Gallic Acid: Beyond the Lab Bench
Historical Development
Centuries before high-tech labs and stainless steel autoclaves, gallic acid already played a role in daily life. Oak galls, those growths on trees caused by insects, gave up their secrets to medieval tanners, scribes, and early chemists. Long before anyone talked about phenolics, communities used gall extracts for dyeing cloth and preparing iron-gall ink — that rich, dark liquid which helped shape the intellectual world from Renaissance manuscripts to constitutional documents. Chemists in the 18th and 19th centuries worked through various extraction and purification techniques, driven by this organic acid’s remarkable ability to bind with metals and colorants. By the early 20th century, scientists began piecing together gallic acid’s structure, tracing it back to the larger family of plant polyphenols, and setting the stage for serious research in pharmaceuticals and food science.
Product Overview
Everyday use of gallic acid looks pretty different now. Instead of scraping oak galls or boiling plant roots, chemical manufacturers typically offer gallic acid as a free-flowing white powder or fine crystals. While some batches originate from hydrolysis of tannins derived from Chinese gallnuts or Tara pods, others roll off lines after synthetic processes for greater purity or consistency. Laboratories and factories list a range of purities, often 98% or higher, catering to research, food, and pharma clients alike. Whether someone is developing an antioxidant blend or a new kind of ink, gallic acid emerges as a dependable core ingredient thanks to its well-characterized nature.
Physical & Chemical Properties
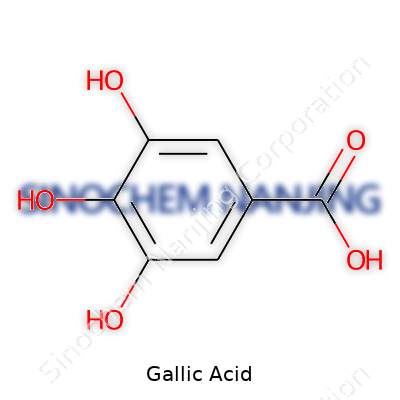
Handling gallic acid never leaves any mystery: it melts around 251°C, dissolves well in water and ethanol, and shows solubility quirks in ether and chloroform. The molecular formula C7H6O5 translates to three adjacent hydroxyls on a benzoic acid ring. Anyone working with it should expect a gentle tartness if accidentally tasted — though nobody recommends that — and a reputation for oxidizing easily, which explains its utility as both an antioxidant and a marker compound in several assays. Despite modern advances, storage still calls for dark bottles and tight lids to avoid gradual degradation from air and light.
Technical Specifications & Labeling
Each container arriving from reputable suppliers tells a clear story. Labels describe the lot number, purity (often backed by HPLC or UV-VIS analysis), appearance, and moisture content. You see storage recommendations — usually “keep cool, dry, and away from light.” Food-grade versions require more detailed batch records, allergen declarations, and in some cases, country of plant origin. For industrial buyers, technical data sheets walk through melting point, particle size, microbiological limits, and even recommendations for lab handling.
Preparation Method
Sourcing gallic acid mainly happens by breaking down tannins: a common route starts with plant material rich in gallotannins, like gallnuts or sumac. The material sits in hot water or dilute acid, releasing gallic acid over time. Technicians separate, filter, and crystallize the solution, often washing it further to boost purity. Large plants sometimes lean into chemical synthesis, pursuing more precise yields and regularity. Each step leaves a different environmental footprint, reminding buyers with sustainability in mind to ask about source and process.
Chemical Reactions & Modifications
Chemists appreciate gallic acid for its lively reactivity. Oxidation leads to quinones, used in inks and photographic developers. Methylation of the hydroxyl groups creates methyl gallate, a handy antioxidant in food and pharma. Esterification with various alcohols produces gallate esters, several of which stabilize fats and oils. The acid function allows easy coupling to other molecules — valuable for creating dyes, tanning agents, or polymers. In medicinal chemistry, one often sees this molecule as a foundation for more elaborate structures or conjugates.
Synonyms & Product Names
The world of chemical nomenclature delivers plenty of options. Some know it as 3,4,5-trihydroxybenzoic acid, others call it C.I. 36936, depending on context or regulatory focus. Trade names crop up among ingredient suppliers, particularly in food and nutraceutical markets, while historical texts simply reference “tannic acid” derivatives. It’s essential for buyers and researchers to double-check documentation — despite sharing a root name, tannic acid and gallic acid differ in formula and behavior.
Safety & Operational Standards
Safe handling marks the difference between a reliable workplace and avoidable accidents. Gallic acid doesn’t come across as hazardous in the dramatic sense, but anyone spending hours with kilos at a time should read the SDS carefully. With dust, mild irritation of skin or eyes sometimes develops. Gloves, goggles, and lab coats do the trick for most users. Industrial settings may install local exhaust to control airborne particulates. In food or pharma, batch testing screens for possible contaminants and residual solvents, lined up with regulations by the FDA, EFSA, and other authorities. Training remains essential, whether in academic labs or factory lines — complacency never pays off when even “mild” hazards exist.
Application Area
Gallic acid travels across industries with surprising ease. In food, its presence boosts antioxidant power, helping preserve oils and processed products. Beverage makers appreciate its ability to fortify red wines and teas, both naturally and via enrichment. Pharmaceutical researchers dig deep into its antimicrobial and anti-inflammatory attributes, testing gallic acid derivatives in formulations for wound care, digestive health, or cardiovascular support. Dye and ink producers find a reliable black or brown base that performs over decades. Analysts working in environmental sciences use gallic acid as a calibration standard or a probe molecule for free radical assays. Even photographers still mention it in discussions of classic developing agents — proof that tradition sometimes circles back into relevance.
Research & Development
Academic and commercial labs dig into gallic acid’s chemistry every year. Current output from medicinal chemistry journals features modifications that improve water solubility or target release in bioactive formulations. Food scientists catalogue its influence on shelf life and interaction with other plant-derived phenolics. Chemical engineers evaluate new ways to extract or synthesize the molecule, weighing greener solvents and energy savings against effectiveness. Collaborations between universities and industry speed up innovations, with patents describing everything from anti-cancer prodrugs to fuel cell additives. Everyone in this pipeline races to prove unique value, yet quality research hinges on robust, transparent experimental documentation and peer review.
Toxicity Research
Nobody wants a “natural” ingredient turning problematic at scale. Toxicology studies on gallic acid stretch back decades, with acute and chronic tests across several animal models. Results show low acute toxicity; issues only emerge at doses far above anything used in food or supplements. Metabolites pass quickly through the body, usually via urine. Scientists pay attention to genotoxicity and potential hormone-disrupting effects, although most respected agencies consider it safe for regulated uses. Periodic reappraisal remains smart, and manufacturers working in food or health stick to documented upper limits and rigorous batch analysis.
Future Prospects
Anyone betting on the future of plant polyphenols places gallic acid near the top of the list. Aging populations want better antioxidants in their diet; researchers keep testing new conjugates for cancer care and chronic inflammation. With green chemistry pushing sustainable synthesis, scientists seek routes from renewable sources that cut down on energy and hazardous solvents. Efforts in packaging and materials science examine gallic acid’s polymerization and cross-linking potential for compostable films and coatings. Tech watch lists pick up on its role in next-gen batteries and bio-based electronics, too. These broad, sometimes surprising uses make gallic acid a worthy topic for any R&D portfolio.
What are the health benefits of Gallic Acid?
What Gallic Acid Brings to the Table
Gallic acid exists naturally in many fruits, teas, grapes, and even oak bark. I first heard about it years ago from a nutrition-savvy friend who swore by herbal teas for chasing away seasonal colds. I dug in, and science backed her up. This simple plant compound has been grabbing researchers’ attention for good reason—its health benefits range far beyond basic nutrition.
Fighting Oxidative Stress
Oxidative stress takes its toll through pollution, processed food, sun damage, and even vigorous exercise. Cells need help to recover and defend against all that daily wear. Gallic acid works as a natural antioxidant. A 2020 review in the journal Antioxidants gathered strong evidence that this compound neutralizes free radicals—those unstable molecules that slowly damage our cells. Less cellular damage means lower risks for chronic conditions, from heart disease to certain cancers.
Anti-Inflammatory Power
Many chronic conditions take root in long-term inflammation. Joint pain, skin issues, even metabolic problems trace back to this. Lab studies show gallic acid tampers down inflammation by blocking chemicals like interleukin and TNF-alpha. One animal study in Phytotherapy Research showed reduced swelling and tissue redness, especially in connection to arthritis. For folks with nagging aches, the science behind herbal teas and gallic-rich berries starts to look pretty appealing.
Supporting Brain Health
Memory lapses and brain fog frustrate anyone who juggles work and family demands. Gallic acid catches attention for its potential to protect nerve cells. Researchers at the University of Queensland outlined this antioxidant’s effect on brain cells, noting slower progression of memory loss linked to Alzheimer’s models in animals. These findings match up with nations reporting lower rates of neurodegenerative diseases when their diets include foods high in plant polyphenols—including gallic acid.
Better Metabolic Function
Gallic acid supports healthier blood sugar levels. Regular berries, tea, and gallic-rich foods affect glucose absorption. In 2019, scientists publishing in Nutrition & Diabetes found less insulin resistance in lab animals given gallic acid extracts. This means less strain on our organs over time. Diabetes prevention barely makes the headlines anymore, but little tweaks—like including gallic-rich foods—actually make a difference.
Natural Defense Against Microbes
Modern life runs into antibiotic resistance too often. Many people turn to plant compounds for extra support. Gallic acid shows antimicrobial activity, especially against bacteria like E. coli and Staphylococcus, as seen in laboratory studies out of the International Journal of Food Microbiology. As someone who spent years working in restaurants, I know the importance of natural food safety. A healthy dose of gallic acid in everyday foods could strengthen defenses at a time when superbugs threaten public health.
Making It Work for Everyday Life
No single nutrient solves every problem, but building a diet around berry fruits, nutritious teas, walnuts, and even certain spices naturally increases gallic acid exposure. A lot of people ignore these options, thinking supplements are easier, but whole foods bring other synergy—fiber, minerals, broader antioxidants. Anyone wanting to try gallic acid-rich foods should stick to real, plant-based sources. If questions linger about drug interactions or serious conditions, it’s always worth checking with a healthcare provider.
Is Gallic Acid safe to consume?
Gallic Acid at a Glance
Gallic acid pops up in quite a few spots. Grapes, green tea, berries, even oak bark. Many people take an interest in it because scientists found strong antioxidant activity. Plenty of folks get exposure through diet. The thing is, naturally occurring gallic acid in fruits or teas comes in small amounts. Food manufacturers and supplement brands also started using isolated gallic acid as an additive—sometimes to boost antioxidant content, sometimes as a preservative.
What Does Research Tell Us About Gallic Acid's Safety?
I dove into studies and expert opinions over the years, and the consensus leans toward safety at common dietary levels. Gallic acid is not some obscure chemical being dumped into foods without oversight. The Joint FAO/WHO Expert Committee on Food Additives includes gallic acid in their reviews and reports that it doesn’t build up or linger in the body. Human bodies break it down and clear it, typically without fuss.
People have been eating foods with measurable amounts of gallic acid forever. Tea drinkers, berry fans, wine lovers—these are everyday sources, and broad population studies don’t flag gallic acid as a threat in this context. One study from the European Food Safety Authority (EFSA) looked at gallates (esters of gallic acid, commonly used as antioxidants), and after reviewing animal and human data, the experts set safe upper limits. Rare cases of allergy or intolerance showed up, especially involving additives, but nothing that raised large-scale red flags.
Where Real Caution Makes Sense
I’ve learned over time: most adverse effects from gallic acid crop up in concentrated, highly purified doses—not from eating fruit or drinking tea. Lab studies sometimes pump huge amounts into animals to hunt for toxic effects. Liver and kidney problems start to appear at those levels, way beyond what most people would encounter outside a research setting or aggressive supplement use.
People with certain health challenges, like chronic kidney issues or a history of gallbladder disease, probably ought to talk with their doctor before loading up on any antioxidant supplement, gallic acid included. The dose, the form, and underlying health bother me more than the source itself. In my experience, supplements advertised as “ultra-concentrated antioxidants” or “super detox boosters” tend to gloss over realistic safety discussions. Regulation on dietary supplements lags behind food ingredient oversight, and that leaves room for risk.
Quality, Transparency, and Keeping an Eye on the Details
One thing I always go back to: context matters. An orange or cup of green tea carries gallic acid in the company of fiber, vitamins, and other plant compounds. Supplements deliver higher doses more quickly, and often without the rest of the package. Brands should list exactly how much gallic acid they use and share their quality testing. Look for third-party certificates and responsible sourcing.
There’s a temptation to reach for the latest supplement, thinking more is better. With gallic acid, there’s little to fear from foods and beverages. Used as a common food preservative or antioxidant in regulated amounts, the safety record looks solid. Problems lurk with large, isolated doses and poorly labeled products.
The Bottom Line for Everyday Decisions
My recommendation? Lean on what’s proven—whole foods and balanced diets. Watch out for supplements promising big health changes. If you have a health issue or take many meds, check with a physician before adding highly concentrated antioxidants to your routine. Gallic acid isn’t a villain in the grocery store or the kitchen, but dialing up the dose with little information invites uncertainty.
What are the main uses of Gallic Acid in supplements or products?
Gallic Acid’s Real-World Appeal
Gallic acid keeps popping up on labels for dietary supplements, skincare products, and even some fancy teas. For years, I saw this ingredient and thought it must just be another antioxidant. After digging into research and talking to experts in nutrition and pharmacy, it’s clear that gallic acid’s value goes far deeper.
Naturally Present, Yet Widely Used
Some of the food and drinks I already loved—berries, green tea, wine—naturally contain gallic acid. Its presence isn’t accidental. Plants produce it to protect themselves from pests and harsh environments. That same protective effect interests researchers and nutritionists.
Supplements have harnessed gallic acid for its antioxidant strength. Oxidative stress—essentially damage caused to our cells from free radicals—has been linked to everything from aging skin to heart problems. Gallic acid has shown with repeated studies, including work published in journals like Phytotherapy Research, that it can neutralize these free radicals. People looking to lower risk from chronic inflammation and age-related health issues often seek out this compound.
More Than Just Antioxidant: Anti-inflammatory Benefits
Athletes and anyone with joint pain will spot gallic acid in some anti-inflammatory blends. My own experience with joint soreness led me to a nutritionist who recommended a supplement containing it. The logic’s simple—the compound blocks enzymes and pathways that ramp up inflammation, providing relief for stressed tissues.
Supporting Gut Health and Immunity
There’s something personal about gut health. I’ve been through rounds of antibiotics and the frustration of digestive imbalance. Scientists have found evidence that gallic acid can help reinforce the gut lining. A 2021 paper in Frontiers in Nutrition highlighted its role in supporting healthy gut bacteria and potentially reducing the risk of ulcers.
On the immunity front, preliminary animal studies suggest gallic acid might strengthen the body’s defenses. It can aid in regulating immune cell activity, important for fending off seasonal bugs.
Protecting Skin and Enhancing Cosmetics
Gallic acid shows up in face serums and creams. Anyone who struggles with redness or brown spots will find it in “brightening” formulas. Its anti-inflammatory power helps keep skin calmer, and the antioxidant effects limit sun and pollution damage. Dermatologists sometimes point to it for people dealing with acne or rosacea, since it soothes irritation and supports skin repair.
Challenges and Safe Use
More products include gallic acid each year, yet there’s a gap between what lab results show and how much actually reaches our tissues after swallowing a capsule or applying a cream. Some supplement brands have started to improve their delivery methods, such as combining gallic acid with other plant extracts, to help the body absorb it better. Following serving suggestions, and consulting with a healthcare professional, stays important—especially for anyone with allergies or chronic conditions.
Practical Steps for Informed Choices
Looking for trustworthy labels, transparency around sourcing, and reviewing third-party testing reports is key. Most folks can safely add gallic acid to their routine through food, but supplements give a measured dose for people with specific concerns. Keeping up with new research helps courses of action adapt, whether for personal health or in recommending products to others.
Are there any side effects associated with Gallic Acid?
What People Want to Know About Gallic Acid
Gallic acid attracts a lot of attention because it's found in plants, teas, berries, and even wine. People turn to it for its antioxidant punch, hoping to sidestep cell damage and inflammation. Research teams have explored its benefits for decades. Still, before jumping on the supplement bandwagon, folks ask whether gallic acid brings side effects or risks.
C hemical Doesn't Mean Harmless
Natural doesn’t always mean safe. Gallic acid belongs to the polyphenol family and carries some proven biological activities. In lab dishes and animal studies, it fights free radicals and slows certain types of inflammation. That sounds promising, but personal experience and years of working in health communication taught me to read beyond the feel-good headlines. Plenty of compounds work in the lab but act differently in real life and across unique bodies.
What Researchers Say About Side Effects
Most studies on gallic acid center on moderate food-based intake. In these amounts, actual side effects rarely show up. Foods like blueberries, nuts, and black tea all supply small, steady doses. People eating a balanced diet almost never experience problems from these sources.
Trouble appears when folks take concentrated gallic acid as a supplement or use extracts far beyond the amounts nature provides. In higher doses, researchers noted mild stomach upset, nausea, or headaches. Sometimes people feel tired or dizzy. In extreme animal tests, huge doses damaged kidneys and liver tissues. Now, nobody eats pure gallic acid by the spoonful, but stories of accidental high intake show that too much can create harm.
Individual Differences Matter
No two people react the same. Someone with a sensitive gut could feel queasy after even low doses. Allergies to related compounds may increase the chances of unwanted reactions. I once tried an herbal blend with high amounts of fruit extracts; within hours, I felt bloated and uneasy. From that day, I became skeptical of mega-dose plant supplements. What works for one person sometimes causes discomfort for another. For many, less really is more.
Supplements and Drug Interactions
Mixing gallic acid supplements with prescription medications carries real risks. Polyphenols can alter how the body processes certain drugs, especially those broken down in the liver. For example, anyone using blood thinners or anti-inflammatory drugs should approach with caution. One study published in the journal Food and Chemical Toxicology raised concerns about gallic acid’s potential to interfere with drug-metabolizing enzymes.
Safer Steps Before Supplementing
Doctors always want to know about new supplements, especially if you already take any daily medications. While most folks can eat foods with gallic acid safely, extra supplements should always be discussed with a licensed healthcare provider. Never trust claims promising miracle cures, whether the bottle shows berries or boasts about antioxidants.
Focusing on Balanced Choices
Gallic acid, like so many nutrients, works best through real food. Folks who eat plenty of fruits, vegetables, nuts, and teas get all the benefits without diving into unknown territory. Simple, steady variety in the diet protects cells, tames inflammation, and cuts the need for risky experiments with concentrated powders or pills. From my years leading community nutrition classes, I saw firsthand how switching to whole foods brings steady improvement, while quick fixes invite new complications.
How should Gallic Acid be stored to maintain its potency?
Potency and Everyday Lab Lessons
In research labs, a chemical’s true value often fades because of simple missteps in storage. Gallic acid looks mild, but its antioxidant punch can deflate fast outside the right conditions. I’ve seen new arrivals, fresh and white, turn tan and lumpy after just a few weeks on a cluttered bench. A couple of common precautions go a long way to keep this compound working the way you need.
Enemies: Light, Air, Moisture, and Heat
Gallic acid doesn’t handle light or air very well, and humidity makes things worse. Exposure means slow breakdown, like fruit left out too long. A bottle left open by accident, or poor seals, lets in moisture from the air. Lab humidity often creeps above 50%—that’s bad news for any phenolic acid. Protecting against these threats isn’t hard, but carelessness sneaks in with busy schedules.
Sealed and Shielded
Best practice calls for an amber glass bottle, or an opaque plastic container that keeps light out. Never use clear glass under fluorescent bulbs. Once open, screw that cap back fast and tight after every use. I’ve learned to watch for caked powder by the bottle neck; that’s a sign moisture found its way in. Sometimes, desiccant packs dropped inside the container can absorb excess water in the air, but these only help so much if people leave caps loose.
Storage Spots: Fridge or Freezer?
Many suppliers recommend refrigeration, holding gallic acid between 2°C and 8°C. That works for small-quantity bottles used regularly. If you’re dealing with larger batches or storing long-term, it pays to go lower—many experienced chemists store bulk gallic acid in -20°C freezers. With both fridges and freezers, condensation can build up after repeated temperature changes. Taking out a cold bottle, letting it sit open, and then returning it adds moisture that can quietly ruin the powder. Some labs use vacuum-sealed pouches or store bottles inside airtight secondary containers to cut this risk.
Quality Checking—Don’t Skip It
Before making a large solution, always check the powder’s color and texture. Faded pigment or clumping usually means trouble with moisture or oxidation. In school, I’d cut corners and skip these checks; wasted hours on failed reactions taught me otherwise. Even with good storage, quality can slip after months. A simple test or a quick sample scan with an IR spectrometer reveals breakdown before it ruins an experiment or product.
Communication: The Overlooked Factor
People are the wildcard. Good containers, clear labels, and a reliable routine only go so far if the rules aren’t shared. In my own work, posting a straightforward sign—date opened, storage temp, reminder to reseal—cuts down on slip-ups. Experienced techs set the tone for newcomers. One-off reminders from colleagues sometimes save months’ worth of stock from being lost. Building these habits, and holding each other to them, has stronger impact than the fanciest storage setup.
Solutions Rooted in Daily Practice
Keeping gallic acid potent comes from respect for its chemistry and a commitment to routine. Light, heat, air, and water all gnaw at sensitive powders. Store it cool and dry, avoid clear containers, never leave the lid loose, and get everyone on board. Labs that get these basics right end up with less waste, more reliable results, and more time to focus on the questions that really matter.
| Names | |
| Preferred IUPAC name | 3,4,5-Trihydroxybenzoic acid |
| Other names |
3,4,5-Trihydroxybenzoic acid Gallicin Pyrogallic acid-5-carboxylic acid |
| Pronunciation | /ˈɡæl.ɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 149-91-7 |
| Beilstein Reference | 359953 |
| ChEBI | CHEBI:30772 |
| ChEMBL | CHEMBL283 |
| ChemSpider | 715 |
| DrugBank | DB04272 |
| ECHA InfoCard | ECHA InfoCard: 100.003.150 |
| EC Number | 3.1.1.11 |
| Gmelin Reference | 4768 |
| KEGG | C00131 |
| MeSH | D005698 |
| PubChem CID | 370 |
| RTECS number | MC5250000 |
| UNII | 9G2MP84A8W |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C7H6O5 |
| Molar mass | 170.12 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 1.7 g/cm³ |
| Solubility in water | 11.9 g/L (20 °C) |
| log P | 0.7 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 4.4 |
| Basicity (pKb) | pKb = 10.37 |
| Refractive index (nD) | 1.554 |
| Dipole moment | 1.83 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 186.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1170.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1241.0 kJ/mol |
| Pharmacology | |
| ATC code | A01AB12 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation |
| GHS labelling | GHS07, GHS Hazard Statements: H302, H319 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | Precautionary statements: "P261, P305+P351+P338 |
| NFPA 704 (fire diamond) | 1-1-0-W |
| Autoignition temperature | 425 °C |
| Lethal dose or concentration | LD50 (oral, rat): 5,000 mg/kg |
| LD50 (median dose) | LD50 (median dose) of Gallic Acid: "5,000 mg/kg (oral, rat) |
| NIOSH | VW5690000 |
| PEL (Permissible) | PEL (Permissible) for Gallic Acid: Not established |
| REL (Recommended) | 300 mg |
| IDLH (Immediate danger) | No IDLH established. |
| Related compounds | |
| Related compounds |
Ellagic acid Pyrogallol Tannic acid Methyl gallate Propyl gallate Ethyl gallate |

