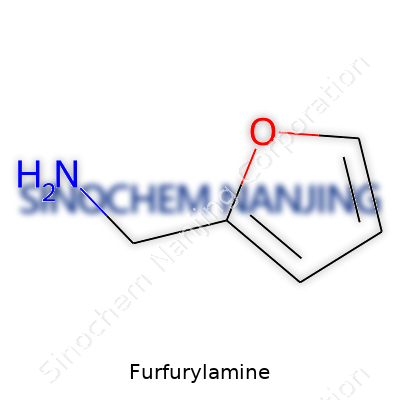
Furfurylamine: An Editorial Commentary
Historical Development
Stories of chemistry often put the spotlight on big names and big discoveries, but behind the scenes, there’s a quieter progress that keeps things moving. Furfurylamine falls into this camp. Its roots trace back over a century to the rise of the furfural industry, which itself is born from the agricultural world — from oat hulls, corn cobs, and sugarcane bagasse, all of which once went to waste. Furfural soon got attention as a platform chemical and it didn’t take clever chemists long to see amines like furfurylamine as the next useful step. Over decades, as markets shifted and regulations changed, furfurylamine kept finding its way into new niches, even though it never grabbed headlines.
Product Overview
As a compound, furfurylamine stands out for its simple structure: a furan ring tagged with a single aminomethyl group. This one small modification is enough to bridge the worlds of natural resources and industrial chemistry. The raw material story speaks to the ongoing need to innovate sustainably, reusing byproducts and closing loops. The presence of a nitrogen atom alongside the aromatic furan ring makes furfurylamine a practical linking agent in making medicines, agricultural chemicals, and specialty polymers. On a daily basis, labs and reactors see it less as a commodity and more as a useful toolkit item for custom synthesis and modification.
Physical & Chemical Properties
Furfurylamine has a pungent odor that’s hard to forget — one whiff marks a day in the lab. It usually shows up as a light yellow liquid. It mixes easily with water and alcohols, and reacts with acids to form salts. The boiling point edges into the mid-170°C range, which tells you that it won’t just take off in open air but also needs care during heating. The furan ring brings aromatic stability but remains reactive toward things like acids or oxidizers. Its primary amine group means it has the ability to participate in a whole range of chemical reactions: acylations, alkylations, and condensations to name a few.
Technical Specifications & Labeling
Regulations and transport guidelines treat furfurylamine with respect, but not alarm. Packaging calls for tight seals, stability against sunlight, and careful labeling. Transport tags highlight it as both flammable and harmful if swallowed or inhaled, but risk management really starts with the person opening the bottle. The amine’s reactivity and volatility mean that even a spill has to be met with gloves and goggles. There’s enough history with the chemical that those in the business know to store it away from acids and oxidizing agents, with good ventilation and clear signage.
Preparation Method
Classic preparation relies on reducing furfural with ammonia. There’s nothing mysterious: furfural’s carbonyl group, meeting hydrogen and an ammonia source, creates the primary amine. Industrial recipes tend to use catalytic hydrogenation, with metals like nickel doing the heavy lifting at raised pressure and moderate temperatures. There’s been plenty of fine-tuning over the years to increase yield, minimize byproducts, and make sure the process keeps up with environmental standards. Smaller labs often opt for milder, batchwise hydrogenation, where the lower scale makes personal oversight easier.
Chemical Reactions & Modifications
Furfurylamine steps up as a building block in many synthetic pathways. The amine function opens doors to coupling with carboxylic acids, leading to amides that show up in drug development or specialty polymer backbones. Reactivity with aldehydes and ketones brings Schiff bases, which provide color and function to dyes and sensors. If you react it with acyl chlorides or anhydrides, you get bespoke amides, valuable in medicinal chemistry. N-alkylated versions serve as custom ligands in catalysis. The furan ring can be oxidized, halogenated, or even opened, creating materials that range from fragrance fixatives to electronic intermediates.
Synonyms & Product Names
Names for furfurylamine pop up depending on the context: 2-furylmethylamine, 2-(aminomethyl)furan, or sometimes just FFA. Certain catalogues list trade names, but most chemists recognize it by its basic descriptors. This can sometimes trip up newcomers, so a close look at chemical structure goes a long way. Naming confusion happens most in global trade, where translation and regulation cross paths, and makes a strong case for sticking to established IUPAC or CAS conventions.
Safety & Operational Standards
Working with furfurylamine demands respect for its volatility and toxicity. Chronic exposure risks aren’t fully mapped, but acute symptoms from inhalation or skin contact show up quickly. Most up-to-date safety sheets point to gloves, goggles, and working in a chemical hood. Industry standards treat it as a hazardous substance, calling for training and clear emergency plans. Cleanup procedures get extra layers: spills need to be contained, not just wiped away, because the amine can linger in the air. Disposal routes pass through licensed hazardous waste processors to avoid contamination of air or water. Regulators check that companies track quantities closely and report releases promptly, showing that public safety stays on the radar.
Application Area
Furfurylamine finds many uses. In pharmaceuticals, it acts as a key intermediate for antihypertensive agents, antimalarial drugs, and sometimes custom peptide modification. In polymer chemistry, it helps produce resin systems with heat and chemical resistance, properties in demand for industrial adhesives and coatings. Agrochemicals make use of its reactivity in designing new pesticides. Electrochemical research, especially on sensors, benefits from the furan ring’s electron-rich character. Even in fragrance chemistry, modified forms of furfurylamine support the creation of new aroma profiles. What connects these uses is the ability to combine carbon, oxygen, and nitrogen into compact, adaptable frameworks.
Research & Development
The pace of R&D picks up every year, with green chemistry goals setting much of the agenda. Synthesizing furfurylamine from renewable feedstocks remains attractive, not only for its resource efficiency but also for a smaller environmental footprint. Academic labs dig into reaction mechanisms, aiming to reduce hazardous side products and find catalysts that work under milder conditions. In medicinal chemistry, there’s growing curiosity about incorporating the furan system into biologically active molecules, with furfurylamine serving as a reliable starting point. Materials science teams use it to design advanced resins with precise electrical and barrier properties, hoping to tap into new markets or upgrade existing ones.
Toxicity Research
Toxicological studies offer a mixed picture. High doses in animals point to organ damage, while human data remains limited, with occasional reports of respiratory or skin irritation. Researchers keep a close eye on potential links to carcinogenicity, particularly given its relationship to furan compounds, some of which have reputations for risk. Long-term inhalation or unprotected handling is discouraged. Regulatory bodies monitor emerging research, reminding users that safety needs regular updates and not just a one-time review. Companies and researchers often share best practices around ventilation, personal protection, and medical surveillance, recognizing that workplace habits make the difference between accident and routine.
Future Prospects
Looking forward, furfurylamine faces stronger regulation and mounting pressure for green, sustainable chemistry. Its ties to agricultural byproducts make it a test case for waste valorization on a global scale: fewer inputs from petroleum, more from farms and forests. Synthetic routes will continue shifting towards lower temperatures, greener solvents, and life-cycle transparency. As markets drive demand for custom amines with furan rings, researchers may unlock new reactions or materials with smaller energy footprints. At the same time, safety data and toxicity insights need to catch up with the compound’s increasing use. With careful stewardship, furfurylamine stands to bridge the gap between greener chemistry and practical application, reminding us that the biggest changes often start with small, adaptable molecules.
What is Furfurylamine used for?
Looking at the Real Uses of Furfurylamine
Furfurylamine sounds like something from a chemistry class, but this compound quietly keeps factories and research labs running. It’s an organic building block, drawn from furfural, which itself is made from plant materials like corncobs and oat hulls. This isn’t just about fancy names or abstract chemistry. When you follow the trail of furfurylamine, you’ll see it woven through countless products.
Pharmaceuticals: More Than a Raw Material
Furfurylamine goes into making medicines—things people count on for their health and comfort. Chemists reshape its structure to produce drugs that treat everything from Parkinson’s disease to high blood pressure. I’ve spoken to researchers who see compounds like this one as tools for fighting tough illnesses, not just places to tinker with molecules. You see a direct impact—patients gain another chance at relief, and sometimes, survival. Drugs based on such amines can reach the market after years of testing, but that chain starts with something as surprisingly humble as furfurylamine.
Agrochemicals and Protecting Food Supplies
Out in the fields, farmers need ways to keep crops healthy and pests in check. Furfurylamine helps make plant protection products, especially fungicides and herbicides. The world loses unimaginably large harvests each year to blight, weeds, and mildew. Take away these protective chemicals, and grocery shelves would start to look pretty bare. Safe and careful use means fewer losses and more predictable food supplies.
Materials and Industry: Resins to Polymers
Furfurylamine also helps create polymers and resins, especially those that need to stand up to heat and chemicals. Foundries use it for special binders in sand casting, giving molds the flexibility and strength to shape molten metal. The coatings on industrial floors and tanks can trace their long-lasting power back to this very ingredient. If you’ve toured an aging factory and wondered why the machinery still works so well, part of the credit belongs to these tough resins.
Challenges and Responsibility
With any chemical, safety brings big questions. Some forms of furfurylamine irritate skin and lungs, and spills can harm workers and the environment. My time on a plant safety team showed me how attentive companies must be—training folks, keeping air handled, running tight spill control. Regulations force the hand, but a culture of care always beats rule-following alone.
Furfurylamine’s origin in bio-based raw materials gives it some green potential, though it doesn’t erase the need for caution. Pushing for improved plant processes and using safer feedstocks lessens risks. Those working in the supply chain benefit from these efforts, and so do people living near factories.
Where We Go from Here
Innovation works best when it includes everyone—chemists, farmers, shop workers, health experts. People should keep finding ways to handle furfurylamine safely, keep emissions in check, and use it where it truly adds value. Sharing what works and what doesn’t, along with inviting outside review, brings better results than any closed-door decisions. That’s how industrial chemistry can support both business and community health, and why watching compounds like furfurylamine makes sense for anyone interested in safer, smarter manufacturing.
What is the chemical formula of Furfurylamine?
Chemistry at the Heart of Everyday Materials
In the world of chemicals, names can sound intimidating, but often the story behind them is pretty straightforward. Take furfurylamine. It crops up in more places than you’d expect, from industrial resins to pharmaceuticals. The chemical formula for furfurylamine is C5H7NO. This formula speaks to the building blocks that make up this compound — five atoms of carbon, seven of hydrogen, one nitrogen, and one oxygen. It’s a simple structure, but it leads to a molecule with some strong real-world uses.
Why Furfurylamine Matters in Industry and Research
Furfurylamine comes straight out of the chemical toolbox, especially in resin manufacturing and the pharmaceutical industry. Companies use it as a starting material to make furfuryl-based resins, which show up in things like foundry sand binders and corrosion-resistant coatings. As someone who has spent time in manufacturing labs, I’ve seen how furfurylamine’s unique combination of amine and furan ring gives it a versatility not found in many other compounds. Those same amine groups help it react easily with acids and aldehydes, making it a reliable workhorse for chemists seeking to build up more complex molecules.
Besides its use in resins, furfurylamine is a stepping stone for drugs and agrochemicals. Its molecular structure allows for tweaks and transformations, helping chemists design molecules that show up in antifungal agents, antihistamines, and even pesticides. Every test I’ve run involving furfurylamine reminds me that chemistry isn’t just theory; it’s hands-on, and every drop matters.
Safety and Environmental Impact
Handling furfurylamine demands respect for both its benefits and its risks. Like many amines, it packs a potent odor, and exposure can irritate skin, eyes, or lungs. From my time in the lab, gloves and fume hoods become non-negotiable. Not everyone outside the chemical field sees the care that goes into using chemicals like furfurylamine. Keeping processes safe means constant vigilance — good protocols keep both workers and the surrounding community protected.
On the environmental front, furfurylamine is synthesized from furfural, which comes from agricultural byproducts like corncobs and oat husks. This gives it a renewable angle that’s pretty rare among chemical feedstocks. Still, disposal calls for attention, since amines can pose hazards to waterways and soil if not neutralized properly. One promising shift I’ve seen involves “greener” synthesis methods that cut down on toxic byproducts and energy use. Some research groups have been working to refine catalysts so that less hazardous waste comes out the other end. This represents the kind of practical innovation that drives both business and public safety forward.
Looking to the Future
With tight supply chains and climate conversations front and center, the push for renewable-source chemicals has gained momentum. Furfurylamine, made from plant waste, fits right in with these goals. Research keeps drilling for better ways to make chemicals safer, cleaner, and less wasteful. That spirit of improvement reflects what drew me to chemistry in the first place — a hands-on promise to mix scientific curiosity and practical progress. Furfurylamine’s simple formula, C5H7NO, sits at the crossroads of creativity and responsibility, showing that even basic molecules can play a part in shaping safer, smarter industries.
Is Furfurylamine hazardous or toxic?
What is Furfurylamine?
Furfurylamine holds a spot on the shelf of industrial chemicals, popping up in labs, manufacturing, and even in chemical research. Its structure comes from furan—a five-membered aromatic ring—and an amine group. On paper, it looks basic, but any chemical with reactive groups deserves a second look. Understanding how it acts around people and the environment goes far beyond memorizing chemical diagrams.
Real Hazards and Health Risks
Anyone who’s dealt with furfurylamine in a lab or read its safety sheets knows it requires careful handling. You smell it first—a strong, ammonia-like odor. The fumes hit your nose hard, hinting that this isn’t something harmless like table salt or sugar.
Getting it on your skin can cause irritation. Eyes burn if a splash catches you off-guard. The liquid seeps through gloves that aren’t chemical-resistant. Workers without proper personal protective equipment end up with red, irritated skin pretty fast. Research papers highlight these common reactions, but every lab tech who’s cleaned a furfurylamine spill remembers the sting in their fingers.
Inhalation risks raise even more concern. The vapors irritate the respiratory tract—coughing and shortness of breath follow direct exposure. High concentrations in the air can cause more severe outcomes, like headaches or dizziness. Accidental swallowing isn’t likely in a workplace, but if it happens, expect nausea, abdominal discomfort, and possibly worse.
Is it a Big Threat to the Environment?
Factories using furfurylamine generate liquid waste, and runoff into waterways can’t be shrugged off. The compound breaks down under certain conditions, but direct environmental release puts fish and aquatic life at risk. Because furfurylamine contains nitrogen species, it encourages unwanted algae blooms and disrupts the natural balance in streams and ponds. Even small leaks from industrial sites can accumulate in sediment.
Evidence and Regulation
Standard chemical safety sheets cite furfurylamine’s classification as an irritant, based on workplace injury reports and animal studies. No major health agency lists it under the most dangerous chemicals, but regulators like OSHA and the European Chemicals Agency require strict hazard labeling. Notably, furfurylamine hasn’t appeared in large-scale toxic exposure lawsuits, but that doesn’t mean the risks are imaginary.
Lack of widespread poisonings doesn’t equal safety for regular users. A handful of chronic exposure studies on lab animals show liver and kidney effects at high doses—proof that taking shortcuts on protective gear brings real risks over time.
How to Lower the Risks
Using a chemical fume hood isn’t optional. Gloves made from nitrile or neoprene keep hands safe, and splash goggles protect eyes. Safety routines and employee training make the difference. Spill control plans and regular ventilation checks also keep exposures low.
Waste must go into sealed containers meant for hazardous chemicals, never down the drain. Environmental monitoring catches leaks early before they can hit streams or groundwater.
Looking Forward with Responsibility
Choices in chemical handling ripple outward: toward coworkers, the community, and the larger ecosystem. Furfurylamine brings benefits in research and industry, but safe use and respect for its hazards aren’t negotiable. Pay attention to those strong odors and warning labels—they’re the frontline defense for worker health and nearby environments.
How should Furfurylamine be stored?
Understanding the Real Risks
Furfurylamine turns up in plenty of chemical labs, often getting used in pharmaceuticals and organic synthesis. If you’re handling it often, you start to care less about generic warnings and more about concrete steps. People learn fast that improper storage causes headaches—literally and legally. I’ve seen what happens when a container leaks its sharp smell across a lab. Even one careless moment leads to contamination, health complaints, or even bigger hazards if sparks or heat are around.
The Importance of Using the Right Containers
Most folks pick chemical-resistant, tightly sealed bottles. Polyethylene or glass works well. Avoid old repurposed jars or rusty tins. Chemical compatibility isn’t just advice; it’s a shield against nasty reactions or leaking. I once worked with a team who kept furfurylamine in a basic plastic jug meant for cleaning fluid; we ended up with a slow, persistent leak that stained a whole lower shelf. Nobody wants to clean up a sticky, toxic mess by surprise. Labels matter too—nothing beats clear, unambiguous identification. Misreading a faded label, especially on a busy morning, leads to mistakes you won’t live down.
Keep It Cool—But Not Icy
Some chemicals beg for refrigeration, but furfurylamine holds up just fine at room temperature. Still, heat kicks up the vapors and increases fire risk. Find a stable, dry place away from heaters, windows, and direct sunlight. I like upper shelves in a ventilated cabinet, away from acids, oxidizers, and ignition sources. Moisture bites too—seeping water invites reactions, and the container can swell, become brittle, or corrode from inside. Investing in a dehumidifier or at least checking for damp corners every so often pays off in less hassle later.
Ventilation Prevents Surprises
Breathing in furfurylamine’s vapors makes you sick. Headaches, nausea, and throat tightness set in pretty quick. That’s why storing it in a chemical fume hood or ventilated chemical storage cabinet gives much more peace of mind. Most labs already have the infrastructure, but it’s surprising how often folk slide bottles onto random open shelving or let fumes wander. I had one mentor who checked airflows religiously, especially after moving chemical stocks. Better habits stop those silent buildup-and-boom types of accidents.
Access Control and Emergency Prep
Leaving hazardous stuff out on an open shelf isn’t just sloppy; it's a security risk. Unauthorized access leads to deliberate misuse or accidental spills. I favor locked cabinets, sign-in logs, and regular audits. Testing fire extinguishers, knowing where eyewash stations sit, and posting spill instructions can make all the difference the one time you really need them. A team that trains together, drills for emergencies, and takes inventory keeps everyone safer and more relaxed.
Why Protocols Matter—And What to Change
Proper storage runs deeper than ticking boxes for compliance. It’s about protecting the people who work near these chemicals and avoiding accidents that can injure, halt research, or wreck expensive equipment. Digital inventory systems help track chemical stocks in real time and remind teams when supplies expire. Double-checking every step, even if it seems tedious, builds confidence and strengthens trust among colleagues. Ignoring simple steps today only brings bigger problems tomorrow.
Final Thoughts on Responsibility
Practicing good storage habits with furfurylamine isn’t just about obeying rules. It’s about building a culture where everyone looks out for each other. Smart choices—secure containers, dry and cool storage, proper ventilation, and strict access—send a message: safety comes first, always. That commitment shows in every quiet, uneventful shift where nothing goes wrong, and that’s how it should stay.
What are the physical properties of Furfurylamine?
Understanding the Basics
Furfurylamine crops up in labs and factories thanks to its structure. This organic compound carries a furan ring fused to a methylamine group. Just looking at it, this clear-to-yellow liquid has a signature, slightly fishy odor. That smell isn’t just quirky — it tells you about its amine roots and reminds folks to treat it carefully.
Getting to Know Its Physical Traits
Pour furfurylamine into a beaker and it flows fast. Its viscosity runs on the low side, so it spreads easily. At room temperature, furfurylamine stays liquid, with a boiling point close to 145 degrees Celsius — not one for open flames or careless heating. That matters for storage and handling, especially since it can give off vapors that irritate eyes and the nose.
The density comes in at about 1.09 grams per cubic centimeter. In practice, that means it feels heavier than water, so spills sink rather than skate on the surface. Many chemists pick up on the ammonia-like tang right away — a signal of that lone amine attached to the ring, and a reminder to watch for skin and respiratory exposure.
Solubility and Mixing Behavior
Put furfurylamine into water, and it mixes well due to the polar amine group. This isn’t like putting oil in water. Furfurylamine blends right in, but in industrial work, it’ll also dissolve in common organic solvents like ethanol or ether. People handling it rely on gloves and fume hoods, not just for spills but for the stubborn vapor that lingers.
Because of that water solubility, furfurylamine makes for a handy building block in chemical syntheses. It’s not just its ability to mix — it reacts with acids to form salts and can jump into more complicated reactions to build pharmaceuticals, resins, and more.
Stability and Everyday Safety
Furfurylamine won’t explode in the lab, but it won’t sit around happily with strong oxidizers or under open air, either. Leave a bottle open and expect the liquid to slowly catch moisture and unwanted byproducts. The color can darken as air creeps in, which tips off anyone watching that aged or contaminated stock should be handled with extra care.
People who’ve worked with amines learn fast that ventilation and protective equipment cut down on headaches and health risks. Even a small splash can irritate skin and eyes, so the safety shower and eye-wash station stay close. The sharp odor also doubles as a pre-warning for leaks and spills.
Environmental and Practical Impact
Because furfurylamine isn’t persistent like some big industrial solvents, it breaks down more easily when exposed to sunlight and microbes. That matters in waste streams, since it doesn’t hang around for years. Simple precautions like storing it in dark, tightly sealed bottles and working with small batches help keep unintended exposure low.
Most people working with it swear by clear labeling and double-checking storage — furfurylamine’s properties don’t care whether you’re a veteran chemist or a new tech prepping samples. Its smell, solubility, and fluidity say a lot about its character and signal why so many industries keep it in stock. Respecting those traits makes for smoother work and safer labs.
| Names | |
| Preferred IUPAC name | (2-furanylmethyl)amine |
| Other names |
2-Furylmethylamine 2-Furfurylamine Furfuran methylamine alpha-Furylmethylamine Furylmethylamine |
| Pronunciation | /ˌfɜːrfjʊrˈɪləmiːn/ |
| Identifiers | |
| CAS Number | 100-58-3 |
| Beilstein Reference | 1209243 |
| ChEBI | CHEBI:5004 |
| ChEMBL | CHEMBL25338 |
| ChemSpider | 54615 |
| DrugBank | DB08395 |
| ECHA InfoCard | 100.011.939 |
| EC Number | 205-999-9 |
| Gmelin Reference | 7307 |
| KEGG | C01745 |
| MeSH | D005693 |
| PubChem CID | 8025 |
| RTECS number | WN8570000 |
| UNII | 9YGH295QQF |
| UN number | UN2676 |
| CompTox Dashboard (EPA) | DTXSID3049183 |
| Properties | |
| Chemical formula | C5H7NO |
| Molar mass | 97.13 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | amine-like |
| Density | 0.981 g/mL at 25 °C |
| Solubility in water | miscible |
| log P | 0.43 |
| Vapor pressure | 0.6 mmHg (20 °C) |
| Acidity (pKa) | 9.03 |
| Basicity (pKb) | 7.44 |
| Magnetic susceptibility (χ) | -7.05e-6 cm³/mol |
| Refractive index (nD) | 1.488 |
| Viscosity | 1.104 cP (25 °C) |
| Dipole moment | 1.96 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 108.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -35.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2105.7 kJ/mol |
| Pharmacology | |
| ATC code | C01CC06 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H314, H317, H411 |
| Precautionary statements | P210, P260, P280, P302+P352, P304+P340, P312, P330, P363, P501 |
| NFPA 704 (fire diamond) | 3-3-2 |
| Flash point | 60°C |
| Autoignition temperature | 555°C |
| Explosive limits | 1.7–9.8% |
| Lethal dose or concentration | LD50 oral rat 214 mg/kg |
| LD50 (median dose) | LD50 (median dose) of Furfurylamine: 205 mg/kg (oral, rat) |
| NIOSH | KW2975000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Furfurylamine: Not established |
| REL (Recommended) | 160 ppm |
| IDLH (Immediate danger) | IDLH: 100 ppm |
| Related compounds | |
| Related compounds |
Furan Furfuryl alcohol Furoic acid Furan-2-carbaldehyde (Furfural) Pyrrole |

