Glutaronitrile: Development, Properties, and Future
Historical Development
Scientists first identified glutaronitrile in the early twentieth century, curious about its role as an aliphatic dinitrile. Chemical manufacturers realized its value as the world embraced new polymers and solvents. Companies gradually scaled up its production during the mid-century surge in plastics and specialty chemicals. The substance helped drive significant advances in organic syntheses—a feat that often doesn’t get much attention outside industry and academic circles. Chemists who spent time in industrial labs or university settings probably recall glutaronitrile popping up as a key intermediate, bridging gaps between raw hydrocarbon feedstocks and the more valuable chemicals in pharmaceuticals, dyes, and plasticizers.
Product Overview
Most people never encounter pure glutaronitrile, but for those working with fine chemicals or custom synthesis, it becomes a staple. Its structure offers chemists two highly reactive nitrile groups linked by a three-carbon chain—simple at first glance, but that simplicity works in its favor for building more complex molecules. In the laboratory, glutaronitrile comes as a colorless to slightly yellowish liquid, common in glass bottles with hazard labels that signal both its reactivity and toxicity.
Physical & Chemical Properties
Glutaronitrile presents as a clear liquid, with a faint, somewhat sharp smell. Its boiling point hovers around 265°C, while it stays liquid at room temperature due to a melting point well below zero. The moderate polarity means it mixes predictably with most organic solvents, from acetone to ether. It doesn’t dissolve in water quite as readily, but enough does to require careful wastewater management. Two nitrile groups confer a degree of chemical stubbornness, resisting hydrolysis unless pushed with acid or base. Air and sunlight barely change it, yet for anyone heating or reacting glutaronitrile, care must be taken not to generate dangerous by-products or allow vapor accumulation.
Technical Specifications & Labeling
Manufacturers typically supply glutaronitrile in technical-grade and high-purity forms, the latter for research and specialty synthesis. Labels mark the CAS number—110-61-2—alongside proper hazard icons for toxicity, flammability, and aquatic risk. Safety datasheets warn of harsh effects on the nervous system with accidental exposure. Shipping drums and laboratory bottles need clear identification and storage away from incompatible materials, such as strong oxidizers or acids.
Preparation Method
Industrially, most glutaronitrile is made by dehydrating glutaric acid or heating glutaryl chloride with ammonia or amines, sometimes catalyzed by phosphorus pentachloride or other dehydrating agents. Older routes involved oxidative ammonolysis of pentamethylenediamine. These reactions call for rigorous temperature control, anhydrous conditions, and management of off-gassing. Any operator must handle both the starting materials and product with gloves, goggles, and proper ventilation, given nitriles’ well-known toxic risks. Scrubbing exhaust gases and treating liquid effluent are not just best practice but also required by most environmental authorities.
Chemical Reactions & Modifications
Chemists value glutaronitrile for its role in synthesizing diamines after full or partial hydrogenation, turning it into compounds like pentamethylenediamine—a building block for polyamides. The reactivity of the two nitrile groups lets organic chemists run a neat spectrum of reactions: hydrolysis to glutaric acid, reduction to diamines, addition reactions with organometallics, and cyclizations to make heterocycles. In the right hands, glutaronitrile unlocks multistep syntheses for bespoke medicines and advanced polymers. These are not just textbook reactions but real-world transformations where controlled conditions determine safety and yield. Researchers always keep material safety at the forefront, as off-gassing and incomplete reactions can generate toxic by-products.
Synonyms & Product Names
Glutaronitrile often turns up under various names, including 1,5-dicyanopentane and pentanedinitrile. Different suppliers might use product codes or catalog names, but in laboratory jargon or industrial procurement, these synonyms refer to the same molecule. The uniformity in chemical identification helps track raw materials along the lengthy chain from manufacture, shipment, through to the lab bench or plant floor.
Safety & Operational Standards
Every technician who’s ever handled glutaronitrile knows safety guidelines are not optional. Without gloves, even brief skin contact can irritate or worse. Fume hoods, face protection, and proper storage away from incompatible chemicals are essentials, not afterthoughts. Inhalation or accidental ingestion brings real health consequences—central nervous system depression, respiratory challenges, even severe long-term effects if exposure repeats or concentrations spike. Companies install emergency showers, eyewash stations, and strict training regimes; regulators enforce exposure limits in workplace air. Disposal follows local hazardous waste protocols, often with incineration or chemical neutralization.
Application Area
The world outside chemistry rarely hears about glutaronitrile, even though it greases the wheels of several industries. Polymer manufacturing relies on it for synthesizing specialty nylons, polyamides, and engineering plastics. Pharmaceutical chemistry makes use of it as an intermediate in advanced heterocycle or active pharmaceutical ingredient (API) synthesis. Pesticide and agrochemical builders look to it for the backbone of modern crop-protection agents. Custom synthesis shops use it in dozens of pathways, sometimes at metric ton scale, sometimes just in a beaker. Not every route works out, but as a versatile intermediate, glutaronitrile keeps finding new roles.
Research & Development
During the past two decades, research teams have explored safer routes to glutaronitrile, aspiring toward green chemistry by eliminating hazardous reagents or harsh conditions. Innovative catalytic systems, solvent-free reactions, and continuous-flow setups tackle both efficiency and worker safety. In specialty polymer labs, new monomers and cross-linkers built from glutaronitrile help drive performance in areas like filtration, insulation, and medical devices. Researchers are deeply invested in finding catalytic systems that handle both yield and selectivity, while industry engineers balance cost, reliability, and regulatory pressure for lower emissions.
Toxicity Research
Toxicologists have tracked glutaronitrile’s health risks since the 1950s, recognizing the central nervous system as its primary target. Rodent models show that acute exposure triggers tremors, convulsions, coma, or even fatal respiratory depression. Chronic skin contact sensitizes or damages tissue, with inhalation risks compounded by volatility. Recent work has focused on biomonitoring production staff for nitrile metabolites and lowering workplace air concentrations. New toxicological studies keep regulators informed on threshold limits, while epidemiological surveillance covers any long-term morbidity in exposed workers. Technology helps, but the onus remains on responsible handling and site engineering.
Future Prospects
Looking forward, glutaronitrile faces both challenges and new opportunities. Environmental rules will only tighten, nudging manufacturers toward cleaner production methods and closed-loop waste handling. As demand rises for advanced polymers and fine chemicals, especially in renewable energy and medical fields, more eco-friendly glutaronitrile routes would unlock broader applications. The push for low-toxicity, biocompatible materials could see it play a background but vital role in everything from next-generation batteries to engineered tissues. It’s easy to overlook intermediates like glutaronitrile, but in chemical innovation and regulatory risk management, its story keeps evolving, shaped by the scientists, engineers, and operators who work with it—including those who never set foot in a traditional lab.
What is glutaronitrile used for?
Inside Glutaronitrile: The Basics
Glutaronitrile doesn’t stand out like household cleaners or plastic containers, but it plays a quiet, vital role in many industries. As someone who has spent years around industrial chemicals and spoken directly with plant technicians, I know that even the toughest-sounding chemicals often hide behind finished products and big medical breakthroughs. Glutaronitrile is one of these behind-the-scenes helpers.
Chemical Building Blocks
Factories use glutaronitrile to help make nylon-6,6. Most folks don’t think about how their stretchy workout gear, carpet fibers, or car airbags are made, but polyamides like nylon often start with a handful of sharp-smelling chemicals like glutaronitrile. Strong and flexible, these synthetic fibers protect, insulate, and last for years. The process to create them relies on safe chemical handling practices, which means people working with glutaronitrile need proper training and gear. This matters, because keeping workplaces safe helps prevent accidents and keeps production running smoothly.
Role in Advanced Chemistry
Pharmaceutical research wouldn’t move forward without small, reactive chemicals like glutaronitrile. Drug discovery needs reliable ways to build complex molecules, and glutaronitrile offers a versatile starting point. It brings two nitrile groups close together, which means chemists can transform it into all sorts of compounds—intermediates for drugs, agricultural chemicals, or specialty additives. According to industry reports, pharmaceutical labs use these nitriles to speed up testing and improve the yield of certain synthetic reactions.
Electronics and Coatings
Electronics depend on specialty coatings that protect circuits against humidity or dust. Manufacturers use chemical intermediates based on glutaronitrile to craft epoxy resins and curing agents. These products keep devices running in challenging environments. If you work anywhere near assembly lines, you’ll see the layers built up, tested, and stressed—reliable protection allows companies to avoid warranty headaches and customer complaints.
Risks and Safety Practices
Glutaronitrile deserves respect. Direct skin contact can cause irritation, and inhaling fumes creates health risks. Facilities that handle this chemical always invest in proper ventilation, leak detection, and employee training. These practical steps pay off. According to data from the National Institute for Occupational Safety and Health (NIOSH), accidental exposure reports dropped in plants that followed guidelines for chemical handling and used personal protective equipment consistently.
Looking Ahead: Balancing Use and Responsibility
Nobody expects specialty chemicals to disappear. Society keeps inventing new materials and therapies. The issue is making sure the people working with glutaronitrile stay safe, and downstream products remain trustworthy. Strong safety standards, real-time monitoring, and independent auditing help keep risks in check. Some research teams look for greener production methods or safer substitutes, though replacing a well-understood molecule with a brand new one creates its own challenges.
Summary: More Than a Name
Most people never encounter glutaronitrile outside of technical paperwork or material safety sheets. Its impact stretches from basic textiles and durable electronics to modern drug development. My experience says the real story with glutaronitrile, and so many other chemicals, runs deeper than recipes or formulas. It’s about responsible stewardship, workplace safety, and supporting innovation by handling the basics well.
What is the chemical formula of glutaronitrile?
Unlocking the Meaning Behind C3H6(CN)2
Most folks won’t come across glutaronitrile in everyday life, unless science or industry draws you in. Still, every piece of chemistry hiding behind industrial doors shapes daily routines, often invisibly. Glutaronitrile, with its chemical formula C3H6(CN)2, stands as one core example. This formula breaks down to three carbon atoms, six hydrogens, and two cyanide (nitrile) groups. Simple numbers tell a bigger story—of logistics, risk, innovation, and the way common items are made.
Industrial Reach: Not Just a Number Game
Glutaronitrile lives in the world of intermediates. Nylon-6,6, for instance, relies on it during production. Nylon shows up in everything from toothbrush bristles to seatbelts. Without a stable supply chain of raw chemicals, daily products vanish from shelves, and factories stall. Each atom in that formula plays its part. If a chemical plant misses the correct arrangement in C3H6(CN)2, the entire batch could turn unusable. Mistakes at the atomic level reach much farther, costing factories lost time or, worse, harming workers or the environment.
Hazards and Health: Trust But Verify
Most people would never spot glutaronitrile’s sharp smell outside the lab, yet for those who handle it, this compound poses real risk. Inhaling vapors, touching skin, or mismanaging waste—these can lead to dangerous outcomes. Cyanide groups demand extra respect, as they break down into toxic substances. Ask anybody in chemical safety, and you’ll hear stories about protocols, spill kits, and emergency drills. Reports from regulatory agencies back this up: the U.S. Occupational Safety and Health Administration (OSHA) and the European Chemicals Agency track solvents and intermediates like glutaronitrile, making sure safety systems go beyond the textbook.
Supply Chains and Global Impact
People often overlook the web of companies involved in producing and moving chemicals. Easily disrupted by politics, weather events, or transport problems, the chain that brings C3H6(CN)2 to factories connects continents. Global trends in demand for plastics or engineering fibers push producers to seek greener methods and alternatives, nudged by rule changes or public pressure. Sustainable chemistry, or green chemistry, steers research into replacements or safer handling strategies. Academic labs and startups chase ways to create key intermediates using less energy or renewable resources. These small adjustments ripple out to lower costs, reduce exposure to hazardous substances, and shrink environmental impact.
Learning through Openness and Quality Control
Trust comes from open information and steady oversight. More suppliers publish safety data and provide details on chemical purity, packing, and shipping standards. Third-party audits and transparent reporting systems help everybody—workers, regulators, buyers—judge quality before hazards arise. Studies led by industry consortia and universities track health effects, suggesting reforms in ventilation or protective gear long before regulations update. Anyone involved in manufacturing or research needs reliable sources for substances like glutaronitrile, not guesses or shortcuts.
Toward Safer, Smarter Practice
Solutions rest in careful handling, honest labeling, and a commitment to continuous improvement. Factories make sure teams understand not just how to run a process, but why certain checks and controls matter. Regulators publish case studies and best practices for safer chemical management. Consumers might never see the molecular details of C3H6(CN)2, but the choices behind every product begin on the atomic scale.
Is glutaronitrile hazardous or toxic?
What Glutaronitrile Actually Does
Glutaronitrile, also called pentanedinitrile, stands out as a basic chemical ingredient in labs and factories. Chemists handle it to make pharmaceuticals, plastics, and specialty chemicals. People working near it rarely see it as something they want to interact with by choice. What draws so much attention isn’t the name; it’s the risks tied to handling it.
Why Its Danger Matters in Real-World Settings
I remember my early days in a research lab, learning to treat every bottle with respect. Reading safety sheets about glutaronitrile, it became obvious that this isn’t one of those chemicals you just splash around. If spilled on skin, it can seep in fast. Many don’t realize that nitriles, the family glutaronitrile belongs to, carry risks much like cyanide. Absorbed doses—even the smallest amounts—can mean quick-onset poisoning. Headaches, dizziness, shortness of breath: these signs appear rapidly and can turn into serious emergencies.
Toxic effects start with irritation to the skin, eyes, and airways. From my experience, even a minor whiff in a closed lab could make your eyes sting and your nose burn. Once inside the body, glutaronitrile converts into compounds that block the ability of your cells to use oxygen. This biological punch is why workplace rules demand gloves, goggles, and fume hoods every single time you move the stuff from its bottle.
Is There Proof That Glutaronitrile Is Hazardous?
Scientific journals and industrial safety regulators all land on the same answer: it isn’t just risky in theory. Circling back to facts, the US National Library of Medicine lists it as “Toxic by all routes of exposure.” Research shows that animals exposed get convulsions, labored breathing, even death at higher doses. In my own lab, a scare from an open container reminded me that a little knowledge backed by careful practice can mean the difference between a normal day and a trip to the emergency room.
There’s more evidence from Europe. The European Chemicals Agency requires it to carry labels for skin, inhalation, and environmental hazards. Nobody wants this accidentally washing into groundwater, and for good reason. Once released, the impact stretches beyond the walls of the lab.
What Real Protection Looks Like
Companies using glutaronitrile need more than the bare minimum. From my time training new lab techs, gloves never left our hands, and nobody opened a container outside strong ventilation. Any accidental skin contact got treated with fast washing and medical observation. Fire chiefs keep material about it close by, since high heat or flame can release poisonous gases.
The best workplaces focus on staff education. I’ve seen too many places take shortcuts, only to scramble during a spill. Responsible employers enforce proper containment, safe storage, and regular drills. Solid record-keeping and clear labeling turn these habits into everyday life instead of once-a-year training.
There’s still work left to do. I believe that regular audits, better spill containment solutions, and open reporting systems can help. After witnessing how fast a routine task can go wrong, I value every extra safety measure people put in place.
Final Word: Respect Means Safety
Handling glutaronitrile means accepting its toxic punch and building safeguards into every part of the process. Respect for its hazards protects people, communities, and the environment from sharp consequences. What keeps everyone safe isn’t luck, but the mix of facts, vigilance, and experience.
How should glutaronitrile be stored?
Understanding the Risks Up Front
Glutaronitrile catches attention for its strong odor and potential health impacts. In my years working around chemicals, I’ve seen what happens when people underestimate substances like this. Even a small spill creates a lingering smell, and contact or inhalation can irritate the nose, throat, and skin. Some people in the chemical industry become sensitive to it after just a few incidents. A glance at its Material Safety Data Sheet (MSDS) makes it clear: this chemical demands respect.
Getting Storage Right From Day One
So, what actually works for storage? The trick is to keep it in a place that discourages carelessness. I’ve relied on tightly sealed containers made of compatible materials—stainless steel holds up, and high-density polyethylene doesn’t react. Glass can work but isn’t practical for larger amounts where breakage creates its own hazard. Containers must stay clearly labeled, not just for legal reasons, but because labeling helps everyone remember what they’re handling and stops mistakes before they start.
Room temperature storage might seem fine, but I stick with a cool, well-ventilated area. Heat makes glutaronitrile even more volatile. I once watched a drum stored in direct sunlight give off such a strong vapor that even the loading dock workers complained. Good airflow does two things: it dilutes any fumes and brings bad storage habits to light faster. Workers notice smells sooner and address leaks before they become dangerous.
Keeping People and Products Separate
One lesson that never leaves me: keep chemicals like this away from acids, strong oxidizers, and food preparation areas. Years ago, a friend told me about a warehouse that kept glutaronitrile near cleaning supplies and didn’t spot a slow drip for days. The mess triggered an evacuation. Segregation in storage isn’t just a checklist item; it’s the foundation for safe chemical management. Always use secondary containment, like bunds or spill pallets, to catch anything that gets out.
Helping Hands: Training and PPE
People forget rules unless training stays fresh. I’ve run plenty of refresher sessions over the years—nothing beats a demonstration for glove checks and respirator fit tests. Cotton won’t help. Use nitrile, neoprene, or PVC gloves. Face shields and chemical-resistant aprons reduce exposure. Anyone who transports or opens these containers needs to understand the risk, not just the procedures.
Treating Storage as an Ongoing Job
Routine checks give peace of mind. I’ve made logbooks a habit, jotting down storage temperatures, checking for leaks, and noting ventilation settings. Leaky containers don’t wait for office hours, so early detection controls damage. Alarm systems and fume sensors add another layer. Quick action matters—protecting health always wins in a cost-benefit analysis.
Solutions for Safer Handling
Digital tracking can help. Inventory programs flag expired materials and keep waste streams predictable. Engineering solutions, like vented storage cabinets, go further, keeping air quality in check and helping pinpoint problems.
Educating teams forms the backbone of chemical safety. Bringing everyone into the process—from cleaning crews to company drivers—stops accidents before they happen. Experience drives these decisions, but the facts remain: strict storage protects both people and property, and it’s the sort of habit nobody regrets.
What are the safety precautions when handling glutaronitrile?
Working around chemicals like glutaronitrile, you quickly learn that attention to safety is more than just following some checkboxes. People assume gloves and goggles will handle any risk, but with this stuff, caution takes on a new meaning. Glutaronitrile, thanks to its volatile nature, demands respect—skin contact can bring about burns, and the vapors carry their own risks. Getting sloppy can lead to headaches, dizziness, or worse, lasting nerve damage. If you’re like me, handling potent substances, missing a simple step can change your day in ways you don’t want.
The Crucial Gear
Picture a lab, just opened a fresh container of glutaronitrile. The smell hits fast—sharp, acrid. Without proper gloves, this clear liquid gets past your skin before you realize. Nitrile gloves, not latex, really make a difference. Add a snug lab coat and protective eyewear. Don’t forget your face shield if there’s a chance of splashes. Many skip the shield thinking safety glasses will cover them. They won’t, not when you’re tipping, mixing, or cleaning.
Fresh Air is Non-Negotiable
Many stories start with someone working in a corner room, fume hood fan humming quietly. Those fans aren’t decoration. Ventilation stops buildup of dangerous vapors. Once, I watched a coworker ignore a clogged fume hood. He paid for that with hours of dizziness and lingering nausea. Air movement matters. Local exhaust, an open window, and regular checks on hood function make a world of difference.
Hygiene Behind the Scenes
After a day around glutaronitrile, every break brings chance for contamination. Even if gloves look clean, residue drifts onto door handles, your phone, even lunchroom chairs. Eating or drinking in the workspace means taking that risk home. Routine hand washing makes a bigger impact than any sign-off sheet. Experienced lab folks keep a second set of clothes handy for a reason. It’s easy to forget, but one careless moment can trail chemical traces into your car or living room.
Spill Plans: Thinking Ahead
Everybody wants to believe spills happen to someone else. The real world says otherwise. A well-prepped chemical spill kit beats improvising with paper towels every time. Know where your eyewash station sits and double check that it hasn’t run dry. In my career, seeing someone scramble during a spill only once reminded me why drills aren’t a waste of time. Training closes the gap between reading a manual and acting fast when things go sideways.
Building a Safety Culture
Glutaronitrile pushes teams to move beyond routine. It’s not about paranoia; it’s about respect for the consequences. Let coworkers see questions as a strength, not a weakness. Encourage double-checks—on equipment, labels, and procedures. Real trust forms when people feel their concerns carry weight, not just for fancy audits, but because their health hangs in the balance. Glutaronitrile doesn’t forgive carelessness. A culture that prizes skill, vigilance, and open communication offers the surest shelter from its risks.
Looking Forward
Proper handling extends beyond rules on a poster. It calls for care in every action, gear that fits the task, a room where air feels clean, and coworkers who check in, not just check off. That’s where safety becomes a daily habit, not just a policy.
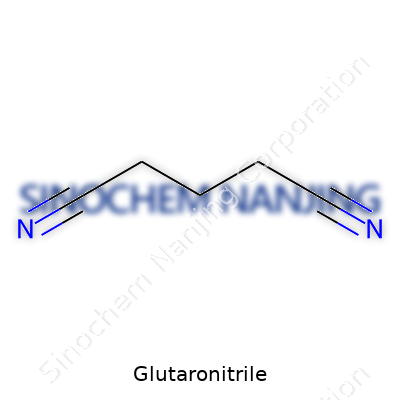
| Names | |
| Preferred IUPAC name | Pentanedinitrile |
| Other names |
1,4-Dicyanobutane 1,4-Butanedinitrile Tetramethylene dinitrile |
| Pronunciation | /ɡluːˈtær.oʊˌnaɪ.trɪl/ |
| Identifiers | |
| CAS Number | 108-13-4 |
| Beilstein Reference | 635940 |
| ChEBI | CHEBI:36641 |
| ChEMBL | CHEMBL2010157 |
| ChemSpider | 8034 |
| DrugBank | DB01982 |
| ECHA InfoCard | 03b9e6b1-88fc-4bb6-8c23-57c89b62c54e |
| EC Number | 203-743-0 |
| Gmelin Reference | 82591 |
| KEGG | C01699 |
| MeSH | D004003 |
| PubChem CID | 8057 |
| RTECS number | MA1400000 |
| UNII | 9G0X5KZ244 |
| UN number | UN1990 |
| CompTox Dashboard (EPA) | DTXSID0035739 |
| Properties | |
| Chemical formula | C5H6N2 |
| Molar mass | 110.14 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Unpleasant odor |
| Density | 0.967 g/mL at 25 °C (lit.) |
| Solubility in water | miscible |
| log P | -0.02 |
| Vapor pressure | 1 mm Hg (20 °C) |
| Acidity (pKa) | pKa = 25.0 |
| Basicity (pKb) | pKb = 4.55 |
| Magnetic susceptibility (χ) | -53.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.435 |
| Viscosity | 1.38 mPa·s (25 °C) |
| Dipole moment | 4.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 179.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -70.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1917 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS05, GHS08 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H312, H314, H317, H331, H411 |
| Precautionary statements | P210, P260, P280, P303+P361+P353, P304+P340, P312, P330, P501 |
| NFPA 704 (fire diamond) | 3-3-2 |
| Flash point | 61°C |
| Autoignition temperature | 730 °F (388 °C) |
| Explosive limits | Explosive limits: 2.8–17.0% |
| Lethal dose or concentration | LD50 oral rat 151 mg/kg |
| LD50 (median dose) | LD50 300 mg/kg (oral, rat) |
| NIOSH | FG9625000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Glutaronitrile is "5 ppm (17 mg/m³)". |
| REL (Recommended) | 10 ppm |
| IDLH (Immediate danger) | 75 ppm |
| Related compounds | |
| Related compounds |
Adiponitrile Succinonitrile Pimelonitrile Glutaric acid |

