Glutaraldehyde: Deep Dive into a Chemical Powerhouse
Historical Development
Early curiosity about simple carbonyl compounds set the stage for discovery and development in the late nineteenth century. Reppe and his team described glutaraldehyde’s chemistry in the 1940s, chasing new classes of aldehydes. The compound caught attention for its cross-linking capability, building on the legacy of formaldehyde in tanning and preservation. My first lab encounter with glutaraldehyde was marked by its sharp odor and potent fixative action, reinforcing lessons that this molecule’s potential emerged not from complexity, but from paired functional groups. Commercial production ramped up after scientists confirmed stronger, faster protein linkages than earlier agents had delivered. Hospitals and manufacturers soon saw its value.
Product Overview
Glutaraldehyde became a staple for industries needing reliable sterilization and biocidal performance. It’s often clear or pale yellow, typically supplied as a 25% or 50% aqueous solution. The disinfectant world carries plenty of risky alternatives, but glutaraldehyde carves its reputation by being lethal to most bacteria, fungi, and viruses. Labs that handle tissue samples for electron microscopy count on this tool for fixing cell structures. Water-treatment plants add it to biofilm control regimens. Companies brandish the chemical under names like Cidex, Metricide, and Glutacide, each pushing toward slightly different end uses.
Physical & Chemical Properties
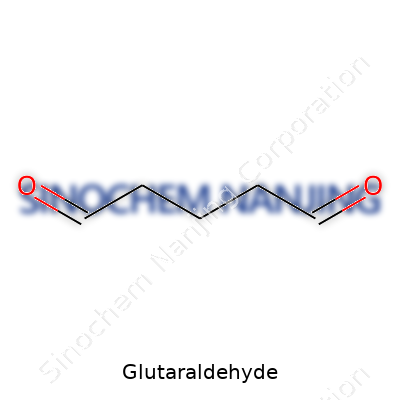
Glutaraldehyde comes as a pungent liquid, with a molecular formula C5H8O2 and molecular weight of 100.12 g/mol. Its boiling point reads about 187 °C. The aldehyde groups at each end of its five-carbon chain define its reactivity, and water blends with it easily, giving off a piercing smell. Years of handling this chemical leave a memory of slippery hands and tight gloves, since it penetrates skin fast and can cause nasty irritation. It typically resists hydrolysis under neutral pH, but strong bases chew it up quickly. Commercial samples show some off-color due to slow self-polymerization, especially on storage.
Technical Specifications & Labeling
Distributors and safety officers home in on concentration, pH value, and stabilizer content. Labeling also mandates hazard statements: “Toxic if inhaled. Causes severe skin burns. May cause allergy or asthma symptoms.” International shipping and procurement channels ask for lot numbers and traceability. Storage and shelf life depend on air-tight packaging, dark glass, and moderate temperatures. Quality testing for purity levels, usually above 99% in elite batches, matters most for research and hospital protocols. My own work with tissue culture always meant double-checking expiry and confirming exact dosing, since solution breakdown can compromise results.
Preparation Method
Industrial synthesis relies on oxidation of cyclopentene or pentane derivatives, with air or other agents ramping up one carbon at a time. Another method condenses 1,5-pentanediol with oxidizers like nitric acid in controlled reactors. Every chemical engineer I know agrees about the hazards of mixing strong acids and aldehydes—one mistake means respiratory distress or worse. Buffering and stabilizing agents step in during manufacturing to slow down self-condensation reactions and extend shelf life. Small-scale academic prep starts with less drastic steps but often trades off for lower purity.
Chemical Reactions & Modifications
The standout property is bifunctionality—the terminal aldehydes attack amino groups in proteins, cross-linking lysine residues. That makes glutaraldehyde a favorite for hardening biological tissues and fixing cells, since it locks proteins exactly where they exist in vivo. With strong bases, the molecule degrades, splitting into carboxylic acids. Reaction with alcohols forms acetals, which sometimes serve as masked forms for more complex industrial reactions. Direct polymerization produces viscous gums or resins. Chemists also tweak it to produce derivatives for bioconjugation, or to graft it onto silica agents in advanced filtration systems.
Synonyms & Product Names
You’ll see glutaric dialdehyde, pentanedial, and 1,5-pentanedial on safety data sheets. Brands like Cidex stand out in hospital stockrooms; laboratory catalogs feature Merck, Sigma-Aldrich, or Fisher Scientific supply lists under several variants, often coded by percentage or purity grade. Agricultural and industrial suppliers push trade names reflecting their biocidal or cross-linking specialties, filling out rows of blue-capped drums in facility stores. Language differences barely matter in this world—chemical structure always tells the real story.
Safety & Operational Standards
Nobody forgets their first accidental exposure—the burning sensation, the tightness in the lungs, the scramble for fresh air. It irritates eyes, nose, skin, and lungs. Gloves, goggles, and fume hoods show up in every protocol for handling, and yet, overconfidence or equipment failure still sends people to emergency showers. Most countries regulate workplace exposure, pegging short-term exposure limits at 0.2 ppm or lower. Hospitals insist on training and strict cleaning routines, rotating staff to minimize repeat exposure. Waste management means deactivation with sodium bisulfite or similar reducing agents, followed by careful drainage or incineration.
Application Area
Sterilization eats up a massive share, especially for heat-sensitive medical tools like endoscopes and dialysis equipment. Animal researchers use glutaraldehyde for tissue preservation, often alongside osmium tetroxide to harden microscopic samples for imaging. Leather tanning and paper manufacturing both lean on its cross-linking, since it helps toughen animal hides and add strength to specialty papers. Oil and gas companies pump diluted solutions downhole to control bacteria. I’ve seen plumbers and building engineers dose HVAC systems with it to control Legionella. Aquaculture and water treatment operators rely on its biocidal punch. It even pops up as a hardener in some adhesives and coatings.
Research & Development
Toxicologists, polymer scientists, and surface engineers constantly search for safer, smarter, and more selective uses. Biomedical research pairs glutaraldehyde-activated matrices with antibodies for diagnostics. Environmental chemists keep testing breakdown products in effluent, looking at risk profiles and searching for alternative disposal strategies. Industry has looked hard at polymer-immobilized formats—capturing the chemical’s benefits while cutting the risks of direct exposure. Efforts also target reducing off-gassing, since glutaraldehyde tends to vaporize and trigger asthma or allergic reactions.
Toxicity Research
The effects of exposure have kept industrial hygienists and clinical researchers busy for decades. Acute contact inflames mucous membranes, causes headaches and lung problems, and can trigger skin blisters. Long-term effects include occupational asthma and persistent dermatitis, especially among hospital staff, veterinary techs, and laboratory workers. Animal studies raise alarms about reproductive toxicity and possible cancer risk, though human evidence remains up for debate. Agencies like the US EPA, IARC, and NIOSH monitor data closely, setting exposure limits and guidance for disposal. In my early training, glutaraldehyde exposures cropped up more often than anyone liked to admit. Once released in a poorly ventilated room during a night shift, it sent three technicians home for days, reinforcing safety culture throughout the facility.
Future Prospects
Demand shows no sign of collapse, but pressure mounts to find greener alternatives that blend powerful disinfection with lower human and environmental risk. Biotechnology giants and university labs look for less reactive, more targeted agents for tissue preservation and cross-linking. Regulatory hurdles grow every time new data surfaces about allergies or toxic waste. Automation may reduce worker exposure, enclosing the process in “glovebox” isolators or shifting to solid-phase systems. Synthetic biology’s advance brings up new cross-linking peptides and enzyme systems, inspired by natural fixatives, which could one day sideline classic aldehydes. End users push for better user training, improved ventilation, and sensors that catch accidental leaks early. Having watched the shift from formaldehyde to glutaraldehyde in pathology suites, I predict the next wave will fuel debates about trade-offs: chemical effectiveness, human safety, and environmental responsibility.
What is glutaraldehyde used for?
More Than Just a Disinfectant
Glutaraldehyde enters the scene in places where clean really means no-nonsense clean. Hospitals count on it to kill germs and keep surgical tools safe, especially the ones that can’t handle the heat of an autoclave. I remember from a hospital internship how central supply rooms would fill with a sharp, acrid smell as trays of scopes came out from soaking. It always impressed me by how even the pickiest surgeons trusted their instruments after glutaraldehyde treatment.
Standing Up Against Bacteria and Viruses
The punch glutaraldehyde packs comes from how well it destroys bacteria and viruses. Compared to plain bleach or hydrogen peroxide, it targets cell walls more aggressively. Its track record covers tough bugs like tuberculosis and a wide mix of viruses. That’s why it finds a home not just in hospitals but in research labs where surprises from new microorganisms are never far off.
Experience in Animal Health
I grew up on a farm, and biosecurity for livestock always ran high on the list. Vets often recommended glutaraldehyde-based solutions for soaking boots and equipment between barns. It helped protect herds from contagious diseases in a way that plain old scrubbing couldn’t. Even now, most modern poultry or pig farms keep a bottle or two near every entryway for this very reason.
Industrial Uses and Water Treatment
Factories also look for something reliable, especially in paper mills and cooling water systems. Algae, fungi, and slime slow down machines and push up costs fast. Glutaraldehyde chips in as a biocide, keeping pipes and filters working smoothly. Municipal water systems use it to control dangerous bacteria, maintaining public safety from the inside out.
Risks and the Call for Safer Handling
Nothing with this level of punch walks away with zero risks. My hands stung after accidental splashes, and even well-ventilated rooms never felt quite fresh while it was in use. Studies tie regular, unprotected glutaraldehyde use to asthma and irritation. Safety data from the CDC highlights wearing the right gloves, face shields, and keeping rooms well-aired to lower exposure.
Regulatory Oversight and the Push for Alternatives
Regulators keep a close eye on glutaraldehyde because of these health concerns. The EPA in the U.S. tracks its use and disposal. OSHA outlines workplace rules around safe handling, helping prevent accidents. The medical field still looks for alternatives, like newer hydrogen peroxide systems, but old habits stay strong because glutaraldehyde delivers results quickly and reliably when stakes are high. A lot of hospitals now review protocols every year to see if options with lower risk might finally fit their needs.
Looking Ahead
People deserve clean tools and safe water, so glutaraldehyde probably remains a fixture for a while. At the same time, it’s worth pushing for safer containers, stronger ventilation, and education on protective gear. Maybe tomorrow’s disinfectant will match its power without trade-offs, but until then, a little caution goes a long way in keeping both people and spaces safer.
How should glutaraldehyde be handled safely?
Why Glutaraldehyde Demands Respect
Glutaraldehyde works as a tough disinfectant in labs, hospitals, and even some water treatment plants. People who have handled it know the fumes can sting your eyes or throat. Touching the liquid has often left folks with burning skin and rashes. I once watched a coworker wipe down a lab bench with bare hands, only to complain about tingling fingers later. These risks aren’t rare stories. Chronic exposure can trigger asthma or allergies. According to the Centers for Disease Control and Prevention, workers exposed regularly face a much higher rate of respiratory illness.
Preparation Builds a Safer Space
Anyone using glutaraldehyde should plan ahead. Many forget about the air itself—ventilation changes everything. Working in a space with strong airflow makes inhalation accidents rare. Fume hoods, or at minimum a strong exhaust fan, keep fumes from hanging in the air. Even with the right airflow, you want long gloves of nitrile or butyl rubber, along with splash-proof goggles. I keep a face shield handy for bigger cleaning jobs, since a single splash changes your whole day.
Small Habits Make a Big Difference
Simple routines work as the best protection. Always label glutaraldehyde clearly. In hospital settings, unmarked bottles often get mistaken for less hazardous cleaners. Labels prevent dangerous mix-ups, especially if staff rotate shifts. After each use, I wash up carefully—no quick rinses. Proper hand soap breaks down what lingers on skin, since water alone misses the mark. I’ve found that storing glutaraldehyde away from acids or strong bases avoids dangerous reactions that can send fumes through a whole room.
Training Supports Safety, Not Just Policies
Companies might push rules, but people learn best from seeing risks firsthand. Demonstrate how to mix solutions slowly, and show what to do if a spill spreads onto a lab bench or floor. Tossing down absorbent pads and opening windows beats calling maintenance. Have a spill kit handy and actually explain it. In my first job, nobody ever practiced spill response, and the first real accident brought panic. Running drills takes a small amount of time and pays off every year.
Preventing Chronic Health Issues
Short contact with glutaraldehyde usually triggers sharp symptoms—the kind that get attention. Long-term exposure works more slowly, with trouble appearing in sinuses or airways. The American Conference of Governmental Industrial Hygienists sets a very low exposure limit: no more than 0.05 parts per million over an 8-hour shift. Monitors can track air concentrations. If someone starts coughing regularly or develops a rash, don’t ignore it; start medical checks sooner, so problems don’t grow unseen.
Smarter Use Means Fewer Accidents
Hospitals found that switching to ready-to-use solutions with lower concentrations often cuts down accidents. It’s tempting to mix stronger batches, but sticking with the minimum strength for disinfection keeps everyone safer. Digital systems for logging chemical use also identify workplace patterns—more spills, complaints, or visits to nurses’ stations point out weak spots in current practices.
Accountability Starts with Everyday Choices
Workers, supervisors, and visitors share the same risks. Creating a culture where glutaraldehyde gets the same respect as lab acids or radiation sets a clear standard. Speak up if you notice shortcuts, and support coworkers who take extra care, even if it slows the pace. Putting safety first, every time, lets everyone leave work in the same shape they arrived.
What are the health risks of exposure to glutaraldehyde?
What Glutaraldehyde Really Means for Health
Glutaraldehyde shows up in places you wouldn’t expect. Hospitals count on it for sterilizing equipment, labs use it for preserving biological samples, and plenty of cleaning companies keep it stocked for its punch against bacteria and fungi. It works hard behind the scenes, but the people working around it end up paying a price far too often. I’ve seen nurses and cleaning staff complain of burning eyes and irritated skin after spending even a couple hours in poorly ventilated decontamination rooms. That’s rarely just a bad day—it’s the glutaraldehyde in the air.
Breathing Risks You Can’t See
The lungs take the biggest hit. When glutaraldehyde vapor hangs in the air, anyone breathing nearby may start coughing and feeling tight in the chest. Over time, workers report shortness of breath and asthma-like symptoms. Research from the National Institute for Occupational Safety and Health points out that repeated exposure raises the odds of developing occupational asthma. The CDC even classifies glutaraldehyde as a "respiratory sensitizer"—meaning your body can become allergic after a few hits, and then every whiff triggers something nasty.
Skin Contact is No Small Threat
Ask anyone scrubbing instruments in a surgical unit—skin contact leads to real trouble. That “chemical burn” feeling isn’t an exaggeration. Prolonged or repeated exposure causes dryness, cracking, and, in some cases, blisters. It’s not rare for workers to develop allergies, where even tiny droplets left on gloves can set off swelling and redness for days. I remember one microbiologist who switched jobs because of recurring eczema after years of handling glutaraldehyde solutions.
Long-Term Health Impact Isn’t Just Hype
Some folks shrug off short-term symptoms, thinking the impact fades after a wash and fresh air. Studies show different patterns. Chronic exposure, even at low levels, leads to permanent asthma in some cases. Eye and throat irritation may persist for hours or days. The National Library of Medicine warns about long-term immune sensitization, which means reacting badly to even tiny exposures far into the future.
Better Solutions Exist
Hospitals can’t just ban substances like glutaraldehyde overnight, but smart steps reduce the dangers. Good ventilation makes a huge difference. In one hospital I toured, adding local exhaust hoods reduced the sick days reported in the decontamination area from four a month to zero. Proper gloves make another big impact—nitrile stands up better than latex, according to published studies from OSHA. Switching to newer sterilizing agents, like hydrogen peroxide-based products, helps where possible. Training staff keeps everybody alert to leaks, spills, and improper handling. One simple improvement—posting clear signage about preparation and storage near sinks—helped a local clinic drop cases of dermatitis to nearly none.
Respect Chemistry—Don’t Get Burned
In the end, relying on glutaraldehyde may be common in clinical settings, but it isn’t something to take lightly. Talk to people cleaning endoscopes or working in dental labs, and you’ll hear stories that match the research findings. Red flags pop up for a reason. Staying aware, investing in protective gear, rotating tasks among workers, and reviewing chemicals used on-site regularly can protect health now and down the road. Those aren’t luxuries—they’re musts.
How is glutaraldehyde stored and disposed of?
Proper Storage: Keeping Risks at Bay
Glutaraldehyde works hard as a disinfectant and sterilizer in hospitals, labs, and various industries. Its strength makes it incredibly useful, but it also brings significant hazards. Anyone who spends time around this chemical knows it can burn the skin and irritate airways. Because of this, storage always demands serious attention.
Secure containers matter most. Glutaraldehyde usually comes in tightly sealed, clearly labeled plastic drums or bottles. Glass risks breakage and metal containers can corrode, so plastics made for chemical resistance make more sense. People who use the chemical day-to-day usually place storage units in dedicated, ventilated rooms far away from patient areas or locations with heavy traffic. There’s peace of mind knowing a spill will get controlled quickly if it happens. Locked cabinets add an extra layer of security, especially since children and untrained staff could accidently put themselves at risk.
Ventilation stands as a basic step toward keeping environments safe. A few times, I’ve seen coworkers underestimate glutaraldehyde’s fumes, learning firsthand how even small complacencies bring on coughing fits and eye irritation. Proper airflow, whether through exhaust fans or windows, can make a tremendous difference. Facilities often keep Material Safety Data Sheets nearby, so anyone can check procedures immediately if something goes wrong.
Disposal: Mistakes Have Long Consequences
Getting rid of old or spent glutaraldehyde isn’t as simple as dumping it down the drain. Waterways suffer from chemical contamination, harming fish and the communities who depend on clean rivers. Environmental protection groups and health authorities set firm rules to limit hazardous waste from entering pipes and landfills.
In most workplaces, staff collect used glutaraldehyde in dedicated containers—often labeled with hazard stickers plus usage logs. Medical settings sometimes neutralize glutaraldehyde with chemicals like glycine or sodium bisulfite before handing over the remnants to certified hazardous waste haulers. This double-check keeps both staff and disposal service workers safe.
Accredited companies transport hazardous waste to treatment centers equipped to break down or contain chemicals safely. Once, I watched a workshop team train new staff on spill kits and emergency protocols with mock drills. Agencies like the EPA and local water authorities regularly audit disposal practices, and fines can cripple smaller operators who cut corners. These checks matter because one carelessly handled drum can pollute soil for years.
Creating a Culture of Accountability
Responsible glutaraldehyde storage and disposal doesn’t grow out of bureaucracy; it grows out of firsthand experience and real harm. Hospitals and labs with strong safety cultures talk openly about exposures and near-misses. Supervisors reward good habits and make safety routines second nature. Fresh employees shadow more experienced ones and gradually earn extra responsibility. It helps new staff build confidence, and mistakes become rare.
As new, less hazardous disinfectants become available, some workplaces choose alternatives to glutaraldehyde altogether. But not every setting can switch so easily, especially when budget constraints or regulatory standards leave little room to experiment. Until better options arrive for everyone, careful storage and rigorous waste handling keep people healthy and water supplies clean.
Local guidelines, industry best practices, and honest conversation shape how people handle glutaraldehyde today. Anyone working with such chemicals needs both the facts and the wisdom of those who’ve spent years keeping themselves and their coworkers out of harm’s way.
What industries commonly use glutaraldehyde?
A Workhorse in Healthcare
Glutaraldehyde has carved out a reputation in the medical world. I first heard about it from a friend who worked as a surgical technologist. Hospitals and dental clinics lean on it for disinfecting medical equipment, especially when items like endoscopes or respiratory therapy gear can’t handle the high temperatures of steam sterilization. Even with new technologies in hospital disinfection, glutaraldehyde stays relevant because of its power against tough pathogens. For instance, studies published in the American Journal of Infection Control keep highlighting its broad antimicrobial action. The downside, and people in the field know this well, involves skin and respiratory irritation. Staff working frequently with glutaraldehyde often use special ventilation systems and personal protective gear. Hospitals could lower these risks by offering better staff training and safer alternatives, but as of now, glutaraldehyde’s survival says quite a bit about trust in its effectiveness.
Essential Role in Laboratories
Anyone who’s done any microscopic analysis or tissue studies recognizes glutaraldehyde. As a fixative, it locks cells in place so researchers can examine tissue under a microscope. Glutaraldehyde works quickly, preserving delicate cell structures that other chemicals often miss or destroy. High school and college biology labs, university research centers, and biotech firms all reach for this solution. I remember the pungent smell from my own time working in a cell biology lab; you get used to the fume, but it reminds you to handle it with care. The chemical also pops up in the production of diagnostic kits. That’s another reason why research and clinical diagnostics often invest in fume hoods and proper waste disposal.
Water Treatment and Environmental Management
Water treatment plants rely on glutaraldehyde’s killing power. The chemical helps control bacteria and algae in cooling towers and industrial water systems. In the oil and gas industry, companies inject glutaraldehyde into pipelines to stop bacterial slime and corrosion that damage equipment and slow down production. Workers operating these pipelines keep a close eye on exposure to avoid headaches or breathing issues. Years of practical use and field reports show that glutaraldehyde beats out many other biocides in effectiveness and stability, even in tough environments. Still, the push for green chemistry could lead to more eco-friendly biocides coming into play, but getting the same performance at a reasonable cost remains a challenge.
Leather and Textiles: Behind the Scenes
Factories treating leather use glutaraldehyde for tanning hides. Its chemical action helps strengthen and soften the leather at the same time. Some textile manufacturers also use it to finish fabrics, giving materials extra durability and reducing shrinkage. This kind of finishing touches everything from car seats to shoes and furniture upholstery. From my experience growing up in a town with a large tanning plant, residents knew the sour scent from miles away, but they also knew these plants meant steady jobs. Change happens slowly here; rules around waste disposal and air quality have become stricter, as they should, to keep the local environment healthy for people and wildlife.
Toward Safer Use and Alternatives
Balancing strong chemical results with worker health and environmental impact turns out to be complicated. Industry can’t just switch overnight to something new—it takes testing, investment, and a steady supply. Using better ventilation, protective equipment, and training all softens the risks right now. At the same time, funding research into safer alternatives gives workers and communities a better future. No easy answers exist, but real change grows from experience, smart oversight, and learning from every industry that uses glutaraldehyde, from city labs to country water plants.
| Names | |
| Preferred IUPAC name | pentanedial |
| Other names |
Glutaral Glutardialdehyde 1,5-Pentanedial Pentane-1,5-dial Cidex Sonacide |
| Pronunciation | /ɡluːˌtær.əlˈdeɪ.haɪd/ |
| Identifiers | |
| CAS Number | 111-30-8 |
| Beilstein Reference | 821805 |
| ChEBI | CHEBI:17718 |
| ChEMBL | CHEMBL25360 |
| ChemSpider | 7120 |
| DrugBank | DB00830 |
| ECHA InfoCard | 100.011.604 |
| EC Number | 203-856-5 |
| Gmelin Reference | Gmelin Reference: 82878 |
| KEGG | C02376 |
| MeSH | D005947 |
| PubChem CID | 3485 |
| RTECS number | MA2450000 |
| UNII | WDT7F5QKQA |
| UN number | UN3265 |
| Properties | |
| Chemical formula | C5H8O2 |
| Molar mass | 100.12 g/mol |
| Appearance | Colorless or pale yellow liquid with a pungent odor |
| Odor | Pungent |
| Density | 1.06 g/cm³ |
| Solubility in water | Miscible |
| log P | -0.37 |
| Vapor pressure | 2 mmHg (20°C) |
| Acidity (pKa) | 7.5 |
| Basicity (pKb) | 8.2 |
| Magnetic susceptibility (χ) | -7.5 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.424 |
| Viscosity | 16 mPa·s (25 °C) |
| Dipole moment | 5.49 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 203.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -504.50 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2431 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D08AX06 |
| Hazards | |
| Main hazards | Toxic by inhalation, ingestion, and skin absorption; causes burns; may cause allergic respiratory and skin reactions. |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS05,GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331, H314, H317, H334, H410 |
| Precautionary statements | P210, P261, P273, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P308+P313, P312, P314, P501 |
| NFPA 704 (fire diamond) | 2-2-0-A |
| Flash point | > 65 °C |
| Autoignition temperature | 210°C (410°F) |
| Explosive limits | Non-explosive |
| Lethal dose or concentration | LD50 oral rat: 134 mg/kg |
| LD50 (median dose) | LD50 (median dose): 134 mg/kg (oral, rat) |
| NIOSH | GN3500000 |
| PEL (Permissible) | 0.2 ppm |
| REL (Recommended) | 0.05% |
| IDLH (Immediate danger) | 40 ppm |
| Related compounds | |
| Related compounds |
Pentane-1,5-dial Glutaric acid 1,5-Pentanediol Succinic aldehyde Formaldehyde |

