Hexachloroacetone: Unpacking a Complex Chemical Story
Tracing the Long Road: How Hexachloroacetone Entered the Scene
Hexachloroacetone didn't just turn up in labs as a curiosity. Chemists chased chlorinated organics for decades across the evolving landscape of industrial chemistry. In the late nineteenth and early twentieth centuries, scientists explored the boundaries of halogenated carbon compounds, searching for new functionalities, reactivity, and insights into molecular structure. Hexachloroacetone emerged as a byproduct from efforts to push acetone’s chemical potential. Unlike its more popular cousin chloroform, it never made splashy appearances in mainstream media or medicine. Companies running heavy synthesis—pesticides, pharmaceuticals, plastics—kept an eye on this dense, pungent liquid, and its niche uses grew from deliberate tinkering and advances in chlorination techniques.
Understanding What Sets This Molecule Apart
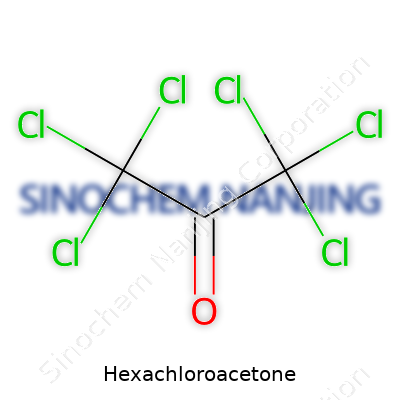
This chemical stands out in a lineup of chlorinated ketones by virtue of having all positions next to the carbonyl group loaded up with chlorine atoms. It ends up as a clear, oily liquid, heavier than water. Its chemical formula, C3Cl6O, tells you just how saturated with chlorine things get. That dense packing influences everything else: volatility, reactivity, smell, and how it behaves with water, solvents, and biological matter. It evaporates slowly at room temperature, almost always carrying a sharp and distinct odor—one you don’t forget after spending time around organohalogens.
Boiling Points, Solubility, and Beyond
If you pour out hexachloroacetone, you’ll notice its weight compared to water. The boiling point stretches into the mid-200s Celsius, much higher than cleaner, lighter acetone. It shows some stubborn resistance to dissolving in water but fares far better in organic solvents such as ether or chloroform. Trickier still, it decomposes if you bring in heat or strong acids, sending off chlorinated fragments that often outpace what you thought was possible under milder conditions. A substance with these characteristics demands respect, not just for its reactivity, but for the way it sneaks into unexpected places in the lab and in processes.
Labeling and Technical Details: More Than Box-Checking
Regulations continue to tighten the screws on how hexachloroacetone gets handled. Detailed chemical labeling requirements don't stem from bureaucratic busywork; they grew out of industry’s hard-learned lessons. Shipments traveling across states or countries need tags that warn about both acute toxicity and potential chronic harm. Labels often note the UN number for hazardous goods, stress the importance of handling precautions, and alert users to firefighting oddities—chlorinated compounds throw nasty fumes when flames break out. Any batch made for industry carries purity specs, with thresholds for permitted impurities, especially given how leftover chloroform or other byproducts can affect downstream utility and safety.
The Craft of Synthesis: Making Hexachloroacetone
Converting everyday acetone into the heavily chlorinated beast that is hexachloroacetone doesn't take a wizard’s touch, but it demands patience and skill. Chlorination of acetone, often carried out by bubbling chlorine gas through cooled solutions, stands as the typical route. Side products build up, corrosion threatens equipment, and environmental risks creep in if vents, seals, or trapping systems fail. Running this process at scale taught generations of chemists about reactor design, temperature control, and the crucial role of quenching and scrubbing systems for off-gas.
Chemistry in Action: What It Does in the Lab
Chemically reactive, hexachloroacetone offers a playground for researchers probing ketone chemistry, organic synthesis, and halogen exchange. If you bring in nucleophiles—think water, amines, or strong bases—you set off a flurry of substitution reactions. That heavy load of chlorine makes the molecule both a potent electrophile and a springboard for adding even trickier groups. Its use as a chlorinating agent lets it transfer those chlorines onto other molecules, which matters when you need to introduce reactivity or manipulate molecular geometry. There's a thrill to watching a familiar group like acetone transformed so deeply by halogenation, opening new paths in synthesis.
So Many Names, So Much Confusion
Ask around in different labs and you might hear it called 1,1,1,3,3,3-hexachloro-2-propanone, parachloracetone, or even by industrial codes tied to supplier catalogs. The proliferation of synonyms crops up from different rational naming systems and decades of chemists giving things their own shorthand. This maze of names frustrates newcomers and muddies the waters in safety databases, supply chains, and research literature. Keeping tabs on synonym lists helps prevent shipping mistakes, research missteps, and even simple miscommunication during procurement.
Not Just a Lab Curiosity: Where It Ends Up Used
Hexachloroacetone never landed headline scientific applications, but it carves out sturdy niches. The agricultural sector once leaned on it for herbicides and pesticides, tapping its reactivity for controlling pests. Chemical manufacturers pull it out when they need to make specialty compounds or harness its unique chlorinating power. Even if not in every chemistry classroom, it finds its way into organic synthesis research as a potent tool for building more complex molecules or preparing intermediates for pharmaceuticals and dyes. Anyone who spent time in industry-scale synthesis recognizes its occasional role despite increased pressure to use greener alternatives.
Working Safely: Operational Realities and Regulations
Handling hexachloroacetone calls for vigilance. Chlorinated ketones don’t play nice if spilled or inhaled. Standards around its use keep changing as health studies surface new risks: skin and lung irritation, worries about carcinogenicity, and unknowns around long-term, low-level exposure. Plant operators, lab managers, and chemical workers know the drill: respirators, chemical-resistant gloves, splash-proof goggles—the works. Developing countries sometimes lag in enforcement, which brings real danger. Facilities that cut corners—on ventilation, waste controls, or staff education—pay dearly in the form of accidents and regulatory crackdowns.
Toxicity: A Shadow Hanging Over Utility
Stories abound of workers knocked back by fumes or skin exposed to accidental splashes that left behind rashes and painful burns. Toxicological research paints a picture that keeps getting darker as more data piles up. Hexachloroacetone’s metabolites likely bind proteins and DNA, raising red flags about chronic disease risks beyond immediate symptoms. Rats exposed to the stuff in labs show damage to the liver and kidneys, while earlier risk assessments sometimes played down dangers in an era less aware of chronic low-dose effects. That growing awareness spurs tighter controls, fresh research, and efforts to substitute safer chemicals where possible.
Chasing Greener, Safer Alternatives: The Future Unfolds
Mounting environmental and public health pressures push chemical companies to pivot away from old chlorinated mainstays. Research into less toxic replacements and greener reaction conditions keeps accelerating. Scientists now hunt for catalysts that remix acetone chemistry without tons of chlorine or for alternative building blocks that lower production hazards. Universities and R&D labs tinker with biodegradable pesticides or medical precursors that demand fewer harsh reagents. As stricter regulation spreads from North America and Europe to other regions, smaller producers face demands to disclose emissions, treat waste streams, and invest in staff safety training. Scaling these changes takes time, but younger chemists want careers built on sustainability and health protection, not just output or profit.
Where R&D Must Focus Next
We need deep dives into bioaccumulation, long-term exposure effects, and breakdown pathways both in the environment and inside living bodies. Modern analytical techniques can trace minute residues in air, soil, and water—data that drive smarter policy and safer handling standards. On top of that, process engineers need to retrofit plants for better containment, recycling, and waste reduction. Scientists have a duty to bridge gaps between bench chemistry and policy, explaining complexities without jargon. By sharing real-world observations and connecting policy changes to on-the-ground improvements in worker safety, researchers can shape a field that respects both innovation and human well-being.
What is hexachloroacetone used for?
What Drives Its Use
Ask anyone working in an industrial lab or specialty chemicals company about hexachloroacetone, and you’ll likely hear stories about its bite—both in terms of its pungent odor and its chemical strength. This compound shows up in niche spots, particularly where powerful reagents are called for. In real life, chemists rely on it as a strong chlorinating agent in the lab. It can strip away hydrogen atoms and replace them with chlorine, helping to build molecules for dyes, pharmaceuticals, and some pesticides.
What stands out is how little most people outside these circles have heard about it. This stuff stays behind closed doors because exposure to hexachloroacetone carries real health risks. The chemical can vaporize easily and irritate the lungs, eyes, and skin. Several years ago, I toured a specialty chemical plant and noticed entire sections isolated behind strict barriers—hexachloroacetone was one of the chemicals behind all that extra effort. Workers wore advanced respirators and protective suits just to handle it safely. It can’t have regular contact with the environment or people without careful management.
Risks Beyond the Lab
Accidents do happen. The EPA keeps track of releases and takes public complaints about chemical smells seriously. Hexachloroacetone can contribute to air and water contamination if a leak goes undetected. It’s toxic to aquatic life and can linger in the environment. A case from the 1980s showed how a small spill into a river affected water supplies for months. It doesn't belong anywhere near public drinking sources, playgrounds, or open warehouses.
This isn’t just paranoia. The Centers for Disease Control has flagged the compound as hazardous, and regulators enforce tight workplace exposure limits. Washing up after handling it is not something to gamble with—chemical burns and severe respiratory symptoms come up in the safety literature. Red flags on the label aren’t just for show.
Seeking Safer Paths
Factories keep looking for greener alternatives. Sustainable chemistry offers hope here. Companies and researchers keep pushing to swap out old-school chlorinated chemicals in manufacturing. Moving away from compounds like hexachloroacetone takes investment in new equipment, dedicated staff training, and stronger process control. The cost can be steep, but the alternative risk to worker health and local communities carries an even higher price.
On a small scale, labs substitute friendlier reagents in experiments where safety trumps tradition. Online resources and journals track every step in safer methods, but the process takes time—old habits and equipment don’t vanish overnight. Change happens, though, especially when regulatory agencies step in or when a high-profile accident reminds everyone why vigilance matters.
Looking Ahead
Experience teaches that narrowly focused chemicals such as hexachloroacetone, with all their hazards, pressure the industry to adapt. I saw a shift among plant managers who started offering more frequent safety briefings and drills. Some labs switched over to completely contained reactors, using remote operation to remove people from harm’s way. It's not just about compliance, but about keeping people healthy and communities safe from chemical threats lingering out of sight.
Is hexachloroacetone hazardous to health?
Looking at the Risks Around Hexachloroacetone
Anyone working with chemicals often faces labels with long, intimidating names. Hexachloroacetone stands out in this list. It’s clear and almost odorless, used in some chemical manufacturing and research labs. Modern health and safety training keeps telling us to treat chemicals with respect, but stories always stick better than safety posters. I remember the sharp chemical smell wafting down the hall of an older chemistry lab—there’s a reason stories about accidents linger long after cleanup wraps up.
Hexachloroacetone doesn’t usually show up outside chemical facilities, but it does bring risks. According to information from the European Chemicals Agency, hexachloroacetone irritates eyes, skin, and the respiratory tract. Short exposure causes stinging or burning sensations, especially in closed spaces with poor ventilation. Inhaling fumes or coming into contact with the liquid can make a bad day worse, leading to headaches, coughing, or skin rashes.
To add to the concern, several toxicology studies outline that high exposure over time might damage the liver and kidneys. This isn’t rare among chlorinated organic compounds, a group that often includes some nasty actors. Hexachloroacetone breaks down into toxic byproducts when heated or mixed with incompatible chemicals, so carelessness in disposal or storage often brings deeper problems. I’ve watched a coworker land in the emergency room after a splash from another chlorinated solvent—something about those compounds makes reactions more dramatic and recovery longer.
It’s not just workers at risk. Environmental authorities routinely monitor wastes containing hexachloroacetone. Spills seep underground, reaching water sources and spreading harm far beyond the original site. Wildlife and people who rely on wells find themselves affected, sometimes years later. Factories in the past rarely worried about these chemicals leaching into groundwater, but contaminated sites often show up in EPA cleanup lists today. Preventing further damage means tightening every link in the handling chain.
No one stays safe by luck. Proper protection—like chemical-resistant gloves, goggles, and strong ventilation—forms the front line against accidental exposure. It feels uncomfortable to suit up fully, but the first splash on unprotected skin turns every skeptic into a believer. Regular training and clear warning signs matter just as much as fancy new equipment. Easier access to solvents with fewer long-term risks cuts down on accidental poisonings both at work and during cleanups.
Laboratory managers face constant pressure to streamline safety protocols and reduce chemical storage. Swapping hexachloroacetone for safer alternatives often makes sense, especially for educational labs or smaller-scale industry. Regulatory agencies already push for stricter exposure limits and better record-keeping. In my experience, an open-door policy between safety teams and staff makes a huge difference. People share concerns sooner and unsafe practices get addressed before trouble starts.
Hexachloroacetone earns its reputation as hazardous, but dangers fade when awareness stays high. Company leaders, regulators, and workers all play a part. The cost of keeping everyone informed and protected looks small compared to the fallout from a single mishap. Over time, the overall workplace becomes less intimidating and downright safer for everyone on the floor.
What are the storage requirements for hexachloroacetone?
Storing Hexachloroacetone Without Cutting Corners
Hexachloroacetone has a reputation that gets folks in labs and warehouses paying special attention the moment a drum shows up. This stuff, built from a backbone of carbon and drowned in chlorine, checks all the boxes for chemicals that ask for respect. I learned early that storing materials like this isn’t just about checking off regulations; it’s about making sure no one ends up dealing with a cleanup that grabs headlines.
Stable Sheltering, Not a Back Shelf Job
Anyone trusting hexachloroacetone to a forgotten shelf in a makeshift storage room is risking more than a slap on the wrist from inspectors. The chemical needs a stable, cool spot—ideally fewer temperature swings and no sunlight streaming in. In my experience, direct light and fluctuating heat don’t just stress the container; they stir up the more volatile personalities within the bottle.
Plastic rarely cuts it; sturdy, compatible glass or select metal containers with a tight, leak-proof cap keep things safe. I’ve seen colleagues tape labels over closure threads and believe that’s “good enough.” The label belongs front and center, clear and unfaded. Fire officials want instant clarity, not a treasure hunt.
Fire Risks and Fume Stories
Flammability gets most of the press, but hexachloroacetone’s fumes tell their own story. Even a slight whiff can send you coughing: that harsh, biting smell doesn’t take much to sting your nose or throat. Keeping the storage room well-ventilated matters. Personal experience—standing next to a leaking container for just minutes—reminded me that ventilation isn’t a checklist item; it’s a barrier between a regular workday and a crisis.
Fire suppression stands by the door for a reason. Water doesn’t play well with chemicals like this, so dry chemical extinguishers stay closest by.
Segregation—A Policy With Teeth
No storage plan works unless it keeps incompatible chemicals on opposite sides of the room—or even in separate vaults. Hexachloroacetone won’t play nice with strong bases, amines, or even some metals, so mixing them turns careless storage into a recipe for disaster. I’ve listened to old-timers at chemical plants recount the messes left behind when corners were cut or labels got mixed up. Segregation isn’t paranoia—it makes common sense.
Daily Checks and Emergency Plans
A manager once told me, “Don’t trust yesterday’s inspection; give it a new look today.” That thinking paid off more than once, especially when seals on containers dried out or corroded faster than expected. Spotting trouble before it spreads can save more money and stress than waiting for alarms to blare.
Good storage of hazardous materials relies on more than locked doors and posted signs. Staff takes training seriously, learns where the nearest eyewash and shower are, and drills the evacuation plan until it’s second nature. If a leak happens, everyone knows their role and acts fast.
Better Storage, Safer Outcomes
Strong policies, regular training, and a respect for how quickly things can go wrong keep people and property safe. Chemical safety only looks like extra work until it’s the line that stands between routine and regrettable headlines. Real-world experience lays out the way forward: focus on fundamentals, keep information current, and never let familiarity turn into complacency.
How should hexachloroacetone be handled safely?
Recognizing the Risk
Hexachloroacetone stands out as a hazardous chemical—its vapor stings your nose, eyes, and skin. Anyone who has worked with chlorinated compounds knows how fast a small mistake becomes a bad day. One breath can make your lungs burn or eyes tear up. There’s no romance in lab accidents or scrambling for the emergency shower, and hexachloroacetone gives even seasoned researchers pause.
Setting Up for Safety
Good practice starts in the lab design. Only work with hexachloroacetone in a well-maintained fume hood. Not every chemical needs this, but hexachloroacetone’s volatility and toxicity leave zero room for shortcuts. Before cracking open a bottle, double check the airflow, and never prop the sash too high. Scrubbing the air isn’t just about avoiding the smell—it’s about protecting everyone sharing that space.
Gloves get a lot of attention, but not all gloves handle solvents like this one. Nitrile or neoprene gloves hold up well, but thin latex fails fast. Even with thicker gloves, swapping them out at signs of wear gives peace of mind you can’t get from old, stretched-out material. Eye protection is non-negotiable—chemical splash goggles keep your eyes safe from a single stray drop or a hidden splash.
No Room for Carelessness
Hexachloroacetone triggers coughing, headache, and worse, but it’s not just about personal comfort. Think about anyone who might walk through that space next. Contamination lingers. Spills need immediate attention. Once, I watched a careless pour spread across a bench, and evaporation filled the air with vapor. Quick response meant grabbing the right spill absorbent—commercial pads, not paper towels—and keeping everyone calm. Only specialized waste bins should take used absorbents and gloves.
Training and Awareness
Every new researcher in my lab got a walk-through: emergency showers, eyewash stations, and where the safety data sheets sit. Nothing beats hands-on training for learning proper chemical transfer, waste disposal, and spill response. Safety meetings can feel routine, yet they prevent confusion when trouble starts. If a protocol stays updated with new best practices, the odds of an ugly accident drop dramatically.
Storing Chemicals with Care
Hexachloroacetone never belonged near water or acids—mixing spells trouble. It lives in a corrosion-resistant cabinet far from incompatible chemicals. Label the storage bottle with the date received and date opened. Open bottles don’t last forever, even capped tight. Routine inventory checks help catch aging containers before leaks or crystal buildup threaten safety.
Solutions that Work
Real protection demands a plan that goes beyond rules on a poster. Air monitoring, regular fume hood inspections, and continuous feedback between staff and supervisors build a culture of safety. Good communication catches trouble before it snowballs. Reach for experience—ask older colleagues for tips, and share what works and what doesn’t. Sometimes, solutions look simple: a clear sign on a cabinet, a buddy check before working late. Little habits like this carry the weight of experience and put fewer people at risk.
What is the chemical formula of hexachloroacetone?
Breaking Down the Formula
Hexachloroacetone slips off the tongue like something out of a science fiction novel, but its footprint is much closer to home. The chemical formula for hexachloroacetone is C3Cl6O. This stark sequence of carbon, chlorine, and oxygen atoms sets off immediate curiosity among chemists and folks dealing with industrial chemistry. Each piece of the formula brings its own trick to the table, shaping a compound that’s more than just a mouthful of syllables.
Why Hexachloroacetone Grabs Attention
I remember walking through a university chemistry lab years ago, the smell of cleaning agents hanging in the air, and hearing someone mention hexachloroacetone. The name stuck, mostly because of its reputation. This is not something you keep under your kitchen sink or find on a store shelf. Hexachloroacetone draws notice for its use in specialized chemical processes, such as serving as an intermediate in pesticide manufacturing and its rare appearance as a chemical warfare agent long ago. While many chemicals fade into obscurity, hexachloroacetone’s formula pops up whenever discussions about serious industrial controls and safety practices take place.
Safety and Handling: Lessons From the Past
Anyone with experience in a professional chemistry lab understands the itching discomfort that comes with mismanaging volatile substances. Hexachloroacetone demands respect. The six chlorine atoms don’t just contribute to its name—they amp up toxicity, volatility, and potential for environmental harm. Labs carry strict rules about how to store and dispose of such substances. I recall a colleague sharing a story about a minor spill; even the smallest quantity threw the entire lab into evacuation mode. This tells its own story: an academic understanding of a formula matters, but implementing rigid safety habits matters even more.
Real-World Uses of C3Cl6O
The everyday world rarely hears about hexachloroacetone, yet its presence in the manufacturing chain of certain chemicals can't simply be ignored. In agriculture, hexachloroacetone has functioned as a building block for some herbicides. The chemistry behind producing these tools for crop protection traces a line running straight through this compound’s formula. During the cold war era, the same chemical structure was pressed into service for far more sinister purposes—proof that the formula’s importance stretches from fields to history books.
Environmental Impact and Alternatives
Satisfying curiosity about hexachloroacetone’s formula means acknowledging consequences. With global concerns over persistent chemical pollutants, compounds like this one are coming under increased scrutiny. Regulators look for pathways to phase out hazardous intermediates in favor of cleaner alternatives. In the past decade, innovations in green chemistry have started to yield new, less toxic compounds, offering options that meet agricultural needs without putting workers, communities, or ecosystems at unnecessary risk. Companies have begun switching to those alternatives, proving that responsible chemistry has no excuse in falling behind.
Why Exact Formulas Matter in Daily Decisions
Studying hexachloroacetone’s formula is far from an abstract exercise. Every chemical equation sits at the crossroad of practical outcome and personal safety. Young scientists who memorize C3Cl6O also carry forward years of lessons—lessons in caution, precision, and deep respect for the impact that one small bottle or barrel can have. It isn’t about rote memory; it is about grasping exactly what we work with and deciding, each time, to put safety and sustainability on the lab bench right next to innovation and discovery.
| Names | |
| Preferred IUPAC name | 1,1,1,3,3,3-Hexachloropropan-2-one |
| Pronunciation | /ˌhɛksəˌklɔːrəʊ.əˈsiːtəʊn/ |
| Identifiers | |
| CAS Number | 116-16-5 |
| 3D model (JSmol) | `3D model (JSmol) string for Hexachloroacetone: "CC(=O)(Cl)(Cl)Cl"` |
| Beilstein Reference | 1206960 |
| ChEBI | CHEBI:39227 |
| ChEMBL | CHEMBL167786 |
| ChemSpider | 22220 |
| DrugBank | DB01840 |
| ECHA InfoCard | 100.014.063 |
| EC Number | 206-048-4 |
| Gmelin Reference | 583 |
| KEGG | C14205 |
| MeSH | D006597 |
| PubChem CID | 15649 |
| RTECS number | AB1925000 |
| UNII | W0P5T0606R |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C3Cl6O |
| Molar mass | 272.75 g/mol |
| Appearance | Colorless liquid |
| Odor | Penetrating odor |
| Density | 1.77 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 1.94 |
| Vapor pressure | 0.4 mmHg (20 °C) |
| Acidity (pKa) | 15.3 |
| Basicity (pKb) | Not basic |
| Magnetic susceptibility (χ) | -98.8×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.546 |
| Viscosity | 1.29 cP (20°C) |
| Dipole moment | 1.97 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 282.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -117.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -469.9 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | D08AX01 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; causes severe skin burns and eye damage; may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. H314: Causes severe skin burns and eye damage. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P210, P260, P273, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P403+P233, P501 |
| NFPA 704 (fire diamond) | 3-1-0 |
| Flash point | 69 °C (156 °F; 342 K) |
| Autoignition temperature | 205 °C |
| Lethal dose or concentration | LD₅₀ (oral, rat): 620 mg/kg |
| LD50 (median dose) | 457 mg/kg (rat, oral) |
| NIOSH | SN1227000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Hexachloroacetone is "1 ppm (10 mg/m³) |
| REL (Recommended) | 10 ppm |
| IDLH (Immediate danger) | 13 ppm |
| Related compounds | |
| Related compounds |
Chloroacetone Hexafluoroacetone Trichloroacetyl chloride Trichloroacetic acid |

