Guanidine Nitrate: A Deep-Dive into Its Present and Future
Historical Development
Guanidine nitrate has a story that mirrors many breakthroughs of chemistry in the late 19th and early 20th centuries. Synthetic guanidine compounds, first isolated in the mid-1800s, quickly found interest due to their versatility. Crystallizing guanidine from calcium carbide byproducts came about as industries searched for value from what used to be waste. Over the decades, chemists optimized routes to produce guanidine salts, focusing on stability, yield, and purity. Large-scale production picked up pace thanks to wartime demands in the twentieth century, since guanidine nitrate provided both chemical energy and nitrogen content. This compound stopped being an obscure laboratory curiosity and started serving as a core material in defense, mining, and specialty processing.
Product Overview
Guanidine nitrate is a white, crystalline solid, best known for its role as a high-energy material. Chemists use it for its strong base properties, and industries look at its nitrogen content and its ability to release energy rapidly. It comes in powder or granule form, usually packed in moisture-tight containers. Laboratories and manufacturers label their guanidine nitrate by minimum purity, moisture content, and particle size. The market classifies it by these specifications for pyrotechnics, propellant manufacturing, and research uses. Sometimes, a shift in grade means the difference between what works in airbag systems and what belongs on a color lab bench.
Physical & Chemical Properties
This compound melts around 213°C and decomposes at higher temperatures, producing nitrogen-rich gases and small organics. It dissolves quite well in water and polar solvents, making it easy to process. At room temperature, guanidine nitrate is stable if kept dry and away from strong acids or bases. Water pulls it out of containers if there's a leak, so packs always carry a warning about storage. The nitrate part gives it oxidizing capability, while the guanidine backbone injects reactivity in synthesis chemistry.
Technical Specifications & Labeling
Manufacturers fix their attention on purity ratings—often quoted as 99% or better for technical applications. Labels on industrial drums must show the batch number, weight, country of origin, date of production, and safety designations. Hazard pictograms highlight its oxidizing nature, and handling sheets often accompany orders above one kilogram. Bulk supply asks for consistent particle profile so large-batch chemical reactions run smoothly. Certain regulatory regions demand childproof seals or tamper-evident bands.
Preparation Method
Classic routes to guanidine nitrate start from dicyandiamide, reacting with ammonium nitrate under carefully controlled temperatures. Some plants use direct ammonolysis of cyanamide with nitric acid. The reaction usually happens in stainless steel reactors because the nitric acid corrodes mild steel. Operators tune reaction times and reagent ratios for maximum yield, watching exotherms closely. After crystallization, rinsing, and drying, they screen the product to meet quality standards. Waste streams require neutralization before disposal.
Chemical Reactions & Modifications
Guanidine nitrate acts as a starting point for a stack of guanidine derivatives—more than a dozen routes branch off from it towards pharmaceuticals, agricultural agents, or dyes. By tweaking the guanidine or nitrate parts, chemists generate tailored salts or mixed oxidizers. It decomposes on strong heating, producing gases like nitrogen and ammonia, which engineers use for inflating airbags. Reacting it with various acids or bases spawns new materials with carefully tuned reactivity or solubility.
Synonyms & Product Names
Folks in the lab sometimes call it "carbamidine nitrate" or, less glamorously, just guanidine salpeter. Some catalogs list it under EC numbers or as guanidinium nitrate, referencing the charged nature of its molecular form. Product sheets often tie these names back to CAS number 506-93-4, to keep things straight. It pops up in synonyms lists on safety data sheets, showing the overlapping naming conventions in the U.S., Europe, and Asia.
Safety & Operational Standards
Guanidine nitrate demands respect in storage and handling, since it supports combustion and may explode if heated strongly or contaminated with certain metals or organics. Companies lock it down in cool, dry, ventilated warehouses far from flammable materials or acids. I’ve watched safety audits checking labels, spill kits, and PPE stocks. Any spill means a quick evacuation and expert cleanup. Regulatory bodies, including OSHA and the European Chemicals Agency, maintain strict guidelines—training, documentation, and inspection cycles form the backbone of safe practice.
Application Area
Most attention lands on its role in airbags, where the material goes from steady solid to instant gas that cushions impacts. Mining outfits use it in explosives to tune detonation power and brisance. Labs deploy it for finding tiny amounts of trace chemicals, since guanidine structures help uncover elusive biological markers. It shows up in rocket propellants and gas generators for emergency systems. Guanidine nitrate’s basicity also lets it step into organic synthesis as a catalyst or a nitrogen source for specialized molecules. This range—engineering, lab, and fieldwork—grows every decade as new processes find a fit for it.
Research & Development
Chemists and material scientists constantly scan for ways to refine guanidine nitrate’s properties. Projects in my network chase better thermal stability or cleaner burning for eco-friendly airbags. Some research teams develop hybrid materials by embedding guanidine nitrate in novel binders, hoping to reduce sensitivity to impact but keep the efficiency. Bioanalytical teams lean into its backbone chemistry for prepping samples in DNA extraction. Each round of R&D papers brings new crystal forms, smarter combinations with other energetic molecules, or safer packaging. General industry trends now push for greener production routes and waste minimization, spurring pilots on renewable feedstocks.
Toxicity Research
Early work established that guanidine nitrate breaks down in the environment, but its effects on aquatic life and long-term soil health call for deeper dives. Toxicologists now spend as much time on the byproducts as the parent compound. Short-term exposure at commercial use levels mostly affects skin and eyes, so plant crews wear gloves and goggles. Chronic exposure studies point to possible effects on liver and kidney in animal models, making monitoring programs priority number one. More recent animal studies boost hopes for low bioaccumulation, but every large user sets up regular workplace air and surface checks. Data from poison control centers remain sparse, though rare ingestion or mishandling accidents keep training requirements in place.
Future Prospects
Pressure to cut emissions and toxic byproducts pushes the next era of guanidine nitrate chemistry. Airbag demand stays strong as global safety standards expand, but manufacturers want compounds that burn cleaner and degrade faster after use. Green chemistry ideas—biocatalysts, solvent reduction, renewable starting materials—figure into research plans. A broader look reveals its role in potential pharmaceutical intermediates or as a step to more effective explosives with lower environmental footprints. Demand for DNA purification and analytical kits gives guanidine nitrate a future-facing role outside heavy industry. My own conversations with students and startups hint that modified guanidine structures could cross into agriculture, carbon capture, or battery electrolytes—as long as safety and sustainability lead the way.
What is Guanidine Nitrate used for?
What Makes Guanidine Nitrate Special?
Walk into a busy industrial plant or a fireworks display, and you’ll find more science at play than you might expect. Chemicals like guanidine nitrate may never make the front page, but they help shape daily life. Guanidine nitrate, a white crystalline substance, pops up across multiple sectors, doing jobs that few notice.
People might know it best for its role in car airbags. When cars crash, there’s no time to think—the airbag must deploy in a blink. Guanidine nitrate triggers a rapid chemical reaction that produces a burst of safe gas, filling the airbag swiftly to cushion the blow. It replaced sodium azide, an older airbag propellant, since it breaks down into gases that don’t stick around in the air or environment.
Beyond Airbags: Safety and Explosives
Its story doesn’t end with airbags. Factories producing explosives and pyrotechnics rely on this compound. Guanidine nitrate offers something valuable: rapid decomposition with reliable energy output. This gives fireworks their lift and helps controlled demolitions proceed according to plan. In mining, it becomes part of the charge that helps break up rock so that minerals and ores can be hauled away.
People often worry about safety with chemicals linked to explosives. Experience shows that companies need tight controls and well-trained staff to avoid problems such as heat buildup or accidental ignition. In the right hands, guanidine nitrate is manageable. Without proper precautions, it becomes a risk, not just for the immediate team but for entire communities nearby.
Medicines, Research, and Everyday Life
Step outside the industrial world, and guanidine nitrate keeps working. Pharmaceutical researchers turn to it during drug development. In the lab, scientists use it to unravel proteins, helping them glimpse how future drugs could work inside the body. It’s tough to imagine heart medicines or certain antibiotics reaching shelves without chemical helpers like this one.
My time in a university lab opened my eyes to the role guanidine nitrate plays in folding or unfolding proteins. Watching lab techs handle it with care drove home how each substance, no matter how specialized, has a ripple effect. Handling and storage demand attention to temperature and humidity, because mistakes can ruin a whole batch of research results or—for companies—an expensive run of product.
The Environmental Angle and Safer Solutions
Questions keep coming about the balance between performance and environmental footprint. Guanidine nitrate wins points for biodegrading more easily than some older propellants. Still, production facilities need to tackle pollution and manage chemical waste. Scrubbing facilities, good oversight, and innovations such as closed-loop systems could shrink the risks.
For families and communities near production sites, vigilance matters. Public agencies run inspections, but the strongest pressure often comes when neighbors join watchdog efforts, call for transparency, and advocate for tech upgrades that reduce emissions. Real progress often grows from this kind of grassroots push.
Looking to the Future
Innovation never stays still. Chemists keep hunting for new ways to replace risky chemicals with something safer, cleaner, and just as effective. Guanidine nitrate may stick around in airbag plants and labs for years to come, but the search for greener chemistry—whether from plant-based sources or new manufacturing techniques—pushes the entire industry forward.
Chemicals like guanidine nitrate help power everyday life, but the most responsible path keeps safety, health, and the planet front and center. Honest conversations between scientists, industry, and the public keep these substances working for everyone—and not just hidden in the background.
Is Guanidine Nitrate hazardous or explosive?
Getting Real About Guanidine Nitrate
Guanidine nitrate pops up in the conversation about chemical safety because it plays a role in industrial and automotive products, especially airbag inflators. Not everybody working with chemicals has a chemistry degree, so the facts need to be clear: is this stuff genuinely risky?
Taking Stock of the Risks
Guanidine nitrate is pretty much a white, odorless powder. Most people won’t bump into it at the supermarket, but if you work at a plant, lab, or factory where airbags get built, you know the name. And for a good reason — safety rules require respect for any chemical that can turn dangerous if it’s mishandled.
The real headache with guanidine nitrate lies in its dual nature. On one hand, it serves as a fuel in airbags thanks to its stability under typical conditions. On the other, it holds enough energy to go off in a blast if misused. According to the European Chemicals Agency (ECHA) and published safety sheets, it’s rated as an “explosive when mixed with combustible material.” Broken down: this substance stays controllable in the right hands, but combining it with organic matter, or exposing it to fire or intense friction, and things can turn ugly.
Stories From the Floor
People I’ve met from the autoworks world treat guanidine nitrate the same way an electrician treats exposed wires. They don’t mess around — gloves, face masks, tight inventory logs, and strict separation from any sources of sparks. The other day, one tech told me he’d seen packets sent back to the supplier just for packaging flaws, highlighting the zero-tolerance attitude for anything that looks amiss.
Outside automotive, research labs also respect guanidine nitrate, but not out of paranoia. It’s just about building good habits, like keeping containers sealed and logging every transfer. Even small spills get reported. I’ve seen people call a time-out to double-check a label. That attention to detail means nobody has to learn the hard way.
Hazardous, Not Hopeless
Calling guanidine nitrate outright “explosive” oversimplifies a more complicated truth. As pure powder, stored in a cool, dry spot and kept away from fuel or flame, it remains stable. The problems begin when it gets hot, meets friction, or comes into contact with other chemicals, especially anything flammable. The fallout from accidents is no joke. The US Chemical Safety Board once detailed several plant incidents involving improper storage. Sometimes, small mistakes snowball: a leaking package, an overloaded shelf, an overheating piece of machinery.
Smarter Habits Keep Everyone Safer
Rules exist for good reason. Facilities need climate controls, non-sparking tools, and plenty of signage. Regular safety drills help calm nerves and build muscle memory. Reporting near-misses and learning from them helps keep the rate of real accidents lower. For smaller outfits or new users, training and mentoring carry a lot of weight. If someone’s nervous about handling guanidine nitrate, there’s usually an old hand who can walk them through storage protocols, record-keeping, and what to do if something goes sideways.
Technology helps too. Sensors for heat or smoke, checklists for chemical inventories, and updated risk assessments reduce the odds of a surprise. Even something as simple as having a separate, locked cabinet adds another layer of protection. None of this is high theory. It’s the daily grind of keeping workers healthy and business up and running.
Taking Responsibility
Guanidine nitrate calls for respect and a clear head. Overconfidence often causes trouble. With the right attitude and focus on detail, people can use this chemical in ways that help — not harm. It’s the difference between disaster and a good day on the job.
How should Guanidine Nitrate be stored and handled?
Understanding What’s at Stake
Guanidine nitrate holds plenty of promise in chemical labs and across several industries. It plays a role in propellants, airbags, and even research work. Lately, its risks attract closer scrutiny, especially for those working up close with this substance. It’s not a product you can shove on a shelf and forget about. Just one wrong storage decision can invite trouble — chemical leaks, fire, or worse.
From Storage Room to Lab Bench: Practical Safety
A reliable storage plan always begins with paying attention to dryness. Guanidine nitrate does not get along with moisture. Any hint of humidity clumping the powder turns it lumpy and, after a while, makes it less effective or flat-out dangerous. A sealed container works best. High-density polyethylene or metal containers with tight lids give peace of mind. Throw a small desiccant pack in there for extra dryness.
Shelving matters. A container should stay off the floor because accidental spills easily become bigger problems if a leak goes unnoticed. Cabinets designed for chemicals — complete with ventilation and away from direct sunlight — help a lot. Heat builds up fast near radiators, windows, or in rooms that don’t get enough breeze. Hot or cold, this compound keeps best under 25°C.
Keeping Guanidine Nitrate Separate
Separation is safety’s best friend. Guanidine nitrate does not like coming into contact with strong acids or reducing agents. If it does, there’s a good chance of hazardous byproducts popping up. Some people use color-coded storage bins to mark where oxidizers, acids, and organics belong. It cuts down confusion, especially during a stressful moment or shift change. Years ago, I misplaced a chemical in storage. The cleanup took all afternoon and nearly caught me a warning from my supervisor. Sorting these containers pays off.
Mindful Handling, Every Time
Working with this material means personal protective equipment is non-negotiable. Gloves, aprons, chemistry goggles — skipping a step here gives problems down the line. Some workers wear nitrile gloves instead of latex for better resistance. A spill kit near every storage closet or workbench helps respond quickly. It’s always easier to grab a spill pad or neutralizer powder than scramble for it once a bottle tips over.
Never scoop with a metal spatula; static electricity builds up unpredictably and can ignite dust. Plastic, non-sparking tools are the right call. After use, washing hands and wiping down benches might seem routine, but those quick habits avoid cross-contamination.
Planning for Emergencies and Training
Training isn’t paperwork to check off and forget. Real-life drills — mock spills, evacuation walks, or first-aid refreshers — show their worth when mistakes threaten. Most chemical labs keep material safety data sheets (MSDS) easy to reach. They show how to handle any problem, from eye splashes to accidental inhalation.
Fire extinguishers rated for chemical fires shouldn’t gather dust either. If guanidine nitrate ever catches fire, it reacts violently with water. Dry chemical extinguishers stay ready by the exit. Some coworkers make sure everyone can operate these, not just supervisors.
Caring for People and Places
Teams that work closely with chemicals like guanidine nitrate often rely on each other’s habits for safety. Backups matter, whether it’s keeping extra gloves in stock or double-checking every label at the start of a shift. Regular audits give everyone a chance to fix mistakes before they turn serious. These good habits don’t just protect property — they create a safer space for everybody who steps into the lab.
What are the physical and chemical properties of Guanidine Nitrate?
What Stands Out About Guanidine Nitrate
Guanidine nitrate draws attention in the chemical world because of its unique mix of energy and versatility. The chemical formula, CH6N4O3, tells you it has both carbon and nitrogen packed tightly together. This makes it a favored ingredient in things like airbag inflators, pyrotechnics, and propellants. I remember helping a friend with automotive research, where finding a gas generant for airbags meant balancing speed, pressure, and temperature—all key reasons this chemical appears in so many safety applications.
Physical Properties: Taking a Closer Look
As a solid, guanidine nitrate usually appears as a white, odorless crystalline powder. It dissolves well in water. At room temperature, it feels similar to regular table salt in texture, though you never want to handle it as casually. It weighs in at about 1.44 grams per cubic centimeter. The melting point hovers around 213°C, and above that, it decomposes—releasing gases and energy. It doesn’t let off fumes or odors in standard conditions, so you often can’t sense its presence unless you see it.
This material doesn’t just mix readily with water; it also holds a low vapor pressure, so it won’t evaporate into the air. That reduces risk from inhalation during storage and transport, a point I always stress in safety training. Guanidine nitrate’s granular or crystalline forms can create dust clouds if mishandled, adding hazards you wouldn’t expect at first glance. Its density and granular texture help control how it packs, flows, and reacts in industrial settings.
Chemical Instincts: How Guanidine Nitrate Reacts
The real story comes from its chemical behavior. Guanidine nitrate combines guanidine, a high-nitrogen compound, with nitric acid, fixing a nitrate group on the molecule. This structure stores chemical energy that can release quickly as heat and gas under the right spark or heat source. Manufacturers favor it in airbags and explosives because the reaction is energetic but controllable. The equation for decomposition looks simple on paper: it breaks down to form water, nitrogen gas, and carbon dioxide. But in practice, those gases produce massive pressure in an instant—just the tool engineers want for lifesaving restraint systems.
This salt stays chemically stable under common conditions, resisting slow breakdown under normal storage. Direct contact with strong bases or acids can set off violent reactions. In my own lab work, even minor contamination has meant halting operations and launching a cleanup. Guanidine nitrate is incompatible with heavy metal salts and strong oxidizers. Mixing these leads to risks that experienced chemists and emergency responders never underestimate.
Risks and Managing Safety
This chemical brings real hazards. Its rapid decomposition can trigger fires or blasts if ignored or mishandled. Exposure to skin, eyes, or lungs can cause irritation—sometimes severe. Personal protective equipment, controlled environments, and strict storage laws play critical roles in keeping people safe. I’ve seen how lapses—like a faulty glove or an open storage bin—lead to accidents that cost money, health, and lives.
Smart handling, regular safety drills, and equipment checks—all rooted in daily hands-on diligence—help manage these risks. Information from the U.S. Environmental Protection Agency and the National Institute for Occupational Safety and Health (NIOSH) confirms the need for well-ventilated areas, robust containers, and standard housekeeping. Safe disposal methods and spill containment plans keep workplaces and communities protected.
What are the safety precautions when working with Guanidine Nitrate?
Respecting What Guanidine Nitrate Can Do
Guanidine nitrate packs a punch. In labs and industries that handle energetic materials, folks treat it with the same respect they’d give to a chainsaw: useful for many jobs but not one to take lightly. The stuff is often used in airbags and propellants, and it isn’t shy about releasing energy. One careless moment can seriously harm people or equipment.
Looking Danger In The Eye
Coming into contact with guanidine nitrate brings a few risks: toxicity, reactivity, and the possibility of fire or explosion. This compound irritates skin, eyes, and lungs. Swallowing just a small amount or breathing its dust can lead to nausea or headaches. If it catches fire or if a spark hits a pile, it doesn’t hesitate to combust.
Basic Gear And Habits That Save Lives
Before I step near any open container of guanidine nitrate, I check my gear. Chemical-resistant gloves and safety goggles form my basic armor. I add a lab coat and work in a well-ventilated area or, even better, under a fume hood. These simple steps cut risk by half or more. I’ve seen co-workers try to skip gloves. Physical discomfort, rashes, and worse come quick. Protecting skin isn’t optional here.
Good habits make the most difference. I never eat, drink, or touch my face before washing up, and I treat contaminated clothing as hazardous waste. If any gets on me, I remove clothes and rinse off, then let a supervisor know. Out in the plant, workers need to change out of their working clothes before heading home—not just for their own sake, but for the health of their family, too.
Respecting Storage And Fire Hazards
Storing guanidine nitrate in a cool, dry spot is basic common sense. One humid shelf led to caked, clumpy powder in my old lab, upping the chances of accidental ignition. Keeping it away from acids, oxidizers, and open flames is more than a rule—it’s personal safety. I learned fast to avoid working near incompatible chemicals. In labs with regular audits, folks drill on checking chemical compatibility charts. A fire extinguisher sits within reach, with staff trained on its use.
Training, Communication, Real Solutions
What keeps everyone from harm is training and speaking up. On every new project, the most experienced hand leads safety walkthroughs. We practice what would happen if there’s a spill or fire. MSDS (Material Safety Data Sheets) get posted where everyone sees them. People know who to call and what to do. Emergency showers and eyewash stations stay uncluttered—these aren’t for show.
Managers can take safe handling up a notch by limiting how much guanidine nitrate sits on a lab bench at one time. Encouraging a culture where nobody’s afraid to say something boosts everybody’s safety. After seeing close calls, I know it’s worth reviewing procedures together. In my time, open conversations, regular safety checks, and real-world drills made our working group stronger and safer. Guanidine nitrate doesn’t forgive mistakes; respectful, informed workers stay out of harm’s way.
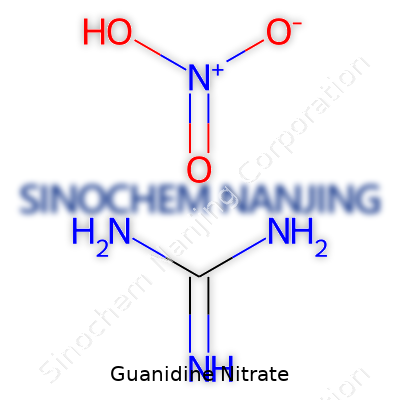
| Names | |
| Preferred IUPAC name | guanidinium nitrate |
| Other names |
Guanyl nitrate Nitraguanyl Guanidinium nitrate |
| Pronunciation | /ɡwəˈnɪdiːn ˈnaɪtreɪt/ |
| Identifiers | |
| CAS Number | 506-93-4 |
| Beilstein Reference | 361722 |
| ChEBI | CHEBI:75756 |
| ChEMBL | CHEMBL1233463 |
| ChemSpider | 15745 |
| DrugBank | DB11472 |
| ECHA InfoCard | ECHA InfoCard: 100.005.186 |
| EC Number | 208-060-1 |
| Gmelin Reference | 85308 |
| KEGG | C01758 |
| MeSH | D017782 |
| PubChem CID | 24045 |
| RTECS number | MU7175000 |
| UNII | 59K5B0PYNV |
| UN number | UN1477 |
| CompTox Dashboard (EPA) | DTXSID4019223 |
| Properties | |
| Chemical formula | CH6N4O3 |
| Molar mass | 59.07 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.44 g/cm³ |
| Solubility in water | soluble |
| log P | -0.77 |
| Vapor pressure | Negligible |
| Acidity (pKa) | pKa ≈ 13.6 |
| Basicity (pKb) | 11.7 |
| Magnetic susceptibility (χ) | -29.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.436 |
| Viscosity | Viscosity: 2.02 mPa·s (at 25 °C, 56% solution) |
| Dipole moment | 2.58 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 146.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -324.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -365 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V03AB16 |
| Hazards | |
| Main hazards | Explosive, oxidizing, harmful if swallowed, causes serious eye irritation |
| GHS labelling | GHS02, GHS03, GHS06, GHS08 |
| Pictograms | GHS01,GHS03,GHS07 |
| Signal word | Danger |
| Hazard statements | H271, H315, H319, H335 |
| Precautionary statements | P210, P220, P221, P264, P280, P305+P351+P338, P337+P313, P370+P378, P402+P404, P501 |
| NFPA 704 (fire diamond) | 1-1-1-OX |
| Flash point | 160 °C |
| Autoignition temperature | 350°C |
| Lethal dose or concentration | LD50 oral rat 1920 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 936 mg/kg |
| NIOSH | UN0143 |
| PEL (Permissible) | PEL: 0.3 mg/m³ |
| REL (Recommended) | 6.5 mg/m3 |
| IDLH (Immediate danger) | 100 mg/m3 |
| Related compounds | |
| Related compounds |
Guanidine Guanidinium chloride Guanidinium carbonate Guanidinium thiocyanate |

