Diving Deep Into Heptafluorobutyric Acid: A Chemical with a Past, Present, and Future
Historical Development—From Laboratory Curiosity to Analytical Mainstay
Heptafluorobutyric acid didn’t just land on the lab bench by accident. Chemists first synthesized perfluorinated acids in the early days of organofluorine chemistry, back when new fluorinated compounds brought new hope for materials science and separation technology. Work on fluorinated carboxylic acids like this one built the foundation for modern applications in chromatography—an essential tool in everything from pharmaceutical analysis to environmental monitoring. Once people saw that heptafluorobutyric acid made separations faster, cleaner, and more reliable, its reputation grew. By the late twentieth century, labs stocked bottles for their LC-MS workflows, transforming the reliability of both clinical and research assays. That kind of shift doesn’t happen unless a compound proves it brings something others don’t.
Product Overview—A Compound with Range
Heptafluorobutyric acid isn’t just a chemical name to memorize; it’s a workhorse for chemists who need consistent results. Recognizable by its sharp, pungent smell and volatile nature, this acid plays a vital part in reverse-phase chromatography. Because it can act as an ion-pairing reagent, it helps resolve complicated mixtures that would otherwise leave analysts pulling their hair out. A bottle stored in a cool, ventilated corner of the lab means analysts are ready for anything from peptide mapping to testing food contaminants.
Physical & Chemical Properties—Why Chemists Trust It
People often ask why so many labs rely on heptafluorobutyric acid. The answer sits in its properties. With the formula C4HF7O2, this colorless liquid boils just above room temperature and packs a strong acidity, much stronger than ordinary carboxylic acids. Its boiling point, hovering well below 100°C, means it evaporates quickly—a blessing and a challenge when handling. High electronegativity from all those fluorine atoms changes everything, making this acid both highly reactive and uniquely persistent in certain contexts. Chemical stability runs high; oxidation and decomposition rarely become issues under standard storage.
Technical Specifications & Labeling—Beyond the Number on the Bottle
There’s an expectation that bottles labeled “heptafluorobutyric acid” meet standards. In practice, reputable producers deliver purity above 99 percent, tested on the same chromatographs where the acid gets used. Labels usually warn about the fume risk—and not without reason. Even a little inhalation brings sharp discomfort. All containers require Teflon or glass linings; this acid chews right through ordinary plastics and starts rusting aluminum in no time. Handling calls for gloves, splash goggles, and solid ventilation. Compliance with chemical standards often reflects decades of industrial learning, not bureaucracy for its own sake.
Preparation Method—How It Goes from Concept to Container
Fluorinated acids like this one don’t grow on trees. Making heptafluorobutyric acid starts with perfluorobutyl iodide, then swaps out that iodine for a carboxylic group through chemical transformations tough enough to humble routine organic chemistry. Electrochemical fluorination—passing electricity through a solution of organic precursors and hydrogen fluoride—delivers the final compound in relatively high yields. Equipment must resist harsh fluorine environments; nickel and Monel often end up as the materials of choice. Safety routines at industrial scale rise to another level, making the final price tag of the compound less surprising for those who have tried to make any short-chain perfluoro acid in the lab.
Chemical Reactions & Modifications—Not Just a By-the-Book Acid
Heptafluorobutyric acid doesn’t play by the rules set by acetic or formic acid. That collection of fluorine atoms locks it into an electron-hungry state, so typical reactions like nucleophilic substitution move sluggishly. On the other hand, that same stability lets the acid act as a stubborn modifier in chromatographic systems—consistently providing peak resolution without breaking down. It won’t get converted or degraded easily, which helps analysts minimize background signals. Derivatization work can attach its acyl group to amines or alcohols, but these reactions demand forceful conditions and clever chemistry.
Synonyms & Product Names—Talking About the Same Chemical
Depending on what you read, heptafluorobutyric acid might show up as HFBA or perfluorobutyric acid. Analytical suppliers might abbreviate or brand it for clarity, but once you spot C4HF7O2, you know what you’re looking at. Standard naming conventions help prevent mix-ups—a real concern, given the dangers of mislabeling and the presence of related acids with vastly different properties. In academic papers and technical literature, keeping synonyms straight streamlines research and supports reproducibility.
Safety & Operational Standards—Why Respect Matters
Working with heptafluorobutyric acid is not for newbies without proper supervision. The toxicity profile can’t be brushed aside: acute inhalation leads to coughing, headaches, and risk of more significant harm, especially in closed spaces. Skin contact leaves burns, and spills on clothing often mean the trash rather than a cleaning attempt. Gloves—nitrile or even better material—plus lab coats and splash shields should become second nature. Chemical hygiene plans demand updated information and proper waste capture. Air handling—down to fume hoods with strong airflow—matters, especially in shared spaces. Accidents from hurried handling serve as reminders for the whole lab. Any improper storage or disposal risks both personal injury and wider environmental damage, a lesson hammered home by regulatory bodies for good reason.
Application Area—A Modern Analytical Tool
Just about every advanced chromatography setup owes something to heptafluorobutyric acid. As an ion-pairing reagent, it pulls polar analytes into the hydrophobic parts of a chromatography column, tidying up peaks and giving clear, identifiable results on complex samples. In clinical laboratories, its role shows up in drug testing, proteomics, and even the assurance of food safety. Environmental chemists turn to it when pulling trace organic acids from dirty matrices. These roles make heptafluorobutyric acid not just useful but essential for meeting guidelines from agencies like the FDA and EPA. Outside of chromatography, some synthetic chemists reach for this acid to temporarily protect reactive functional groups or tweak surface wettability, although its main home remains in separation science.
Research & Development—Always Room to Grow
Interest in heptafluorobutyric acid hasn’t reached a plateau. Scientists with an eye on sustainability look for alternatives or devise methods to recover and recycle perfluorinated acids, aiming to curb accumulation in waste streams. The challenge isn’t small—perfluorinated compounds don’t break down easily. Analytical chemistry keeps moving forward, with improvements in detection technology demanding cleaner, sharper methods. Research sometimes explores replacements or structurally-similar reagents for certain types of separations, but many still count on HFBA to deliver day after day. Industry pushes for higher purity, lower background noise, and less environmental fallout. Grants and private research dollars back studies that measure the breakdown of fluorinated acids in soils and waterways, scrutinizing what happens after analytical use. Responsibility here goes beyond compliance; it’s about stewardship for future labs and ecosystems.
Toxicity Research—Not a Threat to Ignore
A chemical as potent as heptafluorobutyric acid deserves close attention from toxicologists. Real concerns about organofluorine compounds in the environment push teams to track their mobility and persistence. Acute exposure rules the immediate danger in the lab, but chronic risks, including bioaccumulation in aquatic food chains, add another layer. Some studies zero in on its behavior in liver microsomes and on its run-through in cellular toxicity assays. Evidence says it doesn’t degrade quickly once it leaves controlled waste streams, pointing to a need for scrupulous end-of-life handling. Legislative pushes have tightened restrictions in many countries, with limits on industrial emissions and more monitoring of persistent organic pollutants. Labs must balance analytical clarity with environmental caution. Those who handle it daily recognize the implicit challenges and ongoing responsibility to keep personal and public safety front and center.
Future Prospects—Balancing Progress and Responsibility
Looking forward, the fate of heptafluorobutyric acid won’t ride on laboratory science alone. Its strengths in chromatography guarantee it a place in the toolbox for some time, but environmental regulations and sustainability commitments will shape how and when people use it. Research efforts continue to probe for less persistent alternatives without sacrificing performance. As detection methods for perfluorinated compounds sharpen, accountability stretches back to every pill tested, every river sampled, and every chromatogram printed. The hard truth is, scientific success rarely comes without strings attached—a lesson that hits home for chemists as they refine, analyze, and strive to do less harm, not just more science.
What is Heptafluorobutyric Acid used for?
What Sets Heptafluorobutyric Acid Apart
Heptafluorobutyric acid, usually shortened to HFBA, stands out in the lab world. Its name sounds intimidating, but its work is surprisingly practical. Most folks have no reason to hear about it outside a laboratory or chemical supply shelf, yet this compound quietly shapes a lot of the data scientists use in fields like pharmaceuticals, environmental studies, and food safety.
Heptafluorobutyric Acid in Chromatography
Chromatography uses HFBA as an ion-pairing reagent, letting scientists detect and separate compounds with precision. For researchers running high-performance liquid chromatography (HPLC), HFBA bridges the gap between molecules that want to stick together and those that don’t. Charged molecules—think amino acids, peptides, and certain drugs—often resist sticking to the tools scientists use to separate them. Adding a bit of HFBA changes the charge and “behavior” of these molecules, letting scientists run more accurate and efficient tests.
The pharmaceutical industry leans on HFBA to hunt down impurities and confirm drug composition. Subtle differences in chemical makeup matter when lives are on the line. Food scientists use it to track pesticides hiding in produce or residue in animal products. Environmental labs turn to HFBA for water and soil testing because it reveals contaminants that could slip past older methods.
Good Science Depends on Small Details
HFBA isn’t magic. It’s a tool built on the reality that many chemicals don’t separate cleanly without a little help. Its value shows up most strongly with compounds that basic silica or reverse-phase columns struggle to handle. For me, the first real “aha!” moment with HFBA came while running late-night checks on protein fragments. After adding HFBA, peaks on my chromatogram went from muddy and unclear to sharp and reliable. That’s the difference between guessing at your results and trusting them.
Labs worldwide depend on reliable data. That data forms the backbone of drug safety reports, pollution studies, and even forensic evidence in tough court cases. Whenever someone spots a contaminant or a trace compound, HFBA may have played a role in that analysis.
Risks and Solutions
Every chemical comes with pros and cons. HFBA’s benefits don’t erase its toxicity and corrosiveness. Mishandling it can mean burns or environmental spills—a nightmare in a high-traffic lab or municipal water system. The answer starts with education. Everyone in the lab—from the seasoned lead analyst to the new intern—needs practical training, not just the Safety Data Sheet in a binder. Regular workshops and refreshers pay off in safe labs and fewer accidents. Clear labeling and lockup of hazardous chemicals cut down on confusion.
Disposal deserves real attention, too. Putting HFBA waste down the drain puts ecosystems at risk. Labs should standardize their waste collection and work with certified disposal outfits. Insisting on proper disposal protects both waterways and reputations.
Looking Ahead
Some research labs and companies have started exploring less toxic alternatives. These substitutes won’t replace HFBA in every application right away, but they signal a shift toward safer and greener chemistry. Until more options prove themselves, HFBA remains a workhorse—essential, respected, and in need of careful stewardship from those who rely on it most.
Is Heptafluorobutyric Acid hazardous or toxic?
Looking Closer at Heptafluorobutyric Acid
Heptafluorobutyric acid pops up in conversations around chemical safety, especially in labs, pharmaceutical manufacturing, and environmental circles. It’s a colorless liquid, kind of a workhorse in liquid chromatography. With a sharp, pungent odor, the stuff doesn’t hide its presence. Having worked with compounds from this family, I know that even seasoned chemists don’t shrug off these “perfluoro” acids.
Health Effects and Exposure
Heptafluorobutyric acid isn’t your everyday vinegar — its potent acidity and mix of fluorine atoms means trouble if you get careless. Direct skin or eye contact triggers burns, redness, and pain that can linger. Breathing in vapors irritates the nose, throat, and lungs. Industrial Material Safety Data Sheets report that accidental ingestion causes nasty burns in the mouth and digestive tract.
Most legitimate toxicity concerns come from animal studies and data gathered in chemical plants. Accidental splashes, spills, or inhaled mist remain the most common risk. Repeated exposure leads to dermatitis, respiratory irritation, or long-term respiratory symptoms if safety steps get skipped. There’s not a lot of information about cancer or reproductive harm in humans, but given how similar perfluorinated compounds have earned their reputation, it’s wise to draw a line: gloves, goggles, hood, and careful handling should be non-negotiable.
Environmental Impact
Fluorinated chemicals stick around for a long time. Heptafluorobutyric acid belongs to the same broad chemical group as PFAS (per- and polyfluoroalkyl substances), and they share a certain infamy. These molecules don’t break down easily in soil or water, so they slowly add up in the environment. Scientists have measured PFAS in rivers, wildlife, and even drinking water. Repeated releases from industrial plants once seemed like a small risk, but evidence is mounting—these chemicals travel, accumulate, and affect ecosystems in ways we’re only beginning to understand.
Some countries and states are now cracking down on PFAS releases for exactly this reason. Accidentally dumping or washing away heptafluorobutyric acid comes with real consequences, not just for labs but for the entire neighborhood, risking contamination of groundwater and, by extension, public health.
Managing the Risks
Every lab professional I talk to keeps the MSDS nearby for heptafluorobutyric acid, just in case. Standard practice means gloves, chemical splash goggles, and working in a fume hood. Immediate access to clean water for rinsing accidents makes a difference. Strict storage—tightly sealed containers, clearly labeled, away from anything reactive—helps prevent leaks.
Disposal brings its own headaches. Pouring leftovers down the drain does real harm, so hazardous waste procedures make a difference. Having worked in both small and large lab settings, I know that the cost and effort of proper disposal always beats the risk of groundwater contamination or regulatory fines.
Solutions and Alternatives
Some researchers are testing replacements for heptafluorobutyric acid in chromatography. Using less persistent or less toxic ion-pairing agents can help reduce long-term environmental impact. Improving containment—closed systems and advanced fume hoods—lowers workers’ risk, and stricter disposal laws force organizations to rethink old habits.
Education remains crucial. More people in science need to understand the true hazards, not just what’s listed on paper. Knowing the risks, following best protocols, pushing for greener alternatives, and sharing knowledge will play the biggest role in making sure heptafluorobutyric acid doesn’t become tomorrow’s headline environmental problem.
How should Heptafluorobutyric Acid be stored?
Understanding the Substance
Heptafluorobutyric acid shows up in many labs, especially for chromatography work. Its sharp smell and strong acidity mean respect for its hazards isn’t optional. It reacts with metals and moisture and produces fumes that sting your nose and eyes. Dismissing its dangers comes with a price, sometimes a trip to the emergency room.
Tackling Proper Storage
Before someone starts working with this acid, storage gets top priority. Over years of lab work, I’ve noticed how loose caps and cracked bottles almost always lead to headaches—both literal and legal. Sturdy glass bottles with reliable chemical-resistant liners make all the difference here. Polyethylene caps work better than metal; metal gets eaten alive.
Shelf placement matters just as much as the container. Many acids end up left open on a high shelf, where spilled drops become hard to spot until it’s too late. Keep bottles at eye level or lower, inside corrosion-resistant cabinets. Acid cabinets built for strong mineral acids do the job. Store as far as possible from bases, amines, and anything flammable. Combining acids and bases never leads to peace and quiet—only fizzing messes or dangerous fumes.
Temperature and Ventilation
Heptafluorobutyric acid responds badly to warmth. High temperatures boost its vapor pressure; the stink will fill a room fast, along with the risk for eye and lung irritation. Cool storage below normal room temperature always works better. In an old lab of mine, someone once stuck a container of this acid in a hot equipment room—the result lingered in the air for days after cleanup.
Ventilated storage setups help, too. Acid cabinets with venting keep building air safer by drawing vapors away from people. For anyone without built-in ventilation, storing the container inside a fume hood proves effective. Never store in regular office fridges: chemical-grade cold storage exists for a reason. The gaskets on food fridges can’t take this kind of acid.
Labeling and Tracking
One key detail easy to gloss over is clear labeling. Permanent ink goes on every bottle with both name and date. As chemicals age, their stability falls. Lab managers who keep careful logs prevent confusion, and clear logs make inventory easy to audit. This helps track expirations and lets you spot if a bottle’s been sitting too long to trust.
Dealing with Spills and Degradation
A dropped bottle or a leaking cap can turn a clean lab into a mess. Every place handling this acid keeps a spill kit nearby. Their key ingredients: neutralizing agents for acids (sodium carbonate or bicarbonate), plenty of absorbent material, and sturdy gloves. After the first time I watched someone scramble for a paper towel, I swore to never work without proper spill response close at hand.
Frequent checks prevent accidents. Once a month, I’d look at each bottle for stress cracks or corrosion. Anything off—yellowing labels, sticky threads, or pitted glass—means it’s time to get rid of that container under a chemical waste contract. Never dump this material down the drain: it ruins pipes and poisons water.
A Culture of Caution
Storing heptafluorobutyric acid safely comes down to good habits and real respect for its hazards. Years of seeing careless storage lead to damage or injuries shaped the way I treat any strong acid now. Well-kept bottles, cool cabinets, clear labels, and emergency gear—this combination beats out luck every time. People’s health, as well as environmental safety, hang on these simple choices.
What are the handling precautions for Heptafluorobutyric Acid?
Close Calls and Safety Measures
Anyone who has spent time in a chemistry lab probably remembers a tense moment with something strong and volatile. Heptafluorobutyric acid stands out as one of those compounds you learn to handle with care. It's a strong acid, giving off sharp fumes, and contact with liquid or vapor can quickly turn a good day in the lab into a trip to see the nurse. A friend of mine once knocked over a small beaker of it, and even with quick cleanup, the stinging smell left a mark—both on the desktop and in all our memories.
Protecting Yourself First
Good gloves, eye protection, and a proper lab coat aren’t suggestions—they’re non-negotiable. Splash-resistant goggles keep the acid well away from your eyes, and long sleeves stop those surprises that seem to come out of nowhere. Skin makes a poor shield against strong acids like heptafluorobutyric, which cause burns on contact—so a solid apron offers real peace of mind, too.
Where to Work and Ventilation
Sheltering behind a solid, ventilated fume hood helped me through many chemical preparations. The vapors do not belong anywhere near your lungs, and strong ventilation keeps the work area much safer. Airy windows and open doors don’t cut it, so make the fume hood your closest colleague during work with this stuff.
Storing and Moving the Acid
Leaving heptafluorobutyric acid on a bench or carrying it in an open container isn't just risky—it's asking for trouble. Keep it in tightly sealed glass containers with clear, chemical-resistant labels. I learned never to rely on memory, especially on busy days—sharp, permanent markings save lives and prevent confusion. Store it away from bases and anything reactive, well out of the sun and locked up if possible. I've seen what a spill next to a base can cause, and chemical burns heal slowly, if they heal at all.
Spills Happen—Respond Fast
Small spills need quick action with spill kits designed for acid; never stop to improvise with random absorbents. Neutralizing powders work best, but remember to protect your hands and face before tackling any cleanup. Rags soaked with acid can release fumes for hours, so proper disposal in a dedicated, labeled container keeps the risk contained.
Training and Knowledge Matter
I spent a lot of lab hours repeating safety drills, reading those sheets detailing everything from boiling point to toxicity. It's worth the repetition. Even veterans slip when routines get lazy, so refreshers on emergency eyewash stations and spill response cut down on errors. Everyone working with these materials deserves to know exactly what to do if something goes wrong.
Health and Long-Term Concerns
Contact with heptafluorobutyric acid stings, but repeated exposure to vapors can do deeper harm to lungs and even overall health. A single exposure makes the risks clear, yet they add up in the long run. Keeping a tidy, organized workspace, wearing the right gear, and never rushing pays off in healthy years down the line. Watching colleagues teach and lead by example convinced me that, above all, real safety culture lasts longer than any warning sign.
What is the chemical formula of Heptafluorobutyric Acid?
The Make-Up: Formula That Shapes Science
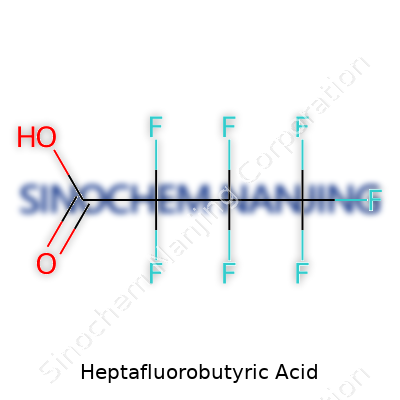
Anyone with an interest in chemistry or a nose for lab work has bumped into a compound with a name that sounds like a spell from a fantasy novel—heptafluorobutyric acid. The chemical formula for this substance reads as C4HF7O2. There’s meaning buried in every part. With a backbone of four carbon atoms, this acid features seven fluorine atoms, a single hydrogen, and two oxygen atoms. The arrangement doesn’t just make it look exotic on paper; it gives the compound a punch when it comes to reactivity and utility.
Where Use Meets Curiosity
Back in my undergraduate days, labs often reeked from the unique, almost biting aroma that everyone learned to respect when working with strong acids. Heptafluorobutyric acid always stood apart. Its robust chemical structure, forged by those seven fluorines, granted it stability and a slightly menacing power when added to separations in liquid chromatography. In practice, this meant it helped keep organic samples from sticking together during analysis. Scientists love it for this; it provides crisp, reliable data during instrument runs—especially for proteins and tricky small molecules.
Safety and Touch: Handling Fluorinated Acids
Dealing with compounds pumped full of fluorine takes a steady hand and a respect for lab rules. Heptafluorobutyric acid asks for gloves made from thick nitrile or even butyl rubber. It’s not the sort of acid that goes easy on the skin or nose, sending sharp warnings with fumes that trigger coughing and watery eyes. Practice, rather than a warning label alone, really drives home why safety matters. Accidental spills become shrill reminders for careful pipetting and proper ventilation.
Environmental Concerns: Not Just a Lab Problem
Stories from industry have drawn a lot more attention to the environmental impact of fluorinated chemicals. Those seven fluorines make the compound resistant to breakdown. Once released, it can stick around in water and soil, mingling with other perfluorinated compounds and raising eyebrows about long-term safety for both wildlife and people. There’s urgency here; research points to some fluorinated acids collecting in living tissues, sometimes disrupting hormones or immune responses.
Seeking Solutions: Smart Chemistry Moving Forward
Regulators and scientists started asking tough questions. Why keep using these acids when some alternatives exist? For now, nothing matches the combination of stability and performance in certain analytical tasks, so heptafluorobutyric acid keeps its place in laboratories. Researchers keep hunting for greener replacements—chemicals that can step up to the job but break down faster without hanging around in ecosystems.
Waste management practices offer part of the answer. Laboratories must treat waste containing fluorinated acids with a commitment to containment and neutralization. There’s no shortcut, only rigorous process. At the same time, developing safer alternatives relies on research funding and cooperation between universities and private companies. It takes a blend of innovation, investment, and a bit of humility as chemists admit some traditional tools need an overhaul.
Science, Curiosity, and Responsibility
Exploring compounds like heptafluorobutyric acid means balancing curiosity against caution. The formula matters not just to those inside the laboratory, but to anyone concerned with what chemicals make their way from bench to environment. From experience, real progress grows from the willingness to question old habits, embrace safer practices, and treat every formula with the respect—and skepticism—it deserves.
| Names | |
| Preferred IUPAC name | 2,2,3,3,4,4,4-Heptafluorobutanoic acid |
| Other names |
Perfluorobutyric acid 1,1,1,2,2,3,3-Heptafluorobutanoic acid HFBA |
| Pronunciation | /ˌhɛp.təˌflʊə.rəˈbjuː.tɪr.ɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 375-22-4 |
| Beilstein Reference | 1721397 |
| ChEBI | CHEBI:4860 |
| ChEMBL | CHEMBL1377 |
| ChemSpider | 85346 |
| DrugBank | DB01970 |
| ECHA InfoCard | 201-991-7 |
| EC Number | 375-22-4 |
| Gmelin Reference | 79022 |
| KEGG | C02658 |
| MeSH | D006523 |
| PubChem CID | 69797 |
| RTECS number | EL8750000 |
| UNII | Q82T16AN9O |
| UN number | UN2810 |
| CompTox Dashboard (EPA) | DTXSID7041488 |
| Properties | |
| Chemical formula | C4HF7O2 |
| Molar mass | 214.048 g/mol |
| Appearance | Colorless liquid |
| Odor | Pungent |
| Density | 1.6 g/mL at 25 °C |
| Solubility in water | Soluble |
| log P | 0.864 |
| Vapor pressure | 41.5 mmHg (20 °C) |
| Acidity (pKa) | 1.57 |
| Basicity (pKb) | 1.17 |
| Magnetic susceptibility (χ) | −32.9 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.252 |
| Viscosity | 0.75 cP (25 °C) |
| Dipole moment | 1.434 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 330.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -960.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1876.9 kJ/mol |
| Hazards | |
| Main hazards | Corrosive, causes severe skin burns and eye damage, harmful if swallowed, harmful if inhaled, may cause respiratory irritation. |
| GHS labelling | GHS05, GHS06 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H314: Causes severe skin burns and eye damage. |
| Precautionary statements | P261, P280, P304+P340, P305+P351+P338, P310, P405, P501 |
| NFPA 704 (fire diamond) | 1-3-1 |
| Autoignition temperature | 310°C |
| Lethal dose or concentration | LD₅₀ Oral Rat 484 mg/kg |
| LD50 (median dose) | LD50 (median dose): 200 mg/kg (rat, oral) |
| NIOSH | MMV595961 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.6 ppm (2.7 mg/m³) |
| IDLH (Immediate danger) | No IDLH established. |
| Related compounds | |
| Related compounds |
Trifluoroacetic acid Pentafluoropropionic acid Nonafluoropentanoic acid Perfluorooctanoic acid Perfluorobutanoic acid |

