it apart, trying to coax something useful from a stubborn structure. Yet, nobody found a way to erase its persistence or turn it into something truly benign once it’s out in the wild. Waste treatment facilities struggle to capture and destroy these kinds of chemicals efficiently.
Other Names in the Game
You don’t always see “Hexachlorobenzene” front and center. Synonyms like HCB or benzene hexachloride crop up in academic papers, legal registers, transport documents. This sometimes hinders international cooperation for clean-up or enforcement—one country uses this synonym, another uses that, and the same chemical slips through regulatory cracks.
Safety: A Moving Target
In the lab or on the field, safety comes down to more than gloves and goggles. Users faced health issues after years of real-world exposure: skin disorders, nerve problems, immune system complications. The biggest danger comes from long-term exposure through air, contaminated soil, or tainted food chains. Rural towns near former factories or dump sites discovered this the hard way, children and livestock showing mysterious symptoms while officials debated data sets. Modern standards ask for closed systems, protective equipment, and strict disposal protocols. That only helps when guidelines reach everyone and resources exist to follow them.
Where People Tried to Use It
Agriculture drove most of the demand—fungicides for wheat and other grains. Chemical factories also used Hexachlorobenzene for specialty rubbers and as intermediates in dye manufacturing. Sometimes it ended up as a byproduct in the making of other pesticides or cleaning agents. Many of these uses dwindled as awareness grew, but stockpiles remain, and there are still ongoing efforts to safely destroy leftover supplies.
Push for Research, Reckoning with the Past
Research on persistent organic pollutants exploded in the 1980s and 1990s, with Hexachlorobenzene sitting high on the watchlist. I remember reading WHO and UNEP reports that pulled no punches about this chemical’s persistence and global reach. Decades after most countries banned widespread use, scientists still document traces in arctic animals, river sediments, and human breast milk. Current research focuses on breaking down accumulated residues, monitoring long-range transport pathways, and holding accountable those responsible for historical contamination. Analytical methods improved, making it easier to detect even trace amounts, which can shed new light on old mysteries, but no silver bullet for remediation emerged.
Toxicity: More Than a Lab Statistic
Toxicity isn’t just a number in a spreadsheet. Communities living near old factories know it as a daily burden—painful skin lesions called porphyria cutanea tarda, mysterious liver problems, reproductive health impacts, links to cancers. Animals and aquatic life suffer too. The Stockholm Convention named Hexachlorobenzene as one of a handful of “dirty dozen” chemicals to eliminate because neglecting toxicity research ended up costing more in sickness, environmental loss, and economic strain than anyone expected. Whole regions lost generations of healthy children and destroyed local ecosystems, all for short-term yield gains.
Paths Forward: Moving Past Legacy Contaminants
At some point, you have to accept old mistakes and get serious about fixing them. Solutions start with tough rules and strong monitoring. Many countries learned this lesson late, watching banned chemicals still circulate in black markets or old stockpiles leaking into groundwater. Innovations in chemical destruction—high-temperature incineration, advanced oxidation, and bioremediation—offer hope, but they need more funding and political will. International cooperation matters, since contamination crosses boundaries. Research and policy must work hand in hand, not just to reduce current risk but to develop methods for cleaning up what we already left behind. Educating the next generation of chemists, farmers, and policymakers about the legacy of chemicals like Hexachlorobenzene remains crucial. People have to see the big picture: the short-term benefits of chemical tools are never worth ignoring their long-term costs.
What is Hexachlorobenzene used for?
Why Hexachlorobenzene Mattered in Agriculture and Industry
Hexachlorobenzene, better known as HCB in scientific circles, once filled a surprising number of roles. Farmers who wanted to spare their wheat crops from fungal blight used it without much hesitation. HCB worked as a reliable fungicide, protecting grains during storage and keeping losses at bay. In industrial settings, folks added it to processes that made fireworks, rubber, even ammunition. The goal was to maximize profits and efficiency, not knowing much about risks.
Health Costs Revealed Through Experience
Things looked different after doctors and environmentalists saw what HCB left behind. Back in the 1950s and 1960s, rural communities in Turkey and Eastern Europe felt its impact first. People fell sick, livestock miscarried, and children suffered life-long illnesses. This wasn’t a small nuisance. Doctors saw children with darkened skin, severe health problems, and whole families struggling to recover.
I remember seeing photographs from these areas in an investigative report from the late 1980s. Sick kids wore the scars of chemicals meant to save crops. Their faces stuck with me more than any technical report could. Stories from real families convinced regulators to look deeper into what HCB actually does beyond killing fungi.
Threats Lingering Long After Use
Communities learned that HCB lingers almost everywhere it lands. Crops, soil, rivers, animals — nothing avoids it. HCB lasts for years. This isn’t just scientific jargon; towns downstream from industrial plants still find it in river mud. Fish caught from contaminated lakes carry traces in fat and organs. People eating those fish take HCB into their bodies, risking damage to their liver, immune system, and nervous system.
Researchers discovered it doesn’t just sit in the ground or water. It heads up into the air, riding wind currents for miles. Arctic wildlife, far from any farm or factory, now shows traces. This isn’t something that disappears quickly. HCB joined the ranks of persistent organic pollutants, or POPs. These chemicals build up in body fat and move up the food chain to humans. For anyone who likes to fish or eat wild game, slow-acting chemicals like this are a real threat.
Policy Responses and Practical Solutions
Few governments now permit HCB in crop protection. The Stockholm Convention, a worldwide treaty signed by more than 180 countries, called for strict limits and safe disposal. Factories must keep close watch to stop HCB from leaking during production. In my own work with environmental groups, I saw towns finish long clean-up projects, dredging contaminated sediment and launching safe disposal programs for chemical waste. It took grit and steady funding but paid off in cleaner water and safer crops.
For people living near old industrial sites, simple steps can help. Test soil and water before planting a garden or drilling a well. Push for local health checks and gather neighbors to demand answers about legacy pollution. HCB proves how easily one generation’s fixes become another generation’s problems. Good policy and honest science can fix a lot, but only with watchful communities and strong oversight.
Moving Forward Together
Hexachlorobenzene’s story shows that what helps crops or industry in the short run can pose bigger risks later. Trust in science, push for tough regulation, and never treat an unknown chemical as harmless. Generations benefit when we care about what chemicals go into our food, water, and soil.
Is Hexachlorobenzene hazardous to health?
What Is Hexachlorobenzene?
Hexachlorobenzene, or HCB, doesn’t crop up in everyday conversation, yet this chemical once found use as a fungicide for crops and in some manufacturing. It doesn’t break down easily. Instead, it sticks around in soil, water, air, and even the food chain. Many people have never heard of it, but that doesn’t mean it’s not a concern.
Where Exposure Happens
Think of wheat farms in the 1950s and 1960s. Farmers relied on HCB to fight plant disease. Those who breathed in dust or touched treated plants picked up more than they bargained for. Over time, HCB made its way into river sediments, and fish turned into another route for people to get exposed. Even though most countries banned its use decades back, leftovers from old stockpiles and spills still show up in surprising places, such as near factories that produce chlorine or burn hazardous waste.
What Research Shows About Health Risks
More than one long-term study and outbreak have pointed to some ugly truths. In the 1950s, people in Turkey faced widespread poisoning because of bread made from wheat treated with HCB. The community saw skin sores, blackish discoloration, and joint pain. Birth defects and liver disease followed. Many of those effects didn’t stop once the exposure ended. The chemical built up in people’s fat tissue and got passed on through breast milk, reaching babies before their first birthdays.
Researchers link long-term HCB exposure to cancer—especially of the liver, kidneys, and thyroid. The International Agency for Research on Cancer classifies hexachlorobenzene as a possible human carcinogen. Studies on animals confirm high doses damage the immune system, mess with hormones, and harm the unborn during pregnancy. Even at low levels, ongoing exposure brings risks nobody should take lightly. That’s why the Stockholm Convention lists HCB as a persistent organic pollutant to phase out worldwide.
Why HCB Stays a Problem
Nobody goes out looking for hexachlorobenzene now. The trouble comes from how long it lasts in the ground or water and the way it climbs its way up the food web. I think about old factory sites—many never cleaned up properly. Sometimes companies try to hide old waste or blame someone else for leaks. Testing water and soil for HCB costs time and money, so rural communities sometimes get stuck living with the risk.
People who fish for their dinners, especially in areas downstream from chemical plants, face steady exposure. Those who live close to sites where hazardous waste gets burned also breathe in trace amounts. Regulations exist, but spotty enforcement leaves gaps. If no one checks, or if budgets get slashed, HCB finds ways to stick around.
How to Cut Down Risks
Dealing with hexachlorobenzene calls for honest tracking and responsible handling of old waste. Governments and local agencies have tools to find and clean up soil and water, but public pressure helps. Communities can ask for regular testing, cleaner fish advisories, and full transparency. Farmers now turn to safer chemicals, and land restoration projects help pull these toxins from the environment.
Education plays a big role. If more people know where HCB might turn up—from older landfills to backyard soil—there’s a better shot at avoiding accidental exposure. Supporting better waste management and pushing for safer industrial processes means fewer dangerous leftovers for future generations. No one should have to worry that their drinking water or local food carries a chemical legacy from decades ago.
What are the safety precautions when handling Hexachlorobenzene?
Understanding the Risks
Hexachlorobenzene (HCB) doesn’t have a place in your average household, but it sometimes pops up in industrial settings as a by-product or old pesticide. I remember reading about its notorious history in Sweden—decades ago, children suffered strange disease outbreaks linked to contaminated grain. Those stories stuck with me, mainly because the consequences stretched across generations. Today, HCB is flagged as a probable carcinogen, and it lingers in the environment for years. It builds up in fat tissue, sticking around in the body, with links to liver disease, immune dysfunction, and skin problems.
Personal Responsibility and Protection
Walking into a lab or factory that deals with HCB, you can’t skip the basics. Ordinary gloves and a dust mask won’t cut it. I’ve worked in facilities with strict chemical safety protocols, and HCB ranks high on the list for precautions. For protection, start with chemical-resistant gloves—nitrile or neoprene work best. Safety goggles with side shields keep the dust and vapors away from your eyes. Don’t just wear any coat; use a fitted lab coat or apron that resists chemical penetration. I know colleagues who suit up in full-body gear because the slightest skin exposure can result in blisters or burns. Shoe covers and face shields aren’t optional extras—they keep residues off your skin at every step.
Ventilation and Handling
Dust from HCB sets off trouble, especially if it becomes airborne. I avoid working in spaces with poor airflow. Fume hoods and extraction fans save a lot of headaches. They filter out particles and vapors that would otherwise settle on every surface. During my time in a research lab, we’d always double-check air systems before starting a task. It’s not paranoia—it’s just common sense after hearing stories of accidental lung injuries caused by just a few minutes of exposure and a lapse in procedure.
Strict Hygiene Practices
Once you finish handling HCB, don’t rush off to lunch or your next task. I learned from a safety officer: always use a proper decontamination area, washing hands and face with soap that can remove oily chemicals. Separate all work clothes from your personal ones; dedicated laundry prevents contamination from hitching a ride home. If your gloves or boots feel sticky or coated, toss them—never try to clean and reuse. Chemical burns don’t wait for a second warning.
Safe Storage and Disposal
Proper storage cuts risk immediately. HCB needs a sealed, labeled container, stashed in a cool and dry area, far from heat or open flames. I remember our storeroom supervisor running routine checks to make sure no containers leaked. Leaks or spills lead to bigger problems fast, so containment kits should be close. If a spill happens, use activated carbon or clay-based absorbents, never sweeping the dust or letting it mix with regular trash. HCB belongs in hazardous waste programs run by specialists. Local regulations demand secure transport and disposal, a point the EPA doesn’t leave up for interpretation.
Health Monitoring and Community Protection
Working with toxic chemicals means regular health screening. It catches signs like chloracne early—something I saw once, where a lab worker noticed skin changes after a minor exposure. Immediate reporting and medical care often limit lasting harm. Places handling HCB owe it to both workers and neighbors to communicate risks, monitor air and water, and keep emergency contacts ready.
Better Safety: Training and Teamwork
No one manages HCB safely on their own. Training makes all the difference. Every worker on site benefits from real-world drills—in my experience, hands-on sessions where you practice decontamination and emergency steps result in fewer mistakes than any instructional video ever could. If you see a shortcut, speak up. Cutting corners rarely saves time or trouble in the long haul. Protect yourself, your team, and the wider community by keeping safety practical and serious.
What is the chemical formula of Hexachlorobenzene?
Understanding HCB: What’s In the Name?
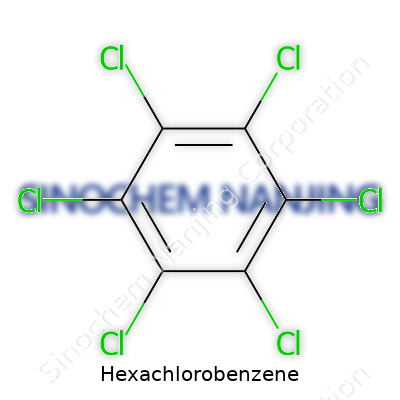
Hexachlorobenzene has the chemical formula C6Cl6. This rings a bell for anyone who remembers basic organic chemistry. Picture a benzene ring, six carbon atoms in a loop, but swap out every hydrogen for a chlorine atom. The result looks simple on paper but cuts a much deeper path in the environment and public health. Nobody hears about hexachlorobenzene unless there’s a story involving pollution or health risk, and that’s not by accident.
Legacy of a Persistent Pollutant
Back in the 1950s and 1960s, farmers used hexachlorobenzene as a fungicide on grain crops. It also cropped up during the manufacture of other chemicals. People didn’t see much harm in it then. But hexachlorobenzene sticks around. It doesn’t break down easily. Reports in places like Turkey (“HCB poisoning epidemic,” 1955) showed how it made whole communities sick. People developed skin sores and liver problems. Some infants died. It’s hard to forget photos from that time—young children with thickened, peeling skin, an image that drives home just how serious chemical exposure can be.
Lingering in the Environment
Hold a handful of soil or look out over a riverbank, and there’s a real chance traces of HCB are present, especially near old factories or in farmland. Once it gets into the ground, it clings to particles and doesn’t move much. It bioaccumulates—showing up in fish and animals, then moving through the food chain. That’s how it enters people, even though many countries banned its use decades ago.
Looking at Exposure and Health Risks
Hexachlorobenzene affects more than just the environment. Studies connect it to cancers, weakened immune responses, and reproductive issues. It’s one of those chemicals that turns up in breast milk samples from around the world, thanks to its persistence. In 2001, the Stockholm Convention listed hexachlorobenzene as a persistent organic pollutant. This wasn’t done lightly; experts weighed the hard evidence over years.
What Does Responsible Management Look Like?
Chemical regulation moves slowly, but after enough evidence mounted, new policies put hexachlorobenzene on the “dangerous” list around the globe. Banning production is one thing, but legacy contamination means the story isn’t over. Cleaning up old sites requires real investment. Technologies exist to break down HCB—like high-temperature incineration or using special bacteria that target chlorinated compounds. Both cost money and need oversight so the fix doesn’t spark fresh problems elsewhere.
Bringing Solutions Into the Everyday
People should know where risks come from, especially folks living near older industrial centers. Community involvement works when cleanups begin. Trust grows when regular health screenings and clear updates roll out. It helps when farmers, local governments, and environmental groups pool their knowledge and advocate for thorough remediation.
Everyday Awareness Counts
The formula C6Cl6 turns up on the label, but it rarely tells people what’s at stake. Protecting future generations goes beyond paperwork. It means looking at the full life cycle of chemicals and building a culture around real, shared responsibility.
How should Hexachlorobenzene be stored and disposed of?
Why Proper Handling Can't Get Overlooked
I remember growing up near a factory town, hearing stories about chemical mishaps. News would travel fast about polluted streams or sick animals after barrels leaked in heavy rain. Those stories stick with you, especially as you learn more about toxic chemicals and their stubborn grip on the environment. Hexachlorobenzene (HCB) stands out as one of those stubborn, lingering threats. Used for years in pesticides and some industrial processes, HCB turns up in the ground and water long after anyone expects it to. Its persistence means there’s no margin for careless storage or disposal. Human health takes a hit when HCB contaminates food and water, and this stuff doesn’t just vanish.
Storage: Not Just Any Barrel Will Do
Deciding where and how to hold onto HCB can’t come down to cost or convenience. Basic sense and public safety demand sealed, corrosion-resistant containers—steel drums with tough linings make a solid choice. Everything stays under cover, away from sunlight and out of the elements, and set on concrete or metal, far from soil or drains. Locked and labeled rooms stop accidental access; dedicated staff guard against slip-ups. I’ve seen what one missed label or distracted worker can spark—confusion, emergency calls, threats to whole neighborhoods. Rules here exist for a reason.
Storing HCB isn’t about just keeping things tidy. One cracked lid during a flood or fire can unleash toxins, hurt workers, and spark costly cleanups that never fix the real damage. No one wants to trust an old, rusting shed with barrels that should stay tightly sealed for the sake of people, pets, and property. Temperature also matters. High heat can set off dangerous reactions, so keeping things cool and stable stops more trouble before it starts.
Disposal: Responsibility Runs Deep
Getting rid of HCB involves a lot more than hauling away trash. High-temperature incineration stands out as the safest way to break down this tough chemical, burning it at over 1,100°C to destroy those cancer-causing compounds. Anything less usually fails to finish the job. Only a few facilities have the equipment to handle that heat, and those operators follow detailed safety procedures from start to finish. Incomplete burning or open dumping only spreads pollution across air, water, and soil.
Some cities and regions face illegal dumping—barrels hidden in forests or buried under construction sites. Groups rooted in local communities, from farmers to anglers, have helped track and stop those abuses. Setting up public reporting hotlines, funding clean-up crews, and pushing for stricter inspections gets the job done. Rewarding whistleblowers can uncover the backroom deals that lead to disasters.
Taking Prevention Seriously
Getting HCB out of regular use and into safe disposal depends on clear, enforceable laws and public oversight. Real progress starts with tighter chemical controls and investment in cleanup research. Training workers, from floor staff to supervisors, builds a wall against accidents or shortcuts. Knowing the full risks builds a culture of care—families shouldn’t have to worry about toxins landing in the backyard because someone cut corners on waste management. If tighter rules, better training, and community watchdogs join up, the next generation might only know HCB as a lesson from the past, not a headline about the present.
| Names | |
| Preferred IUPAC name | 1,2,3,4,5,6-Hexachlorobenzene |
| Other names |
HCB Perchlorobenzene Benzene hexachloride Phenyl hexachloride Hexachlorobenzol |
| Pronunciation | /ˌhɛksəˌklɔːrəˈbɛnziːn/ |
| Identifiers | |
| CAS Number | 118-74-1 |
| Beilstein Reference | 1361149 |
| ChEBI | CHEBI:35527 |
| ChEMBL | CHEMBL14337 |
| ChemSpider | 5059 |
| DrugBank | DB14015 |
| ECHA InfoCard | ECHA InfoCard: 100.000.020 |
| EC Number | 204-825-9 |
| Gmelin Reference | 60627 |
| KEGG | C06738 |
| MeSH | D006520 |
| PubChem CID | 3545 |
| RTECS number | GV9810000 |
| UNII | R8O0ZHT5F7 |
| UN number | UN2729 |
| Properties | |
| Chemical formula | C6Cl6 |
| Molar mass | 284.78 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 1.568 g/cm³ |
| Solubility in water | Insoluble |
| log P | 4.89 |
| Vapor pressure | 0.00011 mm Hg at 25°C |
| Acidity (pKa) | NA |
| Magnetic susceptibility (χ) | -80.0e-6 cm³/mol |
| Refractive index (nD) | 1.569 |
| Viscosity | 1.13 mPa·s (20 °C) |
| Dipole moment | 2.55 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 324.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -18.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3267.8 kJ/mol |
| Pharmacology | |
| ATC code | P50AC03 |
| Hazards | |
| Main hazards | May cause cancer. Causes damage to organs through prolonged or repeated exposure. Toxic if swallowed, in contact with skin or if inhaled. Very toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS07, GHS08, GHS09 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H350, H400, H410 |
| Precautionary statements | P260, P261, P264, P270, P271, P273, P301+P310, P302+P352, P304+P340, P308+P313, P314, P320, P330, P391, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-0-☠️ |
| Autoignition temperature | 654 °C (1209 °F; 927 K) |
| Lethal dose or concentration | LD50 oral (rat) 10,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): rat oral 10,000 mg/kg |
| NIOSH | SN4550000 |
| PEL (Permissible) | 0.2 mg/m³ |
| REL (Recommended) | 0.002 mg/m³ |
| IDLH (Immediate danger) | N/D |
| Related compounds | |
| Related compounds |
Chlorobenzene Dichlorobenzene Trichlorobenzene Tetrachlorobenzene Pentachlorobenzene Benzene Polychlorinated biphenyls |

