Heptanoic Acid: More Than Just a Chemical with Seven Carbons
Historical Development
Heptanoic acid traces its story to a time when chemists still reached for test tubes before computers. These seven-carbon fatty acids appeared on the radar in the 19th century as researchers mapped the value of odd-chain carboxylic acids. Early soap makers and perfumers learned to appreciate it much earlier, although under less scientific names. Over time, as organic synthesis matured, heptanoic acid found itself filling unique roles, especially after large-scale synthesis made it less of a curiosity and more a staple for labs and industries chasing purity and niche properties.
Product Overview
This straight-chain fatty acid, sometimes called enanthic acid, stands out due to its distinctive smell. In the lab, a sample releases a sharp, oily odor that lingers and leaves no one guessing which bottle lost its cap. Heptanoic acid may seem like a side character in the world of everyday chemistry, but for those working in cosmetics, lubricants, plasticizers, or even fragrances, it often plays a quiet but essential supporting role. The molecule carries a carboxyl group at one end and a hydrocarbon tail that helps bridge the gap between shorter, more volatile acids and much longer fatty acids found in oils and fats.
Physical & Chemical Properties
At room temperature, heptanoic acid presents as a colorless to slightly yellow oily liquid. Its boiling point sits around 223°C, making it less volatile than formic or acetic acid but still manageable without extreme lab precautions. The acid dissolves readily in alcohol, ether, and chloroform, while its solubility in water drops off steeply. Its acidity mirrors related carboxylic acids, typically measured around pKa 4.9, so it’s neither the strongest nor the weakest acid among its peers. This balance lets it participate in a range of creative chemical reactions and gives users more options for downstream modifications.
Technical Specifications & Labeling
Chemists in manufacturing settings keep a close eye on purity, especially with heptanoic acid finding its way into flavorings, fragrances, and specialty lubricants. Labels usually announce percentage purity, trace metal content, and residual solvents. The presence of even tiny iron, copper, or phthalate contaminants can mess with end uses ranging from food contact materials to pharmaceuticals. European and US regulatory agencies keep lists of acceptable impurities, and every batch should stay comfortably within those bounds. The acid often arrives in amber glass to avoid light-promoted degradation, though steel drums work for industrial quantities.
Preparation Method
Getting a batch of heptanoic acid typically involves oxidizing heptanal or the hydrolysis of castor oil derivatives. Older methods started with odd-chain alcohols made from petrochemical feedstocks, then slowly scaled up to greener processes as demand grew and regulations tightened. The main route nowadays involves careful oxidation under controlled conditions, monitored by gas chromatography to catch byproducts like heptanol or heptanone. Waste management is another issue, as synthesis sometimes leaves behind organic residues that must be treated before disposal. The push for cleaner chemistry now sees more energy-efficient protocols being tested in both research and production environments.
Chemical Reactions & Modifications
Heptanoic acid joins the long list of carboxylic acids that lend themselves to esterification, amidation, and reduction. In the right hands, the acid becomes heptanoate esters—key ingredients in perfumes and plasticizers—by reacting with alcohols under acidic conditions. Its small but flexible hydrocarbon chain allows for substitution at several points, opening options for creating larger, more functionalized molecules. Reductive reactions turn it into heptanol, adding further value for surfactant and specialty solvent applications. Amidation gives rise to amides used in pharmaceuticals or polymer chemistry, showing the real power packed in such a simple molecule.
Synonyms & Product Names
In different sectors, heptanoic acid goes by enanthic acid, n-heptanoic acid, or heptanic acid. Older industry catalogues sometimes listed it as oenanthic acid, a nod to its use in wine and fragrance. Chemists and product formulators rarely quibble about the name, more focused on the CAS number to avoid confusion, especially in global markets where trade names can proliferate. Most sectors now standardize to “heptanoic acid” for regulatory filings and patent registrations.
Safety & Operational Standards
Heptanoic acid deserves respect in storage and handling. The vapors cause irritation, and the liquid attacks skin and eyes. Labs that follow standard operating procedures use proper ventilation, gloves, and eye protection. Untrained staff sometimes underestimate oily acids compared to their more volatile cousins, but long-term exposure can lead to chronic irritation or mild systemic toxicity. Large spills need proper neutralization, often with sodium bicarbonate, and cleanup teams keep acids far from incompatible chemicals—like strong oxidizers—to reduce the risk of secondary reactions. Regulatory standards, such as those from OSHA and the European Chemicals Agency, lay out concentration limits for workplace exposure.
Application Area
Far beyond the world of beakers and pipettes, heptanoic acid turns up in lubricants, plasticizers, and synthetic esters for perfumes. In cosmetics, some derivatives add that subtle fatty scent or improve skin feel in creams and lotions. The food sector uses related esters for flavors, and paint manufacturers tap into its properties to improve resin stability and drying profiles. Heptanoic acid-based compounds also feature in certain types of biodegradable plastics and in medical applications like antimicrobial agents and barrier creams. Automotive and aircraft lubricants take advantage of the high thermal stability of its esters, providing critical protection in extreme environments where short-chain acids would break down.
Research & Development
Current researchers push heptanoic acid chemistry into new spaces. Green chemistry labs focus on improving yield, reducing toxic byproducts, and finding sustainable feedstocks. Catalysis specialists search for more efficient ways to convert biomass to odd-chain acids using renewable sources. Pharmaceutical teams dig deep into new derivatives, exploring anti-inflammatory and antimicrobial effects, and materials scientists play with copolymer blends for packaging that biodegrades in compost. Peer-reviewed journals fill with new data every year, sharpening understanding and opening practical, scalable pathways for wider use.
Toxicity Research
Animal studies with heptanoic acid show low acute toxicity but consistent irritation with prolonged contact. Human exposure, especially in workplace environments, often leads to dermatitis if protective measures slip. Chronic studies offer reassurance over low-level dietary exposure—especially as related esters occur naturally in some foods—but the emphasis remains on minimizing inhalation or direct skin contact. Regulatory agencies take seriously the reports around repeated exposure, especially in manufacturing or in facilities making consumer-facing products. Emerging studies now track environmental persistence and the potential for bioaccumulation, helping policymakers set future safety standards.
Future Prospects
Demand for heptanoic acid stands poised to grow as industries chase greener, more versatile building blocks. As biorefineries scale up and new feedstocks come online, more sectors lean away from traditional petrochemicals and into molecules like heptanoic acid, which bridge the world of organics and performance materials. Ongoing research into bio-derived variants could cut down carbon footprint and open up markets previously closed due to cost or supply chain challenges. As with many specialty chemicals, market pull will hinge on continued improvements in cost, safety, and versatility, with new applications waiting for chemists and engineers willing to dig deep into the possibilities hidden within that seven-carbon scaffold.
What is Heptanoic Acid used for?
Understanding Heptanoic Acid Beyond Its Chemistry
Heptanoic acid doesn’t show up in everyday conversations, but it makes a mark where fewer people look. It’s a simple fatty acid with seven carbon atoms—hardly something flashy. But in practical jobs, it quietly fuels change and progress. Take its role as a building block. Chemical manufacturers turn to heptanoic acid to make things people rely on without a second thought, like synthetic flavors or fragrances. It gives products that subtle push to smell right, taste better, or last longer on a store shelf.
Lubricants Driven by Precision
Heptanoic acid plays a critical part in developing high-performing lubricants. Whether it’s for the gears in an industrial blender or the engine of a delivery truck, modern lubricants face intense heat and stress. This fatty acid helps create esters, substances that hold up through tough conditions and don’t break down when things heat up. These lubricants keep engines running cleaner, extend the time between changes, and reduce mechanical wear.
I grew up in a house with an old car that always seemed to leak oil. Back then, finding something that prevented gunk from clogging up the works felt out of reach. Now, lubricant chemistry has moved far ahead, and that little boost from heptanoic acid-backed esters—along with advances in base oils—shows how much attention to detail can improve daily life for both hobby mechanics and big industries.
Keeping Food and Fragrance Stable
Preserving foods and creating flavors pulls in heptanoic acid, too. Manufacturers use certain heptanoic acid-derived compounds to lock in freshness or add depth to processed foods, ensuring that what ends up on the table matches the expectations set by advertising and memory. In perfumery, this acid helps certain scents last longer or balance out the harsh edges of other ingredients.
There’s plenty of debate about what goes into foods and perfumes. Using heptanoic acid has passed rigorous checks. Food safety agencies in the US and Europe demand clear answers about what chemicals turn up in finished goods. Approval comes only after layers of research to check for possible health risks.
Special Uses in Pharmaceuticals and Plastics
In some drug formulations, heptanoic acid shows up as an intermediate during the process of building more complex molecules. It pops up in making plasticizers, which soften tough plastics and make things like vinyl flooring or cables more flexible and durable. These uses may not grab headlines, but they matter. Flexible plastics don’t crack under stress, so cables run safely and flooring lasts longer in busy places.
Waste and Environmental Questions
Heptanoic acid enters wastewater during production. Plants that handle this chemical must invest in techniques to reclaim or break it down before discharge. Strong regulations set boundaries, and many companies have begun to view waste as lost profit rather than just a problem. Technology like advanced filtration and better process design cut down on what escapes and help keep local communities safer.
Room for Smarter Solutions
Across all its uses, heptanoic acid reminds me of the value of focusing on small details in big systems. Whether breaking down grease buildup, extending the life of a machine, or holding flavor in a favorite snack, it stands as proof that chemistry touches more parts of modern life than we notice. Continued investment in smarter production and responsible handling can shrink the risk of pollution and boost the quality of everything this unsung workhorse touches.
What are the physical and chemical properties of Heptanoic Acid?
The Nature of Heptanoic Acid
Heptanoic acid, often called enanthic acid, forms part of the family of straight-chain carboxylic acids with seven carbon atoms. This colorless, oily liquid carries a strong and unpleasant odor, similar to old cheese or sweaty socks. Anyone who’s handled it in a laboratory or industrial setting knows that smell; it makes you want to double-check your gloves. The liquid doesn’t stay put—its volatility means it will evaporate fairly quickly at room temperature.
How Heptanoic Acid Behaves
Heptanoic acid boils at around 223°C (433°F) and freezes near -8°C (18°F). This places it right between the shorter and longer chain acids in terms of handling and storage. While it stays liquid under most normal environmental conditions, don’t expect it to pour smoothly in cold weather. Water hardly dissolves heptanoic acid, but organic solvents like alcohol and ether mix well with it. This fact guides many of its uses in chemical labs, especially as a building block for larger molecules.
Working with heptanoic acid brings a certain caution. Inhaling the vapors irritates airways. Skin contact can burn. The substance eats away at some plastics, but stainless steel or glass doesn’t mind it at all. Tough gloves, chemical splash goggles, and a well-ventilated workspace quickly feel necessary if you’re pouring or transferring it.
Chemical Traits that Matter
The carboxylic acid group at the end of the molecule gives heptanoic acid its bite. This group donates hydrogen ions easily, which makes the acid somewhat reactive. Neutralizing it happens with bases like sodium hydroxide or potassium hydroxide, forming heptanoates—useful salts. These heptanoates end up in special lubricants, corrosion inhibitors, and sometimes even the flavors or scents in packaged goods, albeit in controlled amounts.
Most organic chemists value heptanoic acid because of its reactivity and carbon chain length. Seven carbons make it less smelly than butyric or valeric acid but easier to handle than the massive, waxy acids you need to melt just to pour. You can chain it to other molecules or turn it into esters for perfumes, artificial flavors, and plasticizers. Many labs use it as a reference compound or building block to synthesize pharmaceuticals.
Why Understanding Heptanoic Acid Matters
Anyone that spends time in a chemistry lab or plant soon figures out that “just an acid” glosses over key points. Handling a short-, medium-, or long-chain acid changes everything—from storage practices, spill cleanup, all the way to downstream uses. Heptanoic acid, sitting right in the middle lengthwise, gets used for making custom lubricants or even specialty plasticizers. For example, its esters soften plastics, giving flexibility to products like vinyl flooring. In the flavor industry, chemists sometimes use its compounds to give butter- or brie-like notes to processed foods, yet at undetectably low concentrations. High exposure is no joke, but low trace use can be practical.
Managing Safety and Finding Solutions
Anyone storing or transporting heptanoic acid learns the importance of leakage control early. Secondary containment and tight-sealing, corrosion-resistant vessels give peace of mind. Eye washes and safety showers belong nearby in case someone splashes skin or eyes by accident. Analysts check for vapor exposure by monitoring air in storage and handling areas. It’s worth pushing for improved ventilation, as even brief exposure to the harsh vapors can overwhelm the senses.
In recent years, producers have started adopting green chemistry routes for making heptanoic acid from renewable resources, cutting down on emissions and petroleum use. These methods show promise for shaping a more sustainable future in the chemicals sector. If you’ve ever handled a sample or worked near stored drums, you know why safety controls feel just as important as how you use the acid itself.
Is Heptanoic Acid safe to handle and what precautions should be taken?
Not Your Everyday Pantry Item
Heptanoic acid, sometimes popping up in labs and industry, isn’t something most people bump into outside a chemical shelf. The name alone hints at its punch. This oily liquid, with a sharp back-of-the-throat smell, doesn’t seem like much at first glance. Take a closer look, and experience shows that treating it casually invites trouble.
Safety Isn't Optional
Years in the lab taught me to respect chemicals with fiery tempers and sneaky fumes. Heptanoic acid belongs in that camp. Spills sting hands; careless whiffs catch you unprepared. According to the National Center for Biotechnology Information, it irritates skin, eyes, and the respiratory system. Even brief contact can lead to pain or redness. Some colleagues told me how the vapor can overwhelm a workspace if ventilation slips, leaving your nose burning and lungs tight.
Rubber gloves, sturdy goggles, and a face shield aren’t overkill for heptanoic acid; they're the minimum. Chemical-resistant aprons keep splash surprises off clothing and skin. Open bottles only under a chemical hood. If the air draws the odor right out, mistakes shrink. You won’t find shortcuts paying off here.
Spills and Errors: What Experience Teaches
One afternoon, a friend in waste management missed a drop. That faint, almost fruity smell turned acrid in seconds, lingering long after the mess cleared. Heptanoic acid eats through rubber and some plastics—one cheap glove torn and there’s burning in minutes. Not every glove out there will protect your skin either; check chemical compatibility charts, and swap regular latex or vinyl for heavy-duty nitrile.
Eyes need more than everyday safety glasses. Splash goggles close up tight around the face, leaving nothing to chance. Rubbing at stinging skin only pushes the acid deeper. Quick rinses in an eyewash station or safety shower prevent long-term injuries. If your workplace skips these, ask for them. I’ve seen a careless shop with only a garden hose get fined on the spot.
Staying Ready and Informed
Storing heptanoic acid away from heat and open flames matters just as much. Flammable vapors find their way into nearby ignition sources. Fire departments treat heptanoic acid fires with dry chemicals or carbon dioxide. Water spreads the acid. Keep emergency contact numbers near the storage area.
Training isn't bureaucracy—it's survival. Anyone handling this acid needs access to up-to-date Safety Data Sheets. These documents explain how to spot symptoms, respond to spills, and choose materials that keep hands, eyes, and lungs safe. I keep copies on my phone and in print by the door, because nobody thinks clearly during a crisis.
Better Habits Protect People
Learning proper disposal keeps harm out of water systems. Don’t pour heptanoic acid down public drains. Label all containers, so confusion doesn’t cause a dangerous mix-up. Chemical-resistant secondary containment trays prevent leaky containers from seeping into places they shouldn’t go.
Heptanoic acid’s hazards aren’t unique, but they are serious. My years around acids taught me that skipping safety steps to save a minute can lead to months of hard lessons. Practice habits that protect everyone on the job, and treat every bottle with respect. Those who do, stay safe—and earn trust from those working nearby.
How should Heptanoic Acid be stored and transported?
Understanding the Challenges
Handling chemicals like heptanoic acid often takes a back seat in safety talks, but real-world experience teaches that the smallest oversight can lead to big problems. As a colorless oily liquid with a sharp odor, heptanoic acid may seem harmless to the casual observer. Yet spill one drum or crack one seal, and the story changes quickly. The fumes can irritate your lungs and skin, and corrosion can eat through carelessly chosen storage containers.
Proper Storage Starts with the Right Containers
Any chemical-savvy worker can spot trouble when acids get stored in the wrong container. Steel drums might come cheap, but acids chew through unlined metal in no time. Corrosive leaks cause a headache for everyone, risking both worker safety and environmental fines. High-density polyethylene (HDPE) and glass containers hold up best against heptanoic acid, making them the clear choice in chemical warehouses and transport trucks. Staying away from rusty barrels and questionable plastic pays off.
Keep It Cool and Out of Sunlight
Left in a hot, sunny spot, heptanoic acid loses stability and starts giving off stronger vapors. Warehouses with poor ventilation fill with harsh odors. That's more than a comfort issue; breathing in chemical vapors leaves workers coughing and groggy at the end of their shift, and nobody wants that. Smart companies use well-ventilated, shaded storage rooms and label these areas clearly to avoid confusion and keep mishaps to a minimum.
Sealing and Labeling Drums
Old habits die hard, but ignoring proper labeling has always caused trouble. Even a temporary worker notices clear, bold markings on the drums—the name, hazards, and handling instructions—because nobody wants to guess what’s inside. Tight, secure lids stop leaks before they happen. Inspections each week cut down on spills and accidents, and simple routines like checking seals do a lot more good than paperwork ever will. Regulatory compliance aside, these habits just make sense.
Transport Precautions You Can’t Skip
Over the years, stories circulate about spilled drums during loading and unloading, turning routine shipments into cleanup disasters. Trained staff lifts and moves these containers with equipment made for the job—no weak carts, no unbalanced pallets. Most accidents don’t come from wild circumstances, just carelessness or poor planning.
Regulations require proper hazard labels clearly visible during transport, as both emergency responders and drivers need to know what they’re hauling. Equipment like absorbent spill kits and eye-wash stations travel with the load whenever possible. Nobody wants to test their luck miles from a hospital.
Investing in Training and Building a Culture of Awareness
Legislation can push companies to follow guidelines, but day-to-day safety rests in the hands of workers who learn to spot risks and act fast. Training sessions shouldn’t just tick boxes on a compliance form; they should draw on real accidents, near misses, and the stories of workers who’ve seen what goes wrong. Fostering a culture of practical awareness keeps everyone a little sharper and a lot safer.
Prevention and Common Sense Go Hand in Hand
Too often, convenience tempts people into cutting corners—stacking drums too high, skipping a label, holding storage in poorly ventilated sheds. These shortcuts rarely save time in the end. A careful, experienced approach to storing and moving heptanoic acid comes from lessons learned and stories shared on the warehouse floor. In the long run, small steps—proper containers, steady temperatures, clear labels, skilled handling, and real training—protect both people and businesses.
What are the common applications and industries for Heptanoic Acid?
Heptanoic Acid’s Place in the Chemical Toolbox
Walk through the aisles of any auto supply shop or flip over most food packaging, and the ingredients often link back to a small set of chemicals you rarely hear about. Heptanoic acid stands out among these, not just for chemists but for people who care about where things come from. With a formula that points to its simple, seven-carbon backbone, heptanoic acid drives a surprising mix of industries and treatments.
Lubricants: Keeping Engines and Machines Running
Every car or factory machine relies on lubricants to curb friction and extend equipment life. Heptanoic acid’s oily, fatty properties make it a top pick for crafting esters used in high-performance synthetic oils. Engineers value its ability to function reliably across big temperature shifts, which means engines don’t seize up in the cold and don’t fry under pressure. Its stable structure also means longer service intervals and fewer breakdowns, reducing waste and helping budgets stretch further. If you’ve ever owned a car that just runs and runs, chances are some science from this molecule kept it humming.
Food Flavors and Fragrances: Taste and Smell That Linger
The power of smell and taste comes down to chemistry. In the world of flavors and fragrances, heptanoic acid plays a supporting yet crucial role. The food industry leans on its ability to deliver a mild, fatty, slightly fruity note in products where natural fruit or creamy nuances matter. Chewing gum, dairy-based treats, and baked goods gain depth from this acid’s presence. The perfume sector taps into heptanoic acid for its ability to hold floral or green top notes, which means the scent on your skin lasts longer. Without it, some of the most memorable sensory experiences would feel flat.
Pharmaceuticals and Medical Products: Behind-the-Scenes Helper
Medications often demand stable, bio-compatible additives. Heptanoic acid fits this profile, acting as an intermediate in the synthesis of certain drugs and in making antiseptic formulations. Its safety profile, based on established toxicology studies, reassures drug developers and regulators. The medical field also draws on heptanoic acid derivatives in specialty ointments and cream bases, where smooth texture and stability are non-negotiable. This might not grab headlines, but consistent medicine performance improves outcomes for patients worldwide.
Plasticizers and Polymers: Flexible Materials for a Modern World
Modern living depends on plastics and polymers, found in everything from flexible PVC to resilient packaging films. Heptanoic acid-based derivatives deliver stretch, resilience, and durability. Manufacturers see benefits in plastics that resist cracking and stay supple, which translates into products that last longer and cut down on replacement costs. Using heptanoic acid in polymer building gives manufacturers new options for customizing properties without sacrificing quality.
Greener Solutions and Future Focus
For all its roles, heptanoic acid also fits today’s push for more sustainable manufacturing. Plant-based and waste-stream-derived versions lower the environmental footprint, echoing demands from consumers who care about responsible sourcing. Innovators in the field continue to look for safer, smarter, and more renewable ways to use heptanoic acid. My own work with green chemical startups taught me that small changes in the supply chain add up, driving bigger shifts in industry standards.
Tackling Safety and Process Challenges
Safe handling practices keep workplaces and communities secure. Heptanoic acid carries risks if mismanaged, with skin and respiratory irritant properties noted by regulatory bodies like OSHA and ECHA. Companies investing in training, leak detection, and improved personal protection keep workers safe and safeguard the environment. Open communication with local communities about chemical practices also builds trust.
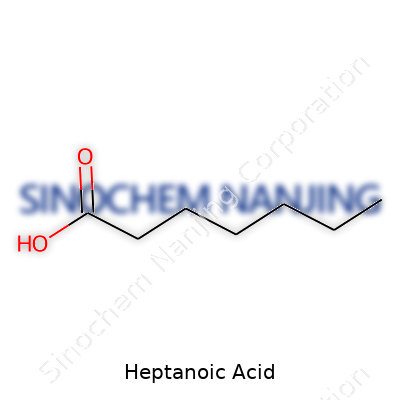
| Names | |
| Preferred IUPAC name | Heptanoic acid |
| Other names |
Enanthic acid Enanthylic acid n-Heptanoic acid Heptylic acid C7 acid |
| Pronunciation | /ˌhɛp.təˈnoʊ.ɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 111-14-8 |
| Beilstein Reference | 1209973 |
| ChEBI | CHEBI:30937 |
| ChEMBL | CHEMBL132 |
| ChemSpider | 4707 |
| DrugBank | DB02957 |
| ECHA InfoCard | 100.003.922 |
| EC Number | 206-761-7 |
| Gmelin Reference | 60538 |
| KEGG | C02679 |
| MeSH | D006523 |
| PubChem CID | 8057 |
| RTECS number | MC5950000 |
| UNII | SY7Q814V8G |
| UN number | UN2810 |
| CompTox Dashboard (EPA) | `DTXSID9020298` |
| Properties | |
| Chemical formula | C7H14O2 |
| Molar mass | 130.18 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Unpleasant rancid odor |
| Density | 0.924 g/mL |
| Solubility in water | slightly soluble |
| log P | 2.39 |
| Vapor pressure | 0.034 mmHg (25°C) |
| Acidity (pKa) | 4.89 |
| Basicity (pKb) | pKb: 15.97 |
| Magnetic susceptibility (χ) | -61.2×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.417 |
| Viscosity | 2.01 mPa·s (25 °C) |
| Dipole moment | 1.950 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 249.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -390.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4204.7 kJ/mol |
| Pharmacology | |
| ATC code | A05AX03 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H314: Causes severe skin burns and eye damage. |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P264, P273, P280, P303+P361+P353, P305+P351+P338, P333+P313, P337+P313, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 2-2-2-A |
| Flash point | 103 °C |
| Autoignition temperature | 440 °C |
| Explosive limits | Explosive limits: 1.1–7% |
| Lethal dose or concentration | LD50 rat oral 1120 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 3750 mg/kg |
| NIOSH | SAF |
| PEL (Permissible) | PEL: 5 mg/m³ |
| REL (Recommended) | 100 ppm |
| IDLH (Immediate danger) | 800 ppm |
| Related compounds | |
| Related compounds |
Caproic acid Octanoic acid Nonanoic acid Pelargonic acid Valeric acid |

