Furoyl Chloride: More Than Just a Building Block in Chemistry
Looking Back: The Story Behind Furoyl Chloride
Furoyl chloride's journey began in the middle of the 20th century when chemists started digging deeper into furan derivatives. The furan ring itself, discovered back in the 1800s, intrigued researchers thanks to its aromatic nature and reactivity. Before industrialists recognized the broader value of functionalized furans, these compounds floated mainly through academic research. The push for new pharmaceuticals, advanced agrochemicals, and unique flavors pushed furoyl chloride from the sidelines into specialized labs and then into routine chemical catalogs. Remembering how hard it was to source reliable intermediates during my own early research days, with only a handful of suppliers offering questionable purity, it’s clear how much progress depended on consistent access to this reactive chloride.
Getting Acquainted: What Furoyl Chloride Brings to the Table
This chemical isn’t something you find on a grocery store shelf. Used mostly as an acylating agent, furoyl chloride introduces a furan ring—a five-membered oxygen-containing ring—into many molecular frameworks. In industry, the draw comes from the versatility the chloride group offers for making more complex compounds. Furoyl chloride typically shows up as a colorless to pale yellow liquid, with a sharp, pungent odor that lingers even after quick handling. That easily recognizable smell brings back memories of hours spent ventilating workspaces to keep the air breathable during acylation experiments.
Digging Into Physical & Chemical Nuances
One of the most striking features lies in the balance of its boiling point and its moisture sensitivity. The chlorine attached to the furan ring makes it an acyl halide that reacts quickly with water, releasing fumes of hydrogen chloride in the process. Its good solubility in many organic solvents, paired with a tendency to decompose if moisture even thinks about getting close, creates challenges and forces chemists to stay sharp about preparations and handling. That reactivity means you can convert all sorts of starting materials into furoyl-based products, but it also means you need well-calibrated glassware and an immune sense of caution.
What’s Written on the Bottle: Technical Specs & Labeling
The label on a furoyl chloride container usually doesn’t mince words. Anyone pouring from a bottle marked "furoyl chloride" knows what’s coming. Safety phrases—wear gloves, avoid inhaling vapors, don’t mix with water—crowd the label with good reason. You usually find details about purity (often over 98 percent from trusted suppliers), typical weights or volumes, and other handling cautions. Back in grad school, there was a four-inch-thick binder documenting every incoming bottle. Furoyl chloride, with its sharp smell and unmistakable effect on mucous membranes, stood out as a constant topic at safety meetings.
A Closer Look at Preparation Methods
Preparation almost always begins with furoic acid, taken from oxidation processes applied to furfural, a chemical sourced from agricultural biomass. Thionyl chloride or phosphorus pentachloride then reacts with furoic acid, replacing the carboxylic group with a far more reactive chloride. As someone who has tried old-fashioned syntheses, the preparation takes both patience and a steady hand: too much heat or a touch of humidity and your yield plummets or side-products take over. Watching a reaction flask bubble aggressively brings home the lesson about venting and temperature control. This isn’t a procedure for careless hands—the slightest misjudgment predicts a ruined batch or worse, a minor lab disaster.
What Happens in the Flask: Chemical Reactions & Tweaks
Furoyl chloride doesn’t like to sit idle. Expose it to alcohols, amines, or water, and acylation or hydrolysis follows almost immediately. Synthetic chemists treat it as a gateway to more elaborate furan-based compounds. Benzyl amines, aniline derivatives, and many other nucleophiles snap up the furoyl group, turning out a string of pharmaceuticals, dyes, and small-molecule probes. It’s been eye-opening to watch experienced hands convert this sharp-smelling liquid into a pharmaceutical precursor with nothing more than basic glassware. Whenever a target required a furan ring for biological activity or stability, this reagent sat front and center on the synthetic bench. Minor modifications around the chloride group also let researchers tweak reactivity or solubility—one more step toward a more workable drug lead or new agrochemical.
Aliases in the World of Chemistry: Synonyms & Trade Names
Over the years, furoyl chloride has picked up a few names—2-furoyl chloride and furan-2-carbonyl chloride pop up most often. In international catalogs, translations or abbreviations sometimes confuse newcomers, but anyone with a little bench experience learns the synonyms quickly. The chemical structure gives away the story: a furan ring with a carbonyl chloride attached. Keeping these aliases clear helps dodge order mix-ups, which every lab has experienced at least once with similar-sounding names.
Playing It Safe: Standards for Handling & Hazard Management
Handling furoyl chloride puts both skill and responsibility on the chemist’s shoulders. Lab coats, tight-fitting goggles, and gloves rated for acid chlorides form the daily uniform. The vapors, heavier than air and irritating to eyes and lungs, demand serious ventilation. During an internship, I once watched a less cautious colleague forgo the fume hood and nearly clear out the entire lab in the process. Repeated dry runs and safety drills made it clear that short-cuts and furoyl chloride never mix. Waste needs alkaline neutralization before disposal, with acid-base indicators and triple-layered containers to prevent leaks or accidental exposure. Anyone who has reached for an emergency eyewash after a fume caught their eyes knows exactly how humbling one slip can feel.
Where It Ends Up: Real-World Applications
Most chemists meet furoyl chloride through its role as a starting material for specialty drugs and fine chemicals. Respiratory medications, anti-inflammatory agents, and certain crop protection products all trace back, sometimes indirectly, to this sharp-smelling compound. The furan core imparts metabolic traits and unique electronic profiles, which medicinal chemists often chase for new activity. Fine fragrance manufacturers, although less publicly, have also dabbled with furoyl derivatives for sweet, earthy notes. Every new paper or patent highlights a slightly different downstream use; visiting trade shows often turns up some as-yet-unreported application bubbling up in a pilot plant or research department.
Staying Ahead: Recent Trends in Research & Development
Much of the current buzz comes from greener preparation methods and more sustainable supply chains. Researchers aim to reduce reliance on harsh chlorinating agents by leveraging electrochemical routes, biotransformations, or less hazardous reagents. Publications cite dozens of new drugs under preclinical review featuring furoyl groups—often in search of better stability or efficacy against tough targets like resistant bacteria or certain cancers. When I catch up with old labmates, the talk often turns to how automated reactors or microfluidic setups can cut waste, contain corrosive vapors, and still produce high yields of these reactive intermediates. With stricter regulations on chemical hazards worldwide, everyone feels the heat to innovate not just the end products but the entire process lifecycle.
Looking Danger in the Eye: Toxicity Research in Perspective
No honest chemist shrugs off health risks around acid chlorides, and furoyl chloride sits on the list for good reason. Direct contact warns instantly, burning the skin within seconds and inflaming mucous membranes at only trace levels. Toxicologists keep tracking the compound’s effect on animal models, noting both acute effects from short exposure and longer-term risks with chronic handling. Recent studies underscore the importance of using sealed apparatus and remote sampling whenever possible, especially as oversight agencies flag older practices for update or overhaul. Safe substitution with less reactive analogs remains limited; the unique chemistry of the furoyl group isn’t always replaceable. For chemists pushed to the edge of discovery, safety studies guide not only policy but the everyday workflow that everyone in the lab grows accustomed to with time.
What’s Next: The Road Ahead for Furoyl Chloride
With green chemistry driving so much of today’s innovation, furoyl chloride sits at a crossroads. Demand keeps ticking up for new flavors, drugs, and specialty materials built on furan scaffolds. To balance progress with responsibility, chemists aim to modernize old batch processes, swap in renewable starting materials, and create less hazardous waste streams. Startups and established players alike invest in new reactor technologies capable of precise control, containment, and real-time monitoring to make working with reactive intermediates safer and more predictable. Over the years, it’s become clear that, while some risks are inherent, most incidents trace back to gaps in training, equipment, or respect for the chemical in question. The future asks for tight-knit partnerships among researchers, regulators, and suppliers, with ongoing education and process upgrades to keep this vital building block accessible, safe, and sustainable. As research pushes into new therapeutic frontiers, furoyl chloride looks set to keep playing a core role—provided that those handling it blend curiosity with care and a clear-eyed view of the hazards and opportunities ahead.
What is Furoyl Chloride used for?
What Is Furoyl Chloride Doing in the Lab?
Furoyl chloride doesn’t show up on household ingredient lists. It usually pops up in chemical labs, pharmaceutical plants, and specialty manufacturing facilities. With a crisp, reactive nature, this chemical has made itself valuable in the world of organic chemistry. Scientists count on it when they want to push science forward and create molecules with a punch. That reactivity lets experts build complex things out of simple building blocks—things like medicines, dyes, and even advanced materials that keep the world moving.
Building Blocks for Big Discoveries
Its claim to fame: furoyl chloride reacts quickly and cleanly with other chemicals to give rise to new compounds. Researchers spend hours in labs trying to come up with something new, and this chemical gives them the flexibility they need to attach a furan ring—an important scaffold found in plenty of bioactive molecules. Some of those molecules turn into pharmaceuticals. Others become agrochemicals or dyes with vibrant colors. The core reason for using this chemical isn’t just that it’s efficient. It’s because it opens the door for discoveries that wouldn’t be possible otherwise.
Making Medicines and More
I’ve chatted with scientists excited about a new synthesis route or a simple solution for getting stubborn chemical groups to react. In the world of drug development, every shortcut has value. Furoyl chloride offers that shortcut in certain situations. Pharmaceutical creators pick it up for making antibiotics, antiviral drugs, and other medications where a furan group is key. Getting the structure right is half the battle, and with a reactive acyl chloride in the toolkit, chemists can build molecules with real therapeutic potential.
Beyond that, the specialty chemical industry calls for furoyl chloride in custom syntheses, making fine chemicals, and crafting compounds destined for everything from new materials to pigments found in paints and plastics. Versatility counts, and this stuff has it.
Handling Risk, Chasing Progress
There’s a flip side. Furoyl chloride is as hazardous as it is useful. It doesn’t play nicely with water, lets off corrosive fumes, and gives lab staff a reason to suit up with gloves and goggles. That extra caution often turns into obstacles for smaller operations, or for researchers who lack access to specialty equipment.
People who work around this chemical need strong training and reliable safety protocols. In my experience, protecting staff isn't just a checkbox on a compliance form—it shapes the daily rhythm of the lab. It can slow progress or make experiments more expensive, but any shortcut risks serious injury or worse. Labs with older equipment or those in low-resource settings sometimes struggle to strike a balance between safety and productivity. Investing in fume hoods and proper chemical storage should be a non-negotiable part of any project. Sharing safety know-how between labs or even across borders can stop accidents before they happen.
Spotting Surprises and Supporting Progress
Furoyl chloride won’t end up in news headlines. Yet, it helps push boundaries in pharmaceuticals, vibrant plastics, and specialty chemicals. Every bottle in the lab carries the promise of something novel—mixed with the responsibility to handle it right. If companies and research teams want the benefits, they have to give safety and environmental protection their due. That’s the deal with progress: embrace the risk, but don’t ignore it. Even the most innovative chemicals need smart stewards to avoid leaving a mess behind.
What is the chemical formula of Furoyl Chloride?
Furoyl Chloride: A Closer Look
Furoyl chloride often pops up in advanced chemical research and pharmaceutical labs. It’s got the formula C5H3OCl. The molecular skeleton builds off the furan ring, which is a five-membered structure made of four carbons and an oxygen atom. Furoyl chloride takes that ring and tacks on a carbonyl group attached to a chlorine atom, turning it into an acid chloride derivative. The structure gets plenty of attention because it’s a member of the heterocyclic aromatic group. Chemists value these structures for their reactivity and versatility.
Why C5H3OCl Matters in Real Labs
In my own experience working with organic reagents, having the right building block saves a lot of headaches. Furoyl chloride is one of those go-to reagents that helps chemists modify molecules in a hurry. In pharmaceuticals, it’s a frequent pick when making compounds with antifungal, antibacterial, or anti-inflammatory potential. Its ability to introduce the furoyl group plays a central role, paving the way for new drug candidates that target stubborn conditions.
If you browse through chemical catalogs from reputable suppliers, you will spot furoyl chloride listed for advanced synthesis. Laboratories depend on its unique chemistry to create molecules that might otherwise take months to build. Its reactivity speeds up the formation of amides, esters, and other valuable compounds.
Making and Handling Furoyl Chloride
Creating furoyl chloride starts with furoic acid. By treating furoic acid with reagents such as thionyl chloride or phosphorus trichloride, chemists coax the replacement of the hydroxyl group with a chlorine atom. This reaction isn’t something to take lightly. Acid chlorides like furoyl chloride will react fiercely—contact with water leads to splashes and fumes, so most chemists handle it under anhydrous, inert conditions. Proper personal protective equipment like goggles and gloves makes a world of difference. I’ve seen new chemists ignore the wisdom of old hands and quickly regret it once the fumes seep out.
The synthesis itself speaks to the evolution of chemical safety. Decades ago, accidents involving corrosive reagents were more common. Today’s safety data sheets spell out just how reactive furoyl chloride can be. It’s good practice to store these bottles in tightly sealed containers, well away from moisture, and in an area with adequate ventilation.
Furoyl Chloride’s Role in Innovation and Solutions for Safer Use
The rush to build new pharmaceuticals and advanced materials pushes chemists to invent new ways to work with reagents like furoyl chloride. Automated synthesis setups and improved fume hoods let researchers push boundaries while reducing risk. Some labs have even shifted toward greener chemistry approaches, swapping out traditional hazardous reagents for less toxic alternatives where possible.
Education sets the foundation. Making sure the next wave of scientists can handle compounds like furoyl chloride safely will keep innovation moving. Training on real-world hazards and knowing how a single chlorine atom changes a molecule’s character helps bring students out of the textbook and into discovery. The urge to invent, tempered with a grounded respect for reactivity, will shape how future chemists unlock the secrets inside every bottle labeled with C5H3OCl.
How should Furoyl Chloride be stored?
Why Storage Matters for Furoyl Chloride
Furoyl chloride plays a role in specialty chemistry, turning up in pharmaceuticals and some advanced materials. One thing stands out: it reacts quickly with moisture and doesn’t take kindly to sloppy storage. I’ve worked around reactive chemicals enough to see how little mistakes become big emergencies. There’s no cutting corners with this one—you stay organized and aware, or pay the price.
Keep the Moisture Out
Furoyl chloride reacts with water in the air, producing corrosive gases and nasty byproducts. Leaving the bottle on a bench for “just a minute” can cause trouble. An airtight container does the job, especially if you’re using glass. Keep the lid on tight, or you might come back to a corroded cap, damaged shelf, or worse.
In my labs, we stored these types of reagents with desiccants—those little packets of silica gel absorb any wayward moisture. Sometimes, special cabinets with dry atmosphere work even better, especially when humidity swings with the weather. Store the container inside a secondary bin in case of leaks: that way, one slip doesn’t flood the whole storage space.
Control the Temperature
This compound starts to decompose if it heats up. Direct sunlight speeds up that process. If you leave a bottle near a sunny window or near a radiator, gas pressure can build inside. The safest spot is a cool, shaded part of the chemical storage room. Aim for temperatures below room temperature, but don’t freeze it—freezing glass bottles brings its own risks.
We used insulated cabinets, and, for large quantities in industrial settings, sometimes cold rooms. Users working at scale have to keep the space well-ventilated, so fumes don’t build up undetected.
Avoid Mixing with Incompatible Materials
Furoyl chloride doesn’t play well with water, alcohols, amines, or strong bases. Make sure shelves are organized—don’t pile everything together just to save space. One splash, and you could have an uncontrolled reaction. Arrange storage areas with clear labels, even in small research settings. In my experience, the difference between a close call and a disaster is clear protocols.
Labeling and Security
Always mark each container clearly. Unlabeled jars often end up forgotten, and discovering one weeks later is a headache nobody wants. In companies where turnovers are common, strong labeling habits save everyone time and nerves. Controlled access matters for substances with serious risks. Only trained staff should get near this reagent.
Keep an inventory and stick to it religiously. Regular checks catch leaks or damage before they spiral. Use spill kits and have emergency showers within reach. If you end up handling a large amount, get input from a safety officer or chemist who’s managed similar hazards before; their advice is worth its weight in gold.
Safer Practices Make a Big Difference
Shortcuts backfire with chemicals like furoyl chloride. Getting storage right keeps colleagues and property safe. Invest in good containers, robust labeling, and dry, cool, separated storage. Most accidents I’ve seen start with someone saying “just for now.” A solid storage routine keeps those words from costing too much.
Is Furoyl Chloride hazardous or toxic?
Why Safety Matters With Chemicals
Having spent years reading chemical labels in busy research labs, one thing quickly becomes clear: ignoring hazard symbols invites trouble. Furoyl chloride isn’t something you’d find under the kitchen sink. It’s a chemical used in specialized applications, mostly by trained chemists or manufacturers making drugs or advanced materials. Of every bottle behind a locked chemical cabinet, this one often sports the kind of warning labels that demand respect.
What Makes Furoyl Chloride Dangerous?
Furoyl chloride packs a punch because it reacts fiercely with water and moisture—including the moisture in your eyes and throat. The reaction produces hydrochloric acid fumes, which sting the nose and send anyone running from the room. Inhaling even a small amount burns. Splash this stuff on your skin and you get a chemical burn that sticks around as an ugly blister. Get a sniff and your lungs start aching. All of that happens fast, without much warning.
The Centers for Disease Control and Prevention, along with organizations like OSHA, flag this compound as hazardous. Acute health impacts include severe eye, skin, and respiratory irritation. Long-term exposure can lead to slowly worsening lung conditions or sensitive skin areas that break out with much less exposure later on. Some sources note the lack of enough human toxicity data to be certain about cancer risk, but the immediate risk is already enough for careful handling.
Why Some Risks Go Unnoticed
Many people don’t consider the route of exposure. Furoyl chloride’s dangers multiply in poorly ventilated spaces. I’ve read accident reports where workers ignored a few precaution steps, leading to avoidable injuries. Chemical goggles, gloves, even a full fume hood belong in the standard gear for handling it. Sometimes, factory workers or casual lab assistants handle containers out of habit, forgetting how easy it is to splash or spill. It only takes one mistake, and the consequences last for a long time.
What Should Change?
The technology already exists to track and store dangerous chemicals more safely. Automated inventories, access logs, and smart cabinets reduce who can get their hands on these substances. Yet accidents still happen because of pressure to work fast or lack of training. Culture matters more than rules. Supervisors can show by example—no shortcuts, no exceptions, and no substitutions for real safety gear.
Looking at published research, companies with excellent training programs keep incident rates low. Government regulations set the baseline, but real safety comes from habits. Labs that drill emergency procedures and run “what-if” safety scenarios build muscle-memory responses. Having a clear plan for every stage—from delivery to disposal—cuts the risk dramatically. Communities living near industrial plants benefit from transparency and preparedness too. If something leaks or spills, everyone needs to know what to do, quickly.
Handling and Disposal: No Room for Half Measures
Disposal causes a final headache. Pouring furoyl chloride down the drain, flushing it without neutralization, or skipping containment turns one local hazard into community risk. Trained professionals pack these materials away for incineration or controlled chemical destruction. Regulations vary regionally, but most call for rigid cradle-to-grave tracking—a smart move that keeps old mistakes from resurfacing as future health problems.
Few people interact with furoyl chloride daily, and most shouldn’t have to. For those who do, a healthy dose of caution protects everyone. Modern workplaces have the tools for safe handling. The next step is making careful handling the default, not the exception.
What are the handling precautions for Furoyl Chloride?
Understanding Furoyl Chloride
Furoyl chloride draws attention in laboratory and industrial settings, mainly for its usefulness as a building block in organic synthesis. The compound stands out because of its aggressive reactivity, especially with water, alcohols, and amines. Yet, the risks tied to its handling leave no room for carelessness. People not only working in chemistry labs but also those in manufacturing plants have learned that working with furoyl chloride can turn a routine day into an emergency if basic precautions get ignored.
Health Hazards and Immediate Risks
Furoyl chloride doesn’t show much mercy on skin or in airways. With direct skin contact, it eats away at tissue, causing burns and lasting scars. Eyes exposed to even a drop run a high risk of permanent damage. Inhalation delivers a burning sensation, cough, chest pain, and, in high doses, breathing trouble. I once saw how a tiny spill in a fume hood created enough fumes to make nearby workers uncomfortable, even though the hood worked properly—this stuck with me as a lesson in vigilance.
Practical Steps for Safe Handling
The best line of defense with furoyl chloride starts with using strong engineering controls. Handling belongs inside a well-ventilated fume hood. Ordinary exhaust fans won’t cut it. Personal protective equipment saves skin and lungs from disaster: splash-resistant goggles, chemical-resistant gloves—typically nitrile or butyl rubber—and a properly fitted lab coat keep accidents from becoming injuries. For those who work with the chemical for any length of time, a face shield and chemical-resistant apron add a smart layer of protection.
Gloves deserve special mention. Ordinary latex gloves give a false sense of safety—they break down fast under organochlorine exposure. I’ve seen colleagues assume all gloves protect equally and regret it with serious skin irritation.
Storage Takes Planning
Furoyl chloride doesn’t mix well with moisture, air, or heat. Keep it in tightly sealed glass containers, never in plastic that may degrade. Store in a cool, dry, and well-ventilated space, away from water sources and out of direct sunlight. Mark every container clearly and lock up storage areas—nobody benefits from an accidental exposure by those not aware of the contents.
Emergency Response and Disposal
Spills call for fast thinking. Only those trained and equipped with full-face respirators and chemical suits should attempt cleanup. Neutralization with sodium bicarbonate works, but reactions become violent if dumped in water. Keep spill control material on hand and rehearse emergency plans—unexpected events move fast, and muscle memory helps stop a mess from becoming a headline. In waste disposal, collect all contaminated materials in sealed, labeled containers and coordinate with certified hazardous waste contractors, no shortcuts or “down the drain” solutions.
Why Vigilance Matters
A few years in laboratory work convinced me that preparation beats luck every time. Reliable safety habits, up-to-date training, and respect for chemical hazards protect people and property. By sticking to well-tested safety routines—never skipping the small stuff—accidents with furoyl chloride remain rare. As chemical technologies push forward, those working on the frontlines face chemicals that demand both respect and knowledge. Each time someone suits up to handle furoyl chloride, they help keep their team and community safer.
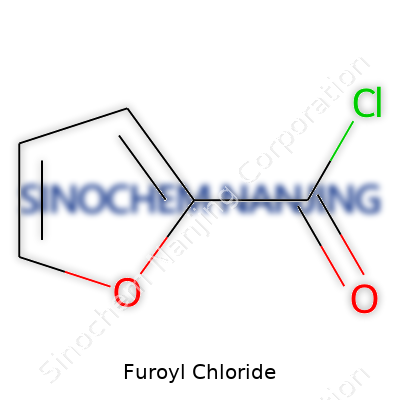
| Names | |
| Preferred IUPAC name | Furan-2-carbonyl chloride |
| Other names |
Furan-2-carbonyl chloride 2-Furoyl chloride Furancarbonyl chloride |
| Pronunciation | /ˈfjʊə.rɔɪl ˈklɔː.raɪd/ |
| Identifiers | |
| CAS Number | 557-20-0 |
| Beilstein Reference | 1207937 |
| ChEBI | CHEBI:51802 |
| ChEMBL | CHEMBL1379 |
| ChemSpider | 14917 |
| DrugBank | DB14004 |
| ECHA InfoCard | 100.007.246 |
| EC Number | 208-789-9 |
| Gmelin Reference | 79246 |
| KEGG | C14316 |
| MeSH | C010804 |
| PubChem CID | 12153 |
| RTECS number | LU5950000 |
| UNII | 5YU9287K2K |
| UN number | UN 3265 |
| CompTox Dashboard (EPA) | `DTXSID2021928` |
| Properties | |
| Chemical formula | C5H3ClO2 |
| Molar mass | 184.57 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Pungent |
| Density | 1.338 g/mL at 25 °C |
| Solubility in water | Reacts violently |
| log P | 0.83 |
| Vapor pressure | 1 mmHg (20 °C) |
| Acidity (pKa) | 1.1 |
| Magnetic susceptibility (χ) | -53.0e-6 cm³/mol |
| Refractive index (nD) | 1.558 |
| Viscosity | 1.62 mPa·s (25 °C) |
| Dipole moment | 2.85 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -322.6 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H302, H314, H317, H334, H335 |
| Precautionary statements | P260, P261, P264, P271, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P363, P405, P501 |
| NFPA 704 (fire diamond) | 1-3-0-W |
| Flash point | 52°C |
| Autoignition temperature | 515°C |
| Lethal dose or concentration | LD50 oral rat 1126 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1330 mg/kg (rat, oral) |
| NIOSH | MW9625000 |
| PEL (Permissible) | PEL for Furoyl Chloride: Not established |
| REL (Recommended) | 0.99 |
| IDLH (Immediate danger) | IDLH: 2 ppm |
| Related compounds | |
| Related compounds |
Benzoyl chloride Thiophosgene Furoyl fluoride Acetyl chloride Furoic acid |

