Hexabromobiphenyl: The Dark Legacy and Lingering Reasons for Vigilance
Historical Development
Hexabromobiphenyl stepped onto the stage in the 1970s, riding the wave of better living through chemistry. This compound appeared as a solution for fire hazards in plastics and resins—those new wonders filling homes and factories. Manufacturers sought to prevent deadly house fires, pushing the industry towards ever-stronger flame retardants. The reach of hexabromobiphenyl spread quickly, especially after its adoption in electrical products and foam materials. Folks working in factories or using everyday electronics might not have known much about what kept their possessions from burning—until a tragedy drew public attention. The 1973 Michigan livestock contamination, caused by a warehouse mix-up, brought this chemical’s dangers right into American kitchens. Dairy, meat, and eggs—suddenly laced with a persistent, invisible threat. The fallout sparked global debates about weighing innovation against knowledge of long-term risks.
Product Overview
Hexabromobiphenyl looks like a fine white powder, a far cry from the terrifying word “toxin” that now follows its mention. It belongs to a bigger family called polybrominated biphenyls (PBBs). These substances are built to resist burning, making them popular in consumer goods. On paper, hexabromobiphenyl appears as a straightforward molecule, but its true impact can’t be summed up by structure alone. Over time, workers and the public experienced the threat lying behind the manufacturer’s promise of “safer” products. In today’s discussions about chemical safety, hexabromobiphenyl functions as a case study in how something designed to keep us safe can, under poor oversight, do the opposite. Industry used the compound for decades and left a pile of regulatory challenges in its wake.
Physical and Chemical Properties
Hexabromobiphenyl doesn’t surprise a seasoned chemist. It won’t dissolve in water, but it disappears easily into fats and oils—a property that turns out to be crucial for understanding its environmental persistence and toxicity. Most living things store fats, which gives this chemical plenty of opportunity to hide and build up over years. Boiling or burning won’t break it down cheaply, so it lingers for ages in landfills and in contaminated sites. Its resistance to breakdown is exactly why it held appeal for fireproofing, but the same quality draws concern among scientists worried about bioaccumulation and long-term health impacts. The physical toughness of hexabromobiphenyl reflects the durability baked into so many synthetic molecules crafted during the 20th century’s chemical boom.
Technical Specifications and Labeling
Industry-grade hexabromobiphenyl usually shows high purity. Packaging and shipment guidelines require clear association with hazardous material regulations. Labels list its risks and storage requirements—locked cabinets, gloves, and goggles stand as barriers between user and harm. Anyone handling the substance in a research or disposal setting faces a level of oversight that didn’t exist during its heavy use decades ago. More than just a technicality, these precautions shape public trust in chemical management. Stories from Michigan and other polluted sites pushed regulatory bodies to demand transparent labeling and detailed instructions long before “right to know” laws swept the globe. The need for better control stands as one of the big lessons hexabromobiphenyl left us.
Preparation Method
Labs used to synthesize hexabromobiphenyl by direct bromination of biphenyl, usually in the presence of a solvent and catalyst. Simple in theory, even elegant, but the process involved large quantities of elemental bromine—a corrosive and hazardous substance in its own right. The drive to produce hexabromobiphenyl as efficiently as possible spurred continuous process tweaks. By the time safety standards caught up to the fire retardant craze, many manufacturers already faced mounting cleanup costs and regulatory scrutiny. Today, the drive to prevent anything like the old methods persists; fewer labs remain willing to take on the risks or the legal baggage.
Chemical Reactions and Modifications
The structure of hexabromobiphenyl makes it stubbornly uncooperative in most common chemical reactions. Its strong bromine-carbon bonds resist attack by acids or bases, heat, and sunlight. Attempts to degrade the molecule have included catalytic hydrogenation and photolytic destruction, but those approaches aren’t practical at the scale required for polluted sites. The failure to quickly break hexabromobiphenyl down into harmless ingredients dogged every major cleanup. Attempts at modifying this structure for easier breakdown have struggled, pointing to a broader problem with brominated flame retardants in general. Chemists researching safer alternatives now take lessons from these struggles, aiming to design materials that protect but don’t persist for generations.
Synonyms and Product Names
Hexabromobiphenyl hides behind a surprising number of names—FireMaster BP-6, PBB 153, HBB, and a string of numbers that only a chemist would love. Each alias links back to various brands or is referenced in scientific literature around the world. The naming maze doesn’t help public understanding, and in the chaos of the Michigan disaster, confusion over labels made tracing contamination tougher than it had to be. Regulators in later decades pressed for unified naming not just for hexabromobiphenyl but for all persistent organic pollutants, hoping to cut through ambiguity and keep both companies and the public on the same page.
Safety and Operational Standards
Handling hexabromobiphenyl in a modern setting means treating it with the gravity it deserves. Producers, labs, and cleanup crews follow international safety guidelines that reflect just how badly things can go wrong. Respirators, chemical suits, sealed containers—these aren’t overkill, but proof that the lessons from contaminated soil and food have stuck. Environmental groups, workers’ advocates, and researchers all keep a close eye on the stuff. Production in most countries has flatlined, replaced with safer flame retardants, and the focus now turns toward legacy sites and preventing future mistakes. Still, cleanup crews report how preparation and vigilance make the difference between a safe day’s work and a health crisis nobody wants to relive.
Application Area
Electronics, textiles, and building materials once relied on hexabromobiphenyl for its flame resistance. Factory owners seeking to meet insurance and safety codes grabbed this substance for wire insulation, plastic casings, and polyurethane foam padding. Most products never carried the chemical’s name—just the promise that children and workers would stay safer. After its dangers became too public to ignore, companies withdrew it from nearly every market. Looking back, the switch was both a win for public health and a testament to what happens when profit comes before precaution. Yet, stockpiles and older goods haven’t disappeared. Every now and then, a demolition crew or scrapper uncovers it in old buildings, and the old risks reappear for a new generation.
Research and Development
In the wake of widespread contamination, research redirected from production to environmental remediation. Scientists turned their attention to monitoring persistence in rivers, soils, and animal tissues—seeking answers to questions that earlier years had overlooked. I remember thesis students spending entire semesters tracking the compound’s spread, learning first-hand how slow progress can feel in toxicology. Current innovation focuses on detecting PBB residues at ever lower levels, understanding how hexabromobiphenyl interacts with other pollutants, and building potent clean-up microbes. No quick fix has emerged, but efforts continue. Once exclusive to fire-protection science, hexabromobiphenyl now underscores the need for chemists and toxicologists to work together in anticipating the downsides of so-called miracle compounds.
Toxicity Research
Toxicologists have traced hexabromobiphenyl through food chains and human tissues. Unlike many fast-acting poisons, its effects creep up over long exposure times. People exposed through contaminated food supplies developed lasting health issues: liver damage, immune suppression, neurological symptoms, and a suspected link to cancer. Researchers learned that the chemical’s fat-loving nature ensures it doesn’t pass through a body quickly but sticks around, stored in organs where it can do damage for years. Local communities fought for answers, pushing lawmakers to fund studies that would eventually prove how deeply this substance could alter human health. The persistence of hexabromobiphenyl in the environment, and the slow but steady march of its adverse effects, forever changed the rules for getting a new industrial chemical approved.
Future Prospects
Bans and strict regulations in the US, EU, and elsewhere have written an ending for new uses of hexabromobiphenyl, but not for its legacy. E-waste dumps in developing economies, contaminated soils, and lingering presence in food chains remind us that long-lived chemicals won’t disappear just because new rules say they should. The future rests on ongoing remediation projects, honest communication with the public about risks and clean-up progress, and investing in alternatives that don’t trade tomorrow’s safety for today’s convenience. Seeing schoolchildren living near former dump sites or workers dismantling old electronics drives home that the work isn’t finished. As more people realize that flame safety’s price tag included decades of health problems, maybe the next chemical “solution” will face tougher questions before it lands in millions of homes. Honest debate, open science, and relentless transparency offer the best way forward—rooted not in fear, but in the lessons learned from hexabromobiphenyl’s long, sobering story.
What is Hexabromobiphenyl used for?
Where Hexabromobiphenyl Showed Up
Hexabromobiphenyl is a chemical that once grabbed plenty of attention in the world of manufacturing. It landed in the spotlight because folks put it to work as a flame retardant, mainly in plastics and electrical equipment. Companies used it in things that might catch fire easily—think televisions, car parts, and kitchen appliances you find in homes and offices. The main job: slow down flames that start from sparks or overheating.
In my younger days, I remember a time when plastic burned fast and spread smoke in seconds. People wanted safer products, less fire risk, and Hexabromobiphenyl promised a way to meet that need. It sounded like a clear win: safer products for homes and businesses, fewer house fires, less havoc in electrical rooms.
Problems Under the Surface
A closer look revealed a much different story. Hexabromobiphenyl belongs to a group of chemicals called polybrominated biphenyls (PBBs). These compounds don’t break down in the environment. Instead, they linger in soil, water, and the bodies of people and animals. Folks who worked in factories or lived nearby carried higher amounts in their blood. Cows grazed on grass near plants that made this chemical, and soon, contaminated meat and milk showed up on kitchen tables.
Researchers started linking the chemical to a pile of health troubles, including cancer and problems with the liver and immune system. Kids were at risk, too. Scientists saw it building up in breast milk and reaching babies. That is hard to shake off. Families just wanted safe food and healthy kids, and now suddenly, something meant to keep them safe brought new worries.
Ripple Effects and Regulations
Once these dangers came to light, things started to change. Governments across Europe and the United States set rules to ban or strictly limit Hexabromobiphenyl. Michigan faced the worst of it in the late 1970s after a chemical mix-up led to widespread PBB contamination of cattle feed. It ended up in crops, milk, cheese—things people eat every day. Many people got sick, and some still deal with long-term health problems from that disaster.
A ban sounds simple, but the real story reveals how chemicals slip into daily life without most folks realizing it. It also reminds us that safety testing of new compounds can never be rushed or skipped. Good intentions—like blocking fires—don’t outweigh the risks if the cure brings a new danger.
Lessons for the Next Generation
Looking back, Hexabromobiphenyl taught a sharp lesson. Before trusting new materials for clothes, electronics, or furniture, companies and regulators need to ask tough questions about long-term impacts. Are there safer alternatives? Can the public understand what’s in the products on their shelves? Today, many firms test non-toxic fire retardants before putting anything on the market, and technology helps track safety faster.
Stronger oversight and better communication between scientists, regulators, and the community protect public health far better than hoping nothing will go wrong. Transparent data, public reports, and easy-to-understand safety labels all build trust. We can’t undo the past, but we can make better choices moving forward—and for those of us who remember the panic of contaminated food, that matters.
Is Hexabromobiphenyl dangerous to human health?
Why We Should Pay Attention to Hexabromobiphenyl
People tend to not think much about chemicals with long names. Hexabromobiphenyl, or HBB, falls into that group. But history gives us plenty of reasons not to ignore it. In Michigan during the 1970s, a massive feed contamination disaster exposed residents to HBB for years. Decades later, health repercussions turned up in workers and families who lived near those contaminated areas. This chemical, used as a flame retardant, sticks around in the environment and the human body for a long time because it doesn’t break down easily.
What Researchers Know About Health Effects
Studies involving HBB point to harmful effects on several systems in the body. People exposed at work or through contaminated food experienced skin rashes, liver problems, and weight loss. Research in animals shows liver tumors, reproductive problems, and developmental effects in the offspring. For humans, the U.S. Environmental Protection Agency and World Health Organization list HBB as an environmental health risk. The Centers for Disease Control and Prevention tracked raised cancer rates in families exposed through contaminated milk and meat in Michigan. Doctors and public health experts advise caution because even small quantities might matter over time.
Why Old Chemicals Still Cause Trouble
Fire safety matters, but introducing chemicals like HBB into consumer products left a legacy that continues today. Soil, water, and even household dust in buildings can hold onto it for decades. Eating fish or animal products from contaminated areas increases risk. One trouble with HBB comes from its ability to build up in fat tissue. So anyone who eats food or drinks milk that contains HBB might store that chemical in their body for years.
Evidence From Past Mistakes
It’s easy to think old chemical bans mean the danger has passed. HBB production stopped in the United States in the late 1970s. Products made before that time still turn up in older buildings, electrical equipment, and landfills, so the risk hasn’t disappeared. People who grew up near contaminated farms in Michigan still live with higher HBB levels in their bodies, decades after the fact. That should raise a flag for people who work in demolition, recycling, or environmental cleanup jobs.
What Can Be Done Now?
Safer choices begin with awareness. Testing soil, water, and food from risky sites can keep new generations safer. Lawmakers have a role—strong rules about toxic chemical use and funding for cleanup projects protect everyone. At home, people can cut back on food from high-risk sources and push for better tracking of old chemicals. Doctors and toxicologists now know more about long-term health monitoring for people exposed to industrial chemicals; keeping up with regular checkups and screenings helps with early intervention.
Looking Ahead
Science around chemicals like HBB keeps changing as new studies emerge. Staying informed, supporting public health research, and making smart regulatory choices matter just as much now as when HBB first came on the scene. Health and safety aren’t just about avoiding what’s trendy or new—they rely on paying attention to what we already know and using that knowledge to protect future generations.
What are the safety precautions when handling Hexabromobiphenyl?
Why Safety Around Hexabromobiphenyl Demands Attention
Hexabromobiphenyl carries a big reputation for more than just being a tongue-twister. This flame retardant, once used heavily in plastics and electronics, showed up in history books after a chemical spill in Michigan back in the 1970s. After that, watching how you handle chemicals like this stopped being optional. Workers and anyone around places still using or decontaminating environments with hexabromobiphenyl—let’s call it HBB—deserve the straight talk on safety. Studies from the CDC and EPA still recognize its toxicity, linking exposure to liver problems, skin irritation, and even possible cancer risks.
What Personal Experience Reminds Us
Standing in a lab in college, hearing your instructor recite chemical risks isn’t the same as seeing that rash spread after a tiny spill you didn’t notice at first. My own run-in with persistent organics taught me that safety goggles and gloves aren’t just aesthetics, and shortcuts in handling cost way more than a few minutes saved. Our skin, lungs, and eyes can’t keep hazardous agents outside just by willpower or luck. Old chemical storerooms or demolition sites can still hide containers that have HBB dust in the air or on surfaces.
Practical Steps for Everyday Protection
Firstly, no one should think about touching HBB without the right gear. Nitrile gloves shield hands better than latex. Eye wash stations and showers should sit close to any work involving this compound. Throwing on a lab coat and goggles is just basic sense.
Ventilation beats fancy air fresheners every time. Lab hoods or industrial exhausts move contaminated air away from anyone breathing it. Teams working with it need formal training on handling spills, cleaning up properly, and disposing of all contaminated disposables in sealed, labeled containers according to hazardous waste rules.
Never eat, drink, or even apply lip balm near active handling areas. That might sound like going overboard, but any contact makes a difference given how long HBB sticks around. Shower after leaving the workspace and change clothes to cut down what leaves the job with you.
Treating and Dealing With Accidents
If HBB contacts skin, immediately washing with soap and water can help halt absorption. Eye contact means minutes under running water, not just a quick splash. Any breathing issues need medical attention, no debate. Having a clear communication plan so everyone knows what to do in an emergency makes the difference in serious situations. Facilities need to report spills and possible exposures right away to environmental and health agencies. Playing down a minor mishap crosses the line from carelessness to putting lives at risk.
Long-Term Solutions and Keeping Hazards Out
Many countries phased out HBB due to its health risks, but it can still lurk in old stockpiles, products, or contaminated soil. Regular audits of chemical inventories remove surprises. Contractors handling demolition or cleanup of legacy sites benefit from treating all unknown dust and waste as toxic until proven otherwise. Regulators and technical advisors urge ongoing training and clarity around personal protective equipment use, spill management, and physical work boundaries.
Every lesson from the past keeps reinforcing one point: real safety depends on habit, respect, and never cutting corners. Following solid safety precautions lets people finish their shift and return home safely, which matters more than any shortcut ever could.
How is Hexabromobiphenyl disposed of safely?
Why Safe Disposal Matters
Hexabromobiphenyl stands out as a toxic industrial substance with a stubborn reputation. Farmers in Michigan back in the ‘70s learned this the hard way after livestock feed got contaminated, spreading across food chains, harming families and wildlife alike. This chemical lingers in the environment and the body, building up over time. Exposure connects to liver damage, nervous system troubles, and an increased risk of cancer. I’ve talked with people raised in that part of Michigan who grew up avoiding farm-fresh eggs. These concerns never really leave a community, especially for families still living near old manufacturing sites.
Real-World Methods to Get Rid of It
Getting rid of hexabromobiphenyl is never as simple as tossing it in the trash or pouring it down a drain. Burning it at a high temperature—in a special incinerator designed for toxic waste—remains the solution most experts turn to. Temperatures above 1,200 °C break those stubborn bonds, turning harmful molecules into ash, water, and basic gases. Every step needs oversight by trained professionals because even a hint of failure means releasing dioxins and other persistent pollutants, which only adds new problems to the old ones.
Some facilities press for chemical treatment methods—like sodium reduction—that break the molecule apart without flames. Sodium reacts with it at high temperature under controlled conditions, destroying the hardest to split bonds. This method avoids the risk of accidental air pollution during incineration. Each approach costs more than dumping, though, and finding the budget to do this cleanup right often challenges communities dealing with bigger priorities.
Landfills and Their Limits
Hazardous waste landfills can accept sealed containers loaded with hexabromobiphenyl. Long-term safety depends on those containers holding up under pressure, moisture, and the slow creep of corrosion. Layers of clay and synthetic liners, constant groundwater monitoring, and legal oversight from environmental agencies all play a role. These barriers only delay, not prevent, contamination. Stories keep surfacing about old landfills leaking after storms or equipment failures. Safe landfill disposal works best as a backup measure instead of a solution anyone should rely on for decades.
Digging Out the Root of the Problem
Most states now ban or tightly control the sale and use of hexabromobiphenyl. Products made thirty or forty years ago still turn up in forgotten warehouses, basements, and abandoned sites. Finding and removing these leftovers offers one chance to cut long-term risks. State and local governments can set up amnesty days, giving people a way to hand in dangerous chemicals without penalties. Funding cleanup crews through public grants or requiring the polluter to pay—if you can find them—puts the financial responsibility where it belongs. Knowledge is power, too. Communities deserve straight talk about what substances like this can do and why a little extra spending to get rid of them safely pays off in the long run.
Building a Safer Future
Learning from disasters pushes better choices. Instead of waiting for another crisis, regulatory agencies should keep pushing for tighter controls, tougher reporting, and open transparency. Making chemical companies pay into cleanup trust funds before things go wrong sounds harsh but creates real incentives for safer production. Exploring greener alternatives to flame retardants—testing them fully before use—helps keep history from repeating. It’s worth spending more on the front end if it means children born fifty years from now won’t need to worry about toxins lurking in their water or food. The fight over chemicals like hexabromobiphenyl isn’t just about today—it’s about setting a better example for the generations following us.
What are the environmental impacts of Hexabromobiphenyl?
Brominated Flame Retardants and Their Mark
Hexabromobiphenyl belongs to a group of chemicals that got wide use in plastics, especially in the 1970s. It served as a flame retardant for electrical devices and certain types of foam used in furniture and building materials. At the time, these products promised safety from fires. People hardly thought about where these chemicals would go after products landed in the dump or when factories released waste. Yet the consequences stretch far beyond a landfill or a smokestack.
Soil and Water Take the Burden
Hexabromobiphenyl doesn’t just disappear. It settles in soil, clings to sediments, and moves through waterways. Rain doesn’t easily wash it away, and bacteria break it down poorly. Researchers found traces in river mud, in the fat of wildlife, and even in crops. Growing up on a farm, I saw up close how certain chemicals—ones with hard-to-pronounce names—can linger long after the harvest. Persistent organic pollutants like this one travel through the food web. Fish, birds, and grazing animals end up with it in their bodies, which means people eating local eggs, milk, or cheese might be exposed too, sometimes without knowing.
Health Links That Can't Be Ignored
Scientific studies tie hexabromobiphenyl to serious health risks. Exposure connects with liver cancer, thyroid problems, reproductive harm, and developmental setbacks in children. The Michigan PBB incident from the 1970s taught us this the hard way—a chemical mix-up contaminated animal feed, which led to one of the state’s most far-reaching chemical disasters. Community members still talk about those years, when farm families dealt with sick animals and uncertainty about what was safe to eat. The link between environmental exposure and human health feels personal for people who remember contaminated water or food sources in their own neighborhoods.
No Simple Way Out
Banned years ago in most countries, hexabromobiphenyl still turns up because products and building materials keep their chemicals for decades. Disposal at landfills doesn’t stop escape into ground or water. The substance travels through old electric appliances, foam, and construction debris. Responsible recycling and improved waste management only solve part of the problem. Legacy pollution remains locked in sediment or riding the wind as dust particles from demolition and landfill sites.
Possible Steps Toward Change
People can push for regular testing near landfills, old factory sites, and in food products from areas with known contamination. Learning from local history and supporting efforts to track pollution help protect the most exposed communities. Farmers and food producers in affected regions should get clear information and monitoring, not left guessing about risks to their products or customers. Governments can prioritize clean-up of contaminated soils and invest in research to find safer alternatives for flame retardancy. Each effort matters because these chemicals hang on for years and pass silently through the food chain.
Everyday Choices and Watchfulness
Consumers play a role, too. Buying refurbished electronics with care, supporting businesses that use safer chemicals, and asking questions about the history of materials inside older homes or workplaces. Stories of long-standing pollution shouldn’t end in silence. Sharing what we know—between neighbors, at local government meetings, through grassroots networks—makes awareness grow. Real protection starts with holding both polluters and regulators to account, drawing on the lessons from communities living with the fallout of unexamined chemical use. Learning from their experience puts science and vigilance into real-world context, shaping better practices for the future.
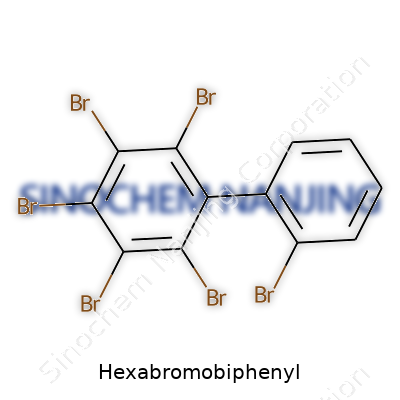
| Names | |
| Preferred IUPAC name | 3,3',4,4',5,5'-Hexabromo-1,1'-biphenyl |
| Other names |
Hexabromobiphenyl Firemaster Hexabromobiphenyl (HBB) Biphenyl, hexabromo- BB-153 |
| Pronunciation | /ˌhɛk.səˌbroʊ.moʊ.baɪˈfiː.nɪl/ |
| Identifiers | |
| CAS Number | 36355-01-8 |
| Beilstein Reference | 1752351 |
| ChEBI | CHEBI:34430 |
| ChEMBL | CHEMBL2107231 |
| ChemSpider | 2078353 |
| DrugBank | DB14055 |
| ECHA InfoCard | 17b8c7c6-73b7-478e-8dd0-8c6cb5e3033e |
| EC Number | 201-799-1 |
| Gmelin Reference | 65143 |
| KEGG | C14015 |
| MeSH | D006530 |
| PubChem CID | 36100 |
| RTECS number | TX9625000 |
| UNII | B9343BOY61 |
| UN number | UN3077 |
| Properties | |
| Chemical formula | C12Br6 |
| Molar mass | 627.524 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.98 g/cm³ |
| Solubility in water | insoluble |
| log P | 5.8 |
| Vapor pressure | 1.06E-8 mmHg at 25°C |
| Acidity (pKa) | 4.5 |
| Magnetic susceptibility (χ) | -99.7×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.781 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 653.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -14.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2711 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | N05CM08 |
| Hazards | |
| Main hazards | Toxic if swallowed, suspected of causing cancer, toxic to aquatic life with long lasting effects |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H351: Suspected of causing cancer. |
| Precautionary statements | P210, P261, P280, P301+P312, P305+P351+P338, P308+P313 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | 163 °C |
| Autoignition temperature | 518 °C |
| Lethal dose or concentration | LD50 (rat, oral): > 5,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2 g/kg (oral, rat) |
| NIOSH | TT4100000 |
| PEL (Permissible) | 1 mg/m³ |
| REL (Recommended) | 0.1 mg/kg |
| IDLH (Immediate danger) | 40 mg/m3 |
| Related compounds | |
| Related compounds |
Polybrominated biphenyl Hexachlorocyclohexane Hexachlorobenzene |

