Hexachloro-1,3-Butadiene: Understanding the Journey, Impact, and Dilemmas
Tracing the Roots
Hexachloro-1,3-butadiene rolls out of a time in chemistry when industries moved fast and regulations walked slowly. Its roots stretch back to the mid-20th century, growing from the demand for solvents and the byproducts of chlorinated hydrocarbon synthesis. During those early chemical booms, factories pulled Hexachloro-1,3-butadiene from the residue left during tetrachloroethylene or trichloroethylene production. What started as industrial waste became a feedstock, a cleaning agent, and sometimes an afterthought in manufacturing. The chemical industry’s history records these shifts in value along with changing priorities and evolving awareness about health.
Getting to Know the Material
Sitting with a bottle of Hexachloro-1,3-butadiene, one notices a thick, colorless-to-yellow liquid, a faintly sweet odor. It doesn’t make a fuss, but that belies a prickly reputation. Its chemical formula, C4Cl6, translates to a stubborn molecule, hardy against fire but less so under sunlight, where it may break down, forming toxic byproducts. Facts about its boiling point, density, and solubility define its handling, but in a storage shed or lab, it's the sense of potential hazard that shapes habits. Folks who work with it rarely become attached; they measure, transfer, and waste no time.
Labels, Specs, and Technical Truths
Labels do a lot of heavy lifting for Hexachloro-1,3-butadiene. Regulatory signs, sharp pictograms, and scrupulous specifications underscore the risks. This molecule’s labeling goes beyond “Toxic” or “Environmental Hazard.” It forces a level of respect, not just during day-to-day use but from anyone responsible for environmental release. Its technical details aren’t just trivia, either—they inform emergency plans and the type of gloves or respirators folks rely on.
Crafting the Chemical
What comes out of a synthesis reactor often depends on intention. For Hexachloro-1,3-butadiene, most lines point back to the chlorination of butadiene or as a secondary product when making tetrachloroethylene. High temperatures, a surplus of chlorine gas, and some watchful eyes shape the reaction course. The yield, tone of impurities, and leftover bits tell a lot about the skill behind the process. Few outside industry realize just how much chemical engineering goes into not just making something, but keeping everything safe on the other end.
What Happens in the Laboratory
Hexachloro-1,3-butadiene rarely stays static. Reactivity with bases and reducing agents leads chemists to develop new mixtures and compounds—although not always by design. Fumes, colored vapors, or a sticky film remind everyone present why chemical reactions involving this material deserve close attention. Anyone who’s tried to clean up a spill remembers the stubborn odor, the slow evaporation, and the persistent reminder that molecules leave traces well after they go down the drain.
By Any Other Name
The chemical goes by several aliases—Perchlorobutadiene, HCBD, and others—which sometimes confuses conversations. Ask around in a warehouse or an older research lab, and you’ll hear all sorts of regional slang or shortened versions. This patchwork of names often shows how a chemical can cut through industry lines—from pesticides to rubber processing, even treading into less savory uses. Each synonym carries subtle baggage, tied to regulations, tradition, or a legacy of uncertainty.
Guardrails in Practice
No one walks blindly into work with Hexachloro-1,3-butadiene anymore. Modern standards knock into place after painful lessons in toxicity and environmental disaster, notably the inclusion of the chemical in protocols like the Stockholm Convention. Anyone pouring, storing, or transporting this material relies on engineered controls, real ventilation, and training that comes from dire environmental case studies. In regions with strong safety rules, handling involves closed systems, negative pressure, and emergency procedures drilled into muscle memory. Where standards slip, accidents and releases enter the news, leaving reminders about why guidelines matter and why investments in real oversight can’t get sidestepped or delayed.
Where It Lands: Uses and the Shadow Beneath
Once, manufacturers welcomed Hexachloro-1,3-butadiene as a versatile solvent, a precursor for making rubber compounds softer, sealing agents tougher. Some pesticide chemistries leaned on it, and even now, legacy applications pop up in older patents or niche industrial flows. These uses, though, funnel almost exclusively to industry. No character floating through daily life gets exposure unless something slips—through a leak, illegal dumping, or mishandling. For communities living near old production sites, this material sometimes showed up in soil or water, long after production lines closed, reminding everyone that chemical legacies hang around well past their intended purpose.
Pushing the Frontiers: Research and Rethink
Barely a decade goes by without a new twist in toxicology research around Hexachloro-1,3-butadiene. Early studies pointed to kidney toxicity and issues for aquatic life. Recent science, boosted by sharper instruments and tighter data, peels away more layers. Researchers piecing together exposure patterns or environmental persistence help regulators understand not just direct toxicity but how breakdown products circulate in wildlife and humans. Environmental advocates push for more sampling, not less, keeping laboratories busy. Questions pile up: What happens when trace residues make their way up food webs? How does chronic exposure reshape risk profiles for workers and communities? Honest, ground-level science draws attention to these puzzles, and nobody who reads these studies comes away unmoved.
Charting the Unknown: Future Outlook
In most chemical stories, future prospects mean new uses or better performance. With Hexachloro-1,3-butadiene, the outlook asks something deeper: how to phase out hazards while reckoning with the past. Policy shifts in many countries already ban or restrict production, nudging industry to safer substitutes and better waste management. Research pivots toward remediation, destruction technologies, and smarter monitoring of contaminated zones. People who grew up near industrial towns know that cleanups take decades, and that trust builds slowly. Solutions rarely feel complete, but steps like public reporting, regulatory teeth, and investment in green chemistry—these tools build a version of progress that measures not just economic output, but real safety and recovery.
What is Hexachloro-1,3-Butadiene used for?
Real-World Uses That Affect Our Everyday Lives
Hexachloro-1,3-butadiene shows up in a handful of industries, each with its own way of using the chemical. Growing up in a small town near an industrial park, I remember my father talking about the strange smells that would sometimes slip into our neighborhood. What we didn’t know then—what a lot of people still don’t—is just how many chemicals float through production sites, and how each one carries its own purpose and risk.
Why Factories Turn to This Chemical
Most often, this compound pops up as part of making rubber. Companies working with chlorinated solvents—think synthetic rubber or specialty plastics—lean on its chemical characteristics. In a job managing safety protocols in a warehouse, I discovered how certain chemicals help keep business costs low or performance high. Some chemicals get tossed into formulas to clean up impurities from other reactions. Hexachloro-1,3-butadiene acts as a separating agent to pull out unwanted byproducts during the refining of chlorinated hydrocarbons, such as in the production of carbon tetrachloride or tetrachloroethylene.
Not Just About What It Makes—But How It Pollutes
Working near waterways, I've grown aware of how easily chemicals move from factory grounds to creeks and soil. Even if most people don’t see it, byproducts of industrial processes have life cycles that don’t stop the moment they leave the plant. Hexachloro-1,3-butadiene, for instance, sticks around. It doesn’t break down quickly in the environment. The United States Environmental Protection Agency (EPA) and the European Chemicals Agency flag it as a concerning pollutant because it builds up in living things. In areas downwind or downstream from manufacturing centers, tests have found this compound in air, water, and sediment. Its tendency to linger and build up in food chains raises questions about health over decades—not days.
Health Risks and Why They Matter
Exposure brings its own set of worries. My cousin spent years working at a chemical plant and had to take regular medical tests because managers knew repeated exposure could damage kidneys or cause irritation. Both the National Toxicology Program and the International Agency for Research on Cancer see it as possibly cancer-causing. Animal studies point to liver and kidney troubles. Even without full human case histories, communities know to stay alert when regulators announce any spike in local readings.
Tackling Exposure—Where Responsibility Starts
Stories from areas near production sites highlight the need for tighter controls. Companies and agencies can invest in closed-loop systems and vapor-capture technology. Instead of just dumping chemical leftovers, recyclers now process waste streams to pull out any leftover value and reduce pollution. Workers should have the right protective gear and training, and communities deserve regular, straightforward communication about what gets used next door. Investing in better reporting, community monitoring, and pollution control holds more weight than apologies after the fact.
Looking for Safer Substitutes
Researchers and advocates continue to scout for less hazardous alternatives. Some firms in Europe and North America now test other solvents that don’t stick around as long in the environment or that don’t collect in our bodies. Solving the problem demands more than swapping out chemicals. From community voices to smarter regulations, everyone has a role in pushing for a safer approach to industrial chemistry.
Is Hexachloro-1,3-Butadiene hazardous to health?
Understanding the Chemical’s Uses
Hexachloro-1,3-butadiene doesn’t pop up in everyday conversation, but it moves through some industrial circles. Factories use it in the process of making rubber compounds and lubricants. Others deploy it to clean up byproducts in the chlorinated solvent industry. Its strong, chemical smell isn’t something you forget once you’ve encountered it.
Where Trouble Can Start
People run into risks mostly when jobs or poor waste management send the chemical into air, soil, or water. Breathing, touching contaminated materials, or drinking polluted water causes most of the harm that the research community sees. Direct contact, especially for workers in certain plants, creates a much higher risk than for the average person.
What the Science Tells Us
Toxicology reports don’t mince words on hexachloro-1,3-butadiene's dangers. Exposure can hurt the kidneys faster than most folks expect. Scientists point to animal studies showing cells breaking down and organs failing with regular or large exposures. The U.S. Environmental Protection Agency labels this substance as likely cancer-causing. That’s a serious flag, considering how cancer rates already challenge public health.
Liver damage and central nervous system effects also show up in some studies. Nausea, headaches, and dizziness creep in early for those working without protection. With enough exposure, the body takes on bigger hits—think chronic organ problems or even nervous system disorders.
Sticking Around a Long Time
Hexachloro-1,3-butadiene doesn’t break down quickly. It hangs around in soil and water. Once it has seeped into the environment, the chemical can stay there for months or years, moving slowly from place to place. Eating fish or crops grown in polluted areas increases people’s exposure, even for folks who don’t work near factories or dumps. The food chain spreads that risk far beyond the original site.
What Protects Us
Governments have slapped controls on how this chemical gets used or tossed. The European Union bans its production and sale. American agencies keep close watch on hazardous waste management involving this compound. For decades, regulators set rules for air and water discharge and require companies to use the right storage and clean-up tools. Most accidents or exposures come from places cutting corners, using old equipment, or ignoring current safety practices.
On the job, the fix starts with proper gear and training. Factories swap out this chemical where possible or use closed systems to keep it from escaping. For people living near industry, local monitoring and strong reporting laws help spot problems before they spiral.
Promoting Safer Practices
Stronger enforcement can do a lot. Regular inspections and meaningful fines for polluters add pressure that changes behavior. Community action holds companies accountable, especially in towns near waste sites. Public education also makes a difference. Knowing what warning signs to watch for and how to handle suspicious spills protects families. Investing in safer alternatives for industry will shrink demand and reduce future legacy pollution.
Experts agree: Better workplace rules and more robust public oversight cut long-term harm. For now, vigilance matters most—in every lab, every storage room, and every patch of earth where these chemicals still hide.
What are the storage and handling requirements for Hexachloro-1,3-Butadiene?
Why Precautions Matter with This Chemical
Hexachloro-1,3-butadiene often catches attention because of its strange name and nasty toxic reputation. It’s not something the average joe stumbles across, but in chemical plants and research labs, people work near it every day. This stuff doesn’t behave like sugar or even gasoline. Workers have to treat it with real respect since it carries both environmental and personal health dangers. Looking back, I remember walking through an old facility, smelling a sharp, harsh chemical odor coming from somewhere behind thick glass. Chemicals like hexachloro-1,3-butadiene make that kind of nerve-wracking impression on anyone who’s spent time in the industry.
Storing It Without Risk
Forget about storing this chemical in makeshift setups or next to incompatible materials. Industry guidelines point out that Hexachloro-1,3-butadiene gets stored in cool, well-ventilated areas, away from both direct sunlight and sources of heat. Sturdy drums or containers engineered for chlorinated solvents work best, since regular steel tanks can corrode and leak. A friend of mine in industrial maintenance shared horror stories about spills caused by rusty containers; one tiny leak taints a whole area and gets way more expensive than simply picking quality storage from the start.
Hexachloro-1,3-butadiene loves to evaporate, releasing toxic fumes. Proper storage hinges on reliable seals and the use of secondary containment systems. Spill pallets with enough capacity should stand ready to catch leaks. The EPA classifies the stuff as a hazardous waste, which means disposal rules get strict; cutting corners only lands a company in regulatory hot water or, worse, puts folks at risk.
Thinking About Handling and Protection
I always say that no amount of experience replaces common sense and gear. Handling hexachloro-1,3-butadiene calls for full chemical-resistant gloves, splash goggles, and tight work clothing. Breathing masks with cartridges made for organic vapors keep workplace air from hurting lungs. Workers must avoid letting it splash on skin or drift into their eyes. Hexachloro-1,3-butadiene absorbs through skin and attacks the liver and kidneys, which makes skipping safety gear just plain reckless.
Proper training changes the story, turning workers into the first line of defense. I’ve seen well-trained crews shut down spills with speed and avoid injuries, while less-prepared teams freeze or try to mop up with rags. Emergency showers and eyewash stations near storage rooms always earn their place, as seconds count when accidents happen. Ventilation plays a big part, too; enclosed spaces can let fumes build up. Good airflow keeps concentrations low and protects both workers and the ventilation system from long-term damage.
Looking for Smarter Solutions
There’s a big push in the chemical world to find substitutes for really hazardous materials, but until then, people have to do the job right. Regular inspections, detailed labeling, and digital monitoring can flag problems before they become disasters. I remember one oil business using electronic sensors to catch vapor leaks before anyone got sick. These sorts of common-sense upgrades don’t just protect health, they help companies avoid heavy fines and legal headaches.
Hexachloro-1,3-butadiene doesn’t belong in the hands of anyone who shrugs at safety. Setting up solid systems for storage and personal protection speaks to a company’s values. I’d trust the operation that invests in safety and training over any high-tech labeling or glossy promises. The stakes go beyond paperwork and reach right into people’s lives—the kind of responsibility you never take lightly once you’ve seen the consequences firsthand.
How should Hexachloro-1,3-Butadiene spills be managed?
The Danger on the Ground
Hexachloro-1,3-butadiene doesn't usually knock at the door of most people's lives, but those living or working near chemical plants know the risks. Picture this: a transparent oily liquid, heavy with chlorine atoms, escapes into the open. Hexachloro-1,3-butadiene isn’t just a tongue-twister; it can stir up major headaches for humans and ecosystems. If the spill isn’t cleaned up fast, it can seep into the soil, slip into groundwater, or drift into the nearest river. From there, it disrupts aquatic life and builds up in the food chain. People who breathe it in or touch contaminated soil might face nausea, dizziness, or worse, liver and kidney damage. The stakes feel personal for workers on site, families downriver, and anyone who counts on clean water.
Why a Strong Plan Matters
Delaying cleanup leads to ruined fishing streams, polluted wells, and unhealthy neighborhoods—most folks in chemical towns have heard stories of what happens without fast action. Chemicals like hexachloro-1,3-butadiene stick around. Federal studies have shown this stuff resists natural breakdown, meaning contamination can linger for years. Our local emergency crews don’t have the luxury of waiting or making guesses, either. The U.S. Environmental Protection Agency lists this chemical as a hazardous air pollutant and flagged it for careful handling. Local responders trust their training and proper equipment for a reason—hexachloro-1,3-butadiene absorbs through the skin and vaporizes enough to fill the air with danger.
Steps for a Safer Cleanup
In the real world, dealing with toxic spills often begins with basic protection. Cleanup teams wear proper suits, gloves, and respirators to avoid breathing in fumes or touching tainted puddles. Experience counts here. Early in my career, I visited a site after a warehouse spill, and I saw two things: folks who suited up and kept safe, and those who gambled with their lungs and skin. It was always the careful ones who went home healthy.
Containing the spill comes next. Crews always need to block drains and use absorbent pads or dikes to keep the chemical from spreading. Anything that keeps hexachloro-1,3-butadiene out of waterways can be a lifesaver—river water feels clean until you hear about what washed in upstream. Mixing up with other chemicals creates bigger risks, so everything that soaks up the mess ends up sealed in steel drums and sent for licensed hazardous waste disposal. Nobody should be tossing this waste in a regular landfill—incinerators with special filters remain the most reliable choice, according to EPA guidelines.
Prevention and Public Involvement
Leaning too hard on luck never works. Safer storage tanks, regular inspections, and clear emergency plans keep these problems from starting. Chemical managers stay sharp by drilling with local firefighters and health officials, not just once but every year. People in nearby neighborhoods need simple information, not legal jargon. If communities understand the risks and see real investments in safety, trust grows. In fact, I remember seeing families at a public meeting asking about air monitors and buffer zones—the companies who answered honestly won more patience during tough times.
Technology with a Human Face
Robots and remote sensors can check areas too risky for people, but machines don’t replace vigilance on the ground. Health workers must be in the loop from day one, checking on clean water and following up if folks start reporting health issues. Real safety always links science with boots-on-the-ground wisdom. Given the stubborn nature of hexachloro-1,3-butadiene, no substitute exists for preparation, clear action, and accountability when something goes wrong.
What are the physical and chemical properties of Hexachloro-1,3-Butadiene?
A Closer Look at the Basics
Hexachloro-1,3-butadiene pops up in the world of industrial chemistry with a serious persona. With a chemical formula of C4Cl6, it wears its chlorine-heavy coat both literally and figuratively—it carries six chlorine atoms on a four-carbon backbone. The stuff shows up as a colorless to yellowish liquid, and you’ll usually catch a strong odor when it’s around, similar to turpentine.
This liquid doesn’t mix well with water, keeping to itself in a way that points to its nonpolar nature. It floats in most oil mixtures and spreads through soil thanks to its density, which goes above 1.6 g/cm³ at room temperature. When left exposed, it moves slowly from liquid to vapor, but the vapor will hang heavy in the air.
Heat, Stability, and How It Breaks Down
Boiling doesn’t start until the temperature hits around 210°C, which makes it tough to get rid of by simple evaporation or open-air processes. It stands up to sunlight surprisingly well, choosing to break down only when the heat or chemical pressure gets serious. Hexachloro-1,3-butadiene doesn’t catch fire easily, but if you do manage to set it alight, expect some harsh chemicals—think hydrogen chloride and phosgene among the release products.
This resistance to breakdown means contamination can last for years. Once it gets into groundwater or soil, removing it takes more than time—it takes action and sometimes a lot of money.
Skin-Deep Hazards and Lasting Impacts
It’s not just the smell that signals trouble. The skin can’t tolerate contact for long, and extended exposure brings liver and kidney risks. Inhalation means more than a cough—it can pose a real danger, especially for workers in chemical plants. Historical cases in manufacturing hubs like Niagara Falls and Germany’s chemical belt in the 1960s paint a clear picture: companies dumping waste without tight controls left a mess for the local water and air quality.
The legacy of this carelessness now fills plenty of environmental science textbooks. Studies from the 1980s found this compound persisting in the Great Lakes long after companies stopped using it, proving that its chemical stubbornness isn’t just theory, but a challenge for anyone who appreciates clean water.
Better Practices, Smarter Solutions
The real value shows up when professionals turn chemical know-how into action. Chemical engineers and site remediators use activated carbon filtration and advanced oxidation to pull hexachloro-1,3-butadiene out of groundwater. These steps aren’t perfect—cost and infrastructure limits force hard decisions—but the commitment to reducing exposure and cleaning old messes proves key for public health.
On the regulatory front, restrictions have forced industries to use better alternatives or limit waste. The U.S. EPA classifies it as a hazardous air pollutant, and European agencies enforce strict reporting on any emissions or use.
Anyone who’s walked through an old industrial district and caught that sharp chemical tang knows why strong oversight of substances like hexachloro-1,3-butadiene matters. It’s clear that a few decades of lax standards produce costs future generations pay many times over.
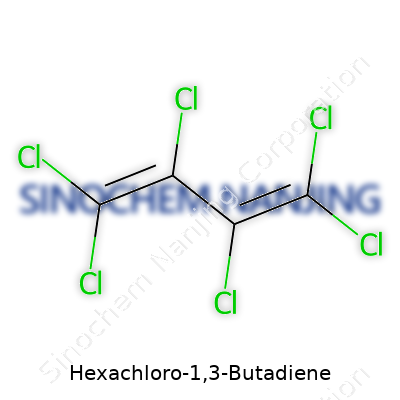
| Names | |
| Preferred IUPAC name | 1,1,2,3,4,4-Hexachlorobuta-1,3-diene |
| Other names |
Perchlorobutadiene Hexachlorobutadiene HCBD 1,1,2,3,4,4-Hexachloro-1,3-butadiene |
| Pronunciation | /ˌhɛk.səˌklɔː.roʊ.wʌn.θriː.bjuː.təˈdaɪ.iːn/ |
| Identifiers | |
| CAS Number | 87-68-3 |
| Beilstein Reference | 633878 |
| ChEBI | CHEBI:39241 |
| ChEMBL | CHEMBL45837 |
| ChemSpider | 10043 |
| DrugBank | DB02573 |
| ECHA InfoCard | ECHA InfoCard 100.003.726 |
| EC Number | 208-660-5 |
| Gmelin Reference | 778 |
| KEGG | C19585 |
| MeSH | D006630 |
| PubChem CID | 8549 |
| RTECS number | FG0525000 |
| UNII | 2TQ4FVM31R |
| UN number | UN2271 |
| Properties | |
| Chemical formula | C4Cl6 |
| Molar mass | 260.76 g/mol |
| Appearance | Colorless liquid |
| Odor | Mild, turpentine-like |
| Density | 1.68 g/cm³ |
| Solubility in water | insoluble |
| log P | 2.97 |
| Vapor pressure | 0.02 mmHg (20°C) |
| Acidity (pKa) | 13.7 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 1.5300 |
| Viscosity | 1.63 mPa·s (20 °C) |
| Dipole moment | 1.06 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 354.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -9.30 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1461.4 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Toxic by inhalation, skin contact and ingestion; may cause cancer; causes damage to organs through prolonged or repeated exposure; very toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS02, GHS06, GHS08, GHS09 |
| Pictograms | GHS06, GHS08, GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. H315: Causes skin irritation. H319: Causes serious eye irritation. H335: May cause respiratory irritation. H351: Suspected of causing cancer. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P201, P202, P260, P264, P270, P271, P273, P280, P281, P302+P352, P304+P340, P305+P351+P338, P308+P313, P314, P321, P330, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-0-0 |
| Autoignition temperature | 632 °C |
| Explosive limits | Lower: 6.4% ; Upper: 14.6% |
| Lethal dose or concentration | LD50 oral rat HCBD: 5800 mg/kg |
| LD50 (median dose) | LD50 (median dose): 5800 mg/kg (oral, rat) |
| NIOSH | DN4550000 |
| PEL (Permissible) | 0.02 ppm |
| REL (Recommended) | 0.02 ppm |
| IDLH (Immediate danger) | 23 ppm |
| Related compounds | |
| Related compounds |
Tetrachloroethylene Carbon tetrachloride Hexachlorobutadiene Chloroform 1,2,3,4-Tetrachlorobutadiene |

