Fluorosulfonic Acid: An Editorial Look at a Molecule That Demands Respect
Historical Development
Long before "superacid" became a buzzword in the world of chemistry, scientists tried to push the limits of what acids could do. Fluorosulfonic acid, with its formula FSO3H, wasn’t just discovered overnight. Early work with sulfuric acid and its close relatives opened the door, but it took chemists digging through reactions with sulfur trioxide and hydrogen fluoride to finally crystallize the compound. By the middle of the 20th century, as research into organofluorine chemistry heated up, lab groups recognized FSO3H’s terrifying strength and began to record its action against molecules assumed to be stable. History shows the acid became a tool not out of necessity, but because of how much it changed the field. It pushed scientists to rethink traditional views on molecule reactivity, stability, and industrial processing.
Product Overview
A single whiff of fluorosulfonic acid reminds anyone in the lab that not all acids belong in the same category. This compound finds use where others simply wither. It cracks open hydrocarbons, helps chemists build up and tear down carbon frameworks, and even serves as a stand-in for sulfuric acid when increased activity is needed. Despite its toxicity and handling challenges, the features that make it dangerous also give it an essential role. Many superacids have come onto the scene, but very few can stand with FSO3H for raw, predictable strength. The acid often carries names like fluorosulfuric acid and, in older texts, hydrogensulfuryl fluoride, reflecting the different routes or intentions chemists pursued in its development.
Physical & Chemical Properties
Clear, colorless, and fuming, fluorosulfonic acid might look innocent, but it packs a potent punch. On contact with air, it gives off a dense, acrid vapor. Its boiling point sits at around 165°C, and it weighs in a little heavier than water, making it easier to separate from less dense liquids in the lab. What sets FSO3H apart is its unique blend of reactivity and willingness to rip protons off even stable substances. It laughs at glassware, chewing it up slowly. Metals buckle under its bite unless protected. Water? Drop even a taste into a glass and it goes off with a violent hiss and splatter, releasing toxic fumes. These properties make even the smallest mishandling a recipe for disaster, dictating a culture of respect wherever it’s used.
Technical Specifications & Labeling
Chemistry doesn’t care about pretty labels, but regulations demand clarity. Strong, clear containers and proper hazard symbols act as the first layer of defense in any storage room. For fluorosulfonic acid, specialized polytetrafluoroethylene, nickel, and silver containers keep it locked away from curious eyes and unsteady hands. The acid never shares storage with water, bases, or organics prone to rapid oxidation—mistakes in segregation feature in many cautionary tales. Key documentation details not just purity and assay, but the presence of water or hydrofluoric acid as impurities, which can tip the balance from dangerous to deadly. That level of transparency offers users a measure of control in a field packed with uncertainty.
Preparation Method
Making FSO3H centers on passing sulfur trioxide over anhydrous hydrogen fluoride. This reaction, usually run under tightly controlled temperatures, yields the acid directly. Scaling up the process means sealed systems, precise temperature monitoring, and corrosion-proof equipment. Laboratory methods can get away with smaller volumes, but production batches must consider purity and recovery, as well as how to handle or recycle leftover hydrogen fluoride—a substance nearly as infamous as FSO3H itself. Operators who run these setups develop a unique sense of caution and respect, knowing every leak or pressure change signals real danger.
Chemical Reactions & Modifications
Fluorosulfonic acid sits among a handful of superacids to teach seasoned chemists humility. It protonates nearly anything with a weak enough bond and shatters the illusions of stability in hydrocarbons and organic molecules. The acid turns ethers to alcohols, demethylates aryl groups, and acts as a catalyst in tough alkylation and isomerization reactions. In combination with antimony pentafluoride, FSO3H yields the legendary “Magic Acid,” a reagent famous for protonating saturated hydrocarbons—something nature itself seems to avoid. The field of carbocation chemistry owes much of its depth to the ability of FSO3H to generate and stabilize these short-lived intermediates. Attempts to modify or control this acid through derivatives or blended superacid mixtures often chase a simple goal: harness some of its raw power while dialing back the chaos it brings.
Synonyms & Product Names
Fluorosulfonic acid wears many masks in the chemical world. Some call it fluorosulfuric acid or hydrogensulfuryl fluoride; others shorten the name to its chemical shorthand FSO3H. Each label carries a bit of the compound’s history, a trace of the paths chemists took to understand or tame it. While trade and regulatory names surface from time to time, the acid’s reputation depends more on its chemical identity than any brand or label. In research labs, the shorthand speaks volumes—a small bottle marked FSO3H signals expertise more than most paperwork.
Safety & Operational Standards
Surviving a day with fluorosulfonic acid in the lab means sticking to strict routines. Chemical splash goggles, acid-resistant gloves, and face shields become daily friends, not options. Fume hoods always run at full tilt whenever the cap twists off the bottle. Every drop gets neutralized, and every spill triggers alarms. Chemical burns from this acid can cut through flesh to the bone, and inhalation risks bring swelling and corrosion down the airways. Emergency drench showers and eye wash stations stay within arm’s reach—sometimes the only defense if things go wrong. Training never stops; stories about old accidents serve as grim reminders for new workers. Complacency breeds disaster faster with FSO3H than almost any other chemical in regular use.
Application Area
Research chemists never reach for FSO3H out of laziness. They pull it down when they want to break ground on tough transformations, open sturdy molecular bonds, or build new frameworks for drugs and materials. Petrochemical breakthroughs, the birth of new carbocation chemistry, and innovations in organofluorine compounds all draw links back to the acid’s unique skill set. Refineries and chemical plants tap its power to cut down processing steps, nudge recalcitrant molecules into action, or strip impurities from valuable starting materials. That reach extends into pharmaceuticals, where superacid chemistry scrambles stubborn functional groups into patterns that translate to new antibiotics and therapies.
Research & Development
Fluorosulfonic acid’s history runs deep in research labs, where the rush to find stronger acids shaped whole careers. Decades ago, the acid's contributions to carbocation chemistry reshaped organic reaction theory. Detailed study of its protonation power opened doors to stable ion intermediates and less predictable transformations. Today’s innovation focuses on both safer handling and new reaction types. Scientists want to keep the reactivity but reduce the threat, looking to blend FSO3H in controlled superacid combos or immobilize it on solid matrices for easier work-up and disposal. Funding skews toward automation, sealed microscale reactions, and continuous processing—all striving to amplify results while paring back risk. Recent research continues to expand its use in fields like proton fuel cells and specialty material synthesis, pushing boundaries in both understanding and application.
Toxicity Research
Serious work on the toxicity of fluorosulfonic acid confirms what intuition screams—this substance can cause more than just surface burns. Animal studies point to severe corrosive effects through skin, lungs, and digestive tract even at low exposures. Chronic inhalation studies remain elusive, mostly because few would stick with a test volunteer long enough to gather tough data. Environmental toxicity follows similar patterns, with acid spills laying waste to plant and microbial communities in short order. Safe disposal and robust neutralization remain strict priorities in both academic and industrial labs, backed up by published accident records and medical data. Modern research tools help detect exposures at much lower thresholds, giving a bit more room to intervene before lasting damage sets in.
Future Prospects
Fluorosulfonic acid will keep a seat at the table of synthetic chemistry, not because it's easy to work with but because its power opens new doors others can barely knock on. Advances in containment and automation can shift more work toward closed system reactions, reducing human contact and lowering risk. Materials science and catalysis offer ripe opportunities, using FSO3H in role of proton conductor for fuel cells or as a building block for high-energy materials. Research on greener, safer acids carries weight, but experience in industry suggests nothing quite matches the crisp efficiency of FSO3H for certain tasks. The future may see applications expand as long as hazard mitigation keeps pace, offering a blend of potential and peril that’s drawn scientists since the earliest days of acid discovery.
What is Fluorosulfonic Acid used for?
The Backbone of Superacid Chemistry
Fluorosulfonic acid makes even seasoned chemists take a breath. I remember my first encounter with it in a university lab—the warning signs alone could give anyone pause. This acid burns through gloves, glass, and careless habits faster than just about anything on the shelf. Its ferocity gives it some unique advantages, especially when other acids just don’t cut it.
Where Industry Relies on Raw Strength
Down at the nuts and bolts, flurosulfonic acid drives reactions you don’t get from weak acids. Drug companies, big plastics plants, and even the folks behind fancy batteries depend on this intimidating chemical. It stands out as one of the strongest acids ever made, stronger than sulfuric or hydrochloric. Engineers and chemists use this quality, especially for making certain advanced pharmaceuticals. You throw a molecule at fluorosulfonic acid and it takes apart bonds most acids barely nudge. I’ve heard colleagues say nothing else can give you the same clean, sharp results when you’re after hard-to-reach chemical targets.
Sparking Complex Reactions
Take catalysis—speeding up reactions in factories. Some processes, like alkylation or isomerization, run best with a superacid. Fluorosulfonic acid pushes stubborn chemical groups into motion. The oil industry, for example, taps into this for making fuels cleaner or more efficient. It isn’t something you pour like vinegar or lemon juice. A misstep brings burns that linger for weeks. So, strict protocols and a culture of safety shape everything about its use.
Taming the Wild Nature
Researchers can’t ignore the environmental and safety headaches. If fluorosulfonic acid gets loose, it chews through lab benches and protective gear and leaves lung-damaging fumes. I once watched an emergency team clean up after a lab spill; you could see the tension in everyone’s faces. This experience sticks with people and shapes their respect for the acid. Factories that use it build in serious ventilation and have their own fire teams. Strict training stands up next to every beaker, and waste disposal follows rules backed by years of hard lessons.
Innovation Meets Caution
Technology keeps risk in line. Labs now have automated systems for handling dangerous acids, and some companies test new materials to build safer pipes and storage tanks. The world’s moving toward greener chemistries, yet for some tasks, there’s no swap. This means ongoing research for alternatives, and engineers sharing knowledge about best practices. Universities and industries have training programs so workers don’t treat this acid like just another chemical. In my experience, teamwork and communication make every difference when the stakes include both progress and people’s safety.
The Balancing Act
Fluorosulfonic acid stands as an example of how chemistry’s strongest tools bring both big breakthroughs and big risks. It takes more than technical skill to handle such hazards; it needs steady oversight, constant learning, and the resolve to put safety above shortcuts. As new challenges in medicine, energy, and manufacturing appear, folks in the field keep searching for smarter, safer ways to wield this chemical giant.
Is Fluorosulfonic Acid dangerous or corrosive?
Real Dangers in the Lab—Far Beyond Ordinary Acids
Fluorosulfonic acid isn’t just another chemical with a scary name. It is one of the strongest acids known, packing a punch far above what most folks ever handle. Most people have seen hydrochloric acid at some point, maybe cleaning concrete or in a high school lab. Fluorosulfonic acid, though, doesn’t play in the same league. Gloves, goggles, even lab coats—these only go so far before this acid finds a way through.
Anyone who’s worked with acids learns respect fast. Some lessons stick quicker than others. Watching a drop of strong acid eat through a bench surface or even start to vaporize on contact with water tends to make an impression. What makes fluorosulfonic acid a real beast is how it reacts with water and organic materials. It releases toxic fumes and heat on contact with even a hint of moisture—the kind of reaction that goes from nothing to dangerous in a blink.
Scars and Stories: Firsthand Encounters Matter
Old chemists like to swap stories about “close calls.” They remember the time a bottle of acid sliced through a glove or the emergency run to the eyewash station. Even a tiny spill of fluorosulfonic acid can cause severe burns and permanent scars. I have watched a thick rubber mat dissolve after a splash, reminding everyone in the lab that some safety gear just doesn’t stand up to this level of corrosion. Not every acid leaves behind that kind of memory.
Immediate Damage—This Is Not a DrillSkin contact means instant burns. Inhalation means coughing, choking, and risking long-term lung injury. Unlike milder acids, this one doesn’t give warnings in the form of irritation or faint smells. Accidents with it don’t just ruin equipment. They threaten lives. The Environmental Protection Agency and the National Institute for Occupational Safety and Health both flag this chemical as a major hazard. Safety data sheets spell it out in bold: only handle this stuff with extreme care, ideally behind thick glass and with serious ventilation.
Not Just a Science Problem—A People Problem
The risk doesn’t end in the lab. Many industries use fluorosulfonic acid for specialized jobs like making chemicals, batteries, or advanced plastics. Improper storage or transportation puts not just workers, but whole neighborhoods in danger. Even a leak in a commercial setting invites disaster if emergency plans fall short or staff don’t know the right response. In 2012, a minor spill forced evacuations at a chemical plant in Europe. Lucky for everyone involved, separation and quick thinking kept things from going much worse.
Addressing the Risk: Real Solutions Needed
No single answer solves the danger. Facilities working with this acid must keep tight controls on who can access it. Routine training, regular safety drills, and near-obsessive attention to proper protective equipment create layers of security. Using automated systems to transfer acid, storing it in reinforced containers, and ensuring every spill response kit is in working order gives workers better odds.
Most important, honest conversations about risk help everyone develop proper respect for chemicals that don’t forgive mistakes. Hiding behind jargon or pretending every danger can be managed with a checklist leads to trouble. Sharing real experiences, reading near-miss reports, and refusing to cut corners ensures this acid’s only legacy is scientific progress—not tragedy. Fluorosulfonic acid deserves every bit of caution. That hard-earned wisdom saves hands, lungs, and sometimes even lives.
How should Fluorosulfonic Acid be stored and handled?
Seeing the Bigger Picture with Fluorosulfonic Acid
Fluorosulfonic acid doesn’t mess around. Just a quick look at its properties tells you this stuff brings out the big guns. With acidity that beats even sulfuric acid and a serious knack for destroying most materials it touches, this chemical reminds science veterans and rookies alike why the lab isn’t for the cavalier. Years in industrial settings have taught hard lessons about treating potent chemicals with direct respect instead of taking shortcuts. A drop in the wrong place, even for a second, can mean disaster.
Choosing the Right Storage Container
The biggest mistake people make is underestimating what this acid can eat through. Standard glass won’t last long, and most plastics just crumble. Fluorosulfonic acid requires its own league of containers: fluoropolymer bottles like PTFE or specialty corrosion-resistant alloys. Ordinary shelves don’t count. Walk into reputable chemical storage areas, and you’ll see these acids isolated, away from organics and moisture. It’s the only way to prevent an accidental reaction or cloud of toxic gas from forming.
Ventilation and Environment
Fluorosulfonic acid fumes can stake out a room in record time if storage gets sloppy. Poor ventilation opens up a lab, warehouse, or plant to real damage—damage I’ve seen firsthand on one unfortunate Monday morning. Fume hoods protect workers, and proper signage stops anyone from getting too curious. Dedicated acid storage cabinets—designed to contain spills—keep people and equipment out of harm’s way. Lined floors and neutralizing agents nearby make a huge difference if a container leaks or tips over unexpectedly.
Personal Protective Equipment: Non-Negotiable
You can spot the veterans by how serious they take their gear—acid-resistant gloves, face shields, and chemical suits. Ordinary lab coats and thin gloves won’t hold up. A colleague once described what happened after a careless splash: rapid burns, uncontrollable pain, and a rush to the emergency station. Only a proper barrier stands between this acid and lasting injury. Eye-wash stations and emergency showers within reach save more than pride in those moments
Training and Vigilance
No two labs operate the same, but the best ones hold regular training for anything as dangerous as fluorosulfonic acid. Anyone who handles it learns emergency protocols, from quick clean-ups to full-evacuation procedures. These drills reduce panic when things go wrong, and they keep new staff from learning lessons the hard way. Fact sheets from agencies like NIOSH and material safety data sheets offer specifics that aren’t optional reading—they’re survival guides.
Safer Transportation and Transfers
Transferring fluorosulfonic acid between containers is where nerves run highest. This task happens over spill trays with absorbent materials on hand, with bottles double-checked for integrity. Rushed jobs light the way for tragedy, so only those signed off by supervisors get the responsibility. Neutralizers like sodium bicarbonate line the benches, ready to go if even a few drops get loose. Transport within facilities sticks to sturdy carts and clear communication—no solo trips, no shortcuts.
Responsibility Above All
Fluorosulfonic acid stands out because it doesn’t offer second chances. From container selection to protective gear to storage practices, every choice matters. Tempering confidence with caution shapes a knowledge-based culture that values health, safety, and community. A good record of safe handling doesn’t happen by luck—it’s built on trust, vigilance, and the lessons others have paid dearly to teach.
What is the chemical formula of Fluorosulfonic Acid?
Getting to Know Fluorosulfonic Acid
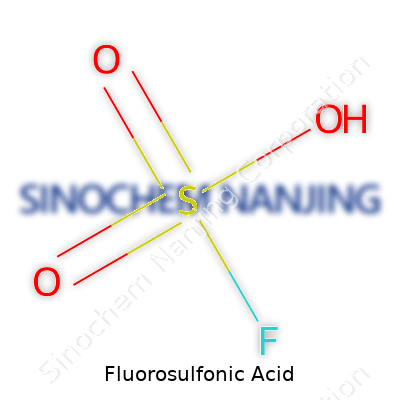
Fluorosulfonic acid ranks as one of the most powerful acids that chemists can handle. Its chemical formula is HSO3F. Unlike some laboratory acids, this one carries a reputation for both strength and unpredictability. The formula carries weight in industrial chemistry, mainly because the combination of hydrogen, sulfur, oxygen, and fluorine packs an intense punch far beyond what people see with sulfuric or hydrochloric acid.
Why This Particular Acid Deserves Attention
Safety always comes up quickly when discussing strong acids. I remember the first time our team brought it into the lab—double gloves, face shields, a fume hood set on full blast. Even a tiny spill on a bench fumed and hissed, warning us not to get comfortable around it. Fluorosulfonic acid’s strength comes from how the fluorine atom draws electrons away, making the attached proton extremely hungry for new bonds. This hunger translates to a level of reactivity you just don’t see in household acids.
The practical impact is clear: fluorosulfonic acid can easily break apart organic molecules, rip off protective groups in pharmaceuticals, and even start reactions that many acids can’t touch. Its industrial uses reflect that. People might use it to make fuel additives, specialty chemicals, or even fluorinated pharmaceuticals that would not work if made with weaker acids.
Health and Environmental Impact
Hazards can’t get ignored. If fluorosulfonic acid comes in contact with skin, it burns deep, far worse than most common acids. Breathing in its fumes could set off coughing, watery eyes, and after substantial exposure, some real medical trouble. I can still remember a seasoned chemist at my old workplace who wore a faded scar along his wrist from a careless moment.
Waste also raises hard questions. Getting rid of used or spilled acid isn’t just about pouring it down the drain—it calls for neutralization with a basic substance, contained handling, and coordination with hazardous waste professionals. If the acid meets water, it reacts violently and can release toxic fumes. Labs and factories have to set up specialized containment systems, so this stuff never ends up in local water supplies.
The Bigger Chemical Picture
People often focus only on strong acids like sulfuric or nitric in their discussions, but fluorosulfonic acid plays in a league above those. Chemists turn to it for unique reactions, often because only this acid can push certain transformations to completion. Its ability to act as a superacid means chemists lean on it for jobs that simply aren’t possible with the basics stored in high school classrooms.
Despite all its usefulness, the real story is respect—for the danger, for the possibilities, for the cost of mistakes. The commitment to strict protocols makes fluorosulfonic acid both powerful and, in the right hands, invaluable. Proper training, rigorous procedures, and constant vigilance are the price of admission for any scientist or manufacturer who dares to work with it.
Moving Forward with Caution
Every time a new process proposes using fluorosulfonic acid, experienced teams ask tough questions about containment, emergency response, and long-term stewardship. Research keeps evolving, looking for substitutes that can match its performance but bring fewer risks to health and environment. Until a safer equivalent turns up, working with this acid stays a realm for the prepared and the vigilant.
What precautions are necessary when working with Fluorosulfonic Acid?
What Fluorosulfonic Acid Brings to the Table
Plenty of people outside the lab have never run into fluorosulfonic acid. Those who have remember it. Stronger than sulfuric—one of the world’s most relentless acids—this liquid smokes in the air and chews through flesh, fabric, and even glass like it found a new hobby. You’ll see it in specialty chemistry, where nothing else cuts it. That kind of power demands real respect, because ignoring safety here leads to scars—sometimes more than just physical ones.
Personal Safety Gear Is Non-Negotiable
Fluorosulfonic acid laughs at most basic gloves and goggles. Anyone working with it needs chemical-resistant gloves, splash-proof goggles, and full-face shields—sometimes all at once. Thin nitrile gloves disappear in seconds. Heavier, specialized ones—think Viton or thick butyl—are your friend. Cotton or polyester is like kindling for the acid. Lab coats made of a chemically resistant material cut down splashes on skin or regular clothes, which don’t stand a chance.
Ventilation: Your Invisible Shield
Fumes turn the air heavy, choking those who forget to mind the air flow. Working with fluorosulfonic acid means staying under a chemical fume hood. These hoods aren’t just furniture—they’re a buffer between you and airborne acid. Standard open-air benches invite accidents and let fumes settle where nobody wants them.
Resistant Equipment Matters More Than Most Realize
Glass breaks, and plastics melt or dissolve. Only PFA (perfluoroalkoxy) or PTFE (polytetrafluoroethylene) holds up reliably in contact with this acid. Some folks try their luck with borosilicate glass, but time wears it thin. Metal? Forget it. Corrosion eats equipment and leaves shrapnel for the next unlucky user. Always check containers for signs of leaks before every use; yesterday’s “good enough” might mean today’s spill.
Spill Response: Ready Beats Sorry
Spills do happen, even for the careful. Baking soda looks good on kitchen spills, but not here. Neutralizing with calcium carbonate or soda ash works, but skip water—violent reactions spray acid everywhere. Keeping a spill kit within arm’s reach saves lives, not paperwork. Training every team member to use it—not just lab techs—keeps accidents from turning into emergencies.
Storage — Out of Sight Is Not Out of Mind
Dark, locked storage for small bottles is a given. Store it somewhere cool and dry. No acids near bases or anything organic; the wrong neighbor kicks off fires or explosions. Label everything clearly, never trust guesswork. Double containment—an acid-resistant secondary tray—catches leaks and prevents chaos on the shelf. Check storerooms every week. The world changes quickly in those bottles.
Medical Help Needs Preparation
Skin contact means a proper drench under running water, throwing away whatever got splashed, and a fast trip to medical care. Eyes exposed mean unlimited rinse time, and nobody should leave it to chance. Having an eyewash station and emergency shower nearby feels like a nuisance unless you or your coworker lands in trouble.
No Shortcut to Safe Chemistry
Working with something as tough as fluorosulfonic acid takes more than nerves. Prioritizing real-world precautions and checking every box on the safety list protects people, projects, and careers. The best chemists I know walk into every shift assuming the acid remembers every mistake. In the end, the most important chemical in the lab is caution itself, measured in seconds and sweat, not milliliters.
| Names | |
| Preferred IUPAC name | fluorosulfonic acid |
| Other names |
Fluorosulphonic acid Fluorosulphuric acid Sulfuric fluoride HSO3F |
| Pronunciation | /ˌflʊəroʊˌsʌlˈfɒnɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 7789-21-1 |
| Beilstein Reference | 3531042 |
| ChEBI | CHEBI:29336 |
| ChEMBL | CHEMBL1232131 |
| ChemSpider | 25258 |
| DrugBank | DB14286 |
| ECHA InfoCard | 100.005.346 |
| EC Number | 016-026-00-0 |
| Gmelin Reference | 676 |
| KEGG | C14147 |
| MeSH | D015226 |
| PubChem CID | 65914 |
| RTECS number | WN5060700 |
| UNII | J9P4OW8358 |
| UN number | UN1777 |
| CompTox Dashboard (EPA) | DTXSID8020093 |
| Properties | |
| Chemical formula | HSO3F |
| Molar mass | HSO3F: 100.07 g/mol |
| Appearance | Colorless to pale yellow fuming liquid |
| Odor | Pungent, suffocating |
| Density | 1.75 g/cm³ |
| Solubility in water | Miscible |
| log P | -2.0 |
| Vapor pressure | 3 mmHg (20 °C) |
| Acidity (pKa) | -10 |
| Basicity (pKb) | -14.2 |
| Magnetic susceptibility (χ) | -48.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.284 |
| Viscosity | 18 cP (25 °C) |
| Dipole moment | 3.49 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 157.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −890.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -969 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Causes severe burns, highly corrosive, toxic if inhaled, harmful if swallowed, reacts violently with water. |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H314, H331, H290 |
| Precautionary statements | H260, H301, H314, P210, P220, P260, P264, P271, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P310, P320, P330, P363, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 4-0-2-W |
| Autoignition temperature | 310 °C (590 °F; 583 K) |
| Lethal dose or concentration | LD50 oral rat 250 mg/kg |
| LD50 (median dose) | LD50 (median dose): 250 mg/kg (rat, oral) |
| NIOSH | LB5425000 |
| PEL (Permissible) | 2 ppm |
| REL (Recommended) | 0.5 ppm |
| IDLH (Immediate danger) | 30 ppm |
| Related compounds | |
| Related compounds |
Sulfuric acid Chlorosulfonic acid Perfluorooctanesulfonic acid Trifluoromethanesulfonic acid |

